Back to Journals » International Journal of Nanomedicine » Volume 14
Slp-coated liposomes for drug delivery and biomedical applications: potential and challenges
Authors Luo G, Yang Q, Yao B, Tian Y, Hou R, Shao A, Li M, Feng Z, Wang W
Received 6 October 2018
Accepted for publication 7 January 2019
Published 20 February 2019 Volume 2019:14 Pages 1359—1383
DOI https://doi.org/10.2147/IJN.S189935
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Linlin Sun
Gan Luo,1,2 Qingliang Yang,1 Bingpeng Yao,1,3 Yangfan Tian,4 Ruixia Hou,1 Anna Shao,1 Mengting Li,1 Zilin Feng,1 Wenxi Wang1
1Department of Pharmaceutics, College of Pharmaceutical Science, Zhejiang University of Technology, Hangzhou, Zhejiang, China; 2Department of Anesthesiology and Intensive Care, The First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, Zhejiang, China; 3Department of Green Pharmaceutics, Jianxing Honors College, Zhejiang University of Technology, Hangzhou, Zhejiang, China; 4Department of Pediatric Surgery, The Children’s Hospital, Zhejiang University School of Medicine, Hangzhou, Zhejiang, China
Abstract: Slp forms a crystalline array of proteins on the outermost envelope of bacteria and archaea with a molecular weight of 40–200 kDa. Slp can self-assemble on the surface of liposomes in a proper environment via electrostatic interactions, which could be employed to functionalize liposomes by forming Slp-coated liposomes for various applications. Among the molecular characteristics, the stability, adhesion, and immobilization of biomacromolecules are regarded as the most meaningful. Compared to plain liposomes, Slp-coated liposomes show excellent physicochemical and biological stabilities. Recently, Slp-coated liposomes were shown to specifically adhere to the gastrointestinal tract, which was attributed to the “ligand–receptor interaction” effect. Furthermore, Slp as a “bridge” can immobilize functional biomacromolecules on the surface of liposomes via protein fusion technology or intermolecular forces, endowing liposomes with beneficial functions. In view of these favorable features, Slp-coated liposomes are highly likely to be an ideal platform for drug delivery and biomedical uses. This review aims to provide a general framework for the structure and characteristics of Slp and the interactions between Slp and liposomes, to highlight the unique properties and drug delivery as well as the biomedical applications of the Slp-coated liposomes, and to discuss the ongoing challenges and perspectives.
Keywords: S-layer protein, liposomes, self-assembly, interactions, drug delivery, biomedical applications
Introduction
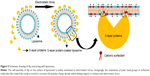
Slp forms a crystalline array of proteins (called S-layer) on the outermost envelope of bacteria and archaea with a molecular weight of 40–200 kDa.1,2 The S-layer, which has a thickness of 5–25 nm, provides capsule-like protection for the maintenance of bacterial cell morphology.3,4 Due to its excellent ability to regularly assemble monomers in aqueous environments, Slp can spontaneously self-assemble on air–water interfaces, solid supports, or liposomes or in suspension and solution, by an entropy-driven process (Figure 1).5,6 This feature enables a wide range of potential applications of Slp in nanomedicine, bioactive drug delivery, and gene drug delivery. As a result, Slp-coated liposomes have received increasing attention in the past decade. Liposomes are nanovesicles composed of phospholipid bilayers that have various beneficial characteristics, such as good cell affinity,7,8 targeting properties,9–11 and sustained drug-release behavior.12 Due to their favorable ability to encapsulate both hydrophobic and hydrophilic drugs, liposomes are widely used as carriers for bioactive drugs, including antineoplastic drugs,13,14 antimicrobial drugs,15 antiparasitic drugs,16 hormonal drugs,17 proteins,18,19 and nucleic acid drugs20,21 such as DNA22–24 and siRNA.25,26 Additionally, liposomes have the capacity to reduce the toxicity of these drugs.27 Unfortunately, conventional liposomes have drawbacks, such as poor stability, drug leakage, short retention time in vivo, and lack of ability to avoid the reticuloendothelial system (RES), which dramatically limit their clinical use. Therefore, increasing attention has been paid to the surface engineering of liposomes using various biological or chemical materials to improve their features.28 Evidence has shown that the membrane stability and gastrointestinal adhesion of liposomes were significantly improved after being coated with Slp.29 Furthermore, some biomacromolecules (such as antibodies) are prone to interaction on the surface of liposomes via binding to the tertiary structure of Slp. Hence, Slp-coated liposomes may be used as a powerful vehicle for drug delivery to resolve problems such as instability, drug leakage, and poor targeting.30 The interactions between Slp and liposomes are crucial elements for the coating of Slp. Therefore, understanding these interactions is necessary and urgent for the development of Slp-coated liposomes. In the current review, after the introduction of the structure and characteristics of Slp, the interactions between Slp and liposomes are summarized. Various advantages of Slp-coated liposomes, including their excellent stabilities, adhesion and immobilization of biomacromolecules, and applications in drug delivery and biomedicine, are reviewed in detail (Table 1). The challenges and perspectives of Slp-coated liposomes are also discussed.
  | Figure 1 Schematic drawing of self-reassembly of Slp on solid supports, in suspension, at an air–water interface, and on liposomes as well as in solution (nanotubes, ribbons, mono- and double-layer sheets). |
  | Table 1 Application of Slp-coated liposomes |
Structure and characteristics of Slp
In 1952, Houwink and Le Poole first observed crystalline arrays of biomacromolecules on the outermost cell surface of Spirillum serpenes by electron microscopy. Since then, many studies have revealed that this biomacromolecule, now named Slp, is also present on the cell surface of other bacteria and archaea.31 To date, Slp has been found on the outermost cell surface of almost 400 species of archaea and bacteria.32 Most Slps are weakly acidic, with a molecular weight of 40–200 kDa and a thickness of 5–25 nm, and their pI values range from 4 to 6 (however, for the Slps isolated from Lactobacilli and some archaea, such as Methanothermus fervidus, pI values between 8 and 10 have been determined).33,34 Notably, most Slps contain 40%–60% hydrophobic amino acids and 15% acidic amino acids, with 10% lysine and few sulfur-containing amino acids.35,36 This composition is the reason why Slps hold together and reassemble spontaneously in aqueous systems.37 Moreover, data from the circular dichroism spectrum indicated that α-helices and β-sheets comprise 10%–20% and 40% of the amino acids of Slp, respectively. In addition, random coils and β-turns comprise 5%–45% of the amino acids in the Slp sequence. These random coils, α-helices, and β-sheets constitute disordered regions in the structure of Slp,38–42 which may play a key role in self-assembly on the phase interface. The disordered regions occupy almost half of the full length of Slp, and simultaneously, the high content of disordered regions results in the flexibility of Slp to alter its conformation for adapting to the phase interface, which has a crucial role in self-assembly on the surface of liposomes.38,42
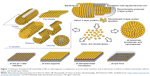
After treatments with detergents/breaking agents (eg, guanidine hydrochloride or urea) or application of chelating agents and cation substitution, Slp can be isolated from the bacterial cell wall due to the weak interactions between Slp subunits.6,43,44 These Slp subunits can hold together and reassemble spontaneously on a two-phase interface or in suspension, forming specific crystalline arrays.45 Specially, in aqueous systems, Slp subunits rapidly hold together to form aggregates, which are prone to arrange on the phase interface and form crystalline arrays.46,47 Regular distribution of self-assembly sites and monomolecular assembly property of Slp tend to result in the formation of oblique (p1, p2), square (p4), or hexagonal (p3, p6) symmetries on the phase interface (Figure 2).2,46,47
  | Figure 2 Schematic drawing of various Slp lattice morphologies containing oblique (p1, p2), square (p4), or hexagonal (p3, p6) symmetries. |
Slp lattices construct the outermost cell surface framework of archaea and bacteria, maintaining the stability of the cell morphology. Simultaneously, Slp lattices can provide cells with protection against changes in extrinsic stress factors (eg, various mechanical forces, osmotic pressure, radiation, and rapid variation in pH values) and the influence of antimicrobial peptides (such as human defensin LL-37)48 as well as lysozyme. The substance-selective crystalline arrays consisting of Slp are able to control the exchange of substances between bacterial cells and the environment.49 In addition, Slp is one of the most important elements for the recognition of host cells and the adhesion of bacterial cells to host cells. For example, Lactobacilli with Slp can adhere to gastrointestinal cells, while the adhesion of Lactobacilli without Slp is decreased by 84%.44 Furthermore, as a virulence factor, Slp possesses heavy metal ion-absorption properties as well as superb immunogenic potential. All of these diverse functions reflect the acclimatization of prokaryotes. Remarkably, the self-assembly property of Slp broadens its application potential in the molecular engineering and nanobiomaterial fields. Following are some of the applications of Slp:
- Production of magnetic ultrafiltration membranes.
- Support and conjugation of biomacromolecules (eg, antigens, antibodies, and enzymes).
- Applications in biosensor systems, such as solid-phase immunoassays.
- Construction of multifunctional nanoparticle drug delivery systems (eg, liposomes and emulsomes).
Surface engineering of liposomes
Liposomes are biomimetic vesicle-like drug delivery systems composed of phospholipid molecules and cholesterol. Due to their good biocompatibility and biodegradability, passive targeting, and slow-release potential, as well as their strong tissue affinity, liposomes have been investigated as carriers of various drugs, such as antitumor drugs, antimicrobial drugs, RNAs, and proteins. Liposomes were first discovered by Bangham in 1969, and in 1971, Rahman proposed that liposomes can be an ideal drug carrier. Later, many related studies were performed. To date, liposomal doxorubicin, liposomal amphotericin B, and liposomal paclitaxel have been applied in the clinic. However, poor stability, drug leakage, short retention time, and undesirable tissue distribution of conventional liposomes limit further clinical applications.50–53 In order to overcome these problems, various chemical and biological materials were adopted to modify liposomes (Figure 3).
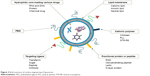  | Figure 3 Brief summary of surface engineering of liposomes. |
PEGylation is the most common strategy which has the ability to reduce clearance by the RES, thus enhancing the in vivo circulating time of liposomes. In addition, PEGylation can delay drug leakage, improving the stability of liposomes. However, PEGylation will affect the normal release of drugs and uptake of cells, thereby decreasing the effect of the drug.54 Chitosan derived from chitin is the only basic amylose in nature. Its exceptional bioadhesive ability can prolong the retention time of liposomes in specific tissues, leading to improved bioavailability.55 Due to its alkalinity, chitosan carries a positive charge after protonation, which could promote the transfection of chitosan-modified liposomes into cells. Hence, a chitosan-modified strategy has been widely used in liposomal delivery of genes.56–58 In addition, many peptides and proteins have been employed to functionalize the liposomes. Collagen protein can enhance the in vitro and in vivo stability of liposomes.59 Transferrin is generally used to allow liposomes or other nanoparticles to cross the blood–brain barrier, achieving delivery to central nervous system.60–62 Other functional proteins or peptides, such as EGF and RGD, also support targeting delivery. Furthermore, membrane-active peptide63,64 and cell-penetrating peptide65,66 facilitate the intracellular delivery of liposomes. Surface-modified liposomes with Slp also have various advantages (eg, enhancing stabilities and promoting gastrointestinal adhesion) and have received increasing attention in recent decades.
Interactions between Slp and liposomes
Due to its capability to self-assemble on the surface of lipid layers, Slp has been employed to functionalize liposomes. In particular, the formation of Slp crystalline arrays on the surface of positively charged liposomes by interaction of exposed carboxyl groups of Slp with positively charged groups on the surface of liposomes can significantly enhance the stability of liposomal membranes,67–70 protecting liposomes from strong mechanical forces and high-temperature environments.6 However, the details of the interactions between liposomes and Slp remain unclear. In recent years, a series of electron microscopy technologies, such as the thin-sectioned, freeze-dried, and freeze-etched methods,30,71–78 have been adopted to elucidate the location and ultrastructure of Slp lattices. In addition, information on lattice constants, such as average size, was provided by atomic force microscopy31,79–83 and small-angle X-ray scattering.73,84,85 Fourier transform infrared (FTIR) spectroscopy was used to validate the secondary structure of Slp lattices.86 Furthermore, the variation of the zeta potential could be utilized to estimate the areas of lipid membranes occupied by Slp.29,87 All of these measurements merely reflected the interaction information indirectly.
In essence, a liposome is a closed lipid membrane with a morphology that is similar to the cell. Hence, some clues on the interactions of Slp with liposomes could be obtained from the interactions between Slp and microbial cells. It was reported1,88 that there are five modes of interaction between Slp and different species of bacteria and archaea: 1) in most archaea, hydrophobic transmembrane domains of Slp can transfix the cell envelope and then combine with hydrophobic groups of phospholipid molecules via hydrophobic forces (Figure 4A), thereby directly attaching to the cytoplasmic membrane; 2) Slp of some archaea possesses lipid-modified glycoprotein subunits, allowing Slp to directly anchor on the surface of the cell envelope (Figure 4B); 3) only a few archaea have a rigid wall layer as an intermediate layer between the Slp and the cell envelope (Figure 4C); 4) in Gram-positive bacteria, Slp links with the rigid peptidoglycan-containing layer by secondary cell wall polymers (Figure 4D); and 5) in Gram-negative bacteria, the interaction mode is more complex: Slp is attached to the lipopolysaccharide (LPS) of the outermost layer (Figure 4E).
  | Figure 4 Schematic illustration of the five modes of interaction between Slp and the bacterial/archaeal cell envelope. |
However, for liposomes, there are no complex components (eg, peptidoglycan and LPS) that can support the interaction between liposomes and Slp. There is mounting evidence that electrostatic forces drive the self-assembly process of Slp. The fact that Slp lattices tend to form on zwitterionic phospholipids and positively charged phospholipids, but not on negatively charged phospholipids, suggests electrostatic interactions between exposed carboxyl groups on Slp lattices and positively charged or zwitterionic lipid head groups. At least two to three structural domains in Slp named contact points have been shown to exist between Slp and lipid film. Meanwhile, 5% of lipid molecules are anchored to contact points, whereas the other 95% of the lipid molecules diffuse freely between anchored lipid molecules. Hence, Slp-coated lipid films are also referred to as semifluid membranes.85 In this case, it is interesting to consider which regions or groups in lipid molecules exactly interact with contact points. This issue was examined by a study on the interaction regions between Slp and liposomes via the monitoring of adiabatic compressibility. An insignificant difference was observed for adiabatic compressibility between Slp-coated liposomes and plain liposomes at a low temperature (T<20°C), whereas at a higher temperature (T>20°C), adiabatic compressibility of Slp-coated liposomes was predominantly lower than that of plain liposomes. Adiabatic compressibility can indicate the contribution of the conformational mobility of polar head groups as well as hydrophobic regions. Analysis of adiabatic compressibility confirmed that such a contribution preferentially came from the conformational variation of polar head groups, suggesting that contact points of Slp interact with polar head groups rather than hydrophobic tail groups.89 FTIR spectroscopy analyses also support this view; the order parameter of phospholipid hydrocarbon chains (hydrophobic regions) did not decrease with the continuous absorption of Slp, proving that contact points did not interpenetrate into the hydrocarbon chains.90 Another FTIR spectroscopy study found no difference between Slp-coated liposomes and a physical mixture of naked liposomes and Slp, suggesting that the interaction force between liposomes and Slp is a non-covalent bond.29 Furthermore, as shown in Figure 5, to accommodate the contact points of Slp, the orientation of polar head groups of phospholipid molecules is preferentially tilted toward the surface normal to increase the positive charge density within the linking regions, and this effect enhances the electrostatic interactions.91 However, there are slight differences between different Slps and liposomes. For example, Hollmann et al92 extracted glycosylated Slp from Lactobacillus kefir and non-glycosylated Slp from Lactobacillus brevis, and then, glycosylated Slp-coated liposomes (GSLs) and non-glycosylated Slp-coated liposomes (nGSLs) were obtained. Laurdan generalized polarization (GP) values of GSLs and nGSLs were determined to investigate the local motion of polar molecules. The results demonstrated that the GP value of GSLs was higher than that of nGSLs, which indicated that the affinity of glycosylated Slp to liposomes was stronger than that of non-glycosylated Slp due to more water molecules penetrating the membrane of nGSLs. Meanwhile, these findings also illustrated that uncharged glycosylated moieties could affect the interactions between Slp and liposomes, and this effect may be mediated by glycosylated moieties changing the charge distribution.
In fact, the formation of Slp lattices on the surface of liposomes depends not only on the interactions between Slp and phospholipid molecules but also on the interactions between Slp subunits. As Figure 6 suggests, there are self-assembly sites in the structure of Slp that support the formation of Slp lattices. The addition of Mg2+ causes a conformational change of Slp, which makes these self-assembly sites to get buried in Slp, thus maintaining monodispersion. Therefore, it is difficult for Slp to form lattices in the presence of Mg2+.38,42 Notably, the uptake of Ca2+ can induce a thorough exposure of self-assembly sites that facilitates the interactions and formation of lattices.93,94 Without any cation addition, the dynamic equilibrium process of the Slp conformation provides opportunities for self-assembly site exposure, allowing lattice formation. However, these lattices are not very stable because this is a dynamic equilibrium process.38,42 Collectively, the cation environment of liposomes and Slp will induce the shape variation of Slp that determines the formation of lattices and the type of lattices. Hence, we hypothesize that the components in the medium, especially the composition of ions, should also be investigated in depth when preparing Slp-coated liposomes.
  | Figure 6 Schematic illustration showing the influence of cations on the formation of Slp lattices. |
In summary, successful S-layer formation on the surface of liposomes mostly relies on two self-assembly modes: 1) the electrostatic assembly between Slp and liposomes; and 2) the intermolecule assembly between Slp monomers. The former helps Slp to anchor onto the lipid membranes, and the latter drives the formation of S-layer. It is known that Slp-coated liposomes possess high stability and it is not easy for Slp to loss from liposomal membranes due to the relatively strong intermolecular assembly between Slp subunits. Of course, the stability depends on the surface charges of liposomes and the species of Slp.
Slp-coated liposomes
Self-assembly properties and interactions between Slp and liposomes are the basis for the development of Slp-coated liposomes. Once Slp is coated on the surface of liposomes, liposomes are endowed with unique features, such as excellent physicochemical and biological stabilities, gastrointestinal adhesion, and immobilization of biomacromolecules. These unique features suggest a series of clinical potential, which lay the foundation for Slp-coated liposomes as an ideal platform for drug delivery and biomedical uses.
Characterization of Slp-coated liposomes
In general, particle size and zeta potential are used to characterize the physicochemical stability of liposomes because these two properties not only reveal stability but also affect the in vivo pharmacokinetic process of liposomes.95,96 In addition, the zeta potential reveals the variation in the ionic microenvironment around the surface of the liposomes and thus contributes to the characterization of adsorption between Slp and liposomes. Compared with plain liposomes, Slp-coated liposomes display an interesting variation in zeta potential. With the increasing ratio of Slp to lipid, the zeta potential decreases significantly29,97,98 until the inversion of the potential.97,98 As noted in the previous section, the adsorption of Slp (carrying negative charges) on the surface of liposomes mainly relies on electrostatic forces. Hence, the Slp-coating efficiency of positively charged liposomes is much better than that of negatively charged or neutral liposomes.29 Indeed, with increasing concentrations of Slp, the area of the positively charged liposome bilayer surface is gradually occupied by Slp, which leads to a decrease in zeta potential. However, the reduction of the potential becomes less, and the value of the potential tends toward invariance with the continuous enhancement of the ratio of Slp to lipid, which is the result of the limited area of liposomes for reassembly of Slp.29,99 For some positively charged liposomes, the inversion of the zeta potential can be observed with the incubation of sufficient Slp. For example, Hollmann et al98 prepared a positively charged liposome composed of soybean lecithin, cholesterol, and stearylamine with a molar ratio of 10:5:1. After incubation for 150 minutes with Slp extracted from L. kefir JCM 5818, the value of the zeta potential showed an inversion from 80 to −15 mV. In summary, the variation of the zeta potential can be employed to characterize the amount of Slp reassembled on the surface of liposomes.
Contrary to the zeta potential, the coating of Slp does not significantly influence the particle size of the liposomes. Research by Hollmann et al98 found that the particle size of liposomes increased by 21 nm (from 160 to 181 nm) after coating with Slp. The slight increase in particle size exactly conforms to the 5–25 nm thickness of the S-layer on bacteria.35,100
Unique properties of Slp-coated liposomes
Stability
Poor stability is known to restrict the development and application of liposomes due to the fusion and membrane disruption propensity of liposomes. Hence, developing a technology to improve the stability of liposomal membranes is critical to expand the application potential of liposomes. Liposomes are assembled by phospholipid molecules via hydrophobic forces. However, this is a weak interaction; hence, the membrane of the plain liposome is weak and can easily be disrupted by environmental stress factors (eg, low pH value, mechanical force, and rapid variation in temperature), leading to disordered phospholipid molecule arrays (Figure 7A), which cause membrane deformation and drug leakage. When Slp self-assembles on the surface of the liposome by electrostatic forces, the arrays of phospholipid molecules would be restored to the ordered state (Figure 7B), and then, the shape of the liposome would tend to be spherical to maintain the lowest energy state, which is the result of the protective function of Slp crystalline arrays for reducing the influence of environmental stress factors. Hollmann et al97 extracted Slp from L. kefir JCM 5818 and L. brevis JCM 1059 and prepared Slp-coated liposomes. To evaluate the stabilities in the conditions simulating the gastrointestinal environment, researchers studied the effects of pancreatic extract and bile salts. The results indicated that after incubation of pancreatic extract for 60 and 120 minutes with bile salts, Slp-coated liposomes retained more carboxyfluorescein than control liposomes (Figure 7A and B). Therefore, Slp-coated liposomes possessed higher physicochemical stability than plain liposomes. Other stress factor evaluation assays, such as changes in pH value (Figure 7C),97 rapid variation in temperature (Figure 7D),101 and strong mechanical forces101 (Figure 7E and F), all proved the higher stability of Slp-coated liposomes relative to plain liposomes. It was also observed that the coating of Slp would lead to a delayed release.
Biological stability in the circulatory system is a core subject in liposomal delivery to the targeted sites. This biological stability mainly reflects the ability of the liposome to avoid the capture of RES. The RES, with key roles in the recognition and clearance of foreign particles, is a major constraint to nanoparticle-based drug delivery systems, such as liposome-based drug delivery system.102 Generally, most liposomes will be recognized by opsonins and complement components after intravenous administration and are captured by the mononuclear phagocyte system (MPS), leading to a clearance effect (Figure 7A).103 The most common strategy to avoid early clearance is surface modification with biocompatible polyethylene glycol, which yields a hydrated steric barrier that restricts interactions of liposomes with blood-borne components (eg, opsonins).104,105 This effect prolongs the blood circulating time of liposomes.106 Compared to PEGylated liposomes, Slp-coated liposomes likely possess a better capability to avoid the RES, which is mainly mediated by the synergistic effect of two mechanisms. On one hand, the existence of a protein corona107 (a complex biomolecular layer) induces biological interactions of liposomes with their surroundings, while the Slp lattices serve as a “protective suit” that can block biological interactions, avoiding the recognition by opsonins,108 and achieving extended circulation (Figure 7β). On the other hand, Slp coating decreases membrane fluidity, which hinders lipid extraction by high-density lipoproteins, reducing the risks of liposome breakdown.106 These two mechanisms cooperate to prolong the blood circulating time and promote the escalation of in vivo biological stability, which facilitates the accumulation in tumor tissue via the enhanced permeability and retention effect. Collectively, Slp-coated liposomes display significantly higher physicochemical and in vivo biological stabilities than plain liposomes through extended circulation, which broaden their applications in tumor-targeting technology.
Adhesion
Specific tissue affinity has traditionally been a continuously explored topic for liposomes. With this property, site-specific drug delivery and targeted drug delivery can be achieved. In particular, if liposomes show specific adhesion to the gastrointestinal epithelial cell line, the drug bioavailability mediated by liposomal oral drug delivery systems will increase.
In recent years, increasing research has been conducted to prove that Slp mediates bacterial adhesion to host cells, especially the gastrointestinal epithelial cell line. Åvall-Jääskeläinen et al109 constructed a surface display cassette to obtain recombinant Lactococcus lactis NZ9000 (containing the SlpA gene of L. brevis ATCC8287), and an in vitro adhesion assay demonstrated that the recombinant L. lactis NZ9000 could adhere to the human intestinal epithelial cell line Intestinal 407; the affinity was significantly better than that of wild-type L. lactis NZ9000. Another study reported that adhesion of Lactobacillus acidophilus ATCC4356 to the human colorectal adenocarcinoma cell line HT-29 was reduced dramatically when Slp was removed.110
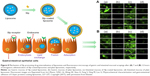
All of these studies support the view that Slp induces adhesion. However, most studies were carried out in vitro with different cell strains, and adhesion to the gastrointestinal mucosa in vivo, especially that by Slp-coated liposomes, has rarely been investigated in depth. Our previous study investigated the effects of Slp coating on the gastric and intestinal mucoadhesion of liposomes, in which the Slp-coated liposomes and control liposomes were all labeled by fluorescein isothiocyanate (FITC). After 7 and 12 hours of oral administration, the stomach and intestine were excised, and the mucosa was scraped off gently with a glass slide. Then, the mucosal scrapings were observed under a fluorescence microscope. The results (Figure 8A and B) showed that the fluorescence in gastric and intestinal mucosa at 7 and 12 hours after Slp-coated liposome oral administration was stronger than that of the control liposomes, which confirmed that Slp could also enhance the adhesion of liposomes to gastrointestinal mucosa.29
Hollmann et al98 found that Slp (from L. kefir)-coated liposomes showed no cytotoxic effects and excellent adhesion to the Caco-2 cell line. Simultaneously, an enhanced ability to transfer cargo molecules into Caco-2 cells from Slp-coated liposomes compared to control liposomes demonstrated that adhesion mediated by Slp was crucial for facilitating drug internalization, as shown by flow cytometry. The mechanism of drug internalization by Slp-coated liposomes is illustrated in Figure 8. Generally, liposomes release drugs by the interaction with target cell via adsorption, fusion, lipid exchange, and/or endocytosis.111 Slp-coated liposomes have longer retention times on epithelial cells and mucosa than plain liposomes, thereby promoting drug internalization. Currently, there is mounting evidence that the epithelial cell surface and extracellular matrix (ECM) express Slp receptors that can bind to Slp, inducing adhesion. For example, CbpA, an Slp of Lactobacillus salivarius REN, has been examined, and the results showed that enolase, its receptor on the surface of HT-29 cells, plays a crucial role in the adhesion of CbpA to HT-29 cells.112 Table 2 lists several Slps with their receptors and target cells or locations. From Table 2, we can see that fibronectin, collagen, and laminin are the most common receptors of Slp. They are primarily found in the ECM and on the surface of intestinal cells. Hence, Slp-coated liposomes can potentially be used in a variety of oral drug delivery systems, particularly for biomacromolecular drugs, such as vaccines.113 In addition to the ECM and intestinal epithelial cells, other tissues in the human body also express Slp receptors, but their function is different. For example, human macrophage cells and bone marrow-derived dendritic cells can express TLR2 and TLR4, which are also Slp receptors. However, interactions of Slp with TLR2 and TLR4 will trigger the expression of proinflammatory factors, thereby stimulating the innate immune system and T helper cell responses rather than inducing adhesion.114,115 The extreme gastrointestinal environment and poor permeability limit the development of biomacromolecule oral delivery systems, while the above Slp-coated liposomes show good physicochemical and gastrointestinal stabilities. Meanwhile, Slp-coated liposomes can significantly facilitate drug internalization. Therefore, Slp-coated liposomes could be a promising tool for the oral delivery of biomacromolecular drugs.
Immobilization of biomacromolecules
Currently, liposomes are typically used in targeted drug delivery systems. Surface modification of liposomes can promote their locations in targeting tissues. In particular, modification by biomacromolecules such as antibodies is very important for targeted drug delivery systems. Slp constructs a crystalline array of self-assembling proteins on the bacterial cell surface layer, and Slp was shown to immobilize biomacromolecules. To functionalize liposomes, researchers have used various approaches to immobilizing biomacromolecules by Slp. Three methods for immobilizing biomacromolecules by Slp have been reported.
The first method is genetic recombination technology (protein fusion technology); the genes encoding Slp and functional domains of biomacromolecules are all ligated into plasmids and heterologously expressed in Escherichia coli. Through this method, Slfp will be obtained. Slfp retains the capability of the Slp moiety to self-assemble on the interface and possesses functional domains of biomacromolecules that exert key effects. For example, Tschiggerl et al130 constructed a recombinant pET28a(+) plasmid that contains the gene sequence encoding SbpA31-1068 from Bacillus sphaericus CCM2177 and a PCR product encoding laminarinase A (LamA) from Pyrococcus furiosus. Then, the SbpA/LamA fusion proteins were overexpressed in E. coli. The fusion protein could self-assemble on glass slides, silicon wafers, and various types of membranes, allowing the orientated and dense surface display of LamA. Catalytic function assays demonstrated that LamA immobilized by SbpA in a periodic and oriented fashion could catalyze twice the glucose release from the laminarin polysaccharide substrate compared to randomly immobilized LamA.
The second method involves biomacromolecule immobilization by Slfp, which contains specific linking domains. First, Slp fuses with the specific linking domains via gene recombination technology. Then, the biomacromolecules interact with the specific linking domains by covalent bonds or non-covalent bonds for the purpose of immobilization. Khang et al131 designed a chimeric protein (EGFP/SlpA/ZZ) composed of EGFP, SlpA from L. brevis KCTC3102, and two copies of Fc-binding Z-domain. Then, rabbit anti-mouse IgG conjugated with peroxidase was mixed with EGFP/SlpA/ZZ, and the mixture was added to HT-29 cells (human epithelial cells). Due to the weak adhesion of IgG to HT-29 cells, the Slp-mediated adhesion of IgG to HT-29 cells can reflect the level of IgG bound to EGFP/SlpA/ZZ. The results indicated that the mixtures (EGFP/SlpA/ZZ and IgG) showed significantly higher adhesion of IgG to HT-29 than that of IgG alone, which proved that IgG was sufficiently immobilized by EGFP/SlpA/ZZ.
Additionally, some biomacromolecules are immobilized by Slp via direct interactions or crosslinkers (the third method). An interesting study132 reported that Slp as an immobilization matrix for aptamers could be applied in biosensors. Thrombin aptamers (ThromApt-SH) and ofloxacin aptamers (OflApt-NH2) were immobilized on the surface of Slp lattices via crosslinkers such as p-maleimidophenyl isocyanate, 1-ethyl-3-(3-dimethylamino-propyl) carbodiimide, and sulfosuccinimidyl-4-(N-maleimidomethyl) cyclohexane-1-carboxylate. Laser-induced fluorescence spectroscopy, IAsys (resonant mirror sensor), and quartz crystal microbalance with dissipation monitoring (QCM-D) analyses proved that aptamers immobilized on solid supports by Slp showed good recognition for thrombin and ofloxacin and could thus have broad applications in biosensor fields. Table 3 lists some biomacromolecules immobilized by Slp and Slfp. Furthermore, the immobilization function of Slp can also be utilized in liposome-based drug delivery systems.
  | Table 3 Summary of biomacromolecules immobilized by Slp and Slfp |
As mentioned above, Slp can self-assemble on the surface of liposomes and immobilize biomacromolecules; that is, Slp promotes the surface engineering of liposomes. For example, EGFP was fused with SbpA; afterward, rSbpA-EGFP also self-assembled on the surface of liposomes (Figure 9A and A1), which displayed green fluorescence (Figure 9A2) when observed under a fluorescence microscope. Hence, the rSbpA-EGFP-coated liposomes can be used to monitor the distribution of liposomal drug delivery systems in vivo.111 Furthermore, emulsomes, a liposome-like supramolecular nanostructure, could be modified by IgG with the help of Slp. Ücisik et al137 developed rSbpA-GG-coated Curcuemulsomes (Figure 9B), in which rSbpA-GG was an Slfp containing SbpA and two protein G domains (IgG-binding domains). ELISAs verified that the fusion of protein G domains with SbpA preserved their IgG-binding feature. Meanwhile, a recrystallization experiment demonstrated that rSbpA-GG could form a square lattice symmetry on the outermost region of the emulsomes, which indicated that rSbpA-GG still possessed the capability to self-assemble on the phase interface. Ultimately, immune colloidal gold technique was employed to confirm the IgG-binding feature of rSbpA-GG-coated emulsomes. The results verified that rSbpA-GG-coated emulsomes (Figure 9B1) exhibited a high affinity for IgG compared with rSbpA (without protein G domains)-coated emulsomes (Figure 9B3). Furthermore, Figure 9B2 shows that p4 symmetry was established.
  | Figure 9 Schematic drawing and TEM images of (A, A1, and A2) rSbpA-EGFP-coated liposomes, (B, B1, B2, and B3) IgG-rSbpA-GG-coated emulsomes, (C and C1) ferritin-rSlp-streptavidin fusion protein-coated liposomes, and (D and D1) ferritin-streptavidin-biotin bridge-Slp-coated liposomes. |
Another typical method for Slp-coated liposome immobilization of biomacromolecules is the use of streptavidin-biotin system. There are two mechanisms involved in the streptavidin-biotin system. Figure 9C and C1 illustrates one mechanism with ferritin as an example. Firstly, Slp fuses with streptavidin to form an rSlp-streptavidin fusion protein, which reassembles on the surface of the liposome. Then, biotinylated ferritin can bind to the streptavidin domain.67 While the other mechanism does not involve protein fusion technology, the “streptavidin-biotin bridge” formed by chemical interactions of streptavidin with biotin binds to Slp via the biotin-terminal region, thus functionalizing the Slp coated-liposomes. Afterward ferritin can bind to the streptavidin-terminal region, achieving the aim of immobilization of the ferritin on the liposomal surface (Figure 9D and D1).138 In general, Slp coating functionalizes the surface properties of liposomes, providing practical value for targeted drug delivery and drug tracing. However, research on Slp-coated liposome immobilization of biomacromolecules remains limited. Hence, this research still has profound academic significance and broad prospects.
Drug delivery
The most promising application of Slp-coated liposomes is drug delivery. Slp-coated liposomes possess strong physicochemical stability that protects their cargo molecules against the mimetic gastrointestinal environment (rapid variations in pH and temperature, mechanical force, pancreatic extract and bile salts). In addition, the gastrointestinal adhesive ability of Slp-coated liposomes could significantly increase the drug’s gastrointestinal retention time, thereby potentially promoting drug internalization. To confirm this point, Hollmann et al98 constructed an Slp-coated liposome carrying calcein and then evaluated whether the Slp-coated liposomes promoted drug internalization in human colon adenocarcinoma Caco-2 cells by flow cytometry. As expected, the Caco-2 cells with calcein fluorescence incubated with control liposomes for 30 minutes at 37°C (Figure 10A) were significantly lower than those incubated with the calcein liposomes coated by Slp extracted from L. kefir JCM 5818 (Figure 10B). Calcein dequenching analysis (Figure 10C) demonstrated that Slp-coated liposomes could transfer approximately 40% more calcein molecules than the control ones. Interestingly, when the systemic temperature decreased to 4°C, the distribution of Caco-2 cells with calcein fluorescence (Figure 10D and E) and calcein dequenching (Figure 10C) decreased dramatically for both liposomes, and these data did not show significant differences between Slp-coated liposomes and control liposomes. A previous study indicated that most endocytosis pathways of cells were inhibited at 4°C. Thus, the researchers theorized that the drug internalization mechanism of Slp-coated liposomes is endocytosis by Caco-2 cells. Furthermore, the results of MTT assays showed that Slp-coated liposomes did not reduce the viability of Caco-2 cells at any of the concentrations tested (Figure 10F).
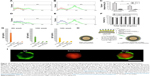  | Figure 10 Evidence of Slp-coated liposomes promoting drug internalization and surface engineering of liposomes and as carriers of vaccines for oral administration. |
Hollmann et al showed that that Slp-coated liposomes indeed display promising potential as drug carriers for oral administration, especially oral delivery of biomacromolecular drugs.98 Currently, the main administration route of biomacromolecular drugs (eg, vaccines and insulin) is injection,140 which causes pain in patients, especially diabetics and children. Hence, a newly developed oral delivery system could eliminate the critical delivery bottleneck of biomacromolecular drugs. Therefore, appropriate gastrointestinal environment and drug internalization are the key factors that should be considered for oral delivery systems of biomacromolecular drugs.141 Enzyme systems and rapid variation of pH in the gastrointestinal tract are lethal for biomacromolecular drugs; meanwhile, it is extremely difficult for biomacromolecular drugs to be absorbed by gastrointestinal epithelial cells compared with plain chemical drugs due to their much higher molecular weight.142 In fact, Sleytr et al had already proposed that Slp-coated liposomes could be potential vaccine carriers in 1999.30 In 2007, Hollmann et al analyzed the feasibility of Slp-coated liposomes as vaccine carriers by physicochemical stability analysis, and concluded that their physicochemical characterization meets the qualification for vaccine carriers.97 Recently, our research group constructed an Slp-coated liposome-entrapped hepatitis B vaccine (HepB) to explore the in vivo feasibility as a vaccine carrier.139 The IgG concentrations in mouse serum were determined by ELISA at the eighth week, 10th week, and 12th week after subcutaneous injection of HepB (Sc-HepB) or intragastric administration of HepB solution (ig-HepB), HepB liposomes (ig-LP-HepB), and HepB liposomes coated by Slp (ig-Slp-LP-HepB). The key results are presented in Figure 10G. The IgG level of the ig-HepB group at the eighth week was 4.00±2.78 ng/mL, indicating this group showed almost no effect, while the IgG levels of the ig-LP-HepB and ig-Slp-LP-HepB groups were 9.44±2.42 and 21.96±7.55 ng/mL, respectively, which strongly indicated that both unmodified liposomes and Slp-coated liposomes could improve the oral delivery of the vaccine. Slp-coated liposomes exhibited a better biological efficiency than unmodified liposomes, but much lower than that of the Sc-HepB group (IgG level 529.13±175.91 ng/mL). However, unlike the Sc-HepB group, the IgG levels of the other three oral administration groups all increased at the 10th and 12th week. The IgG levels of the ig-Slp-LP-HepB group at the 10th and 12th week increased to 41.32±7.50 and 55.24±17.50 ng/mL, respectively. However, it is interesting to note that the IgG levels of the three oral administration groups all showed an upward trend over time, while the IgG levels of the Sc-HepB group showed a downward trend. We inferred that the differences in pharmacokinetic processes between subcutaneous injection and oral administration led to this phenomenon. These findings also suggested that the interval dose and time should be fully considered when developing an oral delivery system for a vaccine. These results showed that the Slp-coated liposomes were able to improve the oral delivery efficiency of vaccines significantly. However, the efficiency was still not strong enough for the clinical application in oral administration, which may be the main cause that limits the publication of the related literature.
As noted in the previous section, immobilization of biomacromolecules is another unique property of Slp-coated liposomes. This feature will drive the functionalization of liposomes to achieve targeted drug delivery. In 2015, Ücisik et al published a research paper reporting that Slp coating as a matrix helped the emulsome-entrapped antitumor drug curcumin (Curcuemulsome) to immobilize and target human antibody IgG.137 This study involved protein fusion technology. Two protein G domains possessing specific IgG affinity were fused with SbpA. The Slfp, rSbpA-GG, could reassemble on the surface of Curcuemulsomes, and the protein G domains could regularly recognize and immobilize IgG (Figure 10H). ELISA indicated that IgG was only bound to rSbpA-GG-coated Curcuemulsomes rather than SbpA-coated or rSbpA-ZZ-coated Curcuemulsomes, which demonstrated that protein G domains fused with Slp preserve their inherent IgG-binding features. Confocal microscopy (Figure 10I) clearly confirmed that FITC-labeled IgG was coated on the surface of rSbpA-GG-coated emulsomes loaded with Sudan β. Emulsomes are a vesicle-based delivery system for the delivery of hydrophobic drugs. As shown in Figure 10H, the emulsome comprises a solid emulsion core surrounded by phospholipid layers. Hence, the structure of the emulsome is like a liposome-entrapped solid emulsion. Thus, the surface characteristics of emulsomes are the same as those of liposomes. The research above also revealed the functionalization potential of Slp-coated liposomes. However, Ücisik et al unfortunately did not further evaluate the in vivo targeted drug delivery efficiency or the cancer therapeutic efficiency of rSbpA-GG-coated Curcuemulsomes.137
As we continue to explore design strategies, our progress in oral delivery of biomacromolecular drugs and targeted drug delivery by Slp-coated liposomes will continue. We also believe that the biomedical engineering uses of Slp combined with the favorable characteristics of liposomes will drive a series of biomedical applications.
Biomedical applications
As an important biomaterial, Slp has already been researched in the biomedical field. A regular crystalline array of Slp provides a firm platform for biomacromolecules, enabling it to be a novel biocatalyst,143,144 a potential agent for biomimetic therapy,145 and a biosensor.132 Back to the subject of this review, Slp-coated liposomes not only exhibit the unique features of Slp but also possess the favorable nanoproperties of liposomes with good biocompatibility and biodegradability. Increasing evidence has confirmed that Slp-coated liposomes display great potential in the biomedical engineering field in addition to drug delivery. In 2004, Ilk et al first reported that uptake of an S-layer-enhanced green fluorescent fusion protein-coated liposome into eukaryotic cells could be visualized by confocal laser scanning microscopy.111 EGFP was labeled on the surface of liposomes by Slp fusion technology. Traditional labeling modes of fluorescent molecules for nanomedicine involve physical adsorption and chemical conjugation.146 However, physical adsorption is a weak interaction that easily causes the shedding of fluorescent molecules. The potentially toxic agents used in the process of chemical conjugation probably diffuse through the lipid bilayer into the interior of the liposome.111,147 Fortunately, Slp fusion technology can avoid the abovementioned issues. In this work, the researchers initially constructed the S-layer-enhanced green fluorescent fusion protein rSbpA31-1068/EGFP and recombinant EGFP (rEGFP) as a control. Additionally, both rSbpA31-1068/EGFP and rEGFP showed nearly the same excitation and emission spectra (Figure 11A), which suggested that the Slp fusion technology did not affect the fluorescence intensity of EGFP. Subsequently, rSbpA31-1068/EGFP maintained the reassembly feature and self-assembled on the surface of liposomes (Figure 11B), and an intense green fluorescence was observed on the surface of rSbpA31-1068/EGFP-coated liposomes via fluorescence microscopy (Figure 11C). For visually monitoring the interactions with eukaryotic cells, the rSbpA31-1068/EGFP-coated liposomes were incubated with HeLa cells, and then, three-dimensional confocal laser scanning microscopy was used to view the kinetic characteristics of rSbpA31-1068/EGFP-coated liposomes. As shown in Figure 11E–G, a single arrow indicates the ongoing interactions between rSbpA31-1068/EGFP-coated liposomes and the HeLa cell membrane, double arrows indicate that the internalization process of liposomes was completed, and the dark arrowheads indicate that liposomes were engulfed by endosomes or located in the cytosol. Overall, confocal images clearly show that most of the liposomes were internalized after 2 hours of incubation mainly via endocytosis, which demonstrated that the rSbpA31-1068/EGFP coating could be successfully used to monitor the behavior of liposomes. This functional liposome has good application potential in the continuous development of liposomes and in vivo imaging technology of specific cells.
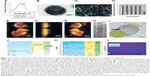  | Figure 11 Biomedical applications of Slp-coated liposomes in imaging technology, as biocatalysts, and in preparation of biomimetic membranes. |
Due to its abundant carboxylic and amino groups and its ability to form regular crystalline arrays, Slp has usually been employed to construct a biocatalytic surface for the immobilization of enzymes.143,144 Immobilized enzymes have significant economic value in the pharmaceutical industry. The immobilization of enzymes can facilitate the separation and reuse of enzymes, thus reducing the cost of industrial production.150,151 Using a combined nanobiotechnological approach, Schäffer et al reported an Slp-coated liposome as a platform for the immobilization of an enzyme constructed as a novel biocatalyst.148 First, the researchers optimized the immobilizing method of glucose-1-phosphate thymidylyltransferase RmlA by SgsE (Slp from Geobacillus stearothermophilus NRS 2004/3a) and obtained rSgsE/RmlA. The catalytic activity of rSgsE/RmlA reached 99.7% compared to the activity of RmlA alone. In addition, rSgsE/RmlA showed better storage stability than RmlA alone. rSgsE/RmlA showed preserved activity at 14 weeks, while RmlA alone lost 70% of its activity after 4 weeks (Figure 11D). Interestingly, rSgsE/RmlA could self-assemble into a nanotube structure as described in Figure 1. However, this rSgsE/RmlA nanotube biocatalyst only possessed 3% of the activity of RmlA. This discrepancy was due to the formation of double-layered Slp on the surface of the nanotube. These double-layer structures with a face-to-face inward orientation of the catalytic epitopes bury the catalytic domains in the intralayer of the nanotube, which dramatically inhibits the efficiency of the reaction (Figure 11H). To overcome this limitation, liposomes were adopted to support rSgsE/RmlA. Compared with the rSgsE/RmlA nanotube biocatalyst, the rSgsE/RmlA-coated liposome biocatalyst showed a significantly higher activity (65.4% of the activity of RmlA). This enhancement was mainly attributed to the full surface display of catalytic epitopes (Figure 11I). For recycling and reuse, the liposomal biocatalyst could be separated by centrifugation, and 61.0% of the original activity was still detectable after one cycle. From this example, we can see the promising application potential and economic value of Slp-coated liposomes in the immobilization of enzymes for constructing novel biocatalysts.
In addition to these biomedical applications, the interactions between Slp and liposomes are typically employed to prepare biomimetic-supported lipid membranes for studying the characteristics and functions of transmembranes and membrane-bound proteins. Furthermore, biomimetic-supported lipid membranes can be used to investigate biomembrane-mediated interactions and cell signal transduction.152,153 Therefore, the construction of biomimetic membranes is crucial. Schrems et al adopted the interactions between liposomes and Slp to prepare biomimetic-supported lipid membranes.149 The preparation process was divided into three steps: in the first step, Slp was recrystallized on the surface of solid supports; in the second step, β-diketone ligand-modified liposomes were anchored to the Slp-coated solid supports via electrostatic forces (as mentioned in the “Interactions between Slp and liposomes” section); and the last step was the addition of europium (III) that caused the formation of the β-diketone ligand-europium (III) complex between adjacent liposomes, thereby facilitating liposomal fusion and the formation of planar lipid membranes. Surface plasmon resonance spectroscopy (Figure 11J) and QCM-D (Figure 11K) were used to characterize this process. Subsequently, the researchers observed that the phospholipid molecules fully covered the surface of solid supports (Figure 11L), which confirmed the successful formation of planar biomimetic membranes. Damiati et al used this method to prepare biomimetic membranes and then investigated the electrical and structural properties of gramicidin (a membrane-active peptide) and α-hemolysin (a transmembrane protein).154
Thus, Slp-coated liposomes have shown promising potential in some biomedical engineering fields, including imaging technology, catalytic chemistry, and biomedical investigation. Although some encouraging progress has been made, developing appropriate biomedical applications of Slp-coated liposomes still requires further research.
Challenges and perspectives
The present review provides an overview of the development of Slp and Slp-coated liposomes. The latter has emerged as an interesting candidate for drug delivery and biomedical uses because of the distinguishing features of Slp-coated liposomes, including favorable physicochemical and biological stabilities, adhesion to specific cells or tissues, and immobilization of biomacromolecules for multifunctionalization of liposomes. However, there are some difficulties in the development of Slp-coated liposomes. Critical challenges that still remain and need to be explored are listed below, which will serve as a roadmap to further elucidate the future beneficial effects of Slp-coated liposomes.
Although Slp-coated liposomes are expected to be suitable candidates for the oral delivery of vaccines, the enhancement of oral delivery of vaccines is not satisfactory. To our knowledge, the oral delivery efficiency of biomacromolecular drugs mostly relies on the gastrointestinal retention time, the uptake by gastrointestinal epithelial cells, and the ability to defend against the extreme gastrointestinal environment. Slp-coated liposomes have shown improvements in the stability and retention time in the gastrointestinal tract. However, absorption by the gastrointestinal tract is not sufficient. Therefore, strategies to improve the penetration of Slp-coated liposomes into epithelial cells should be developed in the future.
As Slp is a virulence factor, the toxicity and side effects of Slp in the circulation system and organs have to be identified. Research by Taverniti et al found that SlpA triggered the expression of the proinflammatory factors TNF-α and COX-2 in the human macrophage cell line U937 by recognition through TLR2, stimulating the innate immune system.115 Slp isolated from Clostridium difficile also induced maturation of bone marrow-derived dendritic cells by expression of MHC class 2, CD40, CD80, and CD86 and production of IL-10, IL-12, and TNF-α via recognition of TLR4, which subsequently generated a T helper cell response.114 These results suggested that Slp could activate innate immunity, which is beneficial for vaccine delivery but should be considered carefully for delivery of other drugs. Further elucidation of the toxicity and side effects of different Slp molecules will lead to an enhanced design of Slp-coated liposome-based drug delivery systems for specific disease therapy.
Furthermore, as described in the “Interactions between Slp and liposomes” section, the interaction of Slp with liposomes is mainly attributed to electrostatic forces. Thus, the usage of positively charged surfactants is crucial for the effective coating of Slp. However, positively charged surfactants display cytotoxicity in our bodies. Positively charged surfactants can disrupt the cell membrane, thereby causing cell death. Hence, the usage of positively charged surfactants in various Slp-coated liposomes should be investigated in depth, and not only should the reasonable coating efficiency be considered, but the side effects or toxicity of the indispensable components in Slp-coated liposomes should also be taken into account.
Compared with those of other supports (such as metal nanoparticles and silicon supports), the rigidity and physicochemical stabilities of liposomes are slightly weak. This disadvantage sometimes limits the use of liposomes as the support for Slp in various biomedical applications. Nevertheless, liposomes composed of phospholipid molecules have a typical biomimetic cell structure with good biocompatibility and biodegradability in vivo, which is not available in the case of other supports or nanoscale materials. Hence, we believe the Slp-coated liposomes have major advantages as in vivo biosensors or biocatalysts as well as diagnostic reagents.
For industrial applications, a conventional liposome is usually produced by an aseptic technique due to its poor resistance to high temperature and pressure. The more complicated structure of the Slp-coated liposome will cause major difficulties in industrial production, such as high technical barriers and high industrial cost. This issue is also an obstacle to the widespread application of Slp-coated liposomes.
Although there are several challenges, given the considerable potential applications, it is worthwhile to devote more efforts to this promising multifunctional platform for drug delivery and biomedical applications. In recent years, Slp has been gradually employed to construct novel biosensors155 for cancer diagnosis,156 detection of chemical ions,157 and nanoelectronic applications.158 We believe this is indeed a signal of the future biomedical development of Slp-coated liposomes.
With the extensive research on Slp, particularly the in-depth depiction of the mechanism of bacterial invasion of host epithelial cells, Slp has already been investigated as an antimicrobial agent for bacteria-induced gastrointestinal disease therapy. Evidence has indicated that Slp can inhibit the adhesion of the enteric pathogen E. coli to human intestinal epithelial tissues145 by competing with pathogens for Slp receptors.159 Interestingly, Slp (from L. acidophilus NCFM) inhibited mitochondrial membrane potential reduction and enhanced Ca2+ levels in HT-29 cells, significantly downregulating pathogen-induced activation of caspase-3 and caspase-9, which prevented pathogen-induced apoptosis of HT-29 cells.160 Furthermore, SlpA extracted from Lactobacillus helveticus MIMLh5 displayed anti-inflammatory effects on the intestinal epithelial Caco-2 cell line by inhibiting the activation of the NF-κB signaling pathway.115 Combined with biomimetic technology and nanotechnology, Slp-coated liposomes with biomimetic structures simulating the morphology of bacterial cells may exhibit a better competitive inhibitory effect than Slp alone. This difference is because the absorption of Slp on the surface of liposomes can significantly improve the local density of Slp, which supports improved competition for binding sites with bacterial cells. Based on the anti-apoptotic and anti-inflammatory effects of Slp, we hypothesized that Slp-coated liposomes carrying other antimicrobial drugs may exert a synergetic effect in intestinal disease therapy. We hope that this review will drive more scientists to join the journey of developing Slp-coated liposomes.
Acknowledgments
This work was supported by Public Welfare Technology Research Project of Zhejiang Province (LGF18H300003), Huahai Pharmaceutical Innovative Program (SROP), and National Undergraduate Training Program for Innovation and Entrepreneurship (2017102).
Disclosure
The authors report no conflicts of interest in this work.
References
Albers SV, Meyer BH. The archaeal cell envelope. Nat Rev Microbiol. 2011;9(6):414–426. | ||
Sára M, Sleytr UB. Crystalline bacterial cell surface layers (S-layers): from cell structure to biomimetics. Prog Biophys Mol Biol. 1996;65(1–2):83–111. | ||
Pum D, Toca-Herrera JL, Sleytr UB, Dietmar P, Luis THJ, Ub S. S-layer protein self-assembly. Int J Mol Sci. 2013;14(2):2484–2501. | ||
Pum D, Sleytr UB. Reassembly of S-layer proteins. Nanotechnology. 2014;25(31):312001. | ||
Teixeira LM, Strickland A, Mark SS, Bergkvist M, Sierra-Sastre Y, Batt CA. Entropically driven self-assembly of Lysinibacillus sphaericus S-layer proteins analyzed under various environmental conditions. Macromol Biosci. 2010;10(2):147–155. | ||
Sleytr UB, Schuster B, Egelseer EM, Pum D. S-layers: principles and applications. FEMS Microbiol Rev. 2014;38(5):823–864. | ||
Yang J, Bahreman A, Daudey G, Bussmann J, Olsthoorn RC, Kros A. Drug delivery via cell membrane fusion using lipopeptide modified liposomes. ACS Cent Sci. 2016;2(9):621–630. | ||
Matos ALL, Pereira G, Cabral Filho PE, Santos BS, Fontes A. Delivery of cationic quantum dots using fusogenic liposomes in living cells. J Photochem Photobiol B. 2017;171:43–49. | ||
Schiffelers RM, Storm G. Liposomal nanomedicines as anticancer therapeutics: beyond targeting tumor cells. Int J Pharm. 2008;364(2):258–264. | ||
Jia X, Han Q, Wang Z, et al. Targeting peptide functionalized liposomes towards aminopeptidase N for precise tumor diagnosis and therapy. Biomater Sci. 2017;5(3):417–421. | ||
Elechalawar CK, Sridharan K, Pal A, et al. Cationic folate-mediated liposomal delivery of bis-arylidene oxindole induces efficient melanoma tumor regression. Biomater Sci. 2017;5(9):1898–1909. | ||
Oku N, Namba Y. Long-circulating liposomes. Crit Rev Ther Drug Carrier Syst. 1994;11(4):231–270. | ||
Lopes SCDA, Guiberti CDS, Rocha TGR, Ferreira DDS, Leite EA, Oliveira MC. Liposomes as Carriers of Anticancer Drugs. 2013. Available from: https://www.intechopen.com/books/cancer-treatment-conventional-and-innovative-approaches/liposomes-as-carriers-of-anticancer-drugs. Accessed October 6, 2018. | ||
Pentak D. Physicochemical properties of liposomes as potential anticancer drugs carriers. Interaction of etoposide and cytarabine with the membrane: spectroscopic studies. Spectrochim Acta A Mol Biomol Spectrosc. 2014;122:451–460. | ||
Siler-Marinkovic S, Mojovic L, Davinic V, Bugarski B. Liposomes as carriers of antimicrobial drugs. Drug Dev Ind Pharm. 1997;23(5):483–488. | ||
Crommelin DJA, Eling WMC, Steerenberg PA, et al. Liposomes and immunoliposomes for controlled release or site specific delivery of anti-parasitic drugs and cytostatics. J Control Release. 1991;16(1–2):147–154. | ||
Schäfer H, Schmidt W, Berger H, Bergfeld J. Pharmacokinetics of gonadotropin-releasing hormone and stimulation of luteinizing hormone secretion after single dose administration of GnRH incorporated into liposomes. Pharmazie. 1987;42(10):689–693. | ||
Fotoran WL, Colhone MC, Ciancaglini P, Stabeli RG, Wunderlich G. Merozoite-Protein Loaded Liposomes Protect against Challenge in Two Murine Models of Plasmodium Infection. ACS Biomater Sci Eng. 2016;2(12):2276–2286. | ||
Gong KJ, Shi AM, Liu HZ, et al. Preparation of nanoliposome loaded with peanut peptide fraction: stability and bioavailability. Food Funct. 2016;7(4):2034–2042. | ||
Xia Y, Tian J, Chen X. Effect of surface properties on liposomal siRNA delivery. Biomaterials. 2016;79:56–68. | ||
Cui S, Wang B, Zhao Y, et al. Transmembrane routes of cationic liposome-mediated gene delivery using human throat epidermis cancer cells. Biotechnol Lett. 2014;36(1):1–7. | ||
Yoshizaki Y, Yuba E, Sakaguchi N, Koiwai K, Harada A, Kono K. pH-sensitive polymer-modified liposome-based immunity-inducing system: effects of inclusion of cationic lipid and CpG-DNA. Biomaterials. 2017;141:272–283. | ||
Majzoub RN, Chan CL, Ewert KK, et al. Uptake and transfection efficiency of PEGylated cationic liposome-DNA complexes with and without RGD-tagging. Biomaterials. 2014;35(18):4996–5005. | ||
Qiao C, Liu J, Yang J, et al. Enhanced non-inflammasome mediated immune responses by mannosylated zwitterionic-based cationic liposomes for HIV DNA vaccines. Biomaterials. 2016;85:1–17. | ||
Yang ZZ, Li JQ, Wang ZZ, Dong DW, Qi XR. Tumor-targeting dual peptides-modified cationic liposomes for delivery of siRNA and docetaxel to gliomas. Biomaterials. 2014;35(19):5226–5239. | ||
Li L, Hou J, Liu X, et al. Nucleolin-targeting liposomes guided by aptamer AS1411 for the delivery of siRNA for the treatment of malignant melanomas. Biomaterials. 2014;35(12):3840–3850. | ||
Petros RA, Desimone JM. Strategies in the design of nanoparticles for therapeutic applications. Nat Rev Drug Discov. 2010;9(8):615–627. | ||
Shehata T, Ogawara K, Higaki K, Kimura T. Prolongation of residence time of liposome by surface-modification with mixture of hydrophilic polymers. Int J Pharm. 2008;359(1–2):272–279. | ||
Wang W, Shao A, Feng S, Ding M, Luo G. Physicochemical characterization and gastrointestinal adhesion of S-layer proteins-coating liposomes. Int J Pharm. 2017;529(1–2):227–237. | ||
Sleytr UB, Messner P, Pum D, Sára M. Crystalline bacterial cell surface layers (S layers): from supramolecular cell structure to biomimetics and nanotechnology. Angew Chem Int Ed. 1999;38(8):1034–1054. | ||
Bahl H, Scholz H, Bayan N, et al. Molecular biology of S-layers. FEMS Microbiol Rev. 1997;20(1–2):47–98. | ||
Claus H, Akça E, Debaerdemaeker T, et al. Molecular organization of selected prokaryotic S-layer proteins. Can J Microbiol. 2005;51(9):731–743. | ||
Chen X, Xu J, Shuai J, Chen J, Zhang Z, Fang W. The S-layer proteins of Lactobacillus crispatus strain ZJ001 is responsible for competitive exclusion against Escherichia coli O157:H7 and Salmonella typhimurium. Int J Food Microbiol. 2007;115(3):307–312. | ||
Sleytr UB, Beveridge TJ. Bacterial S-layers. Trends Microbiol. 1999;7(6):253–260. | ||
Sára M, Sleytr UB, Proteins S-L. S-layer proteins. J Bacteriol. 2000;182(4):859–868. | ||
Boot HJ, Kolen CP, van Noort JM, Pouwels PH. S-layer protein of Lactobacillus acidophilus ATCC 4356: purification, expression in Escherichia coli, and nucleotide sequence of the corresponding gene. J Bacteriol. 1993;175(19):6089–6096. | ||
Sleytr UB, Huber C, Ilk N, Pum D, Schuster B, Egelseer EM. S-layers as a tool kit for nanobiotechnological applications. FEMS Microbiol Lett. 2007;267(2):131–144. | ||
Liu J. Structure and Dynamics of Bacterial S-layer Proteins [Ph.D. thesis]. Hamburg: University of Hamburg; 2014. | ||
Kern J, Wilton R, Zhang R, Binkowski TA, Joachimiak A, Schneewind O. Structure of surface layer homology (SLH) domains from Bacillus anthracis surface array protein. J Biol Chem. 2011;286(29):26042–26049. | ||
Engelhardt H. Are S-layers exoskeletons? The basic function of protein surface layers revisited. J Struct Biol. 2007;160(2):115–124. | ||
Horejs C, Pum D, Sleytr UB, Tscheliessnig R. Structure prediction of an S-layer protein by the mean force method. J Chem Phys. 2008;128(6):065106. | ||
Liu J, Falke S, Drobot B, et al. Analysis of self-assembly of S-layer protein slp-B53 from Lysinibacillus sphaericus. Eur Biophys J. 2017;46(1):77–89. | ||
Sleytr U, Pum D, Egelseer E, Ilk N, Schuster B. Handbook of Biofunctional Surfaces. S-layer proteins. 2013:507–568. | ||
Avall-Jääskeläinen S, Palva A. Lactobacillus surface layers and their applications. FEMS Microbiol Rev. 2005;29(3):511–529. | ||
Sleytr UB, Egelseer EM, Ilk N, Pum D, Schuster B. S-Layers as a basic building block in a molecular construction kit. FEBS Journal. 2007;274(2):323–334. | ||
Sleytr UB, Sára M, Pum D, Schuster B. Characterization and use of crystalline bacterial cell surface layers. Prog Surf Sci. 2001;68(7–8):231–278. | ||
Sára M, Sleytr UB. Relevance of charged groups for the integrity of the S-layer from Bacillus coagulans E38–66 and for molecular interactions. J Bacteriol. 1993;175(8):2248–2254. | ||
de La Fuente-Núñez C, Mertens J, Smit J, Hancock RE. The bacterial surface layer provides protection against antimicrobial peptides. Appl Environ Microbiol. 2012;78(15):5452–5456. | ||
Messner P, Schäffer C, Emem E, Ub S, Sleytr UB. Occurrence, Structure, Chemistry, Genetics, Morphogenesis, and Functions of S-Layers. Berlin: Springer; 2010. | ||
Torchilin VP. Recent advances with liposomes as pharmaceutical carriers. Nat Rev Drug Discov. 2005;4(2):145–160. | ||
Dass CR. Drug Delivery in Cancer Using Liposomes. Clifton, NJ: Humana Press; 2008. | ||
Crommelin DJ, Storm G. Liposomes: from the bench to the bed. J Liposome Res. 2003;13(1):33–36. | ||
Huang SL. Liposomes in ultrasonic drug and gene delivery. Adv Drug Deliv Rev. 2008;60(10):1167–1176. | ||
Allen TM, Cullis PR. Liposomal drug delivery systems: from concept to clinical applications. Adv Drug Deliv Rev. 2013;65(1):36–48. | ||
Takeuchi H, Yamamoto H, Niwa T, Hino T, Kawashima Y. Mucoadhesion of polymer-coated liposomes to rat intestine in vitro. Chem Pharm Bull. 1994;42(9):1954–1956. | ||
Wang B, Zhang S, Cui S, et al. Chitosan enhanced gene delivery of cationic liposome via non-covalent conjugation. Biotechnol Lett. 2012;34(1):19–28. | ||
Liang X, Li X, Chang J, Duan Y, Li Z. Properties and evaluation of quaternized chitosan/lipid cation polymeric liposomes for cancer-targeted gene delivery. Langmuir. 2013;29(27):8683–8693. | ||
Colonna C, Conti B, Genta I, Alpar OH. Non-viral dried powders for respiratory gene delivery prepared by cationic and chitosan loaded liposomes. Int J Pharm. 2008;364(1):108–118. | ||
Shi X, Ma W, Sun C, Wu S. The aggregation behavior of collagen in aqueous solution and its property of stabilizing liposomes in vitro. Biomaterials. 2001;22(12):1627–1634. | ||
Kreuter J. Drug delivery to the central nervous system by polymeric nanoparticles: what do we know? Adv Drug Deliv Rev. 2014;71:2–14. | ||
Ruan S, Qin L, Xiao W, et al. Acid-responsive transferrin dissociation and GLUT mediated exocytosis for increased blood-brain barrier transcytosis and programmed glioma targeting delivery. Adv Funct Mater. 2018;28(30):1802227. | ||
Fan K, Jia X, Zhou M, et al. Ferritin nanocarrier traverses the blood brain barrier and kills glioma. ACS Nano. 2018;12(5):4105–4115. | ||
Kamata H, Yagisawa H, Takahashi S, Hirata H. Amphiphilic peptides enhance the efficiency of liposome-mediated DNA transfection. Nucleic Acids Res. 1994;22(3):536–537. | ||
Kichler A, Mechtler K, Behr JP, Wagner E. Influence of membrane-active peptides on lipospermine/DNA complex mediated gene transfer. Bioconjug Chem. 1997;8(2):213–221. | ||
Ou J, Geiger T, Ou Z, Ackerman AW, Oldham KT, Pritchard KA. AP-4F, antennapedia peptide linked to an amphipathic alpha helical peptide, increases the efficiency of Lipofectamine-mediated gene transfection in endothelial cells. Biochem Biophys Res Commun. 2003;305(3):605–610. | ||
Torchilin VP, Levchenko TS, Rammohan R, Volodina N, Papahadjopoulos-Sternberg B, D’Souza GGM. Cell transfection in vitro and in vivo with nontoxic Tat peptide-liposome-DNA complexes. Proc Natl Acad Sci. 2003;100(4):1972–1977. | ||
Moll D, Huber C, Schlegel B, Pum D, Sleytr UB, Sara M. S-layer-streptavidin fusion proteins as template for nanopatterned molecular arrays. Proc Natl Acad Sci. 2002;99(23):14646–14651. | ||
Ozpolat B, Sood AK, Lopez-Berestein G. Liposomal siRNA nanocarriers for cancer therapy. Adv Drug Deliv Rev. 2014;66:110–116. | ||
Shim G, Kim M-G, Park JY, Oh Y-K, Yk O. Application of cationic liposomes for delivery of nucleic acids. Asian J Pharm Sci. 2013;8(2):72–80. | ||
Küpcü S, Sára M, Sleytr UB. Liposomes coated with crystalline bacterial cell surface protein (S-layer) as immobilization structures for macromolecules. Biochim Biophys Acta Biomembr. 1995;1235(2):263–269. | ||
Györvary ES, Stein O, Pum D, Sleytr UB. Self-assembly and recrystallization of bacterial S-layer proteins at silicon supports imaged in real time by atomic force microscopy. J Microsc. 2003;212(Pt 3):300–306. | ||
Shin S-H, Chung S, Sanii B, Comolli LR, Bertozzi CR, de Yoreo JJ. Direct observation of kinetic traps associated with structural transformations leading to multiple pathways of S-layer assembly. Proc Natl Acad Sci. 2012;109(32):12968–12973. | ||
Toca-Herrera JL, Moreno-Flores S, Friedmann J, Pum D, Sleytr UB. Chemical and thermal denaturation of crystalline bacterial S-layer proteins: an atomic force microscopy study. Microsc Res Tech. 2004;65(4–5):226–234. | ||
Thornley MJ, Glauert AM, Sleytr UB. Structure and assembly of bacterial surface layers composed of regular arrays of subunits. Proc R Soc Biol Sci. 1974;268(891):147–153. | ||
Glaubert AM, Sleytr UB. Analysis of regular arrays of subunits on bacterial surfaces: evidence for a dynamic process of assembly. J Ultrastruct Res. 1975;50(1):103–116. | ||
Beveridge TJ. Bacterial S-layers. Current Opinion in Structural Biology. 1994;4(2):204–212. | ||
Đordić A, Egelseer EM, Tesarz M, Sleytr UB, Keller W, Pavkov-Keller T. Crystallization of domains involved in self-assembly of the S-layer protein SbsC. Acta Crystallogr F Struct Biol Cryst Commun. 2012;68(12):1511–1514. | ||
Waśko A, Polak-Berecka M, Kuzdraliński A, Skrzypek T. Variability of S-layer proteins in Lactobacillus helveticus strains. Anaerobe. 2014;25:53–60. | ||
Sleytr UB, Messner P, Pum D, Sára M. Crystalline Bacterial Cell Surface Proteins. Amsterdam: Elsevier Science; 1996. | ||
Tang J, Ebner A, Ilk N, et al. High-affinity tags fused to S-layer proteins probed by atomic force microscopy. Langmuir. 2008;24(4):1324–1329. | ||
López AE, Pum D, Sleytr UB, Toca-Herrera JL. Influence of surface chemistry and protein concentration on the adsorption rate and S-layer crystal formation. Phys Chem Chem Phys. 2011;13(25):11905–11913. | ||
Chung S, Shin S-H, Bertozzi CR, de Yoreo JJ. Self-catalyzed growth of S layers via an amorphous-to-crystalline transition limited by folding kinetics. Proc Natl Acad Sci U S A. 2010;107(38):16536–16541. | ||
de Sa Peixoto P, Roiland C, Thomas D, et al. Recrystallized S-layer protein of a probiotic Propionibacterium: structural and nanomechanical changes upon temperature or pH shifts probed by solid-state NMR and AFM. Langmuir. 2015;31(1):199–208. | ||
Horejs C, Pum D, Sleytr UB, Peterlik H, Jungbauer A, Tscheliessnig R. Surface layer protein characterization by small angle X-ray scattering and a fractal mean force concept: from protein structure to nanodisk assemblies. J Chem Phys. 2010;133(17):175102. | ||
Kontro I, Wiedmer SK, Hynönen U, Palva AA, Penttilä PA, Serimaa R. The structure of Lactobacillus brevis surface layer reassembled on liposomes differs from native structure as revealed by SAXS. Biochimica Et Biophysica Acta. 1838;2014(8):2099–2014. | ||
Mobili P, Londero A, Maria TMR, et al. Characterization of S-layer proteins of Lactobacillus by FTIR spectroscopy and differential scanning calorimetry. Vibrational Spectroscopy. 2009;50(1):68–77. | ||
Ucisik MH, Küpcü S, Debreczeny M, Schuster B, Sleytr UB. S-layer coated Emulsomes as potential nanocarriers. Small. 2013;9(17):2895–2904. | ||
König H, Claus H, Varma A. Prokaryotic Cell Wall Compounds: Structure and Biochemistry. Berlin: Springer; 2010. | ||
Hianik T, Küpcü S, Sleytr UB, Rybár P, Krivánek R, Kaatze U. Interaction of crystalline bacterial cell surface proteins with lipid bilayers in liposomes. a sound velocity study. Colloids and Surfaces A: Physicochemical and Engineering Aspects. 1999;147(3):331–339. | ||
Diederich A, Sponer C, Pum D, Sleytr UB, Lösche M. Reciprocal influence between the protein and lipid components of a lipid-protein membrane model. Colloids and Surfaces B: Biointerfaces. 1996;6(6):335–346. | ||
Weygand M, Wetzer B, Pum D, et al. Bacterial S-layer protein coupling to lipids: X-ray reflectivity and grazing incidence diffraction studies. Biophys J. 1999;76(1 Pt 1):458–468. | ||
Hollmann A, Delfederico L, de Antoni G, Semorile L, Disalvo EA. Interaction of bacterial surface layer proteins with lipid membranes: Synergysm between surface charge density and chain packing. Colloids and Surfaces B: Biointerfaces. 2010;79(1):191–197. | ||
Baranova E, Fronzes R, Garcia-Pino A, et al. SbsB structure and lattice reconstruction unveil Ca2+ triggered S-layer assembly. Nature. 2012;487(7405):119–122. | ||
Whitelam S. Control of pathways and yields of protein crystallization through the interplay of nonspecific and specific attractions. Phys Rev Lett. 2010;105(8):088102. | ||
Smith MC, Crist RM, Clogston JD, Mcneil SE. Zeta potential: a case study of cationic, anionic, and neutral liposomes. Anal Bioanal Chem. 2017;409(24):5779–5787. | ||
Kono Y, Iwasaki AA, Fujita T. Effect of surface charge, particle size, and modification by polyethylene glycol of liposomes on their association with Caco-2 cells across an unstirred water layer Die Pharmazie. An International Journal of Pharmaceutical Sciences. 2018;73(1):3–8. | ||
Hollmann A, Delfederico L, Glikmann G, de Antoni G, Semorile L, Disalvo EA. Characterization of liposomes coated with S-layer proteins from lactobacilli. Biochimica et Biophysica Acta (BBA) – Biomembranes. 2007;1768(3):393–400. | ||
Hollmann A, Delfederico L, Santos NC, Disalvo EA, Semorile L. Interaction of S-layer proteins of Lactobacillus kefir with model membranes and cells. J Liposome Res. 2018;28(2):117–125. | ||
Carneiro C, Correia A, Collins T, et al. DODAB: monoolein liposomes containing Candida albicans cell wall surface proteins: a novel adjuvant and delivery system. Eur J Pharm Biopharm. 2015;89:190–200. | ||
Ulman A. Formation and structure of self-assembled monolayers. Chem Rev. 1996;96(4):1533–1554. | ||
Mader C, Küpcü S, Sára M, Sleytr UB. Stabilizing effect of an S-layer on liposomes towards thermal or mechanical stress. Biochimica et Biophysica Acta (BBA) – Biomembranes. 1999;1418(1):106–116. | ||
Zhang KL, Zhou J, Zhou H, et al. Bioinspired “Active” Stealth Magneto-Nanomicelles for Theranostics Combining Efficient MRI and Enhanced Drug Delivery. ACS Appl Mater Interfaces. 2017;9(36):30502–30509. | ||
Perche F, Torchilin VP. Recent trends in multifunctional liposomal nanocarriers for enhanced tumor targeting. Journal of Drug Delivery. 2013;2013(1):1–32. | ||
Klibanov AL, Maruyama K, Beckerleg AM, Torchilin VP, Huang L. Activity of amphipathic poly(ethylene glycol) 5000 to prolong the circulation time of liposomes depends on the liposome size and is unfavorable for immunoliposome binding to target. Biochimica et Biophysica Acta (BBA) – Biomembranes. 1991;1062(2):142–148. | ||
Maitani Y, Nakamura A, Tanaka T, Aso Y. Hydration of surfactant-modified and PEGylated cationic cholesterol-based liposomes and corresponding lipoplexes by monitoring a fluorescent probe and the dielectric relaxation time. Int J Pharm. 2012;427(2):372–378. | ||
Immordino ML, Dosio F, Cattel L. Stealth liposomes: review of the basic science, rationale, and clinical applications, existing and potential. Int J Nanomedicine. 2006;1(3):297–315. | ||
Kari OK, Rojalin T, Salmaso S, et al. Multi-parametric surface plasmon resonance platform for studying liposome-serum interactions and protein corona formation. Drug Deliv Transl Res. 2017;7(2):228–240. | ||
Picher MM, Küpcü S, Huang CJ, et al. Nanobiotechnology advanced antifouling surfaces for the continuous electrochemical monitoring of glucose in whole blood using a Lab-on-a-Chip. Lab Chip. 2013;13(9):1780–1789. | ||
Avall-Jääskeläinen S, Lindholm A, Palva A. Surface display of the receptor-binding region of the Lactobacillus brevis S-layer protein in Lactococcus lactis provides nonadhesive lactococci with the ability to adhere to intestinal epithelial cells. Appl Environ Microbiol. 2003;69(4):2230–2236. | ||
Yin QF, Pan DD, Guo YX, Xq Z, Sun YY, Cao JX. Effects of Lactobacillus acidophilus S-layer protein on adhesion to intestinal cells and macrophage proliferation. Modern Food Science and Technology. 2015;31:51–55. | ||
Ilk N, Küpcü S, Moncayo G, et al. A functional chimaeric S-layer-enhanced green fluorescent protein to follow the uptake of S-layer-coated liposomes into eukaryotic cells. Biochem J. 2004;379(Pt 2):441–448. | ||
Wang R, Jiang L, Zhang M, et al. The adhesion of Lactobacillus salivarius ren to a human intestinal epithelial cell line requires S-layer proteins. Sci Rep. 2017;7(1):44029. | ||
Hollmann A, Delfederico L, Miyoshi A, et al. S-layer proteins from lactobacilli as vaccine delivery systems. Int J of Micr Res. 2010;2(2):30–43. | ||
Ryan A, Lynch M, Smith SM, et al. A role for TLR4 in Clostridium difficile infection and the recognition of surface layer proteins. PLoS Pathog. 2011;7(6):e1002076. | ||
Taverniti V, Stuknyte M, Minuzzo M, et al. S-layer protein mediates the stimulatory effect of Lactobacillus helveticus MIMLh5 on innate immunity. Appl Environ Microbiol. 2013;79(4):1221–1231. | ||
Kapczynski DR, Meinersmann RJ, Lee MD, Mmd L. Adherence of Lactobacillus to intestinal 407 cells in culture correlates with fibronectin binding. Current Microbiology. 2000;41(2):136–141. | ||
Sillanpää J, Martínez B, Antikainen J, et al. Characterization of the collagen-binding S-layer protein CbsA of Lactobacillus crispatus. J Bacteriol. 2000;182(22):6440–6450. | ||
Toba T, Virkola R, Westerlund B, et al. A collagen-binding S-layer protein in Lactobacillus crispatus. Appl Environ Microbiol. 1995;61(7):2467–2471. | ||
Antikainen J, Anton L, Sillanpää J, Korhonen TK. Domains in the S-layer protein CbsA of Lactobacillus crispatus involved in adherence to collagens, laminin and lipoteichoic acids and in self-assembly. Mol Microbiol. 2002;46(2):381–394. | ||
Doig P, Emödy L, Trust TJ. Binding of laminin and fibronectin by the trypsin-resistant major structural domain of the crystalline virulence surface array protein of Aeromonas salmonicida. J Biol Chem. 1992;267(1):43–49. | ||
Banerjee A, Tsai CL, Chaudhury P, et al. FlaF is a β-sandwich protein that anchors the archaellum in the archaeal cell envelope by binding the S-layer protein. Structure. 2015;23(5):863–872. | ||
Zhang J, Gao J, Guo Y, Wu Z, Pan D. Extraction of Lactobacillus acidophilus CICC 6074 S-layer proteins and their ability to inhibit enteropathogenic Escherichia coli. Curr Microbiol. 2017;74(10):1123–1129. | ||
Wang Y, Wei Y, Yuan S, et al. Bacillus anthracis S-layer protein BslA binds to extracellular matrix by interacting with laminin. BMC Microbiol. 2016;16(1):183–193. | ||
Bradshaw WJ, Kirby JM, Roberts AK, Shone CC, Acharya KR. Cwp2 from Clostridium difficile exhibits an extended three domain fold and cell adhesion in vitro. Febs J. 2017;284(17):2886–2898. | ||
Hymes JP, Johnson BR, Barrangou R, Klaenhammer TR. Functional analysis of an S-Layer-Associated fibronectin-binding protein in Lactobacillus acidophilus NCFM. Appl Environ Microbiol. 2016;82(9):2676–2685. | ||
Johnson BR, O’Flaherty S, Goh YJ, Carroll I, Barrangou R, Klaenhammer TR. The S-layer Associated Serine Protease Homolog PrtX Impacts Cell Surface-Mediated Microbe-Host Interactions of Lactobacillus acidophilus NCFM. Front Microbiol. 2017;8:1185. | ||
Chinthamani S, Settem RP, Honma K, Kay JG, Sharma A. Macrophage inducible C-type lectin (Mincle) recognizes glycosylated surface (S)-layer of the periodontal pathogen Tannerella forsythia. PLoS One. 2017;12(3):e0173394. | ||
Garrote GL, Delfederico L, Bibiloni R, et al. Lactobacilli isolated from kefir grains: evidence of the presence of S-layer proteins. Journal of Dairy Research. 1999;71(2):222–230. | ||
Golowczyc MA, Mobili P, Garrote GL, de Los Angeles Serradell M, Abraham AG, de Antoni GL. Interaction between Lactobacillus kefir and Saccharomyces lipolytica isolated from kefir grains: evidence for lectin-like activity of bacterial surface proteins. J Dairy Res. 2009;76(1):111–116. | ||
Tschiggerl H, Breitwieser A, de Roo G, Verwoerd T, Schäffer C, Sleytr UB. Exploitation of the S-layer self-assembly system for site directed immobilization of enzymes demonstrated for an extremophilic laminarinase from Pyrococcus furiosus. J Biotechnol. 2008;133(3):403–411. | ||
Khang Y-H, Park H-Y, Jeong Y-S, Kim J-A, Kim Y-H. Recombinant -layer proteins of Lactobacillus brevis mediating antibody adhesion to calf intestine alleviated neonatal diarrhea syndrome. J Microbiol Biotechnol. 2009;19(5):511–519. | ||
Weinert U, Vogel M, Reinemann C, Strehlitz B, Pollmann K, Raff J. S-layer proteins as an immobilization matrix for aptamers on different sensor surfaces. Engineering in Life Sciences. 2015;15(7):710–720. | ||
Varga M. Targeting at the nanoscale: a novel S-layer fusion protein enabling controlled immobilization of biotinylated molecules. Nanomaterials. 2016;6(11):199. | ||
Pleschberger M, Neubauer A, Egelseer EM, et al. Generation of a functional monomolecular protein lattice consisting of an S-layer fusion protein comprising the variable domain of a camel heavy chain antibody. Bioconjug Chem. 2003;14(2):440–448. | ||
Scheicher SR, Kainz B, Köstler S, et al. 2D crystalline protein layers as immobilization matrices for the development of DNA microarrays. Biosens Bioelectron. 2013;40(1):32–37. | ||
Scheicher SR, Kainz B, Köstler S, et al. Optical oxygen sensors based on Pt(II) porphyrin dye immobilized on S-layer protein matrices. Biosens Bioelectron. 2009;25(4):797–802. | ||
Ücisik MH, Küpcü S, Breitwieser A, Gelbmann N, Schuster B, Sleytr UB. S-layer fusion protein as a tool functionalizing emulsomes and CurcuEmulsomes for antibody binding and targeting. Colloids Surf B Biointerfaces. 2015;128:132–139. | ||
Mader C, Küpcü S, Sleytr UB, Sára M. S-layer-coated liposomes as a versatile system for entrapping and binding target molecules. Biochimica et Biophysica Acta (BBA) – Biomembranes. 2000;1463(1):142–150. | ||
Shao AN. The Study on Surface Layer Protein Coating Liposomes as Oral Vaccine Vector [master’s thesis]. Hangzhou: Zhejiang University of Technology; 2017. | ||
Fortuna A, Alves G, Serralheiro A, Sousa J, Falcão A. Intranasal delivery of systemic-acting drugs: small-molecules and biomacromolecules. Eur J Pharm Biopharm. 2014;88(1):8–27. | ||
Mcclements DJ. Encapsulation, protection, and delivery of bioactive proteins and peptides using nanoparticle and microparticle systems: a review. Adv Colloid Interface Sci. 2018;253:1–22. | ||
Tan J, Cheong H, Park YS, et al. Cell-penetrating peptide-mediated topical delivery of biomacromolecular drugs. Curr Pharm Biotechnol. 2014;15(3):231–239. | ||
Weinert U, Pollmann K, Barkleit A, Vogel M, Günther T, Raff J. Synthesis of S-layer conjugates and evaluation of their modifiability as a tool for the functionalization and patterning of technical surfaces. Molecules. 2015;20(6):9847–9861. | ||
Schrems A, Kibrom A, Küpcü S, Kiene E, Sleytr UB, Schuster B. Bilayer lipid membrane formation on a chemically modified S-layer lattice. Langmuir. 2011;27(7):3731–3738. | ||
Horie M, Ishiyama A, Fujihira-Ueki Y, Sillanpää J, Korhonen TK, Toba T. Inhibition of the adherence of Escherichia coli strains to basement membrane by Lactobacillus crispatus expressing an S-layer. J Appl Microbiol. 2002;92(3):396–403. | ||
Hill TK, Mohs AM. Image-guided tumor surgery: will there be a role for fluorescent nanoparticles? Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2016;8(4):498–511. | ||
Hoffman RM. Strategies for in vivo imaging using fluorescent proteins. J Cell Biochem. 2017;118(9):2571–2580. | ||
Schäffer C, Novotny R, Küpcü S, et al. Novel biocatalysts based on S-layer self-assembly of Geobacillus stearothermophilus NRS 2004/3a: a nanobiotechnological approach. Small. 2007;3(9):1549–1559. | ||
Schrems A, Larisch VD, Stanetty C, et al. Liposome fusion on proteinaceous S-layer lattices triggered viaβ-diketone ligand–europium(iii) complex formation. Soft Matter. 2011;7(12):5514–5518. | ||
Bornscheuer UT. Trends and challenges in enzyme technology. Adv Biochem Eng Biotechnol. 2005;100(100):181–203. | ||
Girelli AM, Mattei E. Application of immobilized enzyme reactor in on-line high performance liquid chromatography: a review. Journal of Chromatography B. 2005;819(1):3–16. | ||
Nollert P, Kiefer H, Jähnig F. Lipid vesicle adsorption versus formation of planar bilayers on solid surfaces. Biophys J. 1995;69(4):1447–1455. | ||
Mcconnell HM, Watts TH, Weis RM, Brian AA. Supported planar membranes in studies of cell-cell recognition in the immune system. Biochimica et Biophysica Acta (BBA) – Reviews on Biomembranes. 1986;864(1):95–106. | ||
Damiati S, Schrems A, Sinner EK, Sleytr UB, Schuster B. Probing peptide and protein insertion in a biomimetic S-layer supported lipid membrane platform. Int J Mol Sci. 2015;16(2):2824–2838. | ||
Wong WS, Yung PT. Characterization of a nanoscale S-layer protein based template for biomolecular patterning. Conf Proc IEEE Eng Med Biol Soc. 2014;2014:2781–2784. | ||
Damiati S, Küpcü S, Peacock M, et al. Acoustic and hybrid 3D-printed electrochemical biosensors for the real-time immunodetection of liver cancer cells (HepG2). Biosens Bioelectron. 2017;94:500–506. | ||
Conroy DJR, Millner PA, Stewart DI, Pollmann K. Biosensing for the environment and defence: aqueous uranyl detection using bacterial surface layer proteins. Sensors. 2010;10(5):4739–4755. | ||
Park TJ, Lee SJ, Park JP, Yang MH, Choi JH, Lee SY. Characterization of a bacterial self-assembly surface layer protein and its application as an electrical nanobiosensor. J Nanosci Nanotechnol. 2011;11(1):402–407. | ||
Muñoz-Provencio D, Llopis M, Antolín M, et al. Adhesion properties of Lactobacillus casei strains to resected intestinal fragments and components of the extracellular matrix. Arch Microbiol. 2009;191(2):153–161. | ||
Meng J, Zhang QX, Lu RR, Rr L. Surface layer protein from Lactobacillus acidophilus NCFM inhibit intestinal pathogen-induced apoptosis in HT-29 cells. Int J Biol Macromol. 2017;96:766–774. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.