Back to Journals » Infection and Drug Resistance » Volume 13
Small Molecule Adjuvants Potentiate Colistin Activity and Attenuate Resistance Development in Escherichia coli by Affecting pmrAB System
Authors Kathayat D, Antony L, Deblais L , Helmy YA , Scaria J, Rajashekara G
Received 1 May 2020
Accepted for publication 16 June 2020
Published 10 July 2020 Volume 2020:13 Pages 2205—2222
DOI https://doi.org/10.2147/IDR.S260766
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Dipak Kathayat,1 Linto Antony,2 Loic Deblais,1 Yosra A Helmy,1 Joy Scaria,2 Gireesh Rajashekara1
1Food Animal Health Research Program, Department of Veterinary Preventive Medicine, The Ohio State University, Wooster, OH 44691, USA; 2Animal Disease Research and Diagnostic Laboratory, Department of Veterinary and Biomedical Sciences, South Dakota State University, Brookings, SD 57007, USA
Correspondence: Gireesh Rajashekara 1680 Madison Avenue, Wooster, OH 44691, USA
Tel +1 330-263-3745
Fax +1 330-263-3744
Email [email protected]
Background: Colistin is one of the last-resort antibiotics to treat multi-drug resistant (MDR) Gram-negative bacterial infections in humans. Further, colistin has been also used to prevent and treat Enterobacteriaceae infections in food animals. However, chromosomal mutations and mobile colistin resistance (mcr) genes, which confer resistance to colistin, have been detected in bacterial isolates from food animals and humans worldwide; thus, limiting the use of colistin. Therefore, strategies that could aid in ameliorating colistin resistance are critically needed.
Objective: Investigate the adjuvant potential of novel small molecules (SMs) on colistin.
Materials and Methods: Previously, we identified 11 membrane-affecting SMs with bactericidal activity against avian pathogenic Escherichia coli (APEC). Here, we investigated the potentiation effect of those SMs on colistin using checkerboard assays and wax moth (Galleria mellonella) larval model. The impact of the SM combination on colistin resistance evolution was also investigated by analyzing whole genome sequences of APEC isolates passaged with colistin alone or in combination with SMs followed by quantitating pmrCAB and pmrH expression in those isolates.
Results: The SM combination synergistically reduced the minimum bactericidal concentration of colistin by at least 10-fold. In larvae, the SM combination increased the efficacy of colistin by two-fold with enhanced (> 50%) survival and reduced (> 4 logs) APEC load. Further, the SM combination decreased the frequency (5/6 to 1/6) of colistin resistance evolution and downregulated the pmrCAB and pmrH expression. Previously unknown mutations in pmrB (L14Q, T92P) and pmrA (A80V), which were predicted deleterious, were identified in the colistin-resistant (ColR) APEC isolates when passaged with colistin alone but not in combination with SMs. Our study also identified mutations in hypothetical and several phage-related proteins in ColR APEC isolates in concurrent with pmrAB mutations.
Conclusion: Our study identified two SMs (SM2 and SM3) that potentiated the colistin activity and attenuated the development of colistin resistance in APEC. These SMs can be developed as anti-evolution drugs that can slow down colistin resistance development.
Keywords: colistin, small molecules, pmrCAB, pmrH, resistance, anti-evolution drug
Introduction
Colistin, a cyclic polypeptide antibiotic isolated in 1947 from soil bacterium Bacillus colistinus, is active against many Gram-negative bacteria.1,2 It binds to lipopolysaccharide (LPS) and phospholipids in the outer cell membrane of Gram-negative bacteria and competitively displaces divalent cations from the phosphate groups of membrane lipids, which leads to disruption of the outer cell membrane, leakage of intracellular contents, and bacterial death.3 In particular, it is used as a last-resort antibiotic for the treatment of severe infections in humans caused by multi-drug resistant (MDR) Gram-negative pathogens such as E. coli, Pseudomonas aeruginosa, Klebsiella pneumoniae, and Acinetobacter baumannii.2,4 Further, it has been used in veterinary medicine for decades, mainly as a growth promoter (feed additive) or for the prevention and treatment of Enterobacteriaceae infections, including avian pathogenic E. coli (APEC) in poultry.1,2,4,6 Colistin usage in food animals is widespread, particularly high in low- and middle-income countries, and has been increasing since the mid-twentieth century.1,4 A 13% increase in colistin usage has been reported in Brazil, Russia, India, China, and South Africa from 2000 to 2010.4 In China (the largest user of colistin), 2470–2875 metric tons of colistin was used to treat E. coli and Salmonella infections in chicken and pig farms from 2011 to 2015.4 In the European Union, colistin was the fifth most widely used antibiotic during 2013–2015 to treat and prevent infectious diseases in food animals.2,4 In the USA, colistin (First GuardTM sterile powder; colistimethate sodium) is approved by the Food and Drug Administration (FDA) for the treatment of APEC infection in day-old chicks (Code of Federal Regulations 21 Section 522.468). In the Netherlands, colistin was the most predominant antibiotic used to treat APEC induced E. coli peritonitis syndrome (EPS) in layer flocks in 2013.5 However, with the use of colistin, bacterial resistance to colistin has been reported worldwide.1,7 Consequently, many countries in the world, including USA, European Union, and China, have now banned the growth promotion and prevention uses of colistin in food animals.1,4
Colistin resistance is typically mediated by chromosomal mutations in two-component systems, such as PmrAB and PhoPQ,7 or by horizontal transfer of plasmid carrying mobile colistin resistance (mcr) genes.8 Multiple chromosomal mutations (most commonly, pmrB, pmrA, mgrB, phoP, and phoQ) and mcr genes (mcr-1, mcr-2, mcr-3, mcr-4, mcr-5, mcr-6, mcr-7, mcr-8, and mcr-9) have been detected in bacterial isolates, including E. coli, from food animals (pig, cattle, poultry) and humans worldwide; thus, threatening the use of this last-resort antibiotic.1,4,7,9,11 The chromosomal mutations modify the lipid A moiety of LPS, which is a primary target of colistin, and thereby leads to colistin resistance.7 Specifically, PmrAB and PhoPQ mutations induce the transcription of pmrCAB and pmrHFIJKLM (or arnBCADTEF) operons, which leads to the synthesis of phosphoethanolamine (PEtN) and 4-amino-4-deoxy-L-arabinose (L-ara4N), respectively, and then subsequently transfer it to lipid A.7 The addition of PEtN or L-ara4N reduces the negative charge of lipid A and thereby decreases colistin binding with its target which results in reduced efficacy of colistin.7 Notably, the occurrence of colistin resistance is reported more frequently in food animals (0.9–76.9%) than humans (0.1–8.8%),4,11 which is in accordance with the higher selection pressure posed by the heavy use of colistin in food animals.4,11 Therefore, strategies to decrease colistin use and ameliorate colistin resistance are critically needed to preserve the future use of colistin in treating MDR pathogens.12
One potential approach to ameliorate colistin resistance is the use of antibiotic adjuvants (or potentiators) which can increase the efficacy of colistin, ultimately requiring lower doses of colistin for treatments.13,14 In previous studies, 2-aminoimidazoles (2-AIs),13 (E)-2-hexenal,15 indole,15 N-acetylcysteine,16 resveratrol,17 niclosamide,18 and pentamidine19 were identified as colistin adjuvants against different Gram-negative pathogens. Importantly, colistin adjuvants can also reverse the colistin resistance so that the colistin is effective against the previously resistant bacteria.13,18,20 Specifically, adjuvants can abrogate or reduce the extent of lipid A modification by reducing the expression of mcr and pmrAB/phoPQ genes;13,18,19,21,22 thus, overcoming the colistin resistance. In this study, we investigated the adjuvant potential of previously identified membrane-acting small molecules (SMs)23 with colistin against APEC. Because of the membrane-affecting properties of these SMs,23 similar to previously identified colistin adjuvants which have been shown to potentiate the colistin effect by disrupting as well as permeabilizing the bacterial membrane,19,21,24,27 these SMs can work synergistically with colistin, which is also a membrane-active antibiotic;2 thereby, augmenting the colistin mediated disruption of the bacterial membrane.
APEC, an extra-intestinal pathogenic E. coli (ExPEC) and a model pathogen of this study, is the most common bacterial pathogen of poultry.28 In the USA, it is estimated that at least 30% of commercial flocks are affected by APEC, at any point in time.28 In poultry, APEC causes a wide range of localized and systemic infections; commonly referred to as avian colibacillosis.29 Colibacillosis results in significant morbidity and mortality of chickens, which translates into multi-million dollar annual losses for all facets of the world’s poultry industry.28 Further, recent studies have reported APEC as a foodborne human uropathogen and commonalities in diseases caused by ExPEC strains in humans.28,30 APEC is also considered as a source of antibiotic-resistant genes (ARGs) to human pathogens.31 Therefore, effective control of APEC is beneficial to both human and animal health by reducing the foodborne transmission of ExPECs and ARGs.28
Our study identified two SMs (SM2 and SM3) that potentiated the colistin activity and attenuated the development of colistin resistance in APEC. The SM combination also downregulated the expression of pmrCAB and pmrH genes responsible for colistin resistance. Previously unknown mutations in pmrB and pmrA genes were identified. Mutations were also identified in other genes encoding hypothetical proteins and several phage-related proteins in concurrent with pmrAB mutations, which can help in better understanding of molecular sequelae associated with colistin resistance. These SMs can be developed as anti-evolution drugs that can slow down colistin-resistance development.32 Although our study tested colistin adjuvant potential of SMs only against APEC strains, future studies on the adjuvant potential of these SMs with colistin against human ExPECs and other Gram-negative pathogens can have significant implication in treating MDR pathogens.
Materials and Methods
Bacterial Strains, Culture Conditions and Media
Luria-Bertani (LB) broth (BD DifcoTM) was used for routine propagation of APEC O78, O1, and O2. These APEC isolates were isolated from chickens (O1 – lung, accession number: CP000468; O2 – airsac, accession number: CP006834) and turkey (O78 – lung, accession number: CP004009) clinically diagnosed with various forms of colibacillosis and serotyped using O-serotyping slide agglutination test. M63 minimal media was used in the checkerboard assay as these SMs were identified as growth inhibitors in M63 media.23 Rifampicin resistant (Rifr) APEC O78 generated through spontaneous mutation in our previous study23 was used for the wax moth larvae study.
Small Molecules and Antibiotics
All the SMs (SM 1–11) used in this study were obtained from ChemBridgeTM, dissolved in 100% dimethyl sulfoxide (DMSO) at 100 mM, and stored at −80°C as described previously.23 These SMs were identified as APEC growth inhibitors in our earlier study23 through screening of pre-selected enriched SM library containing 4182 SMs. Moreover, this SM library was assembled through pre-screening of ~81,000 compounds.33 These SMs possess pyrrolidinyl (SM2, SM3, and SM7), imidazole (SM4, SM5, and SM6), piperidine (SM1 and SM11), quinoline (SM8 and SM9), and nitrophenyl (SM10) scaffolds.23 All the antibiotics used in this study (colistin sulfate salt, ciprofloxacin, and tetracycline) were purchased from Sigma-Aldrich. The selected antibiotics belong to different classes of antibacterials with different mechanisms of action and are currently being used in the poultry industry.23 Antibiotic stocks were prepared following CLSI guidelines (M100, 28th edition).34
MIC and MBC Determination
Minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of SMs against APEC serotypes (O78, O1, and O2) were identified in our earlier study.23 MIC and MBC of antibiotics against APEC serotypes were determined by twofold serial dilution in M63 media (Table S1 and S2). Briefly, twofold serially diluted antibiotics were added to 100 µL of 0.05 OD600 adjusted APEC culture and incubated at 37°C for 12 h in Sunrise-Absorbance microplate reader (Tecan Group Ltd. San Jose, CA) with kinetic turbidity measurement set for every 30 min. MIC was indicated by the lowest concentration of antibiotic with non-elevated OD600; whereas MBC was determined by plating the antibiotic-treated culture on the LB agar plate. Two independent experiments were conducted.
Checkerboard Assay
The synergy potential of SMs with antibiotics was assessed using a checkerboard assay as previously described.35,36 Briefly, SMs were diluted (1×, 0.8×, 0.6×, 0.4×, 0.2×, and 0.1× MBC) along the abscissa, whereas; antibiotics were diluted along the ordinate (1×, 0.8×, 0.6×, 0.4×,0.2×, and 0.1× MBC) in 100 µL of 0.05 OD600 adjusted APEC O78 culture in a 96-well plate. We tested MBC in this assay because these SMs were found bactericidal against APEC in our earlier study23 and also MBC can provide more reliable and quantitative data of SM-antibiotic combination as compared to MIC. After the addition of SMs and antibiotics, the plate was incubated at 37°C for 12 h in Sunrise-Absorbance microplate reader and MBC of SM-antibiotic combination was determined as above. The fractional inhibitory concentrations (FICs) of SMs in combination with antibiotics were determined as described previously.37 Synergistic, indifferent, and antagonistic activities were defined by FICs of 0.5, 0.51 to 4.0, and >4.0, respectively. Four SMs (SM2, SM3, SM5, and SM7) that showed synergism with most of the antibiotics tested were selected for further study. However, SM5 belongs to the imidazole group and the potentiating activity of imidazoles on colistin has been already reported;13 therefore, SM5 was not studied further. On the other hand, SM7, which possesses a pyrrolidinyl moiety similar to SM2 and SM3, was not studied due to its unavailability in the ChemBridge library. Therefore, two SMs (SM2 and SM3) which were synergistic with most of the antibiotics tested and possessed novel scaffold (pyrrolidinyl) with no previous report of potentiation effect with colistin were selected for further studies. For the selected SMs, the checkerboard assay was also conducted against APEC serotypes O1 and O2 as above.
In vivo Synergy Studies in Wax Moth Larva
The in vivo synergy potential of SMs (SM2 and SM3) with antibiotics was determined in the wax moth (G. mellonella) larva model as described previously.38 SMs (12.5 µg) were combined with antibiotics: colistin 0.3125 mg/kg; tetracycline 1.5625 mg/kg and ciprofloxacin 2.5mg/kg. The antibiotic dose (the dose that provides minimal protection to the larva against APEC infection) was selected based on the preliminary study (Table S3). Briefly, larvae (n=15 larva/group) were infected with 6.4×104 CFU of Rifr APEC O78 as described previously23 through the right hind pro-leg using an insulin syringe. Larvae were then treated with colistin (0.3125 mg/kg), a combination of colistin and SMs (0.3125 mg/kg+12.5 µg), tetracycline (1.5625 mg/kg), and a combination of tetracycline and SMs (1.5625 mg/kg+12.5 µg) within 30 min of infection as described previously;38,39 or after 2 h of infection for ciprofloxacin (2.5 mg/kg) and the combination of ciprofloxacin and SMs (2.5 mg/kg+12.5 µg), as described previously.40 Uninfected larvae and infected larvae treated with buffer mix (DMSO-15% + PBS) were used as controls. Larval survival was monitored every 12 h for 3 days. Dead larvae were collected at each time point and intra-larval APEC load was quantified as described previously23 by plating the homogenized larval suspension on MacConkey agar plate supplemented with 50 µg/mL rifampicin. APEC load in live larvae was quantified as described above after 72 h post-infection. For the survival analysis, Kaplan-Meier survival curves were generated and statistically analyzed between the treated groups using Log-rank test (P<0.05). The APEC load was statistically compared between the treated groups using a one-way analysis of variance (ANOVA) followed by Tukey’s test (P<0.05). Two independent experiments were conducted.
Impact of SM Combination on Colistin-Resistance Evolution
The impact of SMs (SM2 and SM3) on in vitro colistin-resistance evolution was analyzed using the sub-inhibitory concentration of SMs as described previously.41 Briefly, six parallel independent APEC O78 cultures (DMSO 1–6, Col 1–6, Col + SM2 1–6, and Col+ SM3 1–6) were passaged in M63 media starting with the sub-inhibitory concentration of colistin (0.0625 µg/mL) and in the presence or absence of 0.5× MIC (SM2: 50 µM; SM3: 50 µM) of SMs. Cultures were allowed to grow for 24 h, and 20 µL of grown cultures were transferred to new wells containing fresh M63 media and increasing concentration of colistin (0.125, 0.25, 0.5, 1.0, 1.5, 2.0, 4.0, and 8.0 µg/mL) up to 8 µg/mL (ie, beyond resistance breakpoint). SMs were added at each passage at 0.5× MIC concentrations. APEC cultures containing no colistin and no SM (1% DMSO; DMSO 1–6) were used as controls and passaged similarly (8 passages) to cultures containing colistin or colistin and SMs. The growth of cultures was monitored at each passage and only the grown cultures were selected for further passage. For the non-grown cultures (ie, cultures not grown until the last colistin passage), glycerol stock was made from the previous passage by regrowing the remnant (80 µL) in fresh M63 media containing respective colistin and SM concentrations and stored at −80°C for further analysis. At the end of the experiment, the MIC of colistin for both grown (Col-1, Col-3, Col-4, Col-5, Col-6, Col + SM2-2 and Col + SM3-5) and non-grown (Col-2, Col + SM2-1, Col + SM2-3, Col + SM2-4, Col + SM2-5, Col + SM2-6, Col + SM3-1, Col + SM3-2, Col + SM3-3, Col + SM2-4, and Col + SM3-6) cultures was determined as described above. The selected grown and non-grown cultures (Col-1, Col-3, Col-5, Col-6, Col + SM2-2, Col + SM2-4, Col + SM2-6, Col + SM3-2, Col + SM3-5, and Col + SM3-6), including control cultures (DMSO-1and DMSO-3), were plated on M63 agar plates and single colonies were isolated. Cultures were selected from each group including both colistin-resistant (ColR; Col-1, Col-3, Col-5, Col-6, Col + SM2-2, and Col + SM3-5) and susceptible (ColS; Col + SM2-4, Col + SM2-6, Col + SM3-2, and Col + SM3-6) phenotypes. Control susceptible isolates (DMSO-1 and DMSO-3) were also selected to provide a baseline for genomic analysis. The isolated colonies were grown overnight, DNA was extracted using MasterPureTM DNA Purification Kit (Epicentre) and the whole genome was sequenced.
Whole Genome Sequencing
The Whole genome sequencing (WGS) of APEC isolates (DMSO-1, DMSO-3, Col-1, Col-3, Col-5, Col-6, Col + SM2-2, Col + SM2-4, Col + SM2-6, Col + SM3-2, Col + SM3-5, and Col + SM3-6) was performed on an Illumina MiSeq platform. Briefly, an initial concentration of 0.3 ng/µL DNA was used for library preparation using the Nextera XT Library preparation kit (Illumina Inc.). After dual indexing, the library was bead normalized, and the samples were pooled at equal volume and sequenced using 2×300 paired-end V3 chemistry.
Mutations and Variants Analysis
The chromosomal mutations pertaining to colistin resistance in the genome of APEC isolates (DMSO-1, DMSO-3, Col-1, Col-3, Col-5, Col-6, Col + SM2-2, Col + SM2-4, Col + SM2-6, Col + SM3-2, Col + SM3-5, and Col + SM3-6) were initially searched using PointFinder software 3.1.0 (https://bitbucket.org/genomicepidemiology/pointfinder/src/master/) as described previously.42 Briefly, the quality of the sequence reads was determined using FastQC (version 0.11.8) package (Babraham Bioinformatics). The sequences were then assembled using the SPAdes (version 3.9.0) package. The chromosomal mutations in the assembled genome were identified using the PointFinder database (https://bitbucket.org/genomicepidemiology/pointfinder_db/src/master/). The mutations present in ColR isolates (Col-1, Col-3, Col-5, Col-6, Col + SM2-2, and Col + SM3-5) were compared with ColS isolates (Col + SM2-4, Col + SM2-6, Col + SM3-2, and Col + SM3-6), including control isolates (DMSO-1 and DMSO-3), to identify the unique mutations responsible for colistin resistance. For a further detailed analysis of mutations and variants, read mapping and variants calling method as implemented in CLC Genomics Workbench was used. The variant detection tools in CLC Genomics Workbench implemented is based on the Neighborhood Quality Standard (NQS) algorithm.43,44 Demultiplexing of the data was performed using the Casava pipeline. Further analysis of de-multiplexed sequence data as FASTQ format was performed in CLC Genomics workbench v.12.0. Paired reads were taken through a quality check followed by a trimming step to ensure a minimum average PHRED score of 30. APEC O78 complete genome (GenBank Accession: CP004009.1) was used as a reference for read mapping and annotation. Reads were mapped against the reference genome using local alignment to determine structural variants such as insertions, deletions, inversions, translocations, and tandem duplications in the experimental isolates. This information was used to improve the alignment of the reads in an existing read mapping. Variants were called when there is a minimum coverage of 10× and a minimum frequency of 20%. To avoid false positives, we removed low-quality variants that have an average base quality of less than 20 and forward and reverse read balance of less than 0.2. Variants were then annotated using the coding sequence (CDS) and gene annotation information from the reference genome. Relevant known genomic variants in the ColR isolates were tested for the presence or absence in ColS isolates by comparing the variants in the ColR isolate with the read mappings of ColS isolates, including control isolates. Specific variants that are present only in the ColR isolates and not in ColS isolates, control isolates, and reference genome were taken into account. These variants were then screened for their presence in the CDS as well as for amino acid (AA) change. The complete sequence data used for the above analysis have been deposited in the NCBI sequence read archive (SRA) under the Bioproject number PRJNA591010 (URL: https://dataview.ncbi.nlm.nih.gov/object/PRJNA591010?reviewer=capd9kv55kk447p5if9ucfal0).
pmrCAB and pmrH Expression Analysis
To corroborate the findings of mutation analysis, relative expression of pmrCAB and pmrH in APEC isolates (DMSO-1, DMSO-3, Col-1, Col-3, Col-5, Col-6, Col + SM2-2, Col + SM2-4, Col + SM2-6, Col + SM3-2, Col + SM3-5, and Col + SM3-6) was quantitated using reverse transcription-quantitative polymerase chain reaction (RT-qPCR) as described previously.45,46 APEC isolates frozen in glycerol at −80°C were grown overnight at 37°C with shaking at 200 rpm and total RNA was extracted using RNeasy mini kit (Qiagen) following the manufacturer’s instructions. RNA quantity and quality were measured using nanodrop 2000c spectrophotometer and 1% agarose gel electrophoresis. DNA traces were removed using the genomic DNA elimination mix (Qiagen). Approximately 5 µg of purified RNA was used to synthesize cDNA using RT2 First Strand Kit (Qiagen). The RT-qPCR was performed using Maxima SYBR Green/ROX qPCR master mix (Thermo Fisher Scientific) following the manufacturer’s instructions in a RealPlex2 Mastercycler® (Eppendorf) with 55°C annealing temperature. The primers (Table S4) were designed using PrimerQuest Tool and obtained from Integrated DNA Technologies (IDT). The data were normalized to the house-keeping gene, glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and relative fold change in expression was calculated in each APEC isolate (Col-1, Col-3, Col-5, Col-6, Col + SM2-2, Col + SM2-4, Col + SM2-6, Col + SM3-2, Col + SM3-5, and Col + SM3-6) by comparing with control isolates (DMSO-1 and DMSO-3) using the ∆∆Ct method.47 The data were statistically analyzed using the Student’s t-test (P<0.05). Two-independent experiments were conducted.
Results
Small Molecules Exhibited Synergistic Interaction with Colistin in vitro
Among the 11 SMs tested in combination with antibiotics (colistin, tetracycline, and ciprofloxacin) in checkerboard assay, four SMs (SM2, SM3, SM5, and SM7) reduced the MBC of all three antibiotics by at least 10-fold (Table 1). Interestingly, most of the SMs (SM2, SM3, SM4, SM5, SM6, SM7, SM9, and SM11) reduced 10-fold MBC (2 µg/mL to 0.2 µg/mL) of colistin (Table 1), whereas four SMs (SM2, SM3, SM5, and SM7) reduced 10-fold MBC (128 µg/mL to 12.8 µg/mL) of tetracycline (128 µg/mL to 12.8 µg/mL) (Table 1) and eight SMs (SM2, SM3, SM4, SM5, SM6, SM7, SM9, and SM11) reduced 10-fold MBC (1 µg/mL to 0.1 µg/mL) of ciprofloxacin (Table 1). The MBCs of SMs upon the combination with antibiotics are displayed in Table 2. The combination of colistin also reduced the MBC of all SMs by at least 10-fold. Tetracycline combination reduced the 10-fold MBC of SM3 and SM7, whereas the ciprofloxacin combination reduced only the MBC of SM4 by 10-fold.
 |
Table 1 Minimum Bactericidal Concentration (MBC) of Antibiotics When Tested in Combination with SMs |
 |
Table 2 Minimum Bactericidal Concentration (MBC) of SMs When Tested with Antibiotics |
Among the 11 SMs tested, SM5 showed synergistic interaction (FIC=0.2–0.5) with all three antibiotics tested (Table S5). Interestingly, most of the SMs (SM2, SM3, SM4, SM5, SM6, SM7, SM8, SM9, and SM11) showed synergistic interaction (FIC=0.2) with colistin. Four SMs (SM2, SM3, SM5, and SM7) showed synergism (FIC=0.2–0.5) with tetracycline, and three SMs (SM4, SM5, and SM11; FIC=0.2–0.5) with ciprofloxacin. Four SMs (SM2, SM3, SM5, and SM7) that displayed synergism with most of the antibiotics tested were selected for further study. However, SM5 belongs to the imidazole group and the colistin potentiating activity of imidazoles has been already reported;13 therefore, SM5 was not studied further. SM7, which possess pyrrolidinyl moiety similar to SM2 and SM3, was not studied further due to unavailability in the ChemBridge library.
The selected SMs (SM2 and SM3) also reduced the MBC of colistin by at least 10-fold (4 µg/mL to 0.4 µg/mL) against APEC O1 and O2, similar to O78, and also showed a synergistic interaction with colistin (Table 3).
 |
Table 3 Minimum Bactericidal Concentration (MBC) of Colistin Against APEC O1 and O2 When Tested with Selected SMs |
Small Molecule Combination Increased the Efficacy of Colistin in Wax Moth Larvae
Two selected SMs (SM2 and SM3; 12.5 µg), when combined with colistin, increased (SM2-46.67% and SM3-57.14%) the survival of larvae (Figure 1A) as well as significantly (P<0.001) reduced the intra-larval APEC O78 load (SM2-4.82 logs and SM3-5.24 logs) as compared to colistin treatment alone (Figure 1B). Similarly, these SMs increased (SM2-13.34%) the survival of larvae and reduced the intra-larval APEC O78 load (SM2-0.82 logs and SM3-0.54 logs) when combined with ciprofloxacin as compared to ciprofloxacin treatment alone (Figure S1A and B). However, no difference in the larval survival and intra-larval APEC O78 load was observed when these SMs were combined with tetracycline (Figure S1C and D).
Small Molecule Combination Decreased the Frequency of Colistin-Resistance Evolution
The experimental evolution of colistin resistance in the presence (0.5× MIC) or absence of SMs (SM2 and SM3) was assessed through successive passages of APEC cultures in increasing concentrations of colistin (starting with sub-inhibitory concentration; 0.0625 µg/mL). Most of the APEC cultures (5/6; Col-1, Col-3, Col-4, Col-5, and Col-6) passaged only in the presence of colistin were found resistant, whereas only 1 culture (1/6; Col + SM2-2 and Col + SM3-5) was found resistant to colistin when combined with SM2 and SM3 (Figure 2A). Four of the APEC cultures (Col-1, Col-4, Col-5, and Col-6) passaged only in presence of colistin had MIC 64 µg/mL and the other two APEC cultures had MIC 32 µg/mL (Col-3) and 4 µg/mL (Col-2). On the other hand, only one APEC culture had MIC 64 µg/mL (Col + SM2-2 and Col + SM3-5) and other cultures (Col + SM2-1, Col + SM2-3, Col + SM2-4, Col+ SM2-5, Col + SM2-6, Col + SM3-1, Col + SM3-2, Col + SM3-3, Col + SM3-4, and Col + SM3-6) had MIC ≤4 µg/mL when colistin was combined with SM2 and SM3. Most of the these cultures (Col + SM2-1, Col + SM2-4, Col + SM2-5, Col + SM2-6, Col + SM3-2, Col + SM3-3, and Col + SM2-4) had MIC 2 µg/mL; whereas few (Col + SM2-3, Col + SM3-1, and Col + SM3-6) had MIC 4 µg/mL. The chemical structure and ChemBridge ID of these two SMs are displayed in Figure 2B, Table S6.
Previously Unknown Mutations in pmrB and pmrA Genes Were Identified in ColR APEC Isolates
The PointFinder database mediated comparative analysis of chromosomal mutations revealed point mutations in pmrA and pmrB genes in ColR isolates (Col-1, Col-3, Col-5, Col-6, Col + SM2-2, and Col + SM3-5) as compared to ColS (Col + SM2-4, Col + SM2-6, Col + SM3-2, and Col + SM3-6) and control isolates (DMSO-1 and DMSO-3) (Table 4). The most common mutation was in pmrB p.L14Q (Col-5, Col-6, Col + SM2-2, and Col + SM3-5), where thymine was replaced by adenine (CTG to CAG) resulting in glutamine instead of leucine (L to Q). The mutation was also observed in pmrB p.T92P (Col-3), where adenine was replaced by cytosine (ACC to CCC) resulting in proline instead of threonine (T to P). Similarly, in pmrA p.A80V (Col-1), cytosine was replaced by thymine (GCT to GTT) resulting in valine instead of alanine (A to V).
 |
Table 4 Mutations Observed in ColR APEC Isolates Analyzed Using PointFinder Database |
Consistent with the decreased frequency of colistin-resistance evolution, no pmrA and pmrB mutations were present in most of the sequenced APEC isolates (4/6) which were passaged with colistin and SM combination (absent in Col + SM2-4, Col + SM2-6, Col + SM3-2, and Col + SM3-6; present only in Col + SM2-2 and Col + SM3-5) as compared to colistin alone (present in Col-1, Col-3, Col-5, and Col-6) passaged APEC isolates (Figure 3). Similar to results obtained using the PointFinder database, in reads mapping and variants calling analysis, non-synonymous mutations were observed in ColR isolates in genes encoding sensor protein BasS/PmrB and two-component response regulator BasR/PmrA (Figure 3, Table 5). Single nucleotide variation (SNV; A to T) was observed in gene encoding sensor protein BasS/PmrB in most (4/6; Col-5, Col-6, Col + SM2-2, and Col + SM3-5) of the ColR isolates, compared to ColS isolates which resulted in glutamine (Q) instead of leucine (L) at position 14 of PmrB protein (pmrB p. L14Q). In the same gene, SNV (T to G) was observed in isolate Col-3 which resulted in proline (P) instead of threonine (T) at position 92 (pmrB p. T92P). Additionally, SNV (G to A) was also observed in gene encoding two-component response regulator BasR/PmrA in one of the ColR isolates (Col-1), which resulted in valine (V) instead of alanine (A) at position 80 of PmrA protein (pmrA p. A80V).
 |
Table 5 Non-Synonymous Mutations Observed in ColR APEC Isolates |
Small Molecule Combination Downregulated the pmrCAB and pmrH Expression
Colistin resistance in bacteria is mediated by the upregulation of genes of pmrCAB and pmrHFIJKLM operons;20,45,48 therefore, suppressing the expression of involved genes can prevent the evolution of colistin resistance. Specifically, the upregulated pmrC and pmrH expression results in lipid A modifications; thereby, subsequently decreases the binding and activity of colistin against bacteria.7 Consistent with the absence of pmrA and pmrB mutations in most of the APEC isolates passaged with colistin and SM combination, the level of pmrCAB and pmrH expression was lower in most of the APEC isolates passaged with colistin in combination with SMs (Col + SM2-4, Col + SM2-6, Col + SM3-2, and Col + SM3-6) as compared to APEC isolates (Col-1, Col-3, Col-5, and Col-6) passaged alone with colistin (Figure 4A and B). The level of pmrCAB and pmrH expression in each APEC isolate is displayed in Figure 4A and B. Overall, the expression of pmrCAB and pmrH expression was lower in ColS isolates (Col + SM2-4, Col + SM2-6, Col + SM3-2, and Col + SM3-6) as compared to ColR (Col-1, Col-3, Col-5, Col-6, Col + SM2-2, and Col + SM3-5) isolates. Further, as compared to control APEC isolates (DMSO-1, DMSO-3), the expression level (on average) of pmrC was 17.9±3.8-, 6.4±0.3-, and 3.8±0.5-fold higher in APEC isolates passaged with colistin, colistin and SM2, and colistin and SM3, respectively. Similarly, the expression level of pmrH was 16.2±.25.0-, 5.8±2.5-, and 2.1±0.2-fold higher in APEC isolates passaged with colistin, colistin and SM2, and colistin and SM3, respectively. A similar trend was observed in pmrA and pmrB expression levels. The 3.6±0.7-, 1.9±0.1-, and 2.5±0.1-fold higher pmrB expression was observed in APEC isolates passaged with colistin, colistin and SM2, and colistin and SM3, respectively, as compared to control isolates, whereas 1.4±0.2-, 0.8±0.3-, and 1.3±0.2-fold higher pmrA expression was observed in APEC isolates passaged with colistin, colistin and SM2, and colistin and SM3, respectively. Even though slight upregulation in the expression of pmrCAB and pmrH genes was observed in APEC isolates passaged with colistin in combination with SMs as compared to control isolates, the expression was significantly lower (P<0.05) as compared to isolates passaged with colistin alone.
Mutations in Hypothetical and Several Phage-Related Proteins Were Identified in ColR APEC Isolates Concurrent with pmrAB Mutations
In addition to pmrB and pmrA, non-synonymous and synonymous mutations were also observed concurrently in ColR isolates in other genes encoding hypothetical proteins, several phage-related proteins [putative transposases, phage replication protein O, antitermination protein Q from phage origin, Rz endopeptidase from lambdoid prophage DLP12, lambdoid prophage DLP12 Bor-like protein, bacteriophage lysis protein, phage repressor protein, terminase large subunit, restriction alleviation protein, increased serum survival (Iss), and putative portal protein], uroporphyrinogen III C-methyltransferase, YD repeat protein, and electron transport complex protein RnfC (Figure 3, Table 5, Table S7, Dataset 1, Dataset 2). The details (protein id, type and region of mutations, amino acid changes) of the non-synonymous mutations observed in ColR isolates compared to the ColS isolates, including control isolates, are tabulated in Table 5 and Dataset 1. SNVs, MNV (multi-nucleotide variation), deletion, and insertion were observed in genes encoding hypothetical proteins (AGC85542.1, AGC85972.1, AGC85973.1, AGC85984.1). Interestingly, multiple mutations were detected in ColR isolates in genes encoding several phage-related proteins. SNVs and MNVs were observed in genes encoding putative transposases (AGC87419.1). SNV and insertion were observed in genes encoding phage replication protein O (AGC84960.1). Deletion was observed in genes encoding bacteriophage lysis protein (AGC85980.1) and Iss (AGC85979.1). Likewise, SNVs and MNVs were observed in genes encoding antitermination protein Q from phage origin (AGC86222.1). Similarly, SNVs, deletion, and insertion were observed in genes encoding Rz endopeptidase from lambdoid prophage DLP12 (AGC86226.1), whereas SNV, MNV, and deletion were observed in genes encoding lambdoid prophage DLP12 Bor-like protein (AGC86227.1). Further, SNVs, insertion, and replacement were observed in genes encoding terminase large subunit (AGC87037.1). SNV was also observed in the gene encoding the putative portal protein (AGC87035.1). Similarly, insertion was observed in genes encoding uroporphyrinogen III C-methyltransferase (AGC89286.1), whereas SNV was observed in genes encoding YD repeat protein (AGC86138.1). Similar to non-synonymous mutations, synonymous mutations (only SNVs) were observed in hypothetical proteins, several phage-related proteins (putative transposases, phage replication protein O, antitermination protein Q from phage origin, Rz endopeptidase from lambdoid prophage DLP12, lambdoid prophage DLP12 Bor-like protein, bacteriophage lysis protein, phage repressor protein, terminase large subunit, restriction alleviation protein, and Iss), and electron transport complex protein RnfC (Table S7, Dataset 2). The role of these mutations in colistin-resistant bacteria has not been described before; however, these mutations being simultaneously present with pmrAB system could be adaptive mutations induced to compensate colistin stress or secondary to pmrAB mutations and induced lipid A modifications. Further investigation will aid in an in-depth understanding of molecular sequelae associated with colistin resistance.
Discussion
Colistin is a last-resort antibiotic to treat diverse Gram-negative bacterial pathogens in humans and food animals.1,3 However, chromosomal- and plasmid-mediated resistance to colistin has been reported worldwide, which can limit the future use of colistin.2,4,11 The discovery of colistin activity enhancers or colistin adjuvants as a combination therapy is therefore crucial to extend the lifespan of colistin.49 Strikingly, the highest prevalence of colistin resistance is reported in pathogenic E. coli strains (54%) isolated from food animals,11 which necessitates the development of strategies to attenuate colistin resistance in pathogenic E. coli strains in food animals. Our study has identified SMs (SM2 and SM3) with novel pyrrolidinyl scaffold that potentiated the efficacy of colistin against APEC in vitro and in a wax moth larva model (Table 1, Figure 1). Our previous study had identified that these SMs affect the APEC membrane (by forming membrane blebs)23 which might be the reason behind potentiating the activity of colistin, which is also a membrane-acting antibiotic.2 These SMs also potentiated the activity of tetracycline and ciprofloxacin in vitro which might be due to increased intracellular uptake of tetracycline and ciprofloxacin due to breach in the membrane permeability caused by the SMs.23,50 Similar to our findings, multiple membrane-active compounds with synergistic interaction with colistin have been identified (Table 6) in previous studies.13,15,19,24,25,27 A urea-containing class of 2-aminoimidazole-based adjuvants potentiated colistin activity against ColS A. baumannii with a reduction of colistin MIC by 1000-fold, and increased survival (36–50%) of G. mellonella larva by exhibiting a membrane-permeabilizing effect in the bacteria.13 Eugenol, an essential oil derived from clove, also potentiated the activity of colistin (MIC reduction up to 8-fold) against multiple ColR E. coli isolated from different animals (duck, goose, avian, porcine, zebra, fox, and peacock) by affecting the bacterial membrane integrity.21 Similarly, a combination of pentamidine, a bacterial outer-membrane-active compound, was effective to treat ColR A. baumannii infection in mice.19 Protegrin-1, an antimicrobial peptide derived from porcine leucocytes which disrupts the bacterial membranes, also synergized with colistin against both ColS and ColR A. baumannii strains.25 In the same way, cationic antimicrobial peptides (CAMPs) such as LL-37 (human cathelicidin) and CAMA (cecropin) also reduced the MIC of colistin by at least eightfold against methicillin-resistant Staphylococcus aureus and MDR P. aeruginosa.26 Further, daptomycin, a membrane-acting antibiotic, also showed synergistic interaction with colistin against MDR A. baumannii.24 Moreover, the screening of commercially available FDA-approved library (Prestwick chemical library) identified five compounds (clomiphene citrate, mitoxantrone dihydrochloride, methyl benzethonium chloride, benzethonium chloride, and auranofin) that potentiated the activity of colistin against P. aeruginosa.27 Similarly, four compounds (compounds 1, 2, 3, and 4) identified through screening of substituted 2-aminoimidazole analogs and nitrogen-dense heterocycle libraries reduced the MIC of colistin against ColS and ColR K. pneumoniae, A. baumannii, and E. coli by >32-fold at 30 µM concentrations.22 Niclosamide, an anthelmintic drug, also synergized with colistin against ColR P. aeruginosa, K. pneumoniae, A. baumannii, E. coli, and Enterobacter cloacae with reduction of colistin MIC by 64-fold.18 Resveratrol, a compound derived from plant, synergized with colistin against ColS and ColR K. pneumoniae, A. baumannii, E. coli, E. cloacae, Citrobacter braakii, Stenotrophomonas maltophilia, Serratia marcescens, and Proteus mirabilis.17 In addition, N-acetylcysteine, a mucolytic agent, also synergized with colistin against ColS and ColR A. baumannii grown in planktonic phase and in biofilms.16 These findings suggest that the combination of colistin with other membrane-active compounds or drugs can result in synergistic interaction by facilitating the colistin effect on bacterial membranes.51 In this study, we only tested the synergistic potential of SMs against three APEC strains (O78, O1, and O2); however, these SMs were previously found to be effective against multiple APEC strains, including antibiotic-resistant and biofilm protected APEC;23 therefore, we believe that these SMs may potentiate the activity of colistin against diverse E. coli and possibly against other Gram-negative pathogens. However, this needs further investigation. Further, SMs other than SM2 and SM3 (SM4, SM5, SM6, SM7, SM9, and SM11) which have synergistic interaction with colistin, can be also investigated for their potential as colistin adjuvants.
 |
Table 6 Summary of Colistin Adjuvants Tested Against Different Pathogenic Bacteria |
This study identified mutations in pmrB and pmrA in ColR E. coli O78 (Figure 3, Tables 4 and 5). In this study, substitutions in pmrB p. L14Q, pmrB p. T92P, and pmrA p. A80V were observed. These substitutions are predicted deleterious (PROVEAN score; L14Q: −4.222, T92P: –3.783, and A80V: -–3.818; http://provean.jcvi.org)52 to the functions of PmrB and PmrA proteins. In other studies, different pmrB and pmrA mutations (Table 7) responsible for colistin resistance in E. coli were observed.48,53,58 In ColR E. coli O25 isolated from human clinical specimens in Japan, deletion (∆27-45, LISVFWLWHESTEQIQLFE) in pmrB and substitution (L105P) in pmrA were reported.53 In the same study, substitution (G206D) was observed in pmrB in ColR E. coli O25 and O18 isolates.53 These mutations have led to modification of lipid A with the addition of PEtN and upregulation of eptA (pmrC) and arnT expression and thereby resistance to colistin.53 Likewise, in ColR E. coli LC711/14 isolated from a patient with a urinary tract infection in Italy, substitution (L10P) in pmrB was reported, this mutation upregulated the expression of pmrHFIJKLM operon resulting in lipid A modification and colistin resistance.48 Similarly, in ColR E. coli isolated from human clinical specimens in China, multiple substitutions (A118T, E123D, Y315F, and Y358N) in pmrB were observed, these mutations were predicted to affect phosphate transfer in pmrAB thus altering the function of PmrB protein.54 Likewise, in ColR E. coli isolated from Parisian fecal samples, different substitutions in pmrB (L10P, L10R, C84R, C84Y, P94A, P94L, P94Q, E121K, E121Q, A159P, and A159V) and pmrA (G53A, G53C, G53E, G53R, G53S, G53V, G53W, R81L, and R81S) were observed, these mutations were also predicted to impact the functions of PmrB and PmrA proteins.57 Interestingly, different substitutions in pmrB and pmrA have been observed in ColR E. coli isolated from different food animals.55,58 In ColR E. coli isolated from swine, different pmrB (S138N, E123D, T156A, G160E, V161G, and V351I) and pmrA (G144S, S39I, and R81S) substitutions were observed.55,56 Whereas in ColR E. coli isolated from calf cecum samples, only substitutions in pmrB (D283G and Y358N) were observed.58 Further, in a recent study in Senegalese poultry farms, ColR E. coli isolated from healthy chickens having substitution in pmrB (P94L and D283G) and pmrA (S29G) has been reported.59 However, pmrB and pmrA mutations observed in this study have not been reported yet in any other studies. Based on the studies described above, it is suggested that the different pmrB and pmrA mutations might be responsible for colistin resistance in different E. coli isolates, which might depend upon the E. coli serotype/strain, source of isolation and other factors, including the type of treatment regimen followed in that location. The findings of this study can therefore provide insights for further investigation of potential mechanisms of chromosomal-mediated colistin resistance in poultry E. coli isolates.
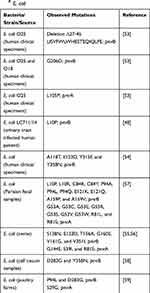 |
Table 7 Summary of pmrB and pmrA Mutations Reported in ColR E. coli |
The SMs identified in this study decreased the frequency of colistin resistance evolution (Figure 2). SM combination also downregulated the pmrCAB and pmrH expression (Figure 4); therefore, these SMs can function as anti-evolution drugs that slow colistin resistance development.32 The upregulation of the expression of operons pmrCAB and pmrHFIJKLM is crucial for bacteria to acquire colistin resistance; therefore, drugs targeting pmrCAB and pmrH expression might help to overcome colistin resistance.45,48 The induced pmrCAB and pmrH expression specifically increases the ability of bacteria to survive in the presence of colistin.45,48 Similar to our SMs, a combination of membrane-acting 2-aminoimidazole-based compounds with colistin also resulted in no resistant A. baumannii and K. pneumoniae, which is mediated by downregulation of pmrCAB expression.20 The lack of resistance observed might be due to reduced selection pressure in combination treatment compared to colistin treatment alone.20 Similarly, antimicrobial peptide glycine-leucine-amide (PGLA), which affects bacterial outer membrane, combination with ciprofloxacin and tobramycin resulted in a 30-fold decrease in E. coli resistance compared to ciprofloxacin and tobramycin alone treatment.41 The combination treatment might reduce the number of resistance-conferring mutations, which ultimately reduces the resistant bacterial population, which is also likely to have occurred in our study.41 Compounds, 1, 2, and 3 (substituted 2-aminoimidazole analogs and nitrogen-dense heterocycles), also suppress the colistin resistance by abolishing or reducing the extent of lipid A modification induced by pmrAB system and mcr-1 gene.22 These findings suggested that the combination of membrane-active adjuvants with conventional antibiotics, including colistin, is a practical way to attenuate colistin and antibiotic resistance as a whole.
Besides PmrA and PmrB, this study also identified other mutations in different hypothetical and phage-related proteins in ColR isolates (Figure 3, Table 5). Similar to our findings, mutations in hypothetical proteins were also found in ColR isolates of A. baumannii,60 K. pneumoniae,61 and P. aeruginosa.62 In our study, we did not obtain any isolates possessing mutations only in genes other than the pmrAB system to determine the relevance of these hypothetical and phage proteins in colistin resistance; therefore, the significance of these mutations is unknown at this time. It is likely that these mutations might just represent adaptive mutations induced to compensate colistin stress or secondary to pmrAB mutations and induced lipid A modifications. The induction of prophage to adapt antibiotic stress was observed when P. aeruginosa was exposed to colistin,63 which can support our finding. Further investigation is necessary to understand the precise role of these mutations in colistin resistance which can help in understanding molecular sequelae associated with colistin resistance.
Conclusion
In conclusion, our study revealed potential mechanisms for chromosomal-mediated colistin resistance in APEC. Two SMs (SM2 and SM3) containing pyrrolidinyl scaffold were identified that potentiated the colistin activity, attenuated the colistin-resistance development against APEC, and downregulated the pmrCAB and pmrH expression. Previously unknown mutations potentially responsible for colistin resistance in pmrB and pmrA were identified in ColR APEC isolates. Mutations were also identified in hypothetical and phage-related proteins; however, further studies are needed to elucidate the relevance of these mutations in colistin resistance. In addition, testing colistin adjuvant properties of these SMs against other APEC related pathogens, including human ExPECs and other Gram-negative pathogens, will have a wider impact on prudent use of colistin.
Data Sharing Statement
All relevant data are contained within the manuscript.
Acknowledgments
We are thankful to Dhwani Parsana and Sochina Ranjit for their technical assistance. We thank Molecular and Cellular Imaging Center (MCIC), Ohio Agricultural Research and Development Centre (OARDC) for providing a platform for bioinformatics analysis of whole-genome sequences.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, agree to the journal in which paper was submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that they have no conflicts of interest.
References
1. Kempf I, Jouy E, Chauvin C. Colistin use and colistin resistance in bacteria from animals. Int J Antimicrob Agents. 2016;48(6):598–606. doi:10.1016/j.ijantimicag.2016.09.016
2. Poirel L, Jayol A, Nordmann P. Polymyxins: antibacterial activity, susceptibility testing, and resistance mechanisms encoded by plasmids or chromosomes. Clin Microbiol Rev. 2017;30(2):557. doi:10.1128/CMR.00064-16
3. Biswas S, Brunel JM, Dubus JC, Reynaud-Gaubert M, Rolain JM. Colistin: an update on the antibiotic of the 21st century. Expert Rev Anti Infect Ther. 2012;10(8):917–934. doi:10.1586/eri.12.78
4. Liu Y, Liu J-H. Monitoring colistin resistance in food animals, an urgent threat. Expert Rev Anti Infect Ther. 2018;16(6):443–446. doi:10.1080/14787210.2018.1481749
5. Landman WJM, van Eck JHH. The incidence and economic impact of the Escherichia coli peritonitis syndrome in Dutch poultry farming. Avian Pathol. 2015;44(5):370–378. doi:10.1080/03079457.2015.1060584
6. Le Devendec L, Mourand G, Bougeard S, et al. Impact of colistin sulfate treatment of broilers on the presence of resistant bacteria and resistance genes in stored or composted manure. Vet Microbiol. 2016;194:98–106. doi:10.1016/j.vetmic.2015.11.012
7. Baron S, Hadjadj L, Rolain J-M, Olaitan AO. Molecular mechanisms of polymyxin resistance: knowns and unknowns. Int J Antimicrob Agents. 2016;48(6):583–591. doi:10.1016/j.ijantimicag.2016.06.023
8. Liu YY, Wang Y, Walsh TR, et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis. 2016;16(2):161–168. doi:10.1016/S1473-3099(15)00424-7
9. Huang X, Yu L, Chen X, et al. High prevalence of colistin resistance and mcr-1 gene in Escherichia coli isolated from food animals in China. Front Microbiol. 2017;8:562. doi:10.3389/fmicb.2017.00562
10. Carroll LM, Gaballa A, Guldimann C, Sullivan G, Henderson LO, Wiedmann M. Identification of novel mobilized colistin resistance gene mcr-9 in a multidrug-resistant, colistin-susceptible Salmonella enterica serotype typhimurium isolate. mBio. 2019;10(3):e00853–00819. doi:10.1128/mBio.00853-19
11. Elbediwi M, Li Y, Paudyal N, et al. Global burden of colistin-resistant bacteria: mobilized colistin resistance genes study (1980–2018). Microorganisms. 2019;7(10):10. doi:10.3390/microorganisms7100461
12. MacNair CR, Stokes JM, Carfrae LA, et al. Overcoming mcr-1 mediated colistin resistance with colistin in combination with other antibiotics. Nat Commun. 2018;9(1):458. doi:10.1038/s41467-018-02875-z
13. Minrovic BM, Jung D, Melander RJ, Melander C. New class of adjuvants enables lower dosing of colistin against Acinetobacter baumannii. ACS Infect Dis. 2018;4(9):1368–1376. doi:10.1021/acsinfecdis.8b00103
14. Bernal P, Molina-Santiago C, Daddaoua A, Llamas MA. Antibiotic adjuvants: identification and clinical use. Microb Biotechnol. 2013;6(5):445–449. doi:10.1111/1751-7915.12044
15. Kubo A, Lunde CS, Kubo I. Indole and (E)-2-hexenal, phytochemical potentiators of polymyxins against Pseudomonas aeruginosa and Escherichia coli. Antimicrob Agents Chemother. 1996;40(6):1438–1441. doi:10.1128/AAC.40.6.1438
16. Pollini S, Boncompagni S, Di Maggio T, et al. In vitro synergism of colistin in combination with N-acetylcysteine against Acinetobacter baumannii grown in planktonic phase and in biofilms. J Antimicrob Chemother. 2018;73(9):2388–2395. doi:10.1093/jac/dky185
17. Cannatelli A, Principato S, Colavecchio OL, Pallecchi L, Rossolini GM. Synergistic activity of colistin in combination with resveratrol against colistin-resistant gram-negative pathogens. Front Microbiol. 2018;9:1808. doi:10.3389/fmicb.2018.01808
18. Domalaon R, De Silva PM, Kumar A, Zhanel GG, Schweizer F. The anthelmintic drug niclosamide synergizes with colistin and reverses colistin resistance in gram-negative bacilli. Antimicrob Agents Chemother. 2019;63(4):4. doi:10.1128/AAC.02574-18
19. Stokes JM, MacNair CR, Ilyas B, et al. Pentamidine sensitizes Gram-negative pathogens to antibiotics and overcomes acquired colistin resistance. Nature Microbiol. 2017;2(5):17028. doi:10.1038/nmicrobiol.2017.28
20. Harris TL, Worthington RJ, Hittle LE, Zurawski DV, Ernst RK, Melander C. Small molecule downregulation of pmrAB reverses lipid A modification and breaks colistin resistance. ACS Chem Biol. 2014;9(1):122–127. doi:10.1021/cb400490k
21. Wang YM, Kong LC, Liu J, Ma HX. Synergistic effect of eugenol with Colistin against clinical isolated Colistin-resistant Escherichia coli strains. Antimicrob Resist Infect Control. 2018;7(1):17. doi:10.1186/s13756-018-0303-7
22. Barker WT, Martin SE, Chandler CE, et al. Small molecule adjuvants that suppress both chromosomal and mcr-1 encoded colistin-resistance and amplify colistin efficacy in polymyxin-susceptible bacteria. Bioorg Med Chem. 2017;25(20):5749–5753. doi:10.1016/j.bmc.2017.08.055
23. Kathayat D, Helmy YA, Deblais L, Rajashekara G. Novel small molecules affecting cell membrane as potential therapeutics for avian pathogenic Escherichia coli. Sci Rep. 2018;8(1):15329. doi:10.1038/s41598-018-33587-5
24. Galani I, Orlandou K, Moraitou H, Petrikkos G, Souli M. Colistin/daptomycin: an unconventional antimicrobial combination synergistic in vitro against multidrug-resistant Acinetobacter baumannii. Int J Antimicrob Agents. 2014;43(4):370–374. doi:10.1016/j.ijantimicag.2013.12.010
25. Morroni G, Simonetti O, Brenciani A, et al. In vitro activity of Protegrin-1, alone and in combination with clinically useful antibiotics, against Acinetobacter baumannii strains isolated from surgical wounds. Med Microbiol Immunol. 2019;208(6):877–883. doi:10.1007/s00430-019-00624-7
26. Geitani R, Ayoub Moubareck C, Touqui L, Karam Sarkis D. Cationic antimicrobial peptides: alternatives and/or adjuvants to antibiotics active against methicillin-resistant Staphylococcus aureus and multidrug-resistant Pseudomonas aeruginosa. BMC Microbiol. 2019;19(1):54. doi:10.1186/s12866-019-1416-8
27. Torres NS, Montelongo-Jauregui D, Abercrombie JJ, et al. Antimicrobial and antibiofilm activity of synergistic combinations of a commercially available small compound library with colistin against Pseudomonas aeruginosa. Front Microbiol. 2018;9:2541. doi:10.3389/fmicb.2018.02541
28. Johnson TJ, Wannemuehler Y, Doetkott C, Johnson SJ, Rosenberger SC, Nolan LK. Identification of minimal predictors of avian pathogenic Escherichia coli virulence for use as a rapid diagnostic tool. J Clin Microbiol. 2008;46(12):3987–3996. doi:10.1128/JCM.00816-08
29. Guabiraba R, Schouler C. Avian colibacillosis: still many black holes. FEMS Microbiol Lett. 2015;362(15):fnv118–fnv118. doi:10.1093/femsle/fnv118
30. Liu CM, Stegger M, Aziz M, et al. Escherichia coli ST131- H 22 as a foodborne uropathogen. mBio. 2018;9(4):4. doi:10.1128/mBio.00470-18
31. Osman KM, Kappell AD, Elhadidy M, et al. Poultry hatcheries as potential reservoirs for antimicrobial-resistant Escherichia coli: a risk to public health and food safety. Sci Rep. 2018;8(1):5859. doi:10.1038/s41598-018-23962-7
32. Baym M, Stone LK, Kishony R. Multidrug evolutionary strategies to reverse antibiotic resistance. Science. 2016;351(6268):aad3292–aad3292. doi:10.1126/science.aad3292
33. Wallace Iain M, Urbanus Malene L, Luciani Genna M, et al. Compound prioritization methods increase rates of chemical probe discovery in model organisms. Chem Biol. 2011;18(10):1273–1283. doi:10.1016/j.chembiol.2011.07.018
34. CLSI.. Performance Standards for Antimicrobial Susceptibility Testing.
35. Orhan G, Bayram A, Zer Y, Balci I. Synergy tests by E test and checkerboard methods of antimicrobial combinations against Brucella melitensis. J Clin Microbiol. 2005;43(1):140–143. doi:10.1128/JCM.43.1.140-143.2005
36. Deblais L, Helmy YA, Kathayat D, Huang H-C, Miller SA, Rajashekara G. Novel imidazole and methoxybenzylamine growth inhibitors affecting salmonella cell envelope integrity and its persistence in chickens. Sci Rep. 2018;8(1):13381. doi:10.1038/s41598-018-31249-0
37. Bremmer DN, Bauer KA, Pouch SM, et al. Correlation of checkerboard synergy testing with time-kill analysis and clinical outcomes of extensively drug-resistant Acinetobacter baumannii respiratory infections. Antimicrob Agents Chemother. 2016;60(11):6892–6895. doi:10.1128/AAC.00981-16
38. Betts J, Nagel C, Schatzschneider U, Poole R, La Ragione RM. Antimicrobial activity of carbon monoxide-releasing molecule [Mn(CO)(3)(tpa-κ(3)N)]Br versus multidrug-resistant isolates of Avian Pathogenic Escherichia coli and its synergy with colistin. PLoS One. 2017;12(10):e0186359. doi:10.1371/journal.pone.0186359
39. Peleg AY, Jara S, Monga D, Eliopoulos GM, Moellering RC, Mylonakis E. Galleria mellonella as a model system to study Acinetobacter baumannii pathogenesis and therapeutics. Antimicrob Agents Chemother. 2009;53(6):2605. doi:10.1128/AAC.01533-08
40. Tsai CJ-Y, Loh JMS, Proft T. Galleria mellonella infection models for the study of bacterial diseases and for antimicrobial drug testing. Virulence. 2016;7(3):214–229. doi:10.1080/21505594.2015.1135289
41. Lázár V, Martins A, Spohn R, et al. Antibiotic-resistant bacteria show widespread collateral sensitivity to antimicrobial peptides. Nature Microbiol. 2018;3(6):718–731. doi:10.1038/s41564-018-0164-0
42. Zankari E, Allesoe R, Joensen KG, Cavaco LM, Lund O, Aarestrup FM. PointFinder: a novel web tool for WGS-based detection of antimicrobial resistance associated with chromosomal point mutations in bacterial pathogens. J Antimicrob Chemother. 2017;72(10):2764–2768. doi:10.1093/jac/dkx217
43. Altshuler D, Pollara VJ, Cowles CR, et al. An SNP map of the human genome generated by reduced representation shotgun sequencing. Nature. 2000;407(6803):513–516. doi:10.1038/35035083
44. Brockman W, Alvarez P, Young S, et al. Quality scores and SNP detection in sequencing-by-synthesis systems. Genome Res. 2008;18(5):763–770. doi:10.1101/gr.070227.107
45. Cannatelli A, Di Pilato V, Giani T, et al. In vivo evolution to colistin resistance by PmrB sensor kinase mutation in KPC-producing Klebsiella pneumoniae is associated with low-dosage colistin treatment. Antimicrob Agents Chemother. 2014;58(8):4399–4403. doi:10.1128/AAC.02555-14
46. Helmy YA, Deblais L, Kassem II, Kathayat D, Rajashekara G. Novel small molecule modulators of quorum sensing in avian pathogenic Escherichia coli (APEC). Virulence. 2018;9(1):1640–1657. doi:10.1080/21505594.2018.1528844
47. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 2001;25(4):402–408. doi:10.1006/meth.2001.1262
48. Cannatelli A, Giani T, Aiezza N, et al. An allelic variant of the PmrB sensor kinase responsible for colistin resistance in an Escherichia coli strain of clinical origin. Sci Rep. 2017;7(1):5071. doi:10.1038/s41598-017-05167-6
49. Tyers M, Wright GD. Drug combinations: a strategy to extend the life of antibiotics in the 21st century. Nat Rev Microbiol. 2019;17(3):141–155. doi:10.1038/s41579-018-0141-x
50. Mingeot-Leclercq M-P, Decout J-L. Bacterial lipid membranes as promising targets to fight antimicrobial resistance, molecular foundations and illustration through the renewal of aminoglycoside antibiotics and emergence of amphiphilic aminoglycosides. MedChemComm. 2016;7(4):586–611. doi:10.1039/C5MD00503E
51. Stowe SD, Thompson RJ, Peng L, et al. Membrane-permeabilizing activity of reverse-amide 2-aminoimidazole antibiofilm agents against Acinetobacter baumannii. Curr Drug Deliv. 2015;12(2):223–230. doi:10.2174/1567201811666140924125740
52. Choi Y, Chan AP. PROVEAN web server: a tool to predict the functional effect of amino acid substitutions and indels. Bioinformatics. 2015;31(16):2745–2747. doi:10.1093/bioinformatics/btv195
53. Sato T, Shiraishi T, Hiyama Y, et al. Contribution of novel amino acid alterations in pmrA or pmrB to Colistin resistance in mcr-negative Escherichia coli clinical isolates, including major multidrug-resistant lineages O25b:H4-ST131- H 30Rx and Non-x. Antimicrob Agents Chemother. 2018;62(9):9. doi:10.1128/AAC.00864-18
54. Luo Q, Yu W, Zhou K, et al. Molecular epidemiology and colistin resistant mechanism of mcr-positive and mcr-negative clinical isolated Escherichia coli. Front Microbiol. 2017;8:2262. doi:10.3389/fmicb.2017.02262
55. Delannoy S, Le Devendec L, Jouy E, Fach P, Drider D, Kempf I. Characterization of colistin-resistant Escherichia coli isolated from diseased pigs in France. Front Microbiol. 2017;8:2278. doi:10.3389/fmicb.2017.02278
56. Quesada A, Porrero MC, Téllez S, Palomo G, García M, Domínguez L. Polymorphism of genes encoding PmrAB in colistin-resistant strains of Escherichia coli and Salmonella enterica isolated from poultry and swine. J Antimicrob Chemother. 2014;70(1):71–74. doi:10.1093/jac/dku320
57. Bourrel AS, Poirel L, Royer G, et al. Colistin resistance in Parisian inpatient faecal Escherichia coli as the result of two distinct evolutionary pathways. J Antimicrob Chemother. 2019;74(6):1521–1530. doi:10.1093/jac/dkz090
58. Rebelo AR, Bortolaia V, Kjeldgaard JS, et al. Multiplex PCR for detection of plasmid-mediated colistin resistance determinants, mcr-1, mcr-2, mcr-3, mcr-4 and mcr-5 for surveillance purposes. Eurosurveillance. 2018;23(6):17–00672. doi:10.2807/1560-7917.ES.2018.23.6.17-00672
59. Vounba P, Rhouma M, Arsenault J, Bada Alambédji R, Fravalo P, Fairbrother JM. Prevalence of colistin resistance and mcr-1/mcr-2 genes in extended-spectrum β-lactamase/AmpC-producing Escherichia coli isolated from chickens in Canada, Senegal and Vietnam. J Glob Antimicrob Resist. 2019;19:222–227. doi:10.1016/j.jgar.2019.05.002
60. Cafiso V, Stracquadanio S, Lo Verde F, et al. Colistin resistant A. baumannii: genomic and transcriptomic traits acquired under colistin therapy. Front Microbiol. 2019;9:3195. doi:10.3389/fmicb.2018.03195
61. Wright MS, Suzuki Y, Jones MB, et al. Genomic and transcriptomic analyses of colistin-resistant clinical isolates of Klebsiella pneumoniae reveal multiple pathways of resistance. Antimicrob Agents Chemother. 2015;59(1):536–543. doi:10.1128/AAC.04037-14
62. Lee J-Y, Na IY, Park YK, Ko KS. Genomic variations between colistin-susceptible and -resistant Pseudomonas aeruginosa clinical isolates and their effects on colistin resistance. J Antimicrob Chemother. 2014;69(5):1248–1256. doi:10.1093/jac/dkt531
63. Mehta HH, Prater AG, Beabout K, et al. The essential role of hypermutation in rapid adaptation to antibiotic stress. Antimicrob Agents Chemother. 2019;63(7):e00744–00719. doi:10.1128/AAC.00744-19
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




