Back to Journals » OncoTargets and Therapy » Volume 13
Six1 Overexpression Promotes Glucose Metabolism and Invasion Through Regulation of GLUT3, MMP2 and Snail in Thyroid Cancer Cells
Authors Yang C, Xu W, Gong J , Chai F, Cui D, Liu Z
Received 14 August 2019
Accepted for publication 28 April 2020
Published 29 May 2020 Volume 2020:13 Pages 4855—4863
DOI https://doi.org/10.2147/OTT.S227291
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yao Dai
Chuanjia Yang,1 Weixue Xu,1 Jian Gong,2 Fang Chai,3 Dongxu Cui,1 Zhen Liu1
1Department of General Surgery, Shengjing Hospital of China Medical University, Shenyang 110004, People’s Republic of China; 2Department of Clinical Pharmacy, School of Life Science and Pharmaceutical University, Shenyang, People’s Republic of China; 3Department of Thyroid Surgery, The First Affiliated Hospital of Jinzhou Medical University, Jinzhou, People’s Republic of China
Correspondence: Chuanjia Yang; Zhen Liu
Department of General Surgery, Shengjing Hospital of China Medical University, Shenyang 110004, People’s Republic of China
Email [email protected]; [email protected]
Introduction: Sineoculis homeobox homolog 1 (Six1) overexpression has been implicated in several human cancers. To date, its clinical significance and potential function in human thyroid cancer remain unclear.
Methods: Immunohistochemistry was used to examine the protein expression of BCAT1 in 89 cases of thyroid cancer tissues. We overexpressed and knockdown Six1 in TPC-1 and B-CPAP thyroid cancer cell lines. Biological roles and potential mechanisms of Six1 were examined using CCK-8, colony formation assay, Matrigel invasion assay, Western blot, PCR, ATP assay, and 2-NBDG uptake assay.
Results: We showed that Six1 protein was upregulated in thyroid cancers and was associated with tumor size and nodal metastasis. Analysis of TCGA dataset indicated that Six1 mRNA was higher in thyroid cancers compared with normal thyroid. CCK-8, colony formation and Matrigel invasion assays demonstrated that Six1 overexpression promoted proliferation, colony number and invasion while Six1 siRNA knockdown inhibited the growth rate, colony formation ability and invasive ability in both cell lines. Notably, Six1 upregulated glucose consumption, lactate production level and ATP level. 2-NBDG uptake analysis showed that Six1 overexpression upregulated glucose uptake while Six1 knockdown inhibited glucose uptake. Further analysis revealed that Six1 overexpression upregulated Snail, MMP2 and GLUT3 at both mRNA and protein levels. TCGA analysis demonstrated positive associations between Six1 and Snail, MMP2 and GLUT3 at the mRNA levels.
Conclusion: Taken together, our data demonstrated that Six1 was upregulated in human thyroid cancers and promoted cell proliferation and invasion. Our data also revealed new roles of Six1 in thyroid cancer development by modulating glucose metabolism and invasion, possibly through regulation of Snail, MMP2 and GLUT3.
Keywords: Six1, thyroid cancer, glucose metabolism, GLUT3
Introduction
Thyroid cancer is a common endocrine cancer world wide and its incidence has increased in the past decade.1 The major subtypes of non-medullary thyroid cancer includes papillary thyroid cancer, follicular thyroid cancer, anaplastic thyroid cancer and poorly differentiated thyroid cancer. Papillary thyroid cancer accounts for more than 80% of all thyroid cancer cases and the development of distant metastases is a major contributing factors to restricted prognosis.2 It is important to understand the mechanism of thyroid carcinogenesis, proliferation, invasion and metastasis, which may provide insights into potential treatment strategies.3
Sineoculis homeobox homolog 1 (Six1) is a homeodomain-containing transcription factor. Six1 is highly expressed during embryo development and plays important roles in mammalian organ development.4–6 Six1 enhances progenitor cell survival and its absence leads to the reduction in size or loss of many organs and increased apoptosis.7–10 The expression of Six1 remains at a low level in adult tissue and its overexpression contributes to tumor initiation.11 It has been found that Six1 was overexpressed in several human malignancies such as breast,12,13 cervical,14 ovarian,15 and pancreatic cancers. Six1 overexpression enhances cancer proliferation, survival and epithelial-mesenchymal transition (EMT).16,17 However, its clinical significance and biological functions in human thyroid cancer remain elusive.
In the present study, we examined its expression pattern in thyroid cancer tissues. We then overexpressed and depleted Six1 in thyroid cancer cell lines. The biological function and underlying mechanism of Six1 in thyroid cancer was also investigated.
Patients and Methods
Patients and Specimens
The study protocol was approved by the Institutional Reviewer Board of Shengjing Hospital. Thyroid cancer tissues were obtained from 89 thyroid cancer patients who received surgical operation between 2013 and 2016 in the Shengjing Hospital. Normal thyroid tissues were from benign thyroid disease such as hyperplasia. Written informed consent was provided by the patients. The study was conducted in accordance with the Declaration of Helsinki. The histological diagnosis were evaluated according to the World Health Organization (WHO) classification guidelines.18
Immunohistochemical Staining
4μm thick sections of formalin-fixed, paraffin-embedded tumor samples were obtained from Pathology Archive. Immunohistochemical staining was carried out using the Elivision Plus staining kit from MaiXin (MaiXin, Fuzhou, China). Briefly, sections were deparaffinized in xylene, and rehydrated in alcohol and water. Antigen retrieval was performed in 0.01 M citrate buffer (pH 6.0) for 2 minutes in an autoclave. Hydrogen peroxide (0.3%) was used to block peroxidase. Then sections were then incubated with Six1 antibody (1:200 dilution rate, Sigma HPA00189, USA) at 4°C for 10 hours. After washing with PBS, section was incubated with HRP polymer from Elivision Plus staining kit at room temperature. Staining was developed using DAB.
We selected five views (containing >80% tumor cells) per section randomly to analyze Six1 intensity. Nuclear Six1 staining score was scored as 0 (negative), 1 (moderate), 2 (strong). The percentage score was divided as 1:1–25%, 2: 26–50% 3: 51–75% and 4: 76–100%. Intensity score and percentage scores were multiplied to obtain a Six1 staining score of 0 to 8. Six1 expression was considered as low when the staining score <4; Six1 expression was considered as high (overexpression) when the staining score ≥4.
Cell Culture and Transfection
Thyroid cancer cell lines B-CPAP and TPC-1 were purchased from Shanghai Cell Bank of Chinese Academy of Sciences. Cells were cultured in RPMI-1640 medium containing 10% fetal bovine serum (FBS). pCMV-SIX1 transfection plasmid was obtained from Origene. siRNA sequences for Six1 and scramble siRNA were purchased from Dharmacon. Lipofectamine 3000 (Invitrogen) was used for plasmid transfection and Dharmafect 1 (Dharmacon) was used for siRNA transfection.
Western Blot
For Western blot, 60ug protein was separated by SDS-PAGE. Then proteins were transferred to PVDF membrane and incubated with primary antibody including Six1 (1:800; Sigma, USA), MMP2, GLUT3, Snail (1:1000; Cell Signaling Technology, USA), and GAPDH (1:2000; Santa Cruz, USA). Membranes were washed with TBS-T, and incubated with HRP-conjugated secondary antibody (1:2000, Santa Cruz, USA) for 1 hour. Images were taken using the DNR Imaging Systems after ECL development (ThermoFisher, USA).
Realtime PCR
RNA was isolated using RNAiso (TAKARA). For quantitative PCR cDNA was synthesized using the iScriptTM Reverse Transcription Supermix (Bio-Rad). Realtime PCR was performed by ABI 7500 PCR system using SYBR Green Master Mix (TAKARA). The thermal profile is 50°C for 2min, 95°C for 2 min, 45 cycles of 95°C for 15 sec and 60°C for 40 sec. β-actin was used as the endogenous calibrator. Relative fold change was calculated using 2-ΔΔCt method. Experiments were done in triplicated. The primer sequences are as follows: Six1 forward, 5ʹ AAGGAGAAGTCGAGGGGTGT 3ʹ, Six1 reverse, 5ʹ TGCTTGTTGGAGGAGGAGTT 3ʹ; MMP2 forward, 5ʹ TGTGTTCTTTGCAGGGAATGAAT 3ʹ, MMP2 reverse, 5ʹ TGTCTTCTTGTTTTTGCTCCAGTT 3ʹ. GLUT3 forward, 5ʹ CCTTTGGCACTCTCAACCAGC 3ʹ, GLUT3 reverse, 5ʹ AACCCAGTAGCAGCGGCCAT 3ʹ. Snail forward, 5ʹ GCCCACCTCCAGACCCAC 3ʹ, Snail reverse, 5ʹ GCAGGGACATTCGGGAGAA 3ʹ. β-actin forward, 5ʹ ATAGCACAGCCTGGATAGCAACGTAC 3ʹ, β-actin reverse, 5ʹ CACCTTCTACAATGAGCTGCGTGTG 3ʹ.
CCK-8 and Colony Formation Assays
CCK-8 assay: Cell viability was assessed using the Cell Counting Kit-8 (CCK-8; Dojindo, Kumamoto, Japan). Cells were seeded into 96-well plates at about 3000 cells per well. 10 μL CCK-8 reagent was added into each well. After incubation at 37°C for additional 2 h, cell proliferation was determined by examining the absorbance at a wavelength of 450 nm using a microplate reader (Bio-Rad, USA). Colony formation was performed using 3000 cells per culture dish. Then the cells were cultured for about 15 days then stained using Giemsa. Experiments were done in triplicated.
Matrigel Invasion Assay
Matrigel invasion assay used a 24 well Transwell chamber (Corning, 8 µm pore size) with 20 µL of Matrigel (BD Bioscience) was added to the chamber at a 1:3 dilution rate. Cells were transferred to the upper chamber in serum-free medium. 10% FBS 1640 medium was added to the lower chamber. After 18 hours cells remaining on the upper chamber surface were removed. The experiments were performed in triplicate.
Glucose Uptake
Glucose uptake was examined with using 2-NBDG. Cells were incubated with 1mM 2-NBDG in 1640 medium at 37⁰C for 30 minute. Cells were washed with PBS and analyzed on flow cytometer at 488 nm excitation. The experiments were performed in triplicate.
Analysis of ATP Production, Glucose Consumption, and Lactate Level
Glucose and lactate assay kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). ATP level was examined with the Beyotime ATP Assay Kit (Guangzhou, China). After transfection, the supernatants were harvested, and glucose and lactate levels in the medium were determined using glucose and lactate assay kits according to the manufacturer’s instructions. The experiments were performed in triplicate.
Statistical Analysis
Statistical analysis was performed using SPSS 17. Six1 mRNA (RNA-seq data) of thyroid cancer was obtained from The Cancer Genome Atlas (TCGA). The Mann–Whitney U-test was used to compare Six1 mRNA in cancers with/without nodal metastasis. Paired t-tests were used to compare Six1 mRNA in cancers and normal thyroid tissue. A χ2 test was used to examine possible correlations between Six1 expression and clinicopathologic factors. Student’s t-test was used to compare data obtained from biological experiments. A p<0.05 was considered as statistically significant.
Results
Six1 Is Upregulated in Human Thyroid Cancers
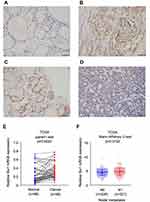
A total of 89 thyroid cancer cases (including 71 papillary carcinoma, 9 follicular carcinoma and 9 anaplastic carcinoma) and 20 normal thyroid tissues were used for analysis of Six1 protein expression. Negative or weak nuclear Six1 staining was observed in normal thyroid tissues (Figure 1A) while upregulated Six1 nuclear staining was found in 46 out of 89 cases of thyroid cancer specimens (Figure 1B–D). The high Six1 level was positively associated with tumor size (p=0.0027) and nodal status (p=0.0330) (Table 1). There was no significant association between Six1 with age (p=0.1561), sex (p=0.713) and histological subtype (p=0.5057). A summary of immunohistochemical results was provided in Supplementary Table 1.
 |
Table 1 Association of SIX1 Expression with Clinicopathological Parameters in Patients with Thyroid Cancer |
We also analyzed of the TCGA data from thyroid cancers. As shown in Figure 1E&F, expression of Six1 mRNA was elevated in thyroid cancers compared with normal tissue (paired t test, p<0.05, Figure 1E). Six1 mRNA levels were higher in cancers with positive nodal metastasis (Mann–Whitney U-test, p<0.05, Figure 1F). Taken together, these data indicated that Six1 was upregulated in human thyroid cancers and correlated with malignant features.
Six1 Promotes Proliferation and Invasion
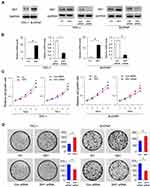
Six1 protein expression was examined in 2 thyroid cancer cell lines (TPC-1, B-CPAP). We transfected both B-CPAP and TPC-1 cells with the Six1 plasmid and siRNA. Transfection efficiency was confirmed by RT-qPCR and Western blots (Figure 2A and B). The CCK-8 assay demonstrated that Six1 depletion downregulated the proliferation rate of both TPC-1 and B-CPAP cells while Six1 overexpression upregulated proliferation rate (Figure 2C). Colony formation results showed that Six1 transfection increased colony numbers while siRNA treatment decreased colony numbers (Figure 2D). To evaluate the effect of Six1 on invasion, Matrigel invasion assay was performed and the results showed that Six1 increased invading ability while its depletion decreased invading ability of thyroid cancer cells (Figure 3A).
Six1 Regulates Glucose Uptake and ATP Levels
Glucose metabolism is important for ATP production, cell survival and proliferation. To investigate whether Six1 could modulate glucose metabolism in thyroid cancer cell lines, we examined several steps involved in glucose metabolism including glucose uptake, consumption and lactate production levels.
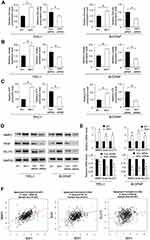
We used 2-NBDG to evaluate the rate of glucose uptake. As shown in Figure 3B, Six1 overexpression increased the rate of glucose uptake in both TPC-1 and B-CPAP cell lines, while Six1 depletion downregulated the rate of glucose uptake in both cell lines. We also examined the levels of glucose and lactate in the medium. As shown in Figure 4A and B, Six1 overexpression increased the level of glucose consumption and lactate production while Six1 depletion exhibited the opposite effects, indicating that Six1 was a positive regulator of glucose metabolism in thyroid cancer cells.
Finally, we examined the role of Six1 on intracellular ATP levels. As shown in Figure 4C. Six1 overexpression increased the level of ATP while Six1 depletion downregulated the level of intracellular ATP in both cell lines, suggesting Six1 enhanced thyroid cancer development through regulation of glucose uptake.
Six1 Regulates GLUT3, Snail and MMP2 Expression
To identify the mechanism of Six1 on glucose metabolism and invasion, we screened potential related genes. Western blot and realtime PCR demonstrated that Six1 positively regulated GLUT3, Snail, and MMP2 protein and mRNA expression in both TPC-1 and B-CPAP cell lines (Figure 4D and E). Next the association of Six1 with GLUT3, Snail and MMP2 mRNA in thyroid cancer tissues was investigated through analysis of RNA-seq data from TCGA dataset. There was a statistically significant association between Six1 mRNA and GLUT3, Snail, MMP2 mRNA expression (Pearson’s correlation, Figure 4F). These data supported our results that Six1 positively regulated glucose metabolism and invasion.
Discussion
Recently, several reports showed that Six1 is upregulated in various human cancers such breast cancer, ovarian cancer, colorectal and hepatocellular carcinoma.15,16,19-22 However, its expression pattern and biological roles in human thyroid cancers remain unexplored. Our data demonstrated that Six1 protein expression was increased in thyroid cancer tissues and was positively correlated with tumor size and nodal status. In addition, our results were supported by TCGA data, suggesting Six1 as a potential thyroid cancer biomarker.
Matrigel invasion results showed that Six1 overexpression promoted thyroid cancer invasion, which was in accord with the TCGA data showing positive correlations between Six1 mRNA and nodal metastasis. We further demonstrated that Six1 could upregulate MMP2 and Snail at mRNA and protein levels. MMP2 is required to mediate cancer cell invasion. Snail is an important regulator of epithelial-to-mesenchymal transition (EMT) which can inhibit cell adhesion by downregulating E-cadherin.23,24 MMP2 and Snail were also reported to promotes thyroid cancer progression through induction of cell invasion. TCGA analysis revealed a positive association between Six1 and MMP2/Snail mRNA in thyroid cancer tissues. These data suggested that Six1 functions as a promoter of thyroid cancer invasion possibly through Snail/MMP2 regulation.
The CCK-8 and colony formation assays confirmed the growth promoting role of Six1 in thyroid cancer cells. Cancer cells are dependent on glucose metabolism to produce ATP, which is essential for the malignant growth.25 Glucose transport is the initial step in glucose metabolism. We found that SIX1 overexpression increased glucose uptake into cancer cells with and elevated ATP production, indicating SIX1 as a positive regulator of glucose metabolism in thyroid cancers. We also found Six1 was able to increase mRNA and protein expression of GLUT3. The GLUT family plays critical roles during glucose transport and has been found to be overexpressed in various cancers.26–28 Upregulation of GLUT family genes has been reported to be correlated with poor prognosis in papillary thyroid cancers.27 Analysis of a TCGA dataset also confirmed the positive correlation between GLUT3 and Six1 mRNA in 501 cases of thyroid cancers, which further support the role of Six1 on glucose metabolism.
In conclusion, our data demonstrated that Six1 is overexpressed in thyroid cancers and is correlated with nodal metastasis and tumor size. We also linked its oncogenic role with glucose metabolism and invasion, possibly through regulation of MMP2, Snail and GLUT3. These findings suggest Six1 as a potential therapeutic target in thyroid cancers.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68(1):7–30. doi:10.3322/caac.21442
2. Xing M. Molecular pathogenesis and mechanisms of thyroid cancer. Nat Rev Cancer. 2013;13(3):184–199. doi:10.1038/nrc3431
3. Lin X, Zhang H, Dai J, et al. TFF3 contributes to epithelial-mesenchymal transition (EMT) in papillary thyroid carcinoma cells via the MAPK/ERK signaling pathway. J Cancer. 2018;9(23):4430–4439. doi:10.7150/jca.24361
4. Xu PX, Zheng W, Huang L, Maire P, Laclef C, Silvius D. Six1 is required for the early organogenesis of mammalian kidney. Development. 2003;130(14):3085–3094. doi:10.1242/dev.00536
5. Zheng W, Huang L, Wei ZB, Silvius D, Tang B, Xu PX. The role of Six1 in mammalian auditory system development. Development. 2003;130(17):3989–4000. doi:10.1242/dev.00628
6. Laclef C, Hamard G, Demignon J, Souil E, Houbron C, Maire P. Altered myogenesis in Six1-deficient mice. Development. 2003;130(10):2239–2252. doi:10.1242/dev.00440
7. Liu Y, Chakroun I, Yang D, et al. Six1 regulates MyoD expression in adult muscle progenitor cells. PLoS One. 2013;8(6):e67762. doi:10.1371/journal.pone.0067762
8. Nord H, Nygard Skalman L, von Hofsten J. Six1 regulates proliferation of Pax7-positive muscle progenitors in zebrafish. J Cell Sci. 2013;126(Pt 8):1868–1880. doi:10.1242/jcs.119917
9. Ikeda K, Kageyama R, Suzuki Y, Kawakami K. Six1 is indispensable for production of functional progenitor cells during olfactory epithelial development. Int J Dev Biol. 2010;54(10):1453–1464. doi:10.1387/ijdb.093041ki
10. Chen B, Kim EH, Xu PX. Initiation of olfactory placode development and neurogenesis is blocked in mice lacking both Six1 and Six4. Dev Biol. 2009;326(1):75–85. doi:10.1016/j.ydbio.2008.10.039
11. Kumar JP. The sine oculis homeobox (SIX) family of transcription factors as regulators of development and disease. Cell Mol Life Sci. 2009;66(4):565–583. doi:10.1007/s00018-008-8335-4
12. Coletta RD, Christensen K, Reichenberger KJ, et al. The Six1 homeoprotein stimulates tumorigenesis by reactivation of cyclin A1. Proc Natl Acad Sci U S A. 2004;101(17):6478–6483. doi:10.1073/pnas.0401139101
13. Reichenberger KJ, Coletta RD, Schulte AP, Varella-Garcia M, Ford HL. Gene amplification is a mechanism of Six1 overexpression in breast cancer. Cancer Res. 2005;65(7):2668–2675. doi:10.1158/0008-5472.CAN-04-4286
14. Zheng XH, Liang PH, Guo JX, et al. Expression and clinical implications of homeobox gene Six1 in cervical cancer cell lines and cervical epithelial tissues. Int J Gynecol Cancer. 2010;20(9):1587–1592.
15. Behbakht K, Qamar L, Aldridge CS, et al. Six1 overexpression in ovarian carcinoma causes resistance to TRAIL-mediated apoptosis and is associated with poor survival. Cancer Res. 2007;67(7):3036–3042. doi:10.1158/0008-5472.CAN-06-3755
16. Smith AL, Iwanaga R, Drasin DJ, et al. The miR-106b-25 cluster targets Smad7, activates TGF-beta signaling, and induces EMT and tumor initiating cell characteristics downstream of Six1 in human breast cancer. Oncogene. 2012;31(50):5162–5171. doi:10.1038/onc.2012.11
17. Radisky DC. Defining a role for the homeoprotein Six1 in EMT and mammary tumorigenesis. J Clin Invest. 2009;119(9):2528–2531. doi:10.1172/JCI40555
18. Kakudo K, Bychkov A, Bai Y, Li Y, Liu Z, Jung CK. The new 4th edition World Health Organization classification for thyroid tumors, Asian perspectives. Pathol Int. 2018;68(12):641–664. doi:10.1111/pin.12737
19. Wang CA, Jedlicka P, Patrick AN, et al. SIX1 induces lymphangiogenesis and metastasis via upregulation of VEGF-C in mouse models of breast cancer. J Clin Invest. 2012;122(5):1895–1906. doi:10.1172/JCI59858
20. Ng KT, Man K, Sun CK, et al. Clinicopathological significance of homeoprotein Six1 in hepatocellular carcinoma. Br J Cancer. 2006;95(8):1050–1055. doi:10.1038/sj.bjc.6603399
21. Ng KT, Lee TK, Cheng Q, et al. Suppression of tumorigenesis and metastasis of hepatocellular carcinoma by shRNA interference targeting on homeoprotein Six1. Int J Cancer. 2010;127(4):859–872. doi:10.1002/ijc.25105
22. Ono H, Imoto I, Kozaki K, et al. SIX1 promotes epithelial-mesenchymal transition in colorectal cancer through ZEB1 activation. Oncogene. 2012;31(47):4923–4934. doi:10.1038/onc.2011.646
23. Baquero P, Sanchez-Hernandez I, Jimenez-Mora E, Orgaz JL, Jimenez B, Chiloeches A. (V600E)BRAF promotes invasiveness of thyroid cancer cells by decreasing E-cadherin expression through a Snail-dependent mechanism. Cancer Lett. 2013;335(1):232–241. doi:10.1016/j.canlet.2013.02.033
24. Yasui K, Shimamura M, Mitsutake N, Nagayama Y. SNAIL induces epithelial-to-mesenchymal transition and cancer stem cell-like properties in aldehyde dehydroghenase-negative thyroid cancer cells. Thyroid. 2013;23(8):989–996. doi:10.1089/thy.2012.0319
25. Wang Y, Wu S, Huang C, Li Y, Zhao H, Yin Yang KV. 1 promotes the Warburg effect and tumorigenesis via glucose transporter GLUT3. Cancer Sci. 2018;109(8):2423–2434. doi:10.1111/cas.13662
26. Pizzuti L, Sergi D, Mandoj C, et al. GLUT 1 receptor expression and circulating levels of fasting glucose in high grade serous ovarian cancer. J Cell Physiol. 2018;233(2):1396–1401. doi:10.1002/jcp.26023
27. Chai YJ, Yi JW, Oh SW, et al. Upregulation of SLC2 (GLUT) family genes is related to poor survival outcomes in papillary thyroid carcinoma: analysis of data from The Cancer Genome Atlas. Surgery. 2017;161(1):188–194. doi:10.1016/j.surg.2016.04.050
28. Cho H, Lee YS, Kim J, Chung JY, Kim JH. Overexpression of glucose transporter-1 (GLUT-1) predicts poor prognosis in epithelial ovarian cancer. Cancer Invest. 2013;31(9):607–615. doi:10.3109/07357907.2013.849722
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.