Back to Journals » Lung Cancer: Targets and Therapy » Volume 12
Spotlight on Mobocertinib (TAK-788) in NSCLC with EGFR Exon 20 Insertion Mutations
Received 2 June 2021
Accepted for publication 21 June 2021
Published 12 July 2021 Volume 2021:12 Pages 61—65
DOI https://doi.org/10.2147/LCTT.S307321
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sai-Hong Ignatius Ou
Shannon S Zhang,1 Viola W Zhu1,2
1University of California, Irvine School of Medicine, Department of Medicine, Orange, CA, USA; 2Chao Family Comprehensive Cancer Center, Orange, CA, USA
Correspondence: Viola W Zhu
University of California, Irvine School of Medicine, Department of Medicine, 200 S Manchester Ave, Suite 410, Orange, CA, 92868, USA
Tel +1 714-456-8105
Fax +1 714-456-2242
Email [email protected]
Abstract: The EGFR exon 20 insertion (EGFRex20ins) mutations are the third most common EGFR mutations seen in non-small cell lung cancer (NSCLC). More than 50 variants of EGFRex20ins mutations have been identified with A767_V769dupASV being the most common variant across multiple surveys. Treatment with currently available EGFR tyrosine kinase inhibitors (TKIs) including osimertinib is generally ineffective. Amivantamab (JNJ-372), a bispecific monoclonal antibody against EGFR and MET, has recently been approved by the US FDA for patients with advanced or metastatic NSCLC harboring EGFRex20ins mutations after disease progression on platinum-based chemotherapy. Among all the TKIs in clinical development, mobocertinib (TAK-788) has been granted priority review by the FDA for the same indication as amivantamab. Here, we provide a concise review on mobocertinib, with a focus on its chemical structure, preclinical data, and phase 1/2 trial results. Future directions will likely focus on combination approach such as TKI plus chemotherapy in the first-line setting, designing drugs with CNS activity, and exploring disease characteristics of various EGFRex20ins mutation variants and how they may affect treatment response.
Keywords: mobocertinib, TAK-788, NSCLC, EGFR exon 20 insertion, TKI, amivantamab
Introduction
The EGFR exon 20 insertion (EGFRex20ins) mutations are the third most common EGFR mutations seen in non-small cell lung cancer (NSCLC), constituting up to 12% of NSCLC cases harboring EGFR mutations.1 This molecular alteration is characterized by in-frame insertions and/or duplications clustered between codons 762 and 774, resulting in constitutive activation of the EGFR pathway.2,3 More than 50 variants of EGFRex20ins mutations have been identified with A767_V769dupASV (ASV) being the most common variant across multiple surveys.1,3 Because most EGFR ex20ins mutations do not have diminished adenosine triphosphate (ATP) binding or increased affinity for EGFR tyrosine kinase inhibitors (TKIs), treatment with EGFR TKIs is generally ineffective.2 Amivantamab (JNJ-372), a bispecific monoclonal antibody against EGFR and MET, has recently been approved by the US FDA for the treatment of advanced or metastatic NSCLC patients with EGFRex20ins mutations after disease progression on platinum-based chemotherapy.4 Mobocertinib (TAK-788) has been granted priority review by the FDA for the same indication. Here we provide a concise review on mobocertinib, with a focus on its chemical structure, preclinical data, phase 1/2 trial results, and future development, in anticipation for its FDA approval in the near future.
Chemical Structure
There is significant overlap in terms of conformation between EGFR ex20ins mutations and wild type (WT) EGFR in the ATP binding pocket, rendering osimertinib’s lack of selectivity against EGFR ex20ins mutations. The amino acid insertion in the exon 20 located at the C-helix or the P-loop of the EGFR kinase pushes the C-helix into a permanent active conformation, which is distant from the ATP binding site.2 Mobocertinib was structurally designed to gain selectivity by targeting proteins in the vicinity of the alpha C-helix, a binding site not exploited by osimertinib. More specifically, in a docking model of osimertinib bound to EGFR ex20ins D770_N771insNPG (NPG) mutation, there remained a vacant pocket accessible by substitution of the pyrimidine ring with mobocertinib’s isopropyl ester. This structure design allowed mobocertinib to have increased binding affinity for EGFR ex20ins mutations than osimertinib. In addition, mobocertinib is an irreversible EGFR TKI that covalently binds to cysteine 797 in EGFR, similar to afatinib and osimertinib.3 The chemical structure is shown in Figure 1.
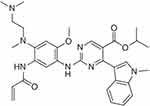 |
Figure 1 Chemical structure of mobocertinib. |
Preclinical Data
Cellular Activity
In Ba/F3 cells, mobocertinib exhibited inhibitory activity against 14 EGFR mutations with IC50 ranging from 2.7 to 22.5 nM while IC50 for WT EGFR was 34.5 nM. More specifically, mobocertinib inhibited all five variants of EGFR ex20ins mutations with IC50 of 4.3 nM for NPG, 10.9 nM for ASV, 11.8 nM for A763_Y764insFQEA (FQEA), 18.1 nM for N771_H773dupNPH (NPH), and 22.5nM for S768_D770dupSVD (SVD).3
The activity of mobocertinib was further explored in two patient-derived cell lines, CUTO14 and LU0387.3 The CUTO14 cell line was derived from a patient with NSCLC harboring the ASV mutation without any prior EGFR TKI. In CUTO14 cells, compared to osimertinib which reached 38% and 63% inhibition of phosphorylated EGFR (p-EGFR) at concentrations of 100 nM and 1000 nM, respectively, mobocertinib was able to reach 80% and 100% inhibition. Additionally, based on the CUTO14 cell viability assay, the IC50 of mobocertinib was 33 nM while it was 2679 nM for erlotinib, 1021 nM for gefitinib, 66 nM for afatinib, and 575 nM for osimertinib, respectively. The LU0387 cell line was derived from a NSCLC patient harboring the NPH mutation. In LU0387 cells, the IC50 of mobocertinib was 21 nM while it was 2793 nM for erlotinib, 364 nM for gefitinib, 20 nM for afatinib, and 195 nM for osimertinib, respectively. Even though the potency of afatinib was similar to that of mobocertinib, the IC50 of afatinib against WT EGFR phosphorylation was 3.9 nM compared to 35 nM for mobocertinib.
Biophysical and Biochemical Assays
The Biacore surface plasmon resistance assay was utilized to assess the binding affinity of mobocertinib in a mutant C797S version of the NPG mutation model. In this assay, the binding affinity of mobocertinib to the NPG mutation was similar to that of afatinib but much lower than that of osimertinib. In contrast, the binding affinity of mobocertinib to WT EGFR was much higher than that of afatinib but similar to that of osimertinib.3
The in vitro kinase assays were utilized to evaluate the kinase selectivity profile of mobocertinib. Briefly, mobocertinib was found to inhibit 28 of 490 kinases by >50% at 1 µM, including all 14 members of the EGFR family. Furthermore, the IC50 of mobocertinib was <2 nM against all these 14 EGFR family kinases including EGFR, HER2, HER4, and 11 variants, as well as BLK.3 These data demonstrated mobocertinib as a potent and selective EGFR inhibitor.
Xenographic Activity
The antitumor activity of mobocertinib in oral dosing was first investigated in two murine models harboring the ASV mutation.3 In the first model using engineered Ba/F3 cells, once daily dosing of mobocertinib at 30 mg/kg and 50 mg/kg induced 77% tumor growth inhibition and 19% tumor regression, while no body weight loss was seen with either dosing. In the second model using CTG-2842 cells derived from a NSCLC patient without prior response to erlotinib, once daily dosing of mobocertinib at 15 mg/kg caused 92% tumor regression without body weight loss.
Next, the efficacy of mobocertinib was explored in mice engrafted with LU0387 tumors containing the NPH mutation.3 Mobocertinib induced 56% tumor growth inhibition at 10 mg/kg daily and 87% tumor regression at 30 mg/kg daily. No body weight loss was observed with either dosing. In comparison, erlotinib at 50 mg/kg daily induced 38% tumor growth inhibition. Osimertinib at 30 mg/kg daily resulted in 3/10 mice with >50% tumor regression with mean tumor regression of 13%, whereas mobocertinib at the same dosing resulted in 10/10 mice with >50% tumor regression with mean tumor regression of 87%.
Furthermore, a genetically engineered mouse model expressing the SVD mutation was created to evaluate the short-term and long-term efficacy of mobocertinib.3 The treatment of two mice with mobocertinib at 30mg/kg daily for 1 week led to remarkable reduction of tumor volumes compared to the vehicle control. The lung tumors were subsequently harvested and stained for p-EGFR and its downstream signaling pathway protein phosphorylated-ERK1/2, both of which showed marked abolishment of staining by immunohistochemistry. The long-term efficacy of mobocertinib was assessed by continuous treatment of eight mice with mobocertinib at 30 mg/kg daily and five with vehicle control. Mice treated with vehicle control had disease progression at 2 and 4 weeks. In comparison, all eight mice treated with mobocertinib showed up to 90% tumor growth inhibition and at least four had sustained responses for 10 weeks.
Clinical Data
Phase 1 Results
The phase 1 study enrolled a total of 73 patients with NSCLC refractory to standard of care, among whom 44 were evaluable for dose-limiting toxicities (DLTs). Patients were excluded if they had asymptomatic cranial nervous system (CNS) metastases requiring corticosteroids for symptomatic control within 7 days prior to the first dose of mobocertinib or symptomatic CNS metastases. By using a 3+3 design, four patients were enrolled in the starting dose of mobocertinib at 5 mg daily cohort, followed by five at 10 mg daily, five at 20 mg daily, six at 40mg daily, seven at 80 mg daily, and 26 at 120 mg daily. Two twice daily dosing cohorts were explored with four patients enrolled in the 40 mg twice daily cohort and six in the 60 mg twice daily cohort. From 120 mg daily dosing, the dose of mobocertinib was escalated to 180 mg daily with four patients enrolled but two developed DLTs with grade 3 diarrhea and missing >25% of planned doses due to treatment-related adverse events (TRAEs). The dose was then decreased to 160 mg daily with six patients enrolled. In addition to two DLTs occurring in the 180 mg daily cohort, one patient each from the 80 mg daily, 120 mg daily, and 160 mg daily cohorts developed grade 3 pneumonitis, grade 5 pneumonitis, and grade 3 mucositis, respectively. Mobocertinib at 160 mg daily was chosen as the maximum tolerated dose and the recommended phase 2 dose (RP2D).5
Pharmacokinetic data showed mobocertinib had a median time to maximum plasma concentrations (Tmax) of 4 hours and a geometric mean effective half-life of 11–17 hours across the 20 to 160 mg daily dosing based on accumulation. Following administration of mobocertinib from 5 to 180 mg orally daily, the area under the concentration-time curve from time 0 to 24 hours (AUC0-24) increased in a dose-dependent manner.5
Phase 2 Results
In the phase 2 study, 130 patients enrolled in seven distinctive cohorts were treated with mobocertinib at the RP2D of 160 mg daily. Combined with six patients treated with 160 mg daily of mobocertinib in the phase 1 portion, a total of 136 patients were pooled for safety evaluation, among whom 131 (96%) patients had TRAEs. The most common TRAEs of any grade were diarrhea (83%), nausea (43%), rash (33%), vomiting (26%), dry skin (22%), decreased appetite (21%), stomatitis (21%), and fatigue (21%). The most common TRAEs of grade 3 or higher were diarrhea (21%), increased lipase (5%), nausea (4%), vomiting (4%), stomatitis (4%), anemia (2%), rash (1%), decreased appetite (1%), fatigue (1%), maculopapular rash (1%), and dermatitis acneiform (1%). Serious TRAEs were seen in 18 (13%) patients, including diarrhea (4%) and vomiting (4%). Dose interruption, dose reduction, and dose discontinuation was seen in 74 (54%), 23 (17%), and 22 (16%) patients, respectively.5
The efficacy of mobocertinib was assessed among 70 previously treated NSCLC patients harboring EGFR ex20ins mutations from both the phase 1 and the phase 2 portions, including 12 patients in 5–40mg daily cohorts, nine in 80 mg total daily (80 mg daily and 40 mg twice daily), 21 in 120 mg daily, and 28 in 160 mg daily.5 More specifically, these 28 patients treated with mobocertinib at 160 mg daily had not received (N = 22) or responded (N = 6) to a previous EGFR TKI. The confirmed objective response rate (ORR) and disease control rate (DCR) by investigator assessment (IA) were 0% and 25% for 5–40mg daily cohorts, 22% and 89% for 80 mg total daily, 19% and 71% for 120 mg daily, respectively. Among 28 NSCLC patients with EGFR ex20ins mutations treated with mobocertinib at RP2D of 160 mg daily, ORR and DCR were 44% (12/28; 95% confidence intervals [CI]: 24–63) and 86% (24/28; 95% CI: 67–96), respectively. The median duration of response (DoR) was 13.9 months (95% CI: 5.0-not reached [NR]), and the median progression-free survival (PFS) was 7.3 months (95% CI: 4.4–15.6). Additionally, the confirmed ORR was 40% (2/5) for the ASV mutation, 50% (2/4) for the NPH mutation, 50% (6/12) for other EGFR ex20ins mutations, and 50% (2/4) for unknown variants, suggesting EGFR ex20ins mutation variants did not seem to confer differential response to mobocertinib. Among 16 out of 28 patients without baseline brain metastases, the confirmed ORR was 56% (9/16; 95% CI: 30–80); the median DoR was 13.8 months (95% CI: 5.0–16.6); the median PFS was 10.2 months (95% CI: 5.6-NR). The rest of the 12 patients had baseline brain metastases, among whom the confirmed ORR, median DoR, median PFS were 25% (3/12; 95% CI: 5–57), 5.5 months (95% CI: 3.9–14.2), and 3.7 months (95% CI: 1.8–15.9), respectively. The inferior outcomes of patients with brain metastases highlight the limited CNS activity of mobocertinib.
Phase 3 Study Design
EXCLAIM-2 (NCT04129502) is a phase 3 clinical trial randomizing treatment naïve patients with locally advanced or metastatic nonsquamous NSCLC harboring EGFR ex20ins mutations at 1:1 ratio to either mobocertinib at 160 mg daily or platinum-pemetrexed chemotherapy for 4 cycles followed by maintenance pemetrexed. For patients on the chemotherapy arm, crossover to the mobocertinib arm upon disease progression is allowed. The primary end point is PFS by independent review committee (IRC). Key secondary endpoints include ORR by IRC, overall survival, PFS by IA, DoR by IRC and IA, time to response by IRC and IA, DCR by IRC and IA, patient-reported outcome, and safety. Subgroup analysis will be conducted on patients with or without baseline brain metastases and Asians versus non-Asians. The trial plans to enroll 318 patients from 168 sites globally. However, enrollment is currently on hold pending interim futility assessment. The trial schema is shown in Figure 2.
Conclusions
The EGFRex20ins mutations represent a distinctive molecular subset of NSCLC with an unmet need for development of effective targeted therapies. Amivantamab is so far the first and only approved treatment for this molecular driver. Among all the available TKIs in clinical trials, mobocertinib is the front runner from a drug development standpoint. Future directions will likely focus on combination approach such as TKI plus chemotherapy in the first-line setting, designing drugs with CNS activity, and exploring disease characteristics of various EGFRex20ins mutation variants and how they may affect treatment response.
Disclosure
VWZ has received honoraria from AstraZeneca, Blueprint, Roche-Foundation Medicine, Roche/Genentech, Takeda, and Xcovery, and had stock ownership of TP Therapeutics until May 2020. The authors report no other conflicts of interest in this work.
References
1. Riess JW, Gandara DR, Frampton GM, et al. Diverse EGFR exon 20 insertions and co-occurring molecular alterations identified by comprehensive genomic profiling of NSCLC. J Thorac Oncol. 2018;13(10):1560–1568. doi:10.1016/j.jtho.2018.06.019
2. Baraibar I, Mezquita L, Gil-Bazo I, et al. Novel drugs targeting EGFR and HER2 exon 20 mutations in metastatic NSCLC. Crit Rev Oncol Hematol. 2020;148:102906. doi:10.1016/j.critrevonc.2020.102906
3. Gonzalvez F, Vincent S, Baker TE, et al. Mobocertinib (TAK-788): a targeted inhibitor of EGFR exon 20 insertion mutants in non-small cell lung cancer. Cancer Discov. 2021. doi:10.1158/2159-8290.CD-20-1683
4. Food and Drug Administration. Available from: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-grants-accelerated-approval-amivantamab-vmjw-metastatic-non-small-cell-lung-cancer.
5. Riely GJ, Neal JW, Camidge DR, et al. Activity and safety of mobocertinib (TAK-788) in previously treated non-small cell lung cancer with EGFR exon 20 insertion mutations from a phase 1/2 trial. Cancer Discov. 2021. doi:10.1158/2159-8290.CD-20-1598
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

