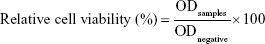Back to Journals » International Journal of Nanomedicine » Volume 14
Silver sulfadiazine nanosuspension-loaded thermosensitive hydrogel as a topical antibacterial agent
Authors Liu X, Gan H, Hu C, Sun W, Zhu X, Meng Z, Gu R, Wu Z, Dou G
Received 18 September 2018
Accepted for publication 1 December 2018
Published 28 December 2018 Volume 2019:14 Pages 289—300
DOI https://doi.org/10.2147/IJN.S187918
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Mian Wang

Xiaoya Liu, Hui Gan, Chaoran Hu, Wenzhong Sun, Xiaoxia Zhu, Zhiyun Meng, Ruolan Gu, Zhuona Wu, Guifang Dou
Department of Pharmaceutical Sciences, Beijing Institute of Radiation Medicine, Beijing 100850, People’s Republic of China
Background: Silver sulfadiazine (AgSD) is widely employed as an antibacterial agent for surface burn management. However, the antibacterial activity of AgSD was restrained because of the lower drug solubility and possible cytotoxicity.
Objective: This study aimed to formulate stable silver sulfadiazine/nanosuspensions (AgSD/NSs) with improved AgSD solubility and prepare a suitable carrier for AgSD/NS delivery. Nanotechnology was used to overcome the low drug dissolution rate of AgSD, while the new carrier loaded with AgSD/NS was assumed to decrease the possible cytotoxicity, enhance antibacterial activity, and promote wound healing.
Methods: AgSD/NSs were prepared by high pressure homogenization method. Poloxamer 407-based thermoresponsive hydrogels were prepared by cold method as carriers of AgSD/NS to obtain AgSD/NS-loaded thermoresponsive hydrogel. Scanning electron microscope (SEM), Fourier transform infrared spectroscopy (FTIR) and X-ray diffraction (XRD) were used to measure the physicalchemical properties of AgSD/NSs and AgSD/NS-loaded gel. The cytotoxicity of the AgSD/NS-loaded gel was evaluated using methyl thiazolyltetrazolium assay with L929 mouse fibroblast cell lines. In vitro antibacterial activities of AgSD/NSs and AgSD/NS loaded gel were also measured.
Results: Stable AgSD/NSs with an average particle size of 369 nm were formulated while 1.5% P407 was selected as a stabilizer. The optimized AgSD/NS thermoresponsive hydrogel exhibited the gelation temperature of approximately 30°C. A significant improvement in solubility was observed for AgSD nanoparticles (96.7%) compared with AgSD coarse powders (12.5%). The results of FTIR and XRD revealed that the physicochemical properties of AgSD/NS were reserved after incorporating into the hydrogel. The cell viability after incubation with AgSD/NS-loaded thermoresponsive hydrogel improved from 60.7% to 90.6% compared with incubation with AgSD/NS directly. Drug release profiles from the thermoresponsive hydrogel increased compared with the commercial AgSD cream, implying less application frequency of AgSD cream clinically. In vitro antibacterial studies manifested that AgSD nanocrystallization significantly enhanced the antibacterial activity compared with the AgSD coarse powder.
Conclusion: The combination of AgSD nanosuspensions and thermoresponsive hydrogel effectively improved the AgSD antibacterial activity and decreased the cytotoxicity. This study also suggested that a poloxamer thermoresponsive hydrogel could be used as a delivery system for other nanocrystals to decrease possible nanotoxicity.
Keywords: cytotoxicity, wound healing infection, nanotechnology
Introduction
It was estimated that 75% of deaths in patients with burns over 40% body surface area were due to infection.1 Every year, about 180,000 people die due to burn wounds in low- to middle-income countries, such as the South-East Asian Region.2 Although many therapeutics have been introduced for wound healing with significant advances in wound care and treatment, the incidence and death rates have increased to a large extent due to ensuing microbial infections in patients with burns. Silver sulfadiazine (AgSD), combining sulfadiazine with silver, is widely employed as an antibacterial agent for surface burn management.3 AgSD has a broad-spectrum action against both gram-negative and gram-positive bacteria by destroying the cell membrane and inhibiting DNA replication.4 However, the application of AgSD is limited due to its poor aqueous solubility.5,6 Besides, AgSD was reported to be cytotoxic toward keratinocytes and fibroblasts.7 The frequent application (usually two to four times daily) was required because of the retarded release of commercial AgSD cream.8,9 Furthermore, frequent removal of the residual cream leads to patient discomfort and pain. AgSD sometimes slows down the wound-healing process owing to these drawbacks.10–12
Nanotechnology has been proved to be an ideal approach in resolving the poor solubility of drugs in previous studies.13,14 Nanosuspensions (NSs) usually contain crystals or amorphous substances with the average particle size ranging from 1 to 1,000 nm.15–17 The larger surface and surface–volume ratio result in a greater interaction with the solvent, and the solubility increases with the decrease in the particle size of drugs. As a result, preparing AgSD NS seems to be an ideal approach to improve its solubility. Moreover, nanosized AgSD with enhanced solubility has been demonstrated to have better antibacterial effects.18 However, the advantages of NSs, including their small size, high reactivity, and great capacity, may become potential lethal factors by inducing toxic effects on cells compared with their micro-sized counterparts.19 Thus, a suitable carrier is required for the wide application of AgSD nanosystems.
Thermoresponsive hydrogel is defined as a hydrogel undergoing phase transition from liquid to semisolid gel on exposure to physiological environments.20 Thermoresponsive hydrogels possess most of the desirable characteristics to be an “ideal dressing”.42 Moreover, the unique in situ formation of thermoresponsive hydrogel enables a thorough coverage of wound site regardless of wound size, shape, and depth. Besides, a thermoresponsive hydrogel was easily applied and removed with remarkably improved compliance in patients.21,22
Poloxamer 407 (P407), also known as Pluronic F127, has been widely used for the formation of a thermoresponsive hydrogel matrix since 1972.23 P407 was demonstrated to possess the property of reverse thermal gelation, which meant that it could remain as a solution at a lower temperature and turn into a gel with the rise in temperature to a low critical solution temperature. In addition, P407 was reported to maintain good tolerability, because it did not induce irritation and sensitivity on the skin, which was proposed to be advantageous in preparing topical, rectal, and ocular formulations.24 No study to date combined AgSD NSs with the P407 thermoresponsive hydrogel as a wound dressing.
The objective of this study was to formulate a delivery system of AgSD NSs loaded within the P407 thermosensitive hydrogel. Nanosized AgSD was prepared by high-pressure homogenization. The most suitable thermosensitive hydrogel was screened by the orthogonal design method to adapt to the skin (32°C). Physicochemical characterizations of AgSD/NS with and within a thermosensitive hydrogel were analyzed using Fourier transform infrared (FTIR) spectroscopy, X-ray diffraction (XRD), and scanning electron microscopy (SEM). Finally, the cytotoxicity and antibacterial activity of AgSD/NS were estimated to ensure its safety and efficacy.
Materials and methods
Materials
AgSD was purchased from Jinyu Pharmacy Company (Nanyang, People’s Republic of China). P407 and poloxamer 188 (P188) were purchased from BASF, Ludwigshafen, Germany. Glycerol (Gly), ethanol, ammonium solution, dimethyl sulfoxide (DMSO), phenol, and phosphoric acid were ordered from Sinopharm Chemical Reagent Co (Beijing, People's Republic of China). Lauryl sodium sulfate was procured from VWR International, Radnor, PA, USA. Methyl thiazolyl tetrazolium (MTT) was purchased from Sigma-Aldrich Co. (St Louis, MO, USA). Acetonitrile and methanol were obtained from Thermo Fisher Scientific (Waltham, MA, USA) with an HPLC grade. All other reagents were of analytical grade. Purified water used in this study was prepared using a Mille-Q (EMD Millipore, Billerica, MA, USA).
Preparation of AgSD NSs
AgSD/NSs were prepared by the high-pressure homogenization method, and 1.5% P407 (w/v%) solution was prepared as a dispersion solution. Then, 20 g of AgSD coarse powder was added into 180 mL of prepared 1.5% P407 solution. To obtain a uniform system, the mixed solution was pre-milled using Ultra Turrax (T25; IKA, Staufen, Germany) at 10,000 rpm for 10 minutes. The uniform suspensions were then circulated at 200, 400, 600, and 800 bar for two circles, followed by 20 circles at 1,200 and 1,400 bar using AH100D high-pressure homogenizer (ATS Engineering Inc., Shanghai, People’s Republic of China) to prepare AgSD NSs.25,26,13 Then, the average particle size (Z-average) and polydispersity indexes (PIs) were detected using Malvern Zetasizer (3000SH; Malvern Instruments, Malvern, UK) to investigate the short-term stability of AgSD NSs. The samples were collected on days 1, 3, 5, 7, 14, 30, 60, and 180 for determining AgSD/NS stability. Day 1 was the day of AgSD/NS production.
Dissolution behavior of AgSD/NS
The dissolution process of AgSD NSs was tested on a rotary shaker, and 100 mL of synthetic serum electrolyte solution (SSES) at 37°C was prepared as dissolution media.27 The sink condition was fulfilled since the saturated solubility was previously determined to be 0.98 mg/mL.28 Samples containing an equal amount of AgSD (5 mg) were dispersed into 100 mL of SSES and shaken at 80 rpm. Then, 1 mL of the medium was withdrawn at predetermined intervals and centrifuged at 12,000 rpm for 5 minutes. Further, 1 mL of SSES was supplemented immediately into each sample. The concentration of AgSD was estimated using Agilent 1290 ultra-HPLC (UHPLC). The chromatographic separation of AgSD was performed with an Agilent Zorab SB C18 (3.5 μm particle size, 3.0 × 100 mm) at 30°C. The mobile phase was the mixture of 0.1% phosphoric acid and acetonitrile with a ratio of 94:6. The flow rate was 0.80 mL/min. The dissolution rate of AgSD coarse power suspensions (AgSD/bulk) was also examined under the same conditions.29
Preparation of AgSD/NS-loaded thermosensitive hydrogel
Gels were prepared on the weight/weight basis using the cold method described by Schmolka.30 In brief, various components such as P407, P188, and Gly were weighed in distilled water to get the final mixture comprising 18% P407, 2% P188, and 10% Gly. The solutions were preserved at 4°C for no less than 24 hours to ensure complete dissolution. To prepare an AgSD/NS-loaded hydrogel (AgSD/NS gel), 10 mL of AgSD NS (100 mg/mL) was added slowly into 90 mL of hydrogel solutions, and the mixture was stirred for 10 minutes. The drug was accurately measured using UHPLC to ensure the homogeneity of the mixture.
Physicochemical characterization
Gelation temperature measurement
The solution–gel transition temperature (Tsol–gel) of thermoresponsive hydrogel within or outside AgSD/NS was measured using a Brookfield digital viscometer (model DV-III; Brookfield Engineering Laboratories, Inc., Middleboro, MA, USA). This viscometer was equipped with a temperature control unit so that the temperatures of samples were adjusted by circulating the water through the thermostated water jacket.
Briefly, 3 mL of samples were added into a small sample adaptor. A spindle (RV14) were used with a rotation speed of 10 rpm. The gelation temperature (Tsol–gel) was graphically determined to be the inflection point of temperature at which the apparent viscosity (cP) witnessed an abrupt change. The experiment was repeated three times, and the average value was obtained.
SEM
The surfaces of hydrogels within or outside AgSD were observed using a scanning electron microscope (Hitachi SU8010; Hitachi Ltd., Tokyo, Japan). The free dried hydrogel samples were spurted with a thin layer of gold under vacuum. The microphotographs of different samples were obtained with electron beam under 30 kV energy. The magnification ranged from 20× to 800,000×.
FTIR spectroscopy
FTIR spectroscopy was applied to identify drug–excipient interactions. The infrared spectrograms of AgSD coarse, dispersion of AgSD NS, copolymer hydrogel, and AgSD-loaded hydrogel were analyzed using FTIR spectroscopy (Nicolet Instrument Corporation, Madison, WI, USA) at the scan range of 450–4,000 cm−1 and resolution of 2 cm−1. KBr was used to prepare the samples. The spectra were baseline corrected with the Omnic version 8.2 software.
XRD
A D8 advance X-ray diffractometer (Bruker AXS Inc., Madison, WI, USA) was used to detect the physical state of AgSD in different formulations (Cu Kα radiation, voltage 30 kV). XRD patterns of the AgSD coarse and AgSD/NS were recorded to evaluate the possible effects of high pressure on the physicochemical property of AgSD. Besides, the characteristics of drugs embedded or not embedded into the copolymer were compared. X-ray scanning was employed at diffraction angles of 2θ ranging from 5° to 50° with a rate of 10°/min.
Drug release behavior
The AgSD release from different formulations was measured using the dialysis bag method in an SSES. The dialysis bag (molecular weight cutoff [MWCO] 8–14 kD; Solarbio Life Science, Beijing, People’s Republic of China) was washed thoroughly with deionized water before use to get rid of the preservative. AgSD/NS, AgSD/bulk, AgSD/NS gel, and commercial AgSD/cream were evaluated in the present study. All samples contained 10 mg AgSD were placed into dialysis bags. The bags were tied with threads at both ends and immersed in a beaker with 250 mL of SSES. The system was shaken at 100 rpm in a water bath at 32°C. Then, 1 mL of the medium was withdrawn at predetermined intervals and centrifuged at 12,000 rpm for 5 minutes. Further, 1 mL of SSES was supplemented immediately into each sample. The concentration of AgSD was estimated using UHPLC. The chromatographic separation of AgSD was performed on a C18 column (3.5 μm, 3.0 × 100 mm) at 30°C. The mobile phase was the mixture of 0.1% phosphoric acid and acetonitrile with a ratio of 94:6 (v/v). The flow rate was 0.80 mL/min. The drug release profiles of different formulations were compared with one-way ANOVA.
In vitro cytotoxicity assays
L929 mouse fibroblasts, obtained from Cell Bank of Chinese Academy of Sciences (Beijing, People's Republic of China), were cultured in Roswell Park Memorial Institute (RPMI) medium supplemented with 10% FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin. The culture was maintained at 37°C in a wet atmosphere containing 5% CO2. The MTT assay was used to evaluate the cytotoxicity. Briefly, the samples were sterilized with ultraviolet radiation for 30 minutes. The hydrogel was immersed into RPMI medium at 37°C for 24 hours to acquire extractants. Meanwhile, 100 μL of L929 fibroblast culture medium with a density of 5×104/mL cells was seeded in a 96-well plate and cultivated for 24 hours at 37°C. The prepared extraction medium of different formulations was added after removing the original culture medium, and another 48-hour incubation was needed. RPMI 1640 medium was taken as the negative control. After that, 20 μL of MTT solution (5 mg/mL) was pipetted into each well following another 4-hour incubation at 37°C. Then, 150 μL of DMSO was added to dissolve the formazan crystals after removing the MTT solution. The OD was measured with the microplate reader (Spectra Max 90; Molecular Devices LLC, Sunnyvale, CA, USA) at 490 nm. The average value of six parallel samples was calculated, and the whole test was repeated thrice. The relative cell viability was calculated using Equation 1, in which ODsamples is the absorbance value for samples and ODnegative is the negative control.
|
|
In vitro antibacterial activity assay
Bacterial strains and cultivation condition
Three bacterial strains used in this study were as follows: Staphylococcus aureus (ATCC 25923), which is a gram-positive bacterium; and Escherichia coli (ATCC 25922) and Pseudomonas aeruginosa (ATCC 27853), which are gram-negative bacteria. The strains were cultivated at 37°C in Luria Bertani (LB) medium or LB agar medium. An isolated colony was picked and inoculated in normal saline for preparing bacterial suspensions of 0.5 McFarland standard. Then, the bacterial suspensions with the concentration of 108 colony-forming unit (CFU)/mL were obtained.
Minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC)
MIC and MBC were measured using the doubling dilution method in line with the guidelines of the Clinical and Laboratory Standards Institute to compare the antimicrobial activity of AgSD coarse powders with that of AgSD nanocrystals. First, 100 μL of LB medium was put into each well of the sterile 96-well microtiter plates. Both AgSD/bulk and AgSD/NS were diluted to 512 μg/mL using LB medium. Diluted solutions of AgSD/bulk and AgSD/NS were added separately to the setting well, which was followed by serial twofold dilutions. Then, 100 μL of prepared bacterial dispersions with concentration of 105 CFU/mL was pipetted into each well, except for the sterility control wells. Finally, the MIC value was evaluated visually by comparing the culture turbidity.
Next, 5 μL of culture medium from the dilutions that showed no visible bacterial growth was picked up to evaluate the MBC of the tested AgSD formulations. The selected incubation medium and two more concentrated dilutions were painted on sterile LB agar medium and incubated for 24 hours at 37°C. After that, the bacterial colonies were counted. The MBC was determined as the lowest concentration at which fewer than five colonies were detected on LB nutrient agar after incubation.
Inhibition zone
The inhibition zone of AgSD/bulk, AgSD/NS, and AgSD/NS-loaded hydrogels against S. aureus, E. coli, and P. aeruginosa was determined for comparing their bactericidal activities. Then, 100 μL of the bacterial suspension (108 CFU/mL) was spread on LB nutrient agar to prepare a confluent ground for bacterial growth. The wells with a diameter of 5 mm were acquired in the agar plates, and 50 μL of different samples were added into these pores. The penicillin and streptomycin solutions were treated as the positive control, and blank hydrogels served as the negative control. The plates were incubated overnight at 37°C, and the diameters of the inhibition zone (mm) were surveyed using a vernier caliper after deducting the original diameter of the well (5 mm) from the total inhibition zone diameter.
Statistical analyses
Data were expressed as mean ± SD. The differences between experimental groups were compared with one-way ANOVA using the SPSS software (version 21.0; IBM Corporation, Armonk, NY, USA). A P-value of <0.05 (95% level) was considered statistically significant.
Results and discussion
Preparation and dissolution evaluation of AgSD NSs
Poloxamer has been extensively used as a nonionic stabilizer for producing NSs because of its low toxicity.13,31–33 It has been applied in several pharmaceutical formulations, such as chemical medicines and polypeptide drugs.20
The average particle size and PI were monitored for 6 months to confirm the stability of AgSD/NS stabilized with 1.5% P407. Figure 1 clearly shows that AgSD/NS exhibited no dramatic change in the particle size even after 6-month storage at 4°C. The average particle size varied from 194.6 to 270.2 nm during this period. No significant increase in particle size was detected. PI usually indicates the stability of NSs, and a lower PI value normally means better long-term stability. According to Singh et al,34 a PI value of >0.5 denotes a wide range of particle size distribution, whereas a PI value of 0.1–0.25 signifies its narrow range. The PI of AgSD/NS fluctuated between 0.18 and 0.28 during 6-month storage, which represented an even distribution. The result indicated that 1.5% P407 was able to stabilize AgSD/NS at least in 6 months. P407, as a nonionic surfactant, was commonly used in topical formulations because it was less irritating compared with ionic surfactants. In addition, a suitable drug-to-working polymer ratio (steric stabilizer) was 20:1 to 2:1.35 As a result, the ratio of 20:3 between AgSD and P407 was selected in this study to prepare stable AgSD/NS. The possible mechanism of action of P407 as a stabilizer in this study was that the nanocrystals were stabilized by the surface adsorption of P407. In other words, the absorbance of P407 onto the surfaces of AgSD nanocrystals provided a steric stabilization effect. Moreover, the viscosity of P407 might also contribute to the stability of AgSD NSs.
Figure 2 shows that AgSD/NS witnessed a significant increase in the drug dissolution rate compared with the AgSD coarse powder. The dissolution of AgSD/NS was so fast that 77.9% AgSD was immediately dissolved in 3 minutes. Moreover, almost all drugs dissolved in SSES within 10 minutes. In contrast, no more than 20% of AgSD dissolved in AgSD/bulk suspensions even after 12 hours. The result indicated that converting the AgSD coarse powder into AgSD nanoparticles could significantly increase its dissolution rate, further improving its bioavailability.36
  | Figure 2 Dissolution rate of AgSD/NS and AgSD/bulk in an SSES. |
Physicochemical characteristics of AgSD/NS gel
Next, 1% AgSD/NSs were added into the blank gel to prepare AgSD/NS-loaded hydrogel. The mixture displayed ideal homogeneity with an average concentration of 9.4 mg/mL. A similar result was obtained after 1-month storage.
Gelation temperature measurement
Gelation temperature is the key parameter in forming an in situ thermosensitive gel. Tsol–gel is the temperature at which the liquid phase undergoes the transition from a solution to a gel. Figure 3 shows the typical viscosity vs temperature curves for both the blank gel and AgSD/NS gel formulations.
  | Figure 3 Effect of temperature on the viscosity of AgSD/NS gel and blank gel. |
The average Tsol–gel values of the two formulations are summarized in Table 1. It is clearly seen that AgSD/NS apparently increased the transition temperature of blank gel. The difference might be explained by one of the proposed mechanisms for the packaging of micelles and micelle entanglements.37 The addition of AgSD/NS might interfere with the P407 micellization process and alter the dehydration of hydrophobic poly(propylene oxide) (PPO) blocks.38,39 If the Tsol–gel of hydrogel is lower than 25°C, gelation occurs at a room temperature, leading to difficulty in administration on the wound. Meanwhile, if the Tsol–gel is higher than 32°C, the mixtures still maintain in a liquid form at physiological temperature, leading to drug leakage.40 This means that the suitable limit of Tsol–gel for surgical dressing is from 25°C to 32°C. The AgSD/NS gel with an average Tsol–gel of 29.8°C is likely to spread easily at ambient temperature but gel quickly when in contact with skin (32°C).
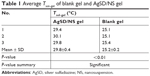  | Table 1 Average Tsol–gel of blank gel and AgSD/NS gel |
SEM
Figure 4A and B shows that AgSD coarse powders were minimized into nanoscales in the presence of the stabilizers. The particle size of AgSD ranged from 200 to 500 nm, which was in accordance with the result of Zetasizer.
Furthermore, Figure 4 shows the SEM photographs of blank gel (Figure 4C and D) and AgSD/NS gel (Figure 4E and F). Both micrographs show that the pores were well interconnected throughout the scaffold matrix like a honeycomb. Interestingly, the blank gel exhibited clean and smooth cross-linked walls, while the drug-loaded hydrogel formed a rough framework. Moreover, the AgSD/NS gel seemed to have a larger cross-linking intensity compared with the blank gel. It might indicate that the AgSD NSs were mostly wrapped in the hydrogel. The packaging of AgSD/NS might have occurred in the process of entanglement and aggregation of poloxamer.41 Besides, the hydrogel containing AgSD/NS still exhibited evenly cross-linked walls, indicating a uniform drug distribution.
FTIR
FTIR spectrophotometry was used to identify functional groups. The possible interactions between AgSD and the polymeric matrix of hydrogel were clarified using FTIR. Figure 5 shows the infrared spectra of AgSD coarse powders (AgSD/bulk) and AgSD/NS. The major characteristic absorption peaks of AgSD in the FTIR spectrum could be identified on the spectra of AgSD/bulk and AgSD/NS at 3,390, 3,340, 1,654, 1,599, 1,560, 1,500, 1,420, 1,233, 1,128, and 1,077 cm−1, although the peaks of AgSD/NS were of lesser intensity compared with those of AgSD/bulk. It was concluded that the homogenization process had no influence on the chemical properties of AgSD.
  | Figure 5 FTIR spectra of AgSD/NS, AgSD bulk, blank gel, and AgSD/NS gel. |
In addition, the blank gel and AgSD-loaded hydrogel (AgSD/NS gel) were compared to identify the possible influence of poloxamer on AgSD. Several characteristic peaks of AgSD at 1,599 cm−1 were observed, which might ascribe to the packaging of poloxamer. The FTIR for blank poloxamer hydrogel showed characteristic peaks at 2,877 and 3,379 cm−1, which were due to the stretching vibration of −CO and −OH. After incorporating AgSD/NS, these characteristic peaks of poloxamer were reserved, which were indicative of no chemical reaction between AgSD and poloxamer. These results demonstrated that the chemical characteristics of the AgSD were preserved during the formation of hydrogel.
These results confirmed that AgSD did not participate in the chemical interaction with the components of the thermosensitive hydrogel, but only loaded uniformly on the hydrogel. The structural integrity of AgSD carried by the poloxamer hydrogel also represented the preservation of antibacterial activity.
XRD
The structural composition and crystalline state of materials were determined by the XRD method. Peaks with high intensity and narrow base width were related to crystalline materials, whereas wide base peaks were related to amorphous substances.42
Figure 6 displays the XRD patterns of AgSD/bulk, AgSD/NS, and blank hydrogel in the range of 2θ 5°–50°. AgSD/NS prepared by high-pressure homogenization showed diffraction peaks with less intensity in the same range, representing a reduction in the crystalline form to the amorphous form. The results of X-ray scattering indicated that the incorporation of AgSD into the poloxamer hydrogel could significantly weaken its crystalline state.
  | Figure 6 X-ray diffractograms of samples of AgSD bulk (AgSD/bulk), AgSD/NS, and AgSD-loaded thermoresponsive hydrogel (AgSD/NS gel). |
Evaluation of the drug release properties
Diffusion through a dialysis membrane is a classic technique to evaluate the release from colloidal dispersions and topical formulations.43,44 The AgSD cream (1.0%) was prepared to compare the release properties of 1.0% AgSD/NS gel with those of the commercial formulation. Figure 7 illustrates the corresponding drug release behaviors of AgSD/NS, AgSD/bulk, AgSD/NS-loaded gel, and commercial AgSD cream. Both AgSD/bulk and AgSD cream exhibited a lower accumulated drug release percentage after 24 hours (36.7% and 48.6%, respectively) because of the lower dissolution rate of AgSD coarse powder. Besides, the incorporation into the poloxamer gel could dramatically decrease the AgSD release compared with AgSD NSs alone. The drug release from AgSD NSs was found to be 92.0% after 10 minutes. However, only 17.2% AgSD was released from the AgSD/NS gel after 10 minutes. Further, 48.2% AgSD was released from the AgSD/NS hydrogel at a constant rate in the first hour and more than 82.4% AgSD was released in 12 hours. The drug release behaviors of AgSD/NS and AgSD/NS gel exhibited a significant difference (P<0.01). This result was in accordance with the microstructure of AgSD/NS gel under SEM. The AgSD might be packaged into a poloxamer gel because of the entanglement of polypropylene monomer of P407 with the increase in temperature.45,46 Lamberti et al47 explained that nanoparticles were more efficiently taken up by cells compared with larger micromolecules. Hence, the decreased drug release rate from the hydrogel might contribute to the increased cell viability compared with AgSD NS alone.
  | Figure 7 In vitro release of AgSD from AgSD/NS gel, commercial AgSD cream, AgSD bulk, and AgSD NS solution. |
Different from the AgSD/NS gel, AgSD was released rather slowly from the marketed AgSD cream. At the end of the study (24 hours), AgSD released from AgSD cream and AgSD/NS gel was 48.6% and 77.7%, respectively. A significant difference (P<0.01) was observed in the drug release profiles of AgSD/NS gel and AgSD cream. This improvement in the drug release behavior of AgSD/NS gel might be explained by the decrease in drug particles and an improvement in the hydrophilic performance of drug carrier.26 The release mechanism was described as a combination of gel erosion and drug diffusion.42 The rapid hydration of the poloxamer and Gly resulted in a significant drug release. The gradual erosion of hydrogel offered a continuous release of AgSD in 24 hours. The faster drug release of AgSD from the AgSD/NS gel might lead to an improved antimicrobial activity compared with the commercial AgSD cream.
Figure 7 shows that combining AgSD/NS with the poloxamer thermoresponsive hydrogel could significantly decrease drug release from NSs. Besides, the AgSD/NS-loaded thermosensitive hydrogel might display a higher antibacterial activity compared with the commercial AgSD cream because of the quick and constant drug release profile.
In vitro cytotoxicity assays
The relative cell viability (%) of L929 fibroblast cells after 48-hour incubation is shown in Figure 8. All formulations with AgSD were at the same concentration of 1% (w/w). Nanosized AgSD exhibited less cell viability compared with AgSD micro-sized bulk particles. It might contribute to the increased interaction between nanoparticles and L929 fibroblasts because of the larger surface area and higher dissolution of AgSD nanoparticles.29 However, the inhibitory effects of AgSD nanoparticles significantly reduced after combination with the thermosensitive hydrogel; the relative cell viability increased from 60.7% to 90.5%. It implied that the release of AgSD nanoparticles was restricted by the poloxamer cross-linked network. This outcome was in accordance with the morphological results of SEM. Hence, the incorporation into the hydrogel decreased the possible cytotoxicity of AgSD. The blank gel proclaimed higher cell viability with zero-degree cell cytotoxicity. A similar result was reported by Schmolka,23 who concluded that the use of poloxamer was not hazardous in medical application. Interestingly, the commercial AgSD cream expressed lower cell viability compared with AgSD bulk powders with the same AgSD concentration. The increased cytotoxicity of AgSD cream might ascribe to the effect of surfactants existing in the cream.48 The cytotoxicity results of AgSD/NS gel and AgSD/NS suggested that the cytotoxicity of AgSD/NS easily decreased by incorporation into the hydrogel. Meanwhile, the poloxamer exhibited fine biocompatibility, which was expected to be vital in the fine-tuning of delivery system characteristics.
In vitro antibacterial activity assay
MIC is defined as the lowest concentration that inhibits the growth of bacteria, while MBC represents the lowest concentration with no bacterial growth after incubation. Table 2 summarizes the MIC and MBC values of AgSD with different particle sizes against both gram-negative bacteria (P. aeruginosa and E. coli) and gram-positive bacteria (S. aureus). The table indicates that AgSD/NS significantly increased the antibacterial activity compared with AgSD/bulk. The MIC of AgSD/bulk was twice as high as that of AgSD/NS for all the three kinds of bacteria. The MBC of AgSD/bulk doubled that of AgSD/NS against S. aureus. However, the MBC of AgSD/bulk against gram-negative bacteria was four times as high as that of AgSD/NS. The antibacterial effect of silver nanoparticles was attributed to their small size and high surface-to-volume ratio, which enabled their close interaction with microbial membranes. Every coin has two sides. Although the cytotoxicity of AgSD increased because of nanosize, the antibacterial activity increased.
The zone of inhibition value was measured further to better compare the bactericidal effect of AgSD/NS with that of coarse powders. Table 3 clearly reveals that the bactericidal effect of nanosized AgSD significantly increased compared with that of AgSD/bulk against all three selected bacteria (P<0.05). Besides, the incorporation of AgSD/NS into the poloxamer hydrogel could partially decrease the bactericidal effect of AgSD/NS against S. aureus and E. coli. Interestingly, the hydrogel only slightly changed the antibacterial activity of AgSD/NS against P. aeruginosa. Since the inhibition zone of AgSD NSs was larger than that of AgSD/Bulk, it was inferred that the superior antibacterial activity of AgSD/NS was probably due to the close interaction of nanosized silver with bacteria.
Conclusion
A new AgSD NS-loaded thermosensitive hydrogel with distinctive advantages over the commercially available AgSD cream was formulated in the present study.
In this study, stable AgSD NSs were formulated using the high-pressure homogenization technique, while 1.5% P407 was added as a stabilizer. AgSD/NS was found to be stable after 6-month storage. Besides, the drug dissolution from nanosized AgSD significantly increased compared with that from AgSD coarse powders, indicating enhanced saturation solubility and antibacterial activity. Further, 18% P407 was selected as the optimized thermoresponsive hydrogel sediment with 2% P188 to control gelation temperature and 10% Gly to retain moisture. The optimized AgSD/NS thermoresponsive hydrogel exhibited the gelation temperature of approximately 30°C. The hydrogel with Tsol–gel of 30°C was perfect for topical application on skin, because it could spread easily at ambient temperature but gel quickly when in contact with skin (32°C). The SEM photograph of AgSD/NS gel clearly revealed the scaffold matrix like a honeycomb with AgSD packaged in it. Besides, the results of XRD and FTIR demonstrated that the physicochemical properties of AgSD were reserved after incorporation into the hydrogel. The results of MTT assay showed that the poloxamer hydrogel could effectively decrease the possible cytotoxicity of nanosized AgSD. More importantly, the AgSD/NS gel exhibited an improved antibacterial activity against common pathogens invading burn wounds. In conclusion, nanosized AgSD could significantly improve the bactericidal effect while the use of in situ gelling vehicles of P407 could effectively and safely decrease the cytotoxicity of AgSD/NS. Further in vivo experiments should be conducted in the future to better compare the antibacterial activity of AgSD/NS gel prepared in this study with that of commercial products. It is a challenge to design drug carriers that maximize antimicrobial activity and minimize cellular toxicity. This poloxamer thermoresponsive hydrogel delivery system together with nanotechnology might be an ideal choice for other poor soluble drugs for burn wound therapy.
Acknowledgments
This work has been funded by the Chinese National Science and Technology Key Projects (number 2016ZX09J16103-001-001). The authors thank Dr Zheng for technical assistance in the measurement of gelation temperature. They also thank Mr Liu Bo for helpful discussion during the manuscript writing.
Disclosure
The authors report no conflicts of interest in this work.
References
Church D, Elsayed S, Reid O, Winston B, Lindsay R. Burn wound infections. Clin Microbiol Rev. 2006;19(2):403–434. | ||
World Health Organization [homepage on the Internet]. Available from: http://www.who.int/zh/news-room/fact-sheets/detail/burns. Accessed December 6, 2018. | ||
Miller AC, Rashid RM, Falzon L, Elamin EM, Zehtabchi S. Silver sulfadiazine for the treatment of partial-thickness burns and venous stasis ulcers. J Am Acad Dermatol. 2012;66(5):e159–e165. | ||
Hoffmann S. Silver sulfadiazine: an antibacterial agent for topical use in burns. A review of the literature. Scand J Plast Reconstr Surg. 1984;18(1):119–126. | ||
Dellera E, Bonferoni MC, Sandri G, et al. Development of chitosan oleate ionic micelles loaded with silver sulfadiazine to be associated with platelet lysate for application in wound healing. Eur J Pharm Biopharm. 2014;88(3):643–650. | ||
Kumar PM, Ghosh A. Development and evaluation of silver sulfadiazine loaded microsponge based gel for partial thickness (second degree) burn wounds. Eur J Pharm Sci. 2017;96:243–254. | ||
Muller MJ, Hollyoak MA, Moaveni Z, Brown TL, Herndon DN, Heggers JP. Retardation of wound healing by silver sulfadiazine is reversed by Aloe vera and nystatin. Burns. 2003;29(8):834–836. | ||
Hoeksema H, Vandekerckhove D, Verbelen J, Heyneman A, Monstrey S. A comparative study of 1% silver sulphadiazine (Flammazine®) versus an enzyme alginogel (Flaminal®) in the treatment of partial thickness burns. Burns. 2013;39(6):1234–1241. | ||
Selçuk CT, Durgun M, Özalp B, et al. Comparison of the antibacterial effect of silver sulfadiazine 1%, mupirocin 2%, Acticoat and octenidine dihydrochloride in a full-thickness rat burn model contaminated with multi drug resistant Acinetobacter baumannii. Burns. 2012;38(8):1204–1209. | ||
Niazi ZB, Essex TJ, Papini R, Scott D, Mclean NR, Black MJ. New laser Doppler scanner, a valuable adjunct in burn depth assessment. Burns. 1993;19(6):485–489. | ||
Bombaro KM, Engrav LH, Carrougher GJ, et al. What is the prevalence of hypertrophic scarring following burns? Burns. 2003;29(4):299–302. | ||
Vloemans AF, Hermans MH, van der Wal MB, Liebregts J, Middelkoop E. Optimal treatment of partial thickness burns in children: a systematic review. Burns. 2014;40(2):177–190. | ||
Deng J, Huang L, Liu F. Understanding the structure and stability of paclitaxel nanocrystals. Int J Pharm. 2010;390(2):242–249. | ||
Mishra PR, Al Shaal L, Müller RH, Keck CM. Production and characterization of Hesperetin nanosuspensions for dermal delivery. Int J Pharm. 2009;371(1–2):182–189. | ||
Merisko-Liversidge E, Liversidge GG, Cooper ER. Nanosizing: a formulation approach for poorly-water-soluble compounds. Eur J Pharm Sci. 2003;18(2):113–120. | ||
Wang L, Du J, Zhou Y, Wang Y. Safety of nanosuspensions in drug delivery. Nanomedicine. 2017;13(2):455–469. | ||
Dizaj SM, Vazifehasl Z, Salatin S, Adibkia K, Javadzadeh Y. Nanosizing of drugs: effect on dissolution rate. Res Pharm Sci. 2015;10(2):95–108. | ||
Venkataraman M, Nagarsenker M. Silver sulfadiazine nanosystems for burn therapy. AAPS PharmSciTech. 2013;14(1):254–264. | ||
Khan I, Saeed K, Khan I. Nanoparticles: properties, applications and toxicities. Arab J Chem. 2017. | ||
Gratieri T, Gelfuso GM, Rocha EM, Sarmento VH, de Freitas O, Lopez RF. A poloxamer/chitosan in situ forming gel with prolonged retention time for ocular delivery. Eur J Pharm Biopharm. 2010;75(2):186–193. | ||
Faulkner DM, Sutton ST, Hesford JD, et al. A new stable pluronic F68 gel carrier for antibiotics in contaminated wound treatment. Am J Emerg Med. 1997;15(1):20–24. | ||
Yim H, Yang HT, Cho YS, et al. A clinical trial designed to evaluate the safety and effectiveness of a thermosensitive hydrogel-type cultured epidermal allograft for deep second-degree burns. Burns. 2014;40(8):1642–1649. | ||
Schmolka IR. Artificial skin. I. Preparation and properties of pluronic F-127 gels for treatment of burns. J Biomed Mater Res. 1972;6(6):571–582. | ||
Chiappetta DA, Sosnik A. Poly(ethylene oxide)-poly(propylene oxide) block copolymer micelles as drug delivery agents: improved hydrosolubility, stability and bioavailability of drugs. Eur J Pharm Biopharm. 2007;66(3):303–317. | ||
Müller RH, Jacobs C. Buparvaquone mucoadhesive nanosuspension: preparation, optimisation and long-term stability. Int J Pharm. 2002;237(1–2):151–161. | ||
Xu H, Yuan XD, Shen BD, et al. Development of poly(N-isopropylacrylamide)/alginate copolymer hydrogel-grafted fabrics embedding of berberine nanosuspension for the infected wound treatment. J Biomater Appl. 2014;28(9):1376–1385. | ||
Tsipouras N, Rix CJ, Brady PH. Solubility of silver sulfadiazine in physiological media and relevance to treatment of thermal burns with silver sulfadiazine cream. Clin Chem. 1995;41(1):87–91. | ||
Morsi NM, Abdelbary GA, Ahmed MA. Silver sulfadiazine based cubosome hydrogels for topical treatment of burns: development and in vitro/in vivo characterization. Eur J Pharm Biopharm. 2014;86(2):178–189. | ||
Gao L, Gan H, Meng Z, et al. Evaluation of genipin-crosslinked chitosan hydrogels as a potential carrier for silver sulfadiazine nanocrystals. Colloids Surf B Biointerfaces. 2016;148:343–353. | ||
Schmolka IR. A comparison of block copolymer surfactant gels. J Am Oil Chem Soc. 1991;68(3):206–209. | ||
Paolicelli P, Cerreto F, Cesa S, et al. Influence of the formulation components on the properties of the system SLN-dextran hydrogel for the modified release of drugs. J Microencapsul. 2009;26(4):355–364. | ||
Gupta S, Samanta MK, Raichur AM. Dual-drug delivery system based on in situ gel-forming nanosuspension of forskolin to enhance antiglaucoma efficacy. AAPS Pharm Sci Tech. 2010;11(1):322–335. | ||
Xiong XY, Tam KC, Gan LH. Effect of enzymatic degradation on the release kinetics of model drug from Pluronic F127/poly(lactic acid) nano-particles. J Control Release. 2005;108(2–3):263–270. | ||
Singh SK, Vaidya Y, Gulati M, Bhattacharya S, Garg V, Pandey NK. Nanosuspension: principles, perspectives and practices. Curr Drug Deliv. 2016;13(8):1222–1246. | ||
Kesisoglou F, Panmai S, Wu YH. Nanosizing-oral formulation development and biopharmaceutical evaluation. Adv Drug Deliv Rev. 2007;59(7):631–644. | ||
Patravale VB, Date AA, Kulkarni RM. Nanosuspensions: a promising drug delivery strategy. J Pharm Pharmacol. 2004;56(7):827–840. | ||
Cabana A, Ait-Kadi A, Juhász J. Study of the gelation process of polyethylene oxidea-polypropylene oxideb-polyethylene oxidea copolymer (poloxamer 407) aqueous solutions. J Colloid Interface Sci. 1997;190(2):307–312. | ||
Dumortier G, Grossiord JL, Zuber M, Couarraze G, Chaumeil JC. Rheological study of a thermoreversible morphine gel. Drug Devel Indust Pharm. 1991;17(9):1255–1265. | ||
Shawesh A, Kallioinen S, Antikainen O, Yliruusi J. Influence of storage time and temperature on the stability of indomethacin Pluronic F-127 gels. Pharmazie. 2002;57(10):690–694. | ||
Koffi AA, Agnely F, Ponchel G, Grossiord JL. Modulation of the rheological and mucoadhesive properties of thermosensitive poloxamer-based hydrogels intended for the rectal administration of quinine. Eur J Pharm Sci. 2006;27(4):328–335. | ||
Liang X, Guo C, Ma J, Wang J, Chen S, Liu H. Temperature-dependent aggregation and disaggregation of poly(ethylene oxide)-poly(propylene oxide)-poly(ethylene oxide) block copolymer in aqueous solution. J Phys Chem B. 2007;111(46):13217–13220. | ||
Jodar KS, Balcão VM, Chaud MV, et al. Development and characterization of a hydrogel containing silver sulfadiazine for antimicrobial topical applications. J Pharm Sci. 2015;104(7):2241–2254. | ||
Dharashivkar SS, Sahasrabuddhe SH, Saoji AN. Niosomally encapsulated silver sulfadiazine gel for burn treatment. J Microencapsul. 2015;32(2):137–142. | ||
Barkat MA, Harshita, Ahmad I, et al. Nanosuspension-based aloe vera gel of silver sulfadiazine with improved wound healing activity. AAPS Pharm Sci Tech. 2017;18(8):3274–3285. | ||
Jindal N, Mehta SK. Nevirapine loaded Poloxamer 407/Pluronic P123 mixed micelles: Optimization of formulation and in vitro evaluation. Colloids Surf B Biointerfaces. 2015;129:100–106. | ||
Lu C, Liu M, Fu H, et al. Novel thermosensitive in situ gel based on poloxamer for uterus delivery. Eur J Pharm Sci. 2015;77:24–28. | ||
Lamberti M, Zappavigna S, Sannolo N, Porto S, Caraglia M. Advantages and risks of nanotechnologies in cancer patients and occupationally exposed workers. Expert Opin Drug Deliv. 2014;11(7):1087–1101. | ||
Nogueira DR, Mitjans M, Morán MC, Pérez L, Vinardell MP. Membrane-destabilizing activity of pH-responsive cationic lysine-based surfactants: role of charge position and alkyl chain length. Amino Acids. 2012;43(3):1203–1215. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.