Back to Journals » Medical Devices: Evidence and Research » Volume 12
Spontaneous-timed versus controlled noninvasive ventilation in chronic hypercapnia – a crossover trial
Authors Kerl J, Höhn E, Köhler D, Dellweg D
Received 14 October 2018
Accepted for publication 18 February 2019
Published 8 May 2019 Volume 2019:12 Pages 173—181
DOI https://doi.org/10.2147/MDER.S190841
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Jens Kerl, Ekkehard Höhn, Dieter Köhler, Dominic Dellweg
Department for Pulmonary Medicine, Intensive Care Medicine and Sleep Medicine, Fachkrankenhaus Kloster Grafschaft GmbH, Schmallenberg, 57392, Germany
Background: There is an ongoing debate about optimal ventilator modes and settings during noninvasive ventilation (NIV).
Objectives: To compare the effect of spontaneous-timed (ST) and controlled NIV on carbon dioxide reduction in patients suffering from chronic hypercapnia.
Methods: Night and daytime blood gas analysis, lung function tests and 6 minute walking distance tests (6MWD) were done before and after every 6-week treatment.
Results: This randomized prospective crossover trial included 42 patients. Pooled data analysis showed a decrease of nocturnal CO2 from 54.8±5.9 mmHg to 41.6±5.5 mmHg during ST ventilation (p<0.01) and from 56.2±7.5 mmHg to 42.7±5.4 mmHg during controlled NIV (p<0.01) with no difference between treatment forms (p=0.30). Daytime CO2 levels decreased from 49.3±5.5 mmHg to 45.6±4.5 mmHg when spontaneous timed ventilation was applied (p<0.01) and from 52.2±6.8 mmHg to 44.9±21114.4 mmHg in case of controlled ventilation (p<0.01) The amount of CO2 reduction was 3.8±5.6 mmHg after ST mode and 7.3±6.8 mmHg controlled ventilation (p<0.05). Nocturnal ventilator use was 5.7±2.1 and 6.7±2.3 hours for ST and controlled ventilation respectively (p=0.02). There was no effect on walking distance and lung function.
Conclusion: Controlled NIV showed improved compliance compared to ST ventilation. We observed similar CO2 reductions during nocturnal ventilation, however controlled ventilation achieved a higher reduction of daytime CO2 levels.
Keywords: noninvasive mechanical ventilation, clinical trials, respiratory failure, reversible hypercapnia, ventilator mode, controlled ventilation
Plain language summary
Noninvasive ventilation (NIV) decreases or even normalizes CO2 levels of hypercapnic patients. Since NIV is usually applied during the night, studies so far have mainly focused on nocturnal CO2 reductions. With increasing respiratory muscle fatigue during the day patients usually experience a CO2 rebound. We investigated if controlled NIV is superior compared to conventional ST-mode NIV in regard to CO2 levels and found a higher reduction of CO2 daytime values with controlled ventilation.
Introduction
Nowadays there is good evidence that most cases of hypercapnic respiratory failure are due to overload of the respiratory muscles1,2 or down-regulation of the central respiratory drive.2 Noninvasive mechanical ventilation (NIV) improves gas exchange, decreases work of breathing and relieves dyspnea in acute and chronic respiratory failure.3–6 The same can be achieved by invasive ventilation in intubated patients but here the mortality is higher.4 NIV improves the patient’s daytime performance and increases endurance7 and mobility.8 In addition NIV reduces mortality in acute as well as in chronic respiratory failure in COPD patients.4,9 NIV can be done by assisted modes of ventilation or controlled ventilation. In the assisted modes the work to trigger the ventilator can be substantial.10–13 For this reason the degree of respiratory muscle unloading can be increased by controlled mode ventilation.14–16 Controlled mode NIV is feasible17 but requires operator experience.
In a short-term study it has been shown that controlled ventilation leads to a much better unloading of the respiratory muscles in comparison to assisted spontaneous (S) mode ventilation.14 Published studies on controlled NIV were short-term measurements in healthy individuals14 or short-term tests in already NIV-adapted patients.18 The aim of this study was to compare the effect of the standard spontaneous-timed (ST) mode to controlled noninvasive ventilation in a mixed cohort of patients who require NIV and who are naïve to NIV in a prospective randomized crossover trial. The primary outcome was CO2 reduction. Lung function parameters, ventilator adherence and 6-minute walking distance were secondary outcome parameters.
Material and methods
Patients had to fulfil indications for NIV according to the national guidelines for mechanical ventilation.19–21 Exclusion criteria were age under 18 years, exacerbations during the previous 3 months, chronic or acute heart failure and creatinine levels >2 mg/dl. Patients had to have chronic hypercapnic failure, had to be naïve to NIV and had to be ambulatory.
Every patient was informed about the protocol and provided written consent. The responsible ethics committee (Medical Association, Wilhelms University Muenster, Germany, application No. 438-f-S) approved the protocol. The study was conducted in accordance with the Declaration of Helsinki.
Study design
The study design was a prospective randomized crossover trial (Figure 1). Randomization was done by means of sealed envelopes. For both groups NIV initiation was done in the hospital by an experienced respiratory therapist. Diagnostic studies were done before and after every treatment period and consisted of daytime as well as nocturnal blood gas analysis, lung function tests and 6-minute walking tests. Ventilator logs were read out after every treatment period. Treatment as well as washout periods lasted 6 weeks. During washout periods patients were discharged home without a ventilator.
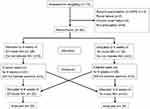 | Figure 1 Consort chart.Abbreviation: NIV, noninvasive ventilation. |
Methods
Ventilation, ventilators and ventilator settings
For every patient a facemask was selected from a number of commercially available masks and adjusted for optimal air tightness and comfort.
Controlled noninvasive ventilation was provided by means of time-adaptive (TA) mode ventilation (Ventilogic®, Weinmann GmbH, Hamburg, Germany). The functioning of this mode was described previously.14 Briefly TA mode ventilation consists of a self-learning algorithm that measures the spontaneous breathing pattern by integration of flow and time. After detection of a stable breathing pattern the ventilator shifts to controlled ventilation. During this phase the ventilator emulates the flow-time curve that was previously measured by adjusting the pressure to match the flow/time profile of the patient.18
In each patient the identical ventilator was either programmed to ST mode or TA mode (Table 1). Inspiratory pressure (Pinsp) and expiratory pressure (Pexp) remained unchanged throughout the treatment phases for each individual patient. During ST mode ventilation the inspiration time (Ti%) was manually set to patient comfort. Inspiratory trigger sensitivity and expiratory trigger sensitivity was manually set to fit the patients respiratory pattern. During ST mode ventilation the respiratory backup rate was adjusted to patient comfort while during TA mode it was set to the spontaneous respiratory rate within a preselected range to be used by the automatic system (Table 1).
 | Table 1 Ventilator settings |
Blood gas monitoring and lung function parameters
Blood gas analysis (ABG) was drawn from the arterialized earlobe and analyzed in the Omni S (Hoffman-La Roche, Basel, Switzerland) blood gas analyzer. Lung function as well as respiratory muscle pressures were measured with a bodyplethysmograph (Masterlab, Jaeger, Hochberg, Germany).
CO2 values during ventilation were measured at midnight (±20 mins) and during spontaneous breathing at noon (±20 mins). As a measure of functional exercise capacity we determined the 6MWD. All of the patients had to perform two practice tests (6MWD) on the day before study measurements were taken.22 The tests were conducted according to current guidelines23,24 under supervision of a respiratory therapist.
Statistical analysis
Sample size calculation: ST-mode ventilation has been shown to reduce CO2 levels by 7.4±2.4% in a large randomized controlled trial.9 To prove a doubling of this effect we calculated an effect size of 0,54. Given an alpha error of 0.05 and a power of 95% with two sided testing, the required sample size was determined to be N=40. Due to design and duration of this study including a washout period of 6 weeks, we expected a drop out rate of 30% and therefore aimed to include 56 patients into the present study.
Data analysis was done with Winstat® for EXCEL®. Normal data distribution was confirmed for all data sets by Kolmogorov-Smirnov test. ANOVA square sum comparison between the groups gave evidence for the assumption that all data share a common database when the p-value was lower than 0.05. The additional Bartlett test showed if the variances of the different groups are within a comparable range when also this p-value was lower than 0.05. The recommended alternative test for a negative Bartlett test result (P>0.05) for comparing groups and their mean values is the nonparametric Kruskal-Wallis test. Both ANOVA and Kruskal-Wallis test were performed for all following data and are presented alternatively. In two-group comparisons the two-sided, paired t-test was used.
Results
Patients
Patients screening, enrolment and dropouts are shown in Figure 1.
Forty-two patients (22 male) completed the study. Patient diseases, body mass index (BMI), 6-minute walking distance, pulmonary function test values as well as ABG results are summarized in Tables 2 and 3 respectively.
 | Table 2 Patient diseases, BMI, walking distance and pulmonary function tests |
 | Table 3 Patient diseases and daytime ABG |
NIV adaptation time
In patients who started with ST mode ventilation (n=20) the adaptation time was 5±2.8 days while the adaptation to TA mode at study entry (n=22) took 6±2.9 days (p=0.91).
Ventilator adherence
Overall mean ventilator usage at home was 6.2±2.2 hours per night (hpn). During ST mode ventilation the average usage was 5.7±2.1 hpn while usage during TA mode was 6.7±2.3 hpn (p=0.02).
CO2 at night
Nocturnal CO2 before therapy was 54.8±5.9 mmHg in the ST group and 56.2±7.5 mmHg in the controlled NIV group (p=0.18, Figure 2). During NIV CO2 decreased significantly (p<0.01) down to 41.6±5.5 mmHg in the ST group and to 42.7±5.4 mmHg in the controlled NIV group with no difference between groups (p=0.30, Figure 2). Crossover studies in general are susceptible to treatment period interaction also known as carryover effects. For this reason we compared the treatment effect of the pooled data (ST plus TA) in every block.25 At study entry (prior to block one) the overall nocturnal CO2 was 57.1±7.3 mmHg. Nocturnal CO2 levels at the end of the washout period and therefore at the beginning of treatment block two however were significantly lower (53.9±5.9 mmHg, p=0.03). Lower CO2 values after the washout period explained a lower efficacy of the post crossover treatment period (11±8.6 mmHg of nocturnal CO2 reduction) compared to the treatment effect prior to crossover (15.7±8.4 mmHg CO2 reduction at night, p=0.013).
 | Figure 2 Mean CO2 values at night and during the day before and after 6 weeks of ST-mode and controlled NIV.Abbreviation: NIV, noninvasive ventilation. |
CO2 at daytimes
Prior to ST-mode treatment the daytime CO2 level was 49.3±5.5 mmHg with significant decrease (p<0.01) to 45.6±4.5 mmHg after 6 weeks NIV (Figure 2). Prior to controlled-mode treatment CO2 was 52.2±6.8 mmHg and was reduced to 44.9±4.4 mmHg after 6 weeks of controlled NIV therapy (p<0.01, Figure 2). The amount of CO2 reduction was -3.8±5.6 mmHg after ST mode NIV and −7.3±6.8 mmHg after controlled NIV (p<0.05, Figure 2). Percentage changes of daytime CO2 were 6.8% for the ST group and 13% for the TA group (p=0.007). Thus regression to the mean appears not to affect our data.
Bicarbonate content (HCO3)
In all treatment groups a significant decrease of HCO3 values was achieved in nocturnal as well as in daytime values (Figure 3, p<0.01) with no difference between groups.
 | Figure 3 Mean HCO3 values at night and during the day before and after 6 weeks of ST-mode and controlled NIV.Abbreviation: NIV, noninvasive ventilation. |
Subgroup analysis of patients with COPD
Since COPD patients represented the largest disease group of our cohort (N=26) we analyzed the course of nocturnal as well as daytime CO2 and HCO3 values separately for this disease entity (Figures 4 and 5). We found similar reductions as we showed in Figures 2 and 3 for the whole cohort.
 | Figure 4 Mean CO2 values at night and during the day before and after 6 weeks of ST-mode and controlled NIV (COPD patients only).Abbreviation: NIV, noninvasive ventilation. |
 | Figure 5 Mean HCO3 values at night and during the day before and after 6 weeks of ST-mode and controlled NIV (COPD patients only).Abbreviation: NIV, noninvasive ventilation. |
Patient-triggered ventilator cycles and breathing frequency
The proportion of patient-triggered breaths as well as the breathing frequencies were read out from the ventilator logs. In the ST group 50.7% (±28.0%) of the breaths were triggered by the patients. In the controlled NIV group the amount of patient-triggered breaths was significantly lower (p<0.01) at a value of 0.9% (±0.7%). Assisted breaths during controlled ventilation are explained by the fact, that the TA mode applied in our study labels breaths during the run in period as assisted breaths. The breathing frequency under ST mode was 16.7/min ±3.8/min while under controlled NIV it was significantly higher at 21.5/min ±2.9/min (p<0.01).
6MWD
The 6MWD at study entry was 395.7±80.5 m in the ST group and improved to 405.1±87.1 m after 6 weeks of therapy (p=0.30). In the controlled NIV group the results were 387.7±88.3 m before NIV and 409.9±94.0 m after 6 weeks of ventilation (p=0.14). No difference was found inbetween the four measurements by means of ANOVA.
Lung function parameters
Lung function parameters are provided in Table 4. There were no significant differences or treatment effects.
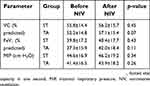 | Table 4 Lung function parameters before and after treatment periods |
Discussion
The major finding of this investigation is that ST-mode as well as controlled NIV ventilation are effective in reducing nocturnal as well as daytime CO2 levels. Daytime CO2 reduction however was significantly higher during controlled ventilation. The observed differences can potentially be explained by more effective respiratory muscle unloading during controlled ventilation. The work to trigger non-invasive ventilators is substantial and can make up to 43% of the total work of breathing.12 During ST-mode ventilation about half of all breaths (50.7%) were triggered by the patient while triggering during controlled ventilation was only possible during the run in period of the system and therefore was negligible (<1%). More effective unloading could either be explained by the significantly higher respiratory rate selected by the ventilator during controlled ventilation compared to a lower respiratory rate that was manually selected for ST mode ventilation or by a more physiological flow profile delivered during TA-mode ventilation. The role of the respirator backup frequency on CO2 levels and respiratory muscle unloading have been investigated previously. Duiverman et al26 found, that increasing the backup rate during high intensity NIV reduces the diaphragmatic electromyographic activity while Lucacsovits27 found similar effects when measuring work of breathing. These findings go along with the work on hand and previous results showing that controlled NIV reduces work of breathing more effectively when compared to ST-mode ventilation.14 Murphy et al28 on the other hand did not find a difference in the CO2 signal at night during high-pressure ventilation with low and high frequencies. The latter is true for our investigation as well, however the effect on daytime CO2 levels in the controlled ventilation group could be suggestive of improved muscle rest during ventilation. A major difference of our findings compared to Murphy’s work is that we achieved normal CO2 values during nocturnal ventilation, thus overall muscle rest was probably more complete within our investigation. Another possible explanation for the daytime CO2 reduction in the controlled ventilation group could be the significantly higher time spent on ventilation during controlled ventilation (6.7 hours compared to 5.7 hours per night during ST-mode ventilation). Ventilator use on the other hand appears to be closely related to therapy efficacy. Dreher et al29 observed in a crossover study that higher inspiratory pressures were not only related to more effective CO2 reduction but also to better ventilator usage. Yazar et al30 found in a cross-sectional prospective study design in 266 patients that higher inspiratory pressures were associated with higher usage times. Thus better adherence as seen during controlled ventilation in the present study could possibly be explained by a higher efficacy of this ventilation mode. As previously shown,17 controlled non-invasive ventilation is feasible and our data suggest, that the time required to institute controlled ventilation is comparable to the ST-mode adaptation time. At least for chronic hypercapnic COPD patients there is common sense, that adequate (high) inspiratory driving pressures are needed to effectively reduce CO228,29 and improve outcomes such as mortality.9 The driving pressure applied in this trial was comparable to previous studies that have been shown to be effective.9 Previous investigations have found different impact of NIV on lung function parameters and walking endurance.9,17,31,32 Neither treatment form in our trial showed to have an impact on lung function parameters or walking endurance, however a 6-week treatment period might have been too short to induce such changes. To our surprise, nocturnal CO2 levels did not return (increase) back to baseline at the end of the 6 weeks washout period. Although previous investigations suggested shorter periods to be sufficient,7,33 therefore a longer washout period would be recommended for future studies.
Limitations
All patients included into this trial met criteria to initiate NIV according to current guidelines. However the heterogeneity of diseases makes our study difficult to compare to other studies. Due to small numbers in some disease groups our study was not powered to allow for subgroup analysis according to all disease groups. Nocturnal CO2 values were lower when post washout values were compared to CO2 values at study entry and treatment effects were lower post crossover compared to pre crossover treatment periods. This is suggestive of a treatment period interaction (or carryover effect) which underestimates the effect post crossover.25
Conclusion
Controlled noninvasive ventilation was feasible with improved compliance compared to spontaneous timed mode ventilation. We observed similar CO2 reductions during ventilation at nighttime, however controlled ventilation achieved lower daytime CO2 levels. Possible mechanisms are economization of the work to trigger the ventilator, longer ventilator usage or both.
Author contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kohler D, Pfeifer M, Criee C. Pathophysiological basis of mechanical ventilation. Pneumologie. 2006;60(2):100–110. doi:10.1055/s-2005-919155
2. Roussos C, Koutsoukou A. Respiratory failure. Eur Respir J Suppl. 2003;47:3s–14s.
3. Brochard L, Isabey D, Piquet J, et al. Reversal of acute exacerbations of chronic obstructive lung disease by inspiratory assistance with a face mask. N Engl J Med. 1990;323(22):1523–1530. doi:10.1056/NEJM199011293232204
4. Osadnik CR, Tee VS, Carson-Chahhoud KV, Picot J, Wedzicha JA, Smith BJ. Non-invasive ventilation for the management of acute hypercapnic respiratory failure due to exacerbation of chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2017;7:CD004104. doi:10.1002/14651858.CD003881.pub4
5. Vitacca M, Nava S, Confalonieri M, et al. The appropriate setting of noninvasive pressure support ventilation in stable COPD patients. Chest. 2000;118(5):1286–1293.
6. Windisch W, Kostic S, Dreher M, Virchow JC
7. Diaz O, Begin P, Andresen M, et al. Physiological and clinical effects of diurnal noninvasive ventilation in hypercapnic COPD. Eur Respir J. 2005;26(6):1016–1023. doi:10.1183/09031936.05.00033905
8. Schonhofer B, Ardes P, Geibel M, Kohler D, Jones PW. Evaluation of a movement detector to measure daily activity in patients with chronic lung disease. Eur Respir J. 1997;10(12):2814–2819.
9. Kohnlein T, Windisch W, Kohler D, et al. Non-invasive positive pressure ventilation for the treatment of severe stable chronic obstructive pulmonary disease: a prospective, multicentre, randomised, controlled clinical trial. Lancet Respir Med. 2014;2(9):698–705. doi:10.1016/S2213-2600(14)70153-5
10. Marini JJ, Capps JS, Culver BH. The inspiratory work of breathing during assisted mechanical ventilation. Chest. 1985;87(5):612–618.
11. Marini JJ, Rodriguez RM, Lamb V. The inspiratory workload of patient-initiated mechanical ventilation. Am Rev Respir Dis. 1986;134(5):902–909. doi:10.1164/arrd.1986.134.5.902
12. Vitacca M, Barbano L, D’Anna S, Porta R, Bianchi L, Ambrosino N. Comparison of five bilevel pressure ventilators in patients with chronic ventilatory failure: a physiologic study. Chest. 2002;122(6):2105–2114.
13. Ward ME, Corbeil C, Gibbons W, Newman S, Macklem PT. Optimization of respiratory muscle relaxation during mechanical ventilation. Anesthesiology. 1988;69(1):29–35.
14. Dellweg D, Barchfeld T, Klauke M, Eiger G. Respiratory muscle unloading during auto-adaptive non-invasive ventilation. Respir Med. 2009;103(11):1706–1712. doi:10.1016/j.rmed.2009.05.003
15. Macklem PT. Respiratory muscles: the vital pump. Chest. 1980;78(5):753–758.
16. Rasche KL-G-G, Weyland W, Braun U, Hüttemann U, Criée CP. Sauerstoffverbrauch der Atemmuskulatur unter kontrollierter bzw. assistierter Beatmung bei Patienten mit chron. Ateminsuffizienz Med Klinik. 1994;89:43–46.
17. Dellweg D, Schonhofer B, Haidl PM, et al. Short-term effect of controlled instead of assisted noninvasive ventilation in chronic respiratory failure due to chronic obstructive pulmonary disease. Respir Care. 2007;52(12):1734–1740.
18. Kohler D, Dellweg D, Barchfeld T, Klauke M, Tiemann B. [Time-adaptive mode, a new ventilation form for the treatment of respiratory insufficiency–a self-learning system]. Pneumologie. 2008;62(9):527–532. doi:10.1055/s-2008-1038157
19. Windisch W, Dreher M, Geiseler J, et al. Guidelines for non-invasive and invasive home mechanical ventilation for treatment of chronic respiratory failure – update 2017. Pneumologie. 2017;71(11):722–795. doi:10.1055/s-0043-118040
20. Windisch W, Geiseler J, Simon K, Walterspacher S, Dreher M,
21. Windisch W, Geiseler J, Simon K, Walterspacher S, Dreher M,
22. Butland RJ, Pang J, Gross ER, Woodcock AA, Geddes DM. Two-, six-, and 12 min walking tests in respiratory disease. Br Med J (Clin Res Ed). 1982;284(6329):1607–1608.
23. Ae H, Ma S, Troosters T, et al. An official European Respiratory Society/American Thoracic Society technical standard: field walking tests in chronic respiratory disease. Eur Respir J. 2014;44(6):1428–1446. doi:10.1183/09031936.00150314
24.
25. Cleophas TJ. A simple method for the estimation of interaction bias in crossover studies. J Clin Pharmacol. 1990;30(11):1036–1040.
26. Duiverman ML, Huberts AS, van Eykern LA, Bladder G, Wijkstra PJ. Respiratory muscle activity and patient-ventilator asynchrony during different settings of noninvasive ventilation in stable hypercapnic COPD: does high inspiratory pressure lead to respiratory muscle unloading? Int J Chron Obstruct Pulmon Dis. 2017;12:243–257. doi:10.2147/COPD.S119959
27. Lukacsovits J, Carlucci A, Hill N, et al. Physiological changes during low- and high-intensity noninvasive ventilation. Eur Respir J. 2012;39(4):869–875. doi:10.1183/09031936.00056111
28. Murphy PB, Brignall K, Moxham J, Polkey MI, Davidson AC, Hart N. High pressure versus high intensity noninvasive ventilation in stable hypercapnic chronic obstructive pulmonary disease: a randomized crossover trial. Int J Chron Obstruct Pulmon Dis. 2012;7:811–818. doi:10.2147/COPD.S36151
29. Dreher M, Storre JH, Schmoor C, Windisch W. High-intensity versus low-intensity non-invasive ventilation in patients with stable hypercapnic COPD: a randomised crossover trial. Thorax. 2010;65(4):303–308. doi:10.1136/thx.2009.124263
30. Yazar EE, Ozlu T, Sariaydin M, et al. Prospective cross-sectional multicenter study on domiciliary noninvasive ventilation in stable hypercapnic COPD patients. Int J Chron Obstruct Pulmon Dis. 2018;13:2367–2374. doi:10.2147/COPD.S164384
31. Windisch W. Quality of life in home mechanical ventilation study g. Impact of home mechanical ventilation on health-related quality of life. Eur Respir J. 2008;32(5):1328–1336.
32. Wijkstra PJ, Lacasse Y, Guyatt GH, et al. A meta-analysis of nocturnal noninvasive positive pressure ventilation in patients with stable COPD. Chest. 2003;124(1):337–343.
33. Petitjean T, Philit F, Germain-Pastenne M, Langevin B, Guerin C. Sleep and respiratory function after withdrawal of noninvasive ventilation in patients with chronic respiratory failure. Respir Care. 2008;53(10):1316–1323.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
