Back to Journals » International Journal of Nanomedicine » Volume 15
Silver Nanoparticles for the Therapy of Tuberculosis
Authors Tăbăran AF , Matea CT, Mocan T , Tăbăran A, Mihaiu M, Iancu C, Mocan L
Received 5 December 2019
Accepted for publication 15 February 2020
Published 31 March 2020 Volume 2020:15 Pages 2231—2258
DOI https://doi.org/10.2147/IJN.S241183
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Alexandru-Flaviu Tăbăran,1,2,* Cristian Tudor Matea,2,* Teodora Mocan,2,3,* Alexandra Tăbăran,4,* Marian Mihaiu,4,* Cornel Iancu,2,5,* Lucian Mocan2,3,*
1Department of Pathology, University of Agricultural Sciences and Veterinary Medicine, Cluj-Napoca, Romania; 2Department of Nanomedicine, Regional Institute of Gastroenterology and Hepatology, Cluj-Napoca, Romania; 3Department of Physiology, University of Medicine and Pharmacy, Cluj-Napoca, Romania; 4Department of Public Health and Food Hygiene, University of Agricultural Sciences and Veterinary Medicine, Cluj-Napoca, Romania; 5Third Surgery Department, University of Medicine and Pharmacy, Cluj-Napoca, Romania
*These authors contributed equally to this work
Correspondence: Teodora Mocan
Department of Physiology, University of Medicine and Pharmacy Cluj-Napoca, Romania 1 Clinicilor Street, Cluj-Napoca 40006, Romania
Tel +40 264 598575
Fax +40 264 599814
Email [email protected]
Abstract: Rapid emergence of aggressive, multidrug-resistant Mycobacteria strain represents the main cause of the current antimycobacterial-drug crisis and status of tuberculosis (TB) as a major global health problem. The relatively low-output of newly approved antibiotics contributes to the current orientation of research towards alternative antibacterial molecules such as advanced materials. Nanotechnology and nanoparticle research offers several exciting new-concepts and strategies which may prove to be valuable tools in improving the TB therapy. A new paradigm in antituberculous therapy using silver nanoparticles has the potential to overcome the medical limitations imposed in TB treatment by the drug resistance which is commonly reported for most of the current organic antibiotics. There is no doubt that AgNPs are promising future therapeutics for the medication of mycobacterial-induced diseases but the viability of this complementary strategy depends on overcoming several critical therapeutic issues as, poor delivery, variable intramacrophagic antimycobacterial efficiency, and residual toxicity. In this paper, we provide an overview of the pathology of mycobacterial-induced diseases, andhighlight the advantages and limitations of silver nanoparticles (AgNPs) in TB treatment.
Keywords: nanoparticles, antimycobacterial, Mycobacterium, tuberculosis, macrophage, granuloma
Introduction
The emergence of multidrug-resistance, the intercurrent immunosuppressive diseases, the relatively low-output and high costs of newly-approved antituberculous antibiotics, and the partially protective vaccines, represents the main cause of the current status of tuberculosis as a regionally re-emerging and global health problem1–4 slowing in the same time the progress towards TB eradication. Tuberculosis infects globally more than one-third of human population,5 and despite the lastest progress, it remains according to the latest WHO report the world’s leading infectious-bacterial cause of deaths among adults, accounting only in 2018 more than 1.5 million deaths and 10 million new cases.6,7 Moreover, in some endemic areas, TB was the first cause of hospital death.8
Tuberculosis (TB) is a zoonotic and anthropozoonotic disease with a complex pathogenesis, produced by bacteria from Mycobacterium tuberculosis complex (MtbC), mainly M. tuberculosis, and in a lesser amount by the infections with other mycobacteria such as M. bovis, M. africanum, M. caprae, M. canetti, and occasionally Mycobacterium pinnipedii or M. microti.9–11 For some newly included members of MtbC as M. mungi,12 the exact role in human tuberculosis is currently poorly understood. MtbC bacteria are nonmotile and non-sporulated bacilli with a distinctively thick and lipid-rich cell wall included in the Actinomycetales order. The emergence of drug-resistant strains of MtbC observed in the last decades gives rise to additional challenges to the anti-TB prevention and control efforts.8
Nanotechnology and nanoparticle science are emerging disciplines connecting interdisciplinary areas of research such as chemistry, physics, and medicine providing innovative approaches and new-practical solutions for several critical-issues, including bacterial-induced infectious diseases.13,14 Metallic silver has a long history in medical applications, but its popularity markedly declined following the introduction and broad-usage of antibiotics.15 Nowadays, in the context of continuous rise in the rate of antibiotics consumption and “antibioresistance crisis”, silver in the form of AgNPs or in combination with classical antibiotics has made a remarkable comeback as a potential antibacterial molecule in the medicine and health care industry.16,17 Intracellular survival represents peculiar pathogenic factors of Mycobacteria, and this combined with the thick, hydrophobic (waxy) bacterial cell wall rich in mycolic acid and arabinogalactan contributes to the “phagocyte sabotage”, failure of the immune system to clear the septic focus, ensures the long-term persistence and furthermore, the local to systemic dissemination of infection.18 Recent reports have shown that AgNPs have a high antimycobacterial effect in both bacterial cultures and within macrophages,19,20 thus, the exploration of this new-concept of antimycobacterial-nanoparticles could change the current optics regarding TB-therapy.
This review explores in detail the main pathological features of mycobacteria and TB-pathogenesis, the AgNPs antibacterial mechanism of action per se and in combination with antibiotics, and not least the advantages and the limitation on using AgNP in TB therapy. Also, we up-to-date review of the main in vitro, in vivo and clinical studies assessing the antimycobacterial potential of AgNPs.
The Emergence of Drug Resistance Tuberculosis
Drug-resistant (DR) (defined as resistant to one or more antituberculosis drugs) and finally Multi-Drug-resistant tuberculosis (MDR) (defined as antibioresistace to at least rifampicin and isoniazid, the two most powerful antituberculosis drugs)7 is the most urgent and difficult provocation in TB treatment, a major public health concern, and an important cause or global TB reemergence noticed in the last three decades.21,22 New cases of both DR and MDR are typically expected to appear following the amplification of TB-resistance patterns through inadequate usage of antituberculosis chemotherapy, mainly the therapeutic use of ineffective-antibiotics formulations as first-line treatment and the premature stoppage of treatment and not last the inter-patient transmission of DR/MDR/XDR (Drug-/Multidrug-/Extensively drug-resistant tuberculosis) strains of TB, especially observed in areas with a high prevalence of DR/MDR-TB infections of following nosocomial transmission.7,23,24 Infection with MDR-TB strains is associated with a high mortality rate (up to 55%, compared to 4.5−17% mortality in infections with nonresistant TB-strains). A low treatment success despite the usage of appropriate second-line treatment, and typically spans a relatively short clinical course from diagnosis to death, especially in cases with concurrent infections like HIV or reduced body mass index.7,25-27
The current antituberculous therapy involves the first-line treatment during a 6 to 9 months, involving four antibiotics in sequential combination (isoniazid, rifampin, pyrazinamide, and ethambutol). In case of relapse or antibioresistace, the second-line therapy-treatment (during 18–24 months) of combination therapy with second-line drugs as aminosalicylic acid, fluoroquinolones, aminoglycosides, cycloserine, linezolid, and clofazimine, which are typically more toxic, more expensive and less efficient.28–30 In addition to poor efficiency in the case of MDR and XDR-strains of M tuberculosis, major adverse reactions (mainly hepatitis, gastrointestinal events) are present in more than 30% of cases following first-line therapy31 and in 83% following the second-line antituberculous therapy.32 Following the second-line antituberculous therapy, the adverse reactions are more severe and include mainly gastrointestinal and hepatic reactions, CNS adverse effects (including reactions raging from insomnia to psychosis and delirium), arthropathies, nephrotoxicity and electrolyte abnormalities, ototoxicity, hypothyroidism and hematological toxicity.30,33
The prolonged antituberculous therapy, limited antibacterial activity and intercurrent diseases are the main reason for patient noncompliance and finally, the induction of DR/MDR/XDR strains MtbC. The DR reaches approx. 20% among the previously treated TB patients, while the MDR tuberculosis appears in 4–10% of the same group population.34
The development of new antimycobacterial drugs and identification of new drug targets must take into account firstly the peculiarity of MtbC pathogeneses35 and the high adaptability of this classically known as an intracellular bacterial pathogen. The most intriguing property of MtbC assured by over 150 virulence factors5,36 is the capacity of MtbC to survive and multiply in certain conditions inside the macrophages, monocytes and dendritic cells.8
Mycobacterial Infection Pathology
Mycobacteria are classified according to their pathogenesis and role in human tuberculous as Mycobacterium tuberculosis complex (detailed above) and non-tuberculous mycobacteria (NTM, previously named “atypical mycobacteria”) (e.g. M. avium, M. kansasii, M. terrae, M. abscessus, etc).37,38
Non-tuberculous mycobacteria are ubiquitous, free-living, acid-fast bacteria, generally with reduced human pathogenicity (most of them are saprophytic) compared with M. tuberculosis complex. Even so, infections with both types of mycobacteria have several common characteristics and some NTM are used as infectious agents in experimental models of tuberculosis (e.g. Mycobacterium marinum in the zebrafish model of tuberculosis).39 This material is mainly intended to review the pathogenesis of bacteria included in the M. tuberculosis complex with few examples of NTM when adequate.
Although a dual intracellular and extracellular-type of infectivity is described for MtbC, the essential mechanism of disease in TB is based on the ability of mycobacteria to inhibit within the cells of the monocyte-macrophage system (MMS) the fusion of the phagosomes (containing microbes) with.18 Modulation of macrophage intracellular organelle compartment is essential not only for MtbC survival but also for its intracellular multiplication. Replication within the MMS-cells leads not only to the destruction of these cells but also of all cell populations surrounding the inflammatory focus. Within the affected organ and regional lymph nodes, this process will result is massive caseous necrosis and formation of a granulomatous reaction (caseating granuloma/tubercle)18,40 with a typical morphology.
Virulence and Pathogenesis Factors of Mycobacterium Tuberculosis
The complex pathogenicity of MtbC is determined by a plethora of virulence factors and literature dedicated to these factors is vast.5,41,42 This is particularly important in the disease process and gives TB a peculiar progression of biological events and interaction with the immune cells.
In a comprehensive review by Forrellad et al5 the MtbC virulence factors were classified in nine groups based on their activity, chemical structure and bacterial location: (1) virulence factors involved in the metabolism of lipids and fatty acids, (2) bacterial-wall proteins and lipoproteins (including secretion systems cell wall), (3) proteins suppressing the antimicrobial effectors of macrophage, (4) proteases (5) protein kinases (6) proteins involved in metal transport, (7) regulator gene, (8) proteins of unknown function and (9) other virulence proteins.5
The main virulence factors and the mechanisms by which they enhance MtbC infectious capability and resistance are summarized in Table 1.
 |
Table 1 A Synopsis of the m Tuberculosis Main Virulence Factor and Their Pathogenic Mechanism |
 |
 |  |
 |
Table 2 A Synopsis on Studies Using AgNPs in the Treatment of Mycobacteria-Induced Diseases |
Entry into Macrophages, Monocytes, and Dendritic Cells
The route of entry into the organism of MtbC is most often by inhalator route; the digestive pathway and other nonrespiratory route are less important for the TB transmission and are often used by other Mycobacteria of MtbC group (e.g. M. mungi is transmitted by an environmental pathway mainly through anal gland secretions and infected urine).76
Following the initial mechanical entrapment in the bilaminar protective mucus covering the respiratory or digestive system, mycobacteria enter in contact with the local macrophages (occasionally suspended in the respiratory mucous blanket) or, rarely, with intestinal M cells. Following mainly a specific ligand–receptor interaction with the membrane receptors (pattern recognition receptors-PPR) of macrophages, mycobacteria are engulfed by phagocytosis (Figure 1). Although macrophages are the main cells responsible for MtbC engulfment, all cells or the MMS, including monocytes and dendritic cells are capable of MtbC phagocytosis.39,77 Other professional-phagocytic cells as neutrophils, although are capable to phagocytose and destroy MtbC,78 have a less-known of the role in TB infection.
There are several phagocytic receptors (surface-expressed PPRs) that assures MtbC recognition and phagocytosis by macrophages/newly-recruited monocytes, such as those for: complement (CR1, CR3, and CR4), macrophage mannose receptors, CD14, surfactant protein receptors (surfactant protein A) (Sp-A), Fc (FcR) and macrophage scavenger receptors.18,36 These receptors recognize different components of MtbC: lipoarabinomannan (LAM) from the bacterial cell wall is recognized by CD14 and macrophage scavenger receptors, mannose, and mannose-capped-LAM by the macrophage mannose receptor, polyanionic macromolecules by the scavenger receptors and mycolyl-arabinogalactan by the intracellular NOD2 receptors.
Typically, the recognition and MMC-internalization of MtbC is mediated through the interaction of several of the PPRs listed above. The active types of PPRs influence the downstream inflammatory events and the fate of MtbC infection. Also, some intracellular PPRs as NOD2 (nucleotide oligomerization domain protein) are able to recognize the MtbC and further regulate the inflammatory process79 (mainly mediated through the NF-kB pathway). The involvement of these receptors could also be sequential, dominating different stages of the MtbC infection (engulfment in the early infections vs phagocytosis in systemically disseminated TB).
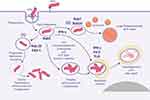 |
Figure 1 Spatiotemporal dynamic model of the possible fates of Mycobacterium tuberculosis (MTb) following macrophage phagocytosis (1) MTb can prevent early phagosome maturation and by the action of Rab20-trafficking, the ESX-1 will destabilize and disrupt the phagosome membrane allowing MTb direct access into the macrophage cytosol, followed in certain conditions by MTb survival and multiplication; (2) Some early phagosomes will undergo normal maturation, will fuse with the lysosomes and MTb will be killed (by reactive nitrogen intermediates, low pH, ROS, antimicrobial peptides and Fe deprivation mediated by iron scavengers, as lactoferrin, and NRAMP1);80 occasionally MTb can survive within the mature phagolysosome; (3) Blocking of the early phagosome maturation (mainly by inhibiting PI3P generation) followed by intravesicular MTb replication; (4) Delivery of the early endosomes or early-endosomes-to autolysosomes, where typically the activity of Mtb will be suppressed. Inspired from Philips et al81 and Schnettger et al.82 Figure 1 was created using BioRender. Abbreviations: NRAMP1, natural resistance-associated macrophage protein 1. |
Replication in Macrophages
Once internalized in macrophages (or other MMC), MtbC resides in a phagocytic vacuole where they are capable to delay or block the fusion of primary/early phagosomes with lysosomes (Figure 1) and thus to prevent the maturation, acidification of lysosomes, MtbC destruction, and activation of other antimycobacterial mechanisms.18,83 This process is actively mediated by MtbC and implies a reduction of proton ATPase amount within the phagosome and inhibition of Ca2 signals84,85 although the exact events that lead to this effect are still controversial. Several MtbC pathogenic factors as sulfolipids, trehalose dimycolate, lipoarabinomannan/mannose-capped-lipoarabinomannan (MC-LAM), tryptophan aspartate coat protein (TACO) and SapM are involved in this process.86–89 Finally, the mycobacterial phagosomes have the biochemical features of the early endosomes90 and are a favorable milieu for MtbC replication and systemic (lymphatic and/or sanguine) dissemination.
Even if these mechanisms seem to robustly block the phagosome-lysosome activity, several acute-phase cytokines (as IL-1 and tumor necrosis factor-TNF) and IFNγ can stimulate the MtbC-infected macrophages to overcome this dysregulation of the intracellular compartments and to regain the antimycobacterial activity (essentially by changing the macrophage polarization state-discussed below).
Tuberculosis Progression: Th1 to Th2 Response Imbalance
The polarization of the immune system activity is critical in the control and evolution of the MtbC infections. The CD4+ T lymphocytes orchestrate by the types of cytokines produced the inflammatory process (including the autoimmune processes) and are responsible for the normal multi-step evolution of a typical inflammation.
In tuberculosis, initially, a TH1 response induces a “classically” activated, M1-bactericidal macrophage (which mainly by secreting IFN-γ is able in certain limits to control the initial MtbC infection). Additional to TH1, also the TH17 cells are considered to induce a protective inflammatory response during MtbC infection.91
By the TH2 response CD4+ T secrete IL-4, IL-5, and IL-13 (promoting an “alternative” M2-activated macrophage); M2-polarised macrophages are commonly responsible for a protective effect against extracellular pathogen.85 The TH2 response typically is not inducing any protective activity against MtbC infection and replication. Moreover, TH2 response is responsible for the development of delayed (Type IV/T cell-mediated) hypersensitivity to MtbC antigens (used as a diagnostic tool – intradermal reaction/tuberculin test), granuloma formation (Figure 2) and progression of clinical tuberculosis.91–93 Although with a relative opposing effect, both TH1/TH2 inflammatory “phenotypes” usually coexists in MtbC infections. The modulation of these two components during the TB evolution is under the influence of several factors, among which individual-genetic variations (“genotype”), immune-system reactivity, microbial products (MtbC strain), intercurrent infections and physiological status.
Persistence of Viable Mycobacteria in Dead Cells and Necrotic Tissue
The capacity of mycobacteria to hijack the type of macrophage calls death is well known,94 but recently a new adaptive mechanism of mycobacteria was found. Mainly, following macrophage necrosis and neutrophil necrosis, a subset of mycobacteria exploits the necrotic cell-debris as a nutrient-rich growing substrate.95 More interestingly is the fact that tissular necrosis tends to enhance the overall mycobacterial replication.96 In a dynamic representation of this pathogenicity, the macrophage and neutrophil necrosis represents the starting point for a vicious cycle which continues with the uptake of the Mtb-infected cell debris from the newly recruited monocytes and neutrophils, de novo Mtb replication, sustained infection and finally the induction of cell death.96,97 The mycobacteria can utilize this growing niche for enhanced replication and survival, contributes to the success of mycobacteria to resist host defense and antibacterial therapy.95,97
Metallic Nanoparticles as Antiinfective Agents
Due to the increasing capacity of bacterial pathogens to acquire resistance to classical anti–infectious agents, nosocomial infections become a major cause of morbidity in patients of all age groups.98 Metallic nanoparticles have unique antiviral, antibacterial, and antiparasitic properties, making them promising candidates for future applications in the treatment of infectious diseases.99 From this class of molecules, zirconium oxide (ZrO2NPs)100 and Co3O4@ZrO2 (CoZ) core/shell NP101 proved to have an antibacterial effect against both gram-negative (E. coli and Pseudomonas aeruginosa) and positive bacteria (Bacillus subtilis and Staphylococcus aureus), copper oxide nanoparticles (CuONP) have shown antifungal (Candida albicans) and antibacterial effect against gram-positive (Staphylococcus aureus and Staphylococcus epidermidis) and gram-negative (E. coli and Proteus vulgaris) bacteria13,102 and iron oxide nanoparticles (FeONP) bactericidal effect against E. coli, Klebsiella pneumoniae, and Staphylococcus aureus.103 Gold nanoparticles (AuNP) have shown broad antibacterial effect against both gram-positive (Staphylococcus epidermidis) and gram-negative (E. coli) bacteria,104 and following appropriate functionalization a selective antibacterial effect against methicillin-resistant Staphylococcus aureus.105 Also, gold nanoparticles (AuNP) synthesized from marine seaweed Gracilaria verrucosa and Gelidium pusillum shows good biocompatibility to human embryonic kidney cells even at high concentrations of 100 and 150 μgmL−1.106,107
Additionally to their antimicrobial properties, some form of nanoparticles posses also antiproliferative-antitumoral effect as was recently shown for magnesium oxide nanoparticles (MgONPs) synthesized from the brown algae Sargassum wighitii,108 for titanium dioxide (TiO2) nanoparticles109,110 and for AgNPs synthesized from Enteromorpha compressa.111 Also, TiO2 nanoparticles show immunomodulatory effects,112 having a hypothetical application in infectious diseases with a hypersensitive component (including some phases of TB).
Moreover, some nanoparticles as copper nanoparticles (CuNPs) show catalytic degradation of organic dyes with application in wastewater treatment113 and interestingly, some nanoparticles as CuO and CuO/Cu(OH)2 show multimodal effects including in addition to antibacterial effects against E. coli and S. aureus also photocatalytic activity with potential application in wastewater management114 and a dose-dependent anticancer activity against tumor rat C6 cell line.115 A similar photocatalytic activity was shown also for zinc oxide nanoparticles synthesized from Cyanometra ramiflora.116
From the metallic nanoparticles, AgNPs are the most popular choice as anti–infectious nanoparticle-adjuvants.17 In conjunction with appropriate-drug delivery systems as chitosan117 AgNP per se or in combination with proanthocyanidin shown also a good in vitro antitumoral effect, against HT 29 human adenocarcinoma cells.118,119 The antibacterial properties of AgNP, their mechanism of action and especially their antimycobacterial effects will be further detailed.
Silver Nanoparticles as an Emerging Therapeutic Approach in Mycobacterial Infections
Silver per sei or incorporated in different compounds has long been used empirically as antimicrobial agents and tested since the XIX century as a natural antibiotic.15,120
In the quest for more efficient antimycobacterial drugs that are able to overcome the “classical” issues discussed above and partially responsible for the global TB status, the antibacterial peptides and nanoparticles gained recently special attention.19,121 Several classes of nanoparticles with intrinsic antibacterial and antibiofilm effects are proven,122 including metallic nanoparticles (e.g copper,123 iron,124 gold125 or silver-based126), carbon nanotubes,127 polysaccharides as chitosan128 and chitosan in conjunction with polycationic polymer129 or combinations of the above-mentioned antibacterial molecules as chitosan-gold NP.130 Among these antibacterial nanoparticles, due to their strong antibacterial activity and long-history of using silver as antiseptic, AgNPs have received most of the attention.131,132 This new paradigm in antituberculous therapy is based on the fact that the efficiency of Ag was already proven for many classes of bacteria and their microorganisms, the long tradition in using Ag salts as disinfectants,15 and due to the fact that unlike antibiotic drugs, most of the currently known pathogenic bacteria rarely develop resistance to metallic nanoparticles. The conditions under which this phenomenon can appear will be discussed in a separate section.
Antibacterial Effect and Mechanism of Silver Nanoparticles
Antibacterial action of AgNPs is mediated by several, generally, accepted-mechanisms (depicted in Figure 3) which in a biological context have a complementary action: 1) Direct contact with the bacteria components (biofilm and bacterial cell wall); 2) Release of bioactive ions (ex. Ag+ ions); 3) Disruption of several metabolic pathways; 4) Generation of reactive oxygen species (ROS); 5) Genotoxicity; 6) Alteration of cell wall and cytoplasm; 7) Inhibition of bacterial DNA replication; 8) Alteration of bacterial membrane permeability and ionic change.133–140
These effects are mediated mainly by the primary action of the AgNP, or by the release of Ag+ species and ROS will further disrupt the metabolic pathways and DNA. Although the main antibacterial effect of AgNPs is believed mediated by the release of bio-active Ag+ ions,141 more exactly, the AgNPs antibacterial mechanisms employ targeting multiple components in the bacterial cell,142 including bacterial wall (disruption and/or increasing the membrane permeability), tRNA (transfer ribonucleic acid), inactivating the respiratory chain (ATP depletion), enzyme and protein synthesis and DNA-binding (resulting cleavage, inhibition of replication).138–140
The overall bactericidal effect of AgNPs depends, in addition to the rate of Ag+ production, also on the AgNPs size and shape, overall NP surface area, type of coating/corona, and rate of Ag+ generation.133–140 The difference in the efficiency of AgNPs against Gram-positive, Gram-negative or acid-fast bacteria is believed to be mainly dependent on the structural and thickness differences of their cell walls. Usually, acid-fast bacteria due to the presence of a thicker-waxy cell wall have a stronger defense-system against Ag-NPs. As in the case of the Gram-positive bacteria, this structural particularity prevents the action of Ag-NPs rendering acid-fast and gram-positive bacteria more resistance to the antimicrobial activity of Ag-NPs comparatively with Gram-positive bacteria.143,144 For example, the Gram-positive Bacillus subtilis, have a cell wall of 55.4 nm, the acid-fast M tuberculosis a 20.2 nm145 while the Gram-negative Pseudomonas aeruginosa, has a cell wall of only a 2.4 nm.146 Interestingly, although there are important functional differences between the mycobacteria cell wall and gram-positive bacteria, the DNA-based molecular taxonomy of bacteria based on the high similarity to genes, groups the classical acid fast-mycobacteria as gram-positive bacteria.147 But this overall generalization regarding the susceptibility towards AgNPs has many exceptions, thus, AgNPS synthesized from Bacillus brevis has a maximum antibacterial effect against the Gram-positive, multi-drug resistant for Staphylococcus aureus and moderate for the Gram-negative Salmonella typhi.148
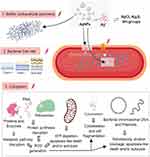 |
Figure 3 The three most important routes of antimicrobial action of AgNPs. 1. Accumulation and disruption of the extracellular polymers of the bacterial biofilm; silver ions (Ag+) could also biochemically alter the biofilm overall adherence, structure, and porosity.2. AgNPs adhere to bacterial cell surface (documented for Gram-positive, negative and also for the acid-fast bacteria) resulting in microbial membrane disruption, altered transmembranar transport, cellular content leakage (mainly electrolytes dysregulation) and bacterial death (apoptosis/lysis); as for the biofilm, Ag+ generated extracellularly contribute to the microbial cell wall disruption by biochemical alteration of the SH– groups. 3. AgNPs penetrate bacterial cell wall and access microbial cytoplasm where can interact with the organelles, cytosolic molecules (as free amino acids, peptides, and enzymes) and bacterial cytoskeleton; By direct action of AgNPs and Ag+ results the alteration of several metabolic pathways, bacterial organelles dysfunction (mainly mitochondria), ROS generation and bacterial DNA alteration ultimately causing cell death apoptosis/lysis). Figure 3 was created using BioRender. Abbreviations: AgNPs, silver nanoparticle; ROS, reactive oxygen species: Ag+, silver ions. |
Role of Ag in Particle State (Ag0) and Ag+ Species in Mediating the Bactericidal Effect of Silver Nanoparticles
This is the first (direct or “primary”), of antibacterial effect of AgNPs and is considered to be due to: (1) nanoparticles damage the bacterial wall and on (2) entrance of particles into the bacterial cytosol and directly interact with the intrabacterial environment.149–151 The adherence of the AgNPs on the bacterial surface and formation of particle agglomerates is followed by disruption of bacterial membrane integrity by induction of cell-wall pits and gaps, and alteration in membrane selectivity and permeability, including ionic transport.151–154 This first, step is dominated by the wall changes is followed by bacterial-cytosol leakage, lost the intracellular contents and finally the collapse of the cell or apoptotic-like bacterial cell death and formation of an amorphous mass of cell debris.149,150,155 Due to the massive loss of the bacterial content the “ghost cells” morphology is used to describe lysed bacteria following this process.150,156
Nanoparticles have the property to be adsorbed at the bacterial membrane mainly by electrostatic adhesion, a process mediated by surface charge of the particle- the zeta (ζ)-potential – and the outer layers of the bacterial cell wall.151 Thus, a study designed to explore this surface-interaction between AgNP and bacteria (Bacillus spp), El Badawy et al found that positively charged BPEI-caped AgNPs were the most bacteriotoxic NPs, mainly due to the local agglomeration. The negatively charged citrate-caped AgNPs were the least bacteriotoxic.151 The outer layer of bacteria (G+) is negatively charged due to the presence of carboxyl, phosphate and amino groups,157 thus influencing the electro repulsion between bacteria and negatively charged AgNP. The highly-negatively charged bacterial wall is believed to be an important fact in explaining the superior activity of AgNP against G- compared with G+ bacteria which is frequently reported.158
In a similar study, positively charged AgNP by functionalization with PHMB functionalized exhibited superior antibacterial effects against E. coli. Also, the bactericidal activity of PHMB was enhanced by the combination with AgNPs.159 Indeed, this hypothesis was further confirmed by Ivask et al,160 which observed that the pathways involved in G- bacterial responses to AgNP are highly dependent on the surface characteristics of the Ag composite, including zeta (ζ)-potential.
At least partially the enhancement of the antibacterial effect of observed in AgNPs with surface-modified by surfactants (SDS) and polymers (PVP 360),161 in addition to stabilization of particles against aggregation, can be attributed to this facilitated-adhesion to the bacterial wall.
Additionally to the wall thickness and structure, the difference in resistance of different classes of bacteria can be explained by the fact that due to the high-presence of LPS the cell wall, Gram-negative bacteria has a higher negative charge, which promotes local adhesion and membrane-clustering of particles and finally enhances the antibacterial effect of Ag-NPs.144,162,163 Therefore, electrostatic interaction between bacterial cells (charged negatively) and AgNPs (charged positively) is critical for the antibacterial activity of NPs.144,161,164
Moreover, to the above-mentioned action against the bacterial wall, AgNPs have the ability to enter inside bacteria's cytosol, to form cytoplasmic precipitates and to disrupt several bacterial-physiological processes. The type of bacterial- metabolic pathways disrupted directly by the Ag in particle state is largely unknown.
Role of Ag+ Species
The antibacterial effect of AgNP is complementary enhanced by the local elimination of Ag+ species which have high affinity especially for thiols, selenols, organic amines and phosphates and forms strong covalent bonds.141 The formation of this covalent bonds (e.g. silver thiolate) in which Ag act as a bridging agent linking several thiols-groups for different molecules can irreversibly alter their tridimensional structure and function141,165 and finally will disrupt simultaneously several enzymatic pathways and constitutive cell-structure elements (DNA, cytoskeleton, plasmatic and organelle membrane, etc.). This multi-molecule disruption mediated by a broad chemical affinity and not by a targeted-element is the main cause of the complex antibacterial mechanism in comparison with classical antibiotics which typically target a narrow groups of molecules, as for example, restricted to cell membrane (beta-lactamides) or interfere with molecules synthesis and also broad spectrum of micro-organisms sensible to Ag.15 Regarding the involvement of different metabolic pathways following the above-described mechanism, probably one of the most important is the disruption of the ROS-regulation system (by interfering with reductase enzymes and other cofactors) and thus increasing their intracellular oxidative stress and triggering cell senescence or death. The ROS-generation as a mechanism of AgNP/Ag+ action will be separately discussed.
In the AgNP/Ag+ model of action, AgNPs acts as a nanoparticulate reservoir for the continuous release of Ag+ species. The rate of release of Ag+ is dependent on many factors including NP size, surface, porosity, O2 amount in the environment, and is mediated by release (“desorption”) of chemisorbed ions from the particulate surface, oxidative dissolution (which is the main way to release Ag+ in the aqueous environment).166
In a dynamic presentation of the plausible effect, the AgNP adherent on the bacterial-cell wall or entrapped inside the bacterial cytoplasm (“Trojan horse effect”) will release in the adjacent environment large amounts of Ag+ species generating a locally high concentration of antibacterial ions.160,167
Generation of Reactive Oxygen Species (ROS)
The generation of ROS is considered a second mechanism by which AgNPs can induce bactericidal or bacteriostatic effects. The ROS generation is due to (1) particle–cell interactions (alteration of local cell activity, e.g. inflammation-driven enhancement of oxygen respiration and oxidative/antioxidative imbalance) or due to (2) in situ production of hydroxyl radicals due to an Ag-mediated Fenton-like reaction168,169 (acellular induction of ROS). The presence of transition metals including Fe, Cu, or Cr as synthesis contaminants enhances ROS generation via direct catalytic Haber–Weiss and Fenton-type reactions.170 This in situ production of free radicals by AgNPs is usually enhanced by exposure to light-sources of variable wavelengths, this feature is currently explored also for photocatalytic degradation of pigments.171,172
The ROS generation within the activated cells is mediated by enhancement of: 1. cytoplasmic ROS (cytoROS) production by NADPH oxidase family of enzymes (e.g. endothelial, neuronal and inducible nitric oxide synthases)(eNOS, nNOS, iNOS) during inflammation; 2. Peroxisome ROS generation as a by-product of enzymatic activity (as hypoxanthine and β-oxidation, polyamine synthesis and amino acid deamination); 3. mitochondrial ROS (mitoROS) as a byproduct of metabolic-enzyme activity and mitochondrial respiration, activity upregulated, for example, by complex I NADH reductase and dehydrogenase via RET (reverse electron transfer) during inflammation. 4. lysosomal and phagolysosomal ROS mediated mainly by NADPH oxidase and myeloperoxidase produced mainly within the professional phagocytic cells (neutrophils, monocytes, and macrophages) during the intracellular destruction of microbes and removal of cell debris.85,173 AgNPs were shown to interact with all of the above systems, including increased expression of iNOS and generation of NO,174 impairment of mitochondrial function and ROS generation,175,176 upregulation of peroxisome oxidative stress-related genes, such as catalase,177 enhancement of phagolysosomal activity (reduction of lysosomes pH)178 and macrophage and neutrophil activation and stimulation of ROS generation.179
Another clear advantage of using AgNPs is based on the well-known fact that NP persists much longer in the body (even years) compared with the small molecule used currently in antibacterial therapy. This would increase the long term releasing of active compounds and thus the sustained therapeutic effects.142,180 Although AgNPs can have a direct effect on the microorganisms, the main effect is considered to be mediated through the biochemical interactions of Ag+.181,182
Presence of Antibacterial – Active Products in Biosynthesized AgNPs, a Possible Source of Antibacterial Synergy?
The antibacterial effect of AgNP, especially in the green-synthesis context (plant, viral, bacterial, fungic, and algal extracts or biomimetic compounds as reducing agents),183,184 can be, at least partially enhanced by the extra bio-active component introduced in the particle synthesis.185,186 AgNP can be prepared using elements that possess per se an antibacterial activity. The synergistic effect between AgNP and other and bioactive phytocompounds can be expected in the green-synthesis, leading to antibacterial effects via different mechanisms as those described above.185 Also, the concentrations of antibacterial-active compounds can be observed below the minimal dose of individual compounds. Therefore, the enhanced antimicrobial effect of NP synthesized by green-extraction which can be occasionally observed can also be determined by the presence of the bioactive molecules of the synthesis attached on the surface of nanoparticles as stabilizing agent.186,187
In the green-synthesis of AgNP, the biological extracts are mixed with the metal salt solutions, the bio-extract (containing starch, steroids, sapogenins, flavonoids, terpenoids, amino cellulose, etc.)188 acts in situ as reducing agents of the silver salts (Ag+) to form metallic silver Ag0, and also as capping agents to provide stability of silver nanoparticles in solution186,189 and partially can be further be found in the structure of the AgNP as surface-stabilizing ligands.190
Indeed, in a recent study, Shaik et al190 tested the efficiency of AgNP synthesized from Origanum vulgar against various bacteria (Escherichia coli, Shigella sonnei, Micrococcus luteus), and fungi (Aspergillus flavus, Alternaria alternate, Paecilomyces variotii, Phialophora alba). The bactericidal and antifungal efficiency was proportional to the amount of plant extract employed for the preparation of AgNPs. Similar findings of obtaining biogenic NP with broad antibacterial effect were reported by Pugazhendhi et al from AgNP synthesized from red algae Gelidium amansii.191
Also, a study which compares the antibacterial effect of AgNP produced by green synthesis (from S. persica) versus chemical against Gram-negative (E. coli and P. aeruginosa) and Gram-positive (M. luteus and S. aureus) bacteria shows that the green synthesized Ag-NPs exhibited slightly higher antimicrobial activity in comparison to the chemically synthesized Ag-NP.158 The conclusion of the study was that, although S. persica root has antibacterial properties per sei, due to the small amount of active compound included in the synthesis process, the increased activity of the green-synthetized Ag-NPs was mainly due to the improved solubility of the Ag-NPs rather than the microbicidal potential of plant-derived compounds used for the synthesis of NPs.
In a study designed to assess the antibacterial effect of AgNPs from Asparagusspp. against 4 mycobacterium species (M.tuberculosis, M.pheli, M.avim, and M. smegmatis), Kote et al185 found a direct connection between the green approach of AgNPs synthesis (mainly due to the enhanced stability) and the antimycobacterial effect. Also, some forms of biosynthesized AgNPs have multimodal action, proving simultaneous antibacterial, antimycotic and antitumoral effects as was recently shown for AgNPs produced from Phoenix dactylifera.192
In the above-mentioned studies, the enhancement of the antibacterial efficiency of AgNP produced by green synthesis is more likely mediated by the uniformity of the dispersion and a better stabilizing of molecules in aqueous solution compared with chemical synthesis.
In a study exploring comparatively the antimycobacterial effects of green-synthetized vs chemically produced AgNPs found that chemically AgNP exhibited greater efficiency in terms of mycobacterial inhibition, specificity and selectivity compared with bio-AgNPs193
Thus, although the presence of co-synthesis products in the green-synthesis of AgNP definitely have a role in determining and fine-tuning the biological activity of the obtained nanoparticles,194 the exact mechanisms and the possible synergism with Ag in mediating antibacterial activity should be further explored.
Enhancement of Antibacterial Efficiency Antibiotics by AgNP
An emerging practice in antituberculous experimental therapy is to combine a metallic nanoparticle (TiNP, CuNP, AuNP, AgNP, ZnNP, etc.) with antibiotics (“nano-antimicrobials”) to enhance their antimycobacterial efficiency, especially in the context of bacterial antibioresistance.195,196 Also, antibacterial-AgNP synthesis using tetracycline as co-reducing and a stabilizing agent was described by Djafari et al.197
It is postulated that combining AgNPs and an antibiotic can synergistically inhibit both Gram + and Gram - multidrug-resistant bacteria.198–200 But this synergism is observed only for certain types of antibiotics, thus Deng et al198 showed AgNP/antibiotic synergistic growth inhibition against the multidrug-resistant bacterium Salmonella typhimurium for enoxacin, kanamycin, neomycin, and tetracycline, while ampicillin and penicillin did not show any enhancement of the antibacterial activity. Regarding the mechanisms of synergy (depicted in Figure 4), the presence of tetracycline enhances the bacterial binding of Ag, followed by an enhancement in Ag+ release which finally leads to a high local-concentration of Ag+ near the bacteria cell wall which leads to bacterial-growth inhibition and death.198 Enhanced positive synergistic response against S. aureus and E. coli was observed also for AgNPs synthesized from Argyreia nervosa associated with seven commercial antibiotics (streptomycin, vancomycin, tetracycline, amoxicillin, gentamicin, erythromycin and ciprofloxacin).201 Similarly, enhanced antibacterial efficiency of ceftriaxone against ceftriaxone-resistant human pathogens was reported following conjugation with biogenic AgNP.202
Another mechanism of AgNPs/antibiotics synergy was described by Hwang et al.203 and is the anti-biofilm effect. This was observed following a combination of AgNPs with ampicillin, chloramphenicol, and kanamycin against various pathogenic bacteria (Enterococcus faecium, Staphylococcus aureus, Streptococcus mutans, E. coli, and P. aeruginosa) inhibits the formation of biofilm which is a major resistance mechanism for several types of bacteria. This antibacterial effect can be related to the high surface to volume ratio of NPs which can permit their deep infiltration into mature biofilms.17 Recently by Farooq et al.204 showed an enhancement of antibiofilm efficiency of rifampicin following conjugation with silver (Rif-AgNPs) in methicillin-resistant K pneumoniae and S aureus.
Other mechanistic studies exploring the antibacterial effect of NPs, shown that AgNPs and amoxicillin, in addition to their intrinsic antibacterial activity, can form a new complex in which amoxicillin-molecules surround the AgNPs metallic core.205
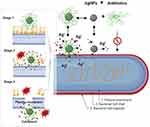 |
Figure 4 Schematic diagram showing, in a step by step fashion, the synergistic pathways and mechanisms of AgNP and antibiotics against multidrug-resistant bacteria (depicted in G- bacteria). Enhancement of the accumulation of the AgNPs conjugates with antibiotics within the bacterial cell membrane is associated with potentiation of Ag+ release and damage of the bacterial capsule, cell wall, and plasma membrane components. In this paradigm, the pathway mediated by AgNPs is a minor antibacterial mechanism, and the activity mediated by antibiotics-only is not effective due to antibacterial resistance. In a step by step diagram of the bacterial membrane destabilization (depicted for AgNPs/nisin conjugates), the interaction between AgNP/antibiotic complexes with bacterial cell membrane (stage 1) will results in enhancement Ag+ release, in situ ROS generation, membrane-insertion of nisin (methyl)-lanthionine rings, followed by local dissolution of lipidic molecules, membrane-pore formation, and internalization of AgNPs/nisin complexes within the bacterial cytoplasm. Inspired from Deng et al198 and Arakha et al206 schematic concepts of AgNP/nisin and AgNPs/tetracycline complexes-mediated antibacterial activity. Figure 4 was created using BioRender. Abbreviations: AgNPs= silver nanoparticle; ROS, reactive oxy gen species: Ag+=silver ions. |
Antimycobacterial Effect of Silver Nanoparticles
Nanotechnology brings a novel and promising therapeutic approach to improve the current antimycobacterial treatments. This include improvement of the efficiency of the currently used first-or second-line antibiotics following generation of different formulations (e.g. liposomes, solid lipid nanoparticles, alginate nanoparticles, niosomes, dendrimers)207 or by adding new antituberculous compounds which can synergies the classical therapy, as metallic metal-based nanoparticles (mainly silver, iron oxide, gold, copper oxide, aluminum oxide, zinc oxide, titanium dioxide, etc.).99,187 Several of the therapeutical advantages of such nanoparticle-based therapy of tuberculosis are, among others: a) prolonged time of action, b) a high carrier ability; c) flexibility of various routes of administration, d) possibility of multiple drugs-encapsulation in the matrix, e) fewer side effects and improved compliance (especially important in prolonged anti-TB therapy).207
In vitro Studies
Multiple experiments carried recently determined the antimycobacterial effect of AgNP.133,208 For example, a good activity against mycobacteria and low cytotoxicity (10 times the dose established as MIC for Mtb) on infected macrophages was recently reported by Singh et al (2016)180 for phytogenic AgNPs.
One of the earliest reports on the antimycobacterial effect of AgNP came from Song et al.209 who tested in vitro small, non-biogenic AgNP measuring <10 nm to several bacteria species, including beside M. tuberculosis, also E. coli, S. aureus, and Salmonella typhi. The antimycobacterial effect was observed at 10 ppm, and the proposed mechanism is based on the presence of AgNPs in the cytoplasm of mycobacteria and the following bacterial-metabolic disturbances.209
A good in vitro antimycobacterial effect, observed mainly by inhibition of the mycobacterial growth, was reported also by studies employing biogenic AgNP produced from Plumbago auriculata,210 Coriandrum sativum,211 Catharanthus roseus,212 Asparagus race,185 Psidium guajava,213 Ipomoea carnea,214 Rhizopus stolonifer,215 and Cucumis sativus.216
In vitro inhibition of MDR and XDR strains of M. tuberculosis was found for physicochemically (“non-green”) synthesized AgNP in doses as low 1 µg/mL.217 Overall, no bactericidal effect was found, and although the AgNP are internalized within THP-1 macrophages, the intramacrophagic antimycobacterial effect was modest. A similar effect of multimetallic nanoparticles (MMN) including AgNP for intramacrophagic mycobacteria was reported by Ellis et al.218 Although internalized within the phagolysosomal apparatus, the AgNP have a limited antitubercular effect for the intracellular bacteria, but increase the antitubercular effect of rifampicin. The co-administration of rifampicin led to a reduction of 68% of M. tuberculosis colony-forming units. Using spherical AgNP measuring 13 nm, Jafari et al.219 observed no antibacterial effect for intramacrophagic M. tuberculosis, but the addition of Zn in the molecule is inducing an anti-tubercular effect. Additionally, the 5Ag:5ZnO report was found to have both an intracellular antibacterial effect and also no significant toxicity to normal lung (MRC-5) cell lines. By contrary, a good antitubercular effect against intramacrophagic M. marinum and M. smegmatis was observed by Mohanty et al.19 for spherical, biogenic AgNP combined with antimicrobial peptides in doses of 0.1 and 0.5 ppm. The tested particles measured 50–100 nm and were synthesized from Alstonia macrophylla and Trichoderma sp. The enhanced antitubercular effect was not correlated with high levels of NO, thus the proposed antibacterial mechanism was associated with superoxide radicals formation and the activation of macrophages by cytokines. In the same study, an increased antibacterial effect against M. smegmatis was observed following the combination of NPs with the classical-antituberculosis drug rifampin.19 Intramacrophagic killing of M. smegmatis internalized in RAW264.7 macrophages (in both pre/and postexposure treatments) was reported for spherical chitosan-coated AgNP (CS-AgNPs) in 3 ppm dose.220 The bactericidal effect was time and concentration-dependent and most of the antibacterial effect was observed in the first hour of incubation. The hypothesized antibacterial mechanism was cell membrane disruption or chemical inactivation of thiol-containing molecules. Also, CS-AgNPs were noncytotoxic on RAW264.7 macrophages at the bactericidal concentration. An increased antitubercular activity was observed following the addition of gentamicin. In the same study, in addition to antimycobacterial effect, CS-AgNPs were found to be also active against other bacteria like Staphylococcus aureus, Pseudomonas aeruginosa, and Salmonella typhi.220
In addition to the direct antitubercular activity in doses beginning with 5 mg/l, AgNPs measuring 10–150 nm were shown to potentiation of the antibacterial effect of isoniazid, rifampicin, ethionamide, levofloxacin, ofloxacin and kanamycin against clinical isolates of M. tuberculosis by Kreytsberg et al.133
In another study employing the antimycobacterial effect of physicochemically synthesized, tetrahedral and spherical AgNP measuring 50 nm, an antibacterial effect against field isolates and standard strains of M. tuberculosis and M. bovis were reported. The minimal inhibitory concentration (MIC) was found to be 1 and 4 µg/mL for standard cultures of M. tuberculosis and M. bovis. Higher doses were needed to inhibit the clinical isolates, being in the range of 4–32 µg/mL for M. bovis and 1–16 µg/mL for M. tuberculosis.221
Promising antimycobacterial results against both M. tuberculosis and M. smegmatis are also observed for AgCl-NP produced from commercial yeast extract in doses of 37 µg/mL concentration.222 In addition to the Ag effect, in this study, the antibacterial effect could be also mediated by the activity of Cl, a potent and broad antiseptic.223
Using biogenic spherical (20–56 nm) AgNP synthesized from Sesbania grandiflora, Patel et al showed that MIC for standard cultures of M. tuberculosis is 12.5 µg/mL. This dose was half of the MIC observed for silver nitrate and approximately 30% of the MIC of Rifampicin. Also, the Sesbania grandiflora extract shown an antimycobacterial effect, but much higher compared with the AgNPs (100 µg/mL).224 A similar MIC was obtained for M.tuberculosis by Punjabi et al.
An interesting strategy is combining AgNP with peptides or chitosan for antibacterial/antitumoral effect. Thus in a recent study, Abdel-Aziz et al150 shown that spherical N,N,N-trimethyl chitosan chloride (TMC)/AgNP in a 0.98 to 125 mg/mL dose have an antibacterial effect on M. tuberculosis mainly by disrupting the bacterial cell wall. Also, the same nanocomposite was found to have a cytotoxic effect against A-549-lung adenocarcinoma cells in 12.3 µg/mL dose and to have reduced toxicity against normal lung cells.
Also, spherical PVP and BSA-caped AgNP measuring 5–9 nm (BSA-AgNP) and 6–45 nm (PVP-AgNP) were shown to have antibacterial effects against clinically isolated and standard M. tuberculosis cultures. The antibacterial effect was mediated by mycobacterial cell membrane injury, followed by bacterial lysis.225
Although differences regarding the sensitivity towards AgNPs were reported between different species of Mycobacteria, most of the tested materials have a simultaneous antibacterial effect against multiple species, including M. tuberculosis, M. pheli, M. avium and M. smegmatis.185
A synopsis of the in vitro studies using AgNPs in the treatment of mycobacteria-induced diseases, including the species and strain of mycobacteria tested, the experimental model, the type of AgNP and the main results are presented in Table 2.
In vivo Preclinical and Clinical Studies
Compared with the large number of in vitro studies exploring the potential used of AgNp as antimycobacterial drugs only a few in vivo studies were up to date carried.
In a clinical trial carried on 50 human patients with ages from 26 to 55 years suffering from with laryngeal tuberculosis, including cases with DR-tuberculosis, an AgNP aqueous suspension (Argovit-C, 10 mg/mL silver, in a concentration of 3.3%) was tested for 2 months as local therapy.236 The AgNP group (n=30) received the treatment topically by inhalation for 2 times a day for 10 mins. The control group (n=20) received classical TB therapy. The suspension was previously characterized as containing spherical AgNP with bimodal size distribution (14.1 ± 9.9 and 50.1 ± 40.3 nm).237
After 60 days of therapy, the sputum was negative for M. tuberculosis in 93.3% of patients enrolled in the AgNP group compared with 70% of patients who received the standard anti-TB treatment. Also, the patients enrolled in the AgNP group – showed faster healing of the laryngeal TB-lesion, including ulcerations and voice function compared to standard tuberculosis drugs.236
An experiment designed to investigate the effect of isoniazid combined with AgNPs on MDR strains of M. tuberculosis was carried by Zakharov et al in 68 BALB/c inbred mice. Spherical AgNP measuring 3–60 nm were tested initially in vitro in ascending concentrations (5;25;50 μg/mL) in 651 MDR strains of M. tuberculosis. In the rodent study, based on the survival rates and histopathology of the lung, the combination of isoniazid and silver nanoparticles was preferable compared to the single-use of the above components.238
In a recently published study carried out by the same author, the effect of isoniazid combined with AgNPs was tested in 3 experimental groups of MDR-TB-infected mice: group 1 received only isoniazid (50 mg/kg); group 2 received intramuscularly AgNPs in doses of 12.5 to 125 µg/kg; group 3 received a combination of the treatments detailed for groups 1 and 2. Based on the histopathologic grading of lesions, the use of AgNPs in the treatment of TB induced by MDR strains enhances the efficiency of isoniazid.239
In an in vivo study carried out in 65 mice experimentally infected with MDR strains of Mycobacterium tuberculosis isolated from human patients, the efficiency of AgNP as a single therapeutic molecule or in combination with isoniazid was tested. The AgNP measured 10–150 nm. The survival rate of TB-infected animals following the combined treatment with isoniazid and AgNP was 95% and 35% in the group receiving AgNP only, compared with 100% mortality in the TB-infected control group.133
The Main Limitation of the Usage of AgNP in the Treatment of Tuberculosis
Potential Toxicity of AgNPs
One of the potential drawbacks of AgNPs, as four most of the inorganic nanoparticles, is their toxicity which may limit their usage in a biological context,240–242 but despite the extension of use in the last decades, the evidence for the toxicity of AgNPs is still unclear.243 However, an in depth discussion regarding the toxicity of AgNP is something that goes beyond the purpose of this manuscript.
The increased production of ROS which is presented above as one of the antibacterial mechanisms of AgNP can be harmful to the normal cells if the cellular protective antioxidative mechanisms are overcome, which will trigger several detrimental biological effects like inflammation, autophagia, apoptosis, necrosis or irreversible DNA-damage followed by mutations and possible oncogenesis.244 There are several studies in which a good antibacterial efficiency and a low-toxicity for the explored doses were observed. Thus, for AgNPs produced from Phenerochaete chrysosporium, a good antibacterial effect against Pseudomonas aeruginosa, Klebsiella pneumoniae, Staphylococcus aureus, and Staphylococcus epidermidis was observed but no in vitro toxic effect on mouse embryo fibroblasts for doses up to 12.5 μg/mL AgNPS.245
Moreover, biogenic AgNPs measuring 50–100 nm synthesized from Alstonia macrophylla and Trichoderma sp showed no cytotoxic effects on macrophages at the mycobactericidal dose (0.1 and 0.5 ppm), but the exposure to higher doses of AgNPs induced cytotoxicity and DNA-damage.19
Low Penetrability in Tuberculous Granulomas
A clinical limitation of the usage of AgNP in TB therapy is based on the low tissue penetrability of large molecules following a non-intravenous route of administration.246 Also in a lack of proper functionalization following an intravenous route of administration, a low intra-lesional accumulation is predictable considering the poorly vascularization of tuberculose-lung cavities present in chronic cases of tuberculosis.247 Additionally, the persistence of mycobacteria in the nonvascularized necrotic material within the granuloma center can assure the persistence of a reservoir of viable mycobacteria in a biological environment largely inaccessible for large molecules and immune cells.18
The Immunomodulatory Effect of AgNP
Especially important in the clinical context of MtbC infection and macrophage polarization, Sarkar et al.248 observed an upregulation of macrophage Hsp72 by AgNP, which is possible to be linked with further suppression of NF-κB pathway and reduction of the macrophage antimycobacterial effect. In vivo following 28 days repeated dose toxicity study in rats, AgNP in high doses induce a marked suppression of natural killer cell (NK) activities and decreased interferon-γ and interleukin (IL)-10 release in response to Concanavalin A (ConA)- mediated activation of the spleen cells.249 Interestingly, in the same study, the lipopolysaccharide (LPS) stimulation was associated with increased IL-1β and decreased IL-6, IL-10 and TNF-α production in the spleen, proving a complex immunomodulatory effect of AgNP. The exposure of human NK cells to AgNPs also resulted in reduced viability and altered function enhancement of expression of the inhibitory receptor CD159a.250
The conclusion drawn by their observation brings new perspectives regarding the drug-designs which intend using AgNP in obligate intra-macrophage pathogens. But this immunomodulatory effect seems to be more complex and interferes with specific immune cell activities. Thus, although exposure of neutrophils to AgNP (50 µg/mL) was associated with reduced neutrophilic degranulation (elastase release) and oxidative burst, the overall phagocytic activity was enhanced.251 In the same study, pre-exposure of macrophages (RAW 264.7) to AgNP, stimulates the release of inflammatory cytokine interleukin-6 as well as enhancement of phagocytic ability in response to lipopolysaccharide stimulation.251 Also, the exposure of J774 A1 murine macrophage to AgNPs resulted in early activation of the inflammatory response by up-regulation of IL-1, IL-6, and TNF-a genes, but in a lesser amount compared with AuNPs.252 Therefore, taking into account the divergent data regarding the immune impact of AgNPs, a better understanding of activation or suppression of immune cell pathways and functions following AgNPs needs to carry before a clinical-functional significance in the context of the complex inflammatory environment associated with mycobacterial infections to be drawn. As a solution to the above-mentioned issues in AgNP usage the utilization of a gold-silver alloy NP promise to be a viable solution in overcoming the macrophage function suppression due to the significant improvement of AgNP biocompatibility following the introduction of gold NP (AuNP) as alloy.253 Additionally, AuNP produced from Terminalia arjuna show important antioxidant and anticholinesterase effects254 which could antagonize the AsNP toxicity.
Development of Bacterial Resistance Towards Silver Nanoparticles
Although rarer and less studied compared with the classical antibioresistance, the mutation in bacteria to resist Ag is similar in certain limits to the pathway that led to chemoresistant bacterial strains.255–258 The widespread use of Ag- and Ag-ions containing nanomaterials and nanocomposites is considered to be the main determinant in a possible bacterial selection and evolution towards a biological resistance to Ag NP and/of Ag ions.258,259 The resistance to antibacterial Ag is reported among nosocomial infections,260 in bacteria present within wounds, including burns,261–264 diabetic foot ulcers,265 dental bacteria,266,267 or exterior natural-environments containing high amounts of Ag.268 Occasionally, the resistance towards Ag is developed in parallel with the multidrug-resistance, as shown in Staphylococcus aureus, klebsiella pneumoniae, Acinetobacter baumannii, and Enterococcus faecium.269 This cross-resistance to antibiotic and metal resistance are typically mediated when genes for different resistant phenotypes (metal/chemioresistant) are located on the same mobile genetic elements as plasmids and conserved regions of integrons.270
Generally observed in fast-growing bacteria, Ag-resistance was described also in Mycobacteria, as Mycobacterium smegmatis,255 M avium, M fortuitum, and M mucogenicum.271 Resistance to both silver nanoparticle and AgNO3 was observed for saprophytic bacteria (as Mycobacterium smegmatis)255 and could be proven also for the other pathogenic classes of bacteria.
As mechanism, the Ag resistance can be associated with elimination and neutralization of ionic forms of silver, as active efflux of Ag+ from bacteria (e.g by P-type ATP/SilP, membrane potential-dependent three-polypeptide cation/proton antiporter or multidrug resistance/MDR efflux pumps), increased capacity for reducing Ag+ to a neutral-oxidation state which are typically less bacteriotoxic.272–274 Recently, in gram-negative bacteria, the resistance to AgNP was induced by overexpression of bacterial flagellum protein-flagellin, which induced aggregation of particles at the surface of bacteria and reduction of AgNPp antibacterial effect.192
The difficulty in gaining such a resistance against AgNPs s is due to the fact that the antibacterial effect of nanoparticles is more complex (illustrated in Figures 2 and 3) compared with classical antibiotics.
Conclusion
Tuberculosis is still a major public health issue, but currently, nanotechnology and nanoparticle research offers several exciting concepts which may prove to be valuable tools in improving the TB-therapy, especially in the context of broad-antibioresistant stains of MtbC.
There is no doubt that AgNPs per sei or in conjunction with different biomolecules as peptides and chitosan have good antimycobacterial effects, but this effect is limited following macrophagic internalization of mycobacteria. A promising strategy is combining AgNPs with classical anti-TB therapeutics which synergically enhance the antimycotic activity both extra and intracellularly. Despite the encouraging in the in vitro-stage of research, still, there are few in vivo studies exploring the anti-TB potential of AgNPs. As a result of the peculiar structure and visceral distribution and of TB-induced lesions, the on-going research efforts for the synthesis of novel anti-TB nanoparticles should be focused on strategies for enhancing the local availability of antibacterial nanoparticles. Increased local availability, associated with good intra-macrophagic disponibility, a potent antimycobacterial effect, and a low-immunosuppressive and toxic effect should be the cumulative characteristics of a good nanoparticle candidate for future therapy of tuberculosis.
Acknowledgments
This work was supported by a grant of Ministery of Research and Innovation, CNCS – UEFISCDI, project numbers PN-III-P1-1.1-PD-2016-1840, PN-III-P1-1.1-PD-2016-1831 and PN-III-P1-1.1-TE2016-2161within PNCDI III.
All figures presented in this work were created with BioRender.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Thoen CO, LoBue PA, Enarson DA, Kaneene JB, de Kantor IN. Tuberculosis: a re-emerging disease in animals and humans. Vet Ital. 2009;45(1):135–181.
2. De Lorenzo S, Tiberi S. Tuberculosis a re-emerging disease. Intern Emerg Med. 2012;7(S3):185–187. doi:10.1007/s11739-012-0822-9
3. World Health Organization. Global Tuberculosis Report 2015, 20th Ed. World Health Organization; 2015.
4. Schneider E, Moore M, Castro KG. Epidemiology of Tuberculosis in the United States. Clin Chest Med. 2005;26(2):183–195. doi:10.1016/j.ccm.2005.02.007
5. Forrellad MA, Klepp LI, Gioffré A, et al. Virulence factors of the Mycobacterium tuberculosis complex. Virulence. 2013;4(1):3–66. doi:10.4161/viru.22329
6. who.tb.99.260.pdf. Available from: https://www.who.int/docstore/gtb/publications/mdrtb/PDF/who.tb.99.260.pdf.
7. 9789241565714-eng.pdf. Available from: https://apps.who.int/iris/bitstream/handle/10665/329368/9789241565714-eng.pdf.
8. Saravanan M, Niguse S, Abdulkader M, et al. Review on emergence of drug-resistant tuberculosis (MDR & XDR-TB) and its molecular diagnosis in Ethiopia. Microb Pathog. 2018;117:237–242. doi:10.1016/j.micpath.2018.02.047
9. Richter E, Weizenegger M, Rüsch-gerdes S, Niemann S. Evaluation of genotype MTBC assay for differentiation of clinical Mycobacterium tuberculosis complex isolates. J Clin Microbiol. 2003;41(6):2672–2675. doi:10.1128/JCM.41.6.2672-2675.2003
10. Thoen CO, Steele JH, Kaneene JB. Zoonotic Tuberculosis: Mycobacterium Bovis and Other Pathogenic Mycobacteria. John Wiley & Sons; 2014.
11. Kiers A, Klarenbeek A, Mendelts B, Van Soolingen D, Koëter G. Transmission of Mycobacterium pinnipedii to humans in a zoo with marine mammals. Int J Tuberc Lung Dis off J Int Union Tuberc Lung Dis. 2008;12(12):1469–1473.
12. Alexander KA, Laver PN, Michel AL, et al. Novel Mycobacterium tuberculosis complex pathogen, M. mungi. Emerg Infect Dis. 2010;16(8):1296–1299. doi:10.3201/eid1608.100314
13. Sathiyavimal S, Vasantharaj S, Bharathi D, et al. Biogenesis of copper oxide nanoparticles (CuONPs) using Sida acuta and their incorporation over cotton fabrics to prevent the pathogenicity of Gram negative and Gram positive bacteria. J Photochem Photobiol B. 2018;188:126–134. doi:10.1016/j.jphotobiol.2018.09.014
14. Shankar PD, Shobana S, Karuppusamy I, et al. A review on the biosynthesis of metallic nanoparticles (gold and silver) using bio-components of microalgae: formation mechanism and applications. Enzyme Microb Technol. 2016;95:28–44. doi:10.1016/j.enzmictec.2016.10.015
15. Silvestry-Rodriguez N, Sicairos-Ruelas EE, Gerba CP, Bright KR. Silver as a Disinfectant. In: Ware GW, editor. Reviews of Environmental Contamination and Toxicology. Vol. 191. New York: Springer New York; 2007:23–45. doi:10.1007/978-0-387-69163-3_2
16. Paladini F, Pollini M. Antimicrobial silver nanoparticles for wound healing application: progress and future trends. Materials. 2019;12(16):2540. doi:10.3390/ma12162540
17. Shanmuganathan R, Karuppusamy I, Saravanan M, Muthukumar H, Ponnuchamy K, Pugazhendhi VSR. Synthesis of silver nanoparticles and their biomedical applications - a comprehensive review. Current Pharmaceut Design2019. doi: 10.2174/1381612825666190708185506
18. Sakamoto K. The pathology of Mycobacterium tuberculosis infection. Vet Pathol. 2012;49(3):423–439. doi:10.1177/0300985811429313
19. Mohanty S, Jena P, Mehta R, et al. Cationic antimicrobial peptides and biogenic silver nanoparticles kill mycobacteria without eliciting DNA damage and cytotoxicity in mouse macrophages. Antimicrob Agents Chemother. 2013;57(8):3688–3698. doi:10.1128/AAC.02475-12
20. Montelongo-Peralta LZ, León-Buitimea A, Palma-Nicolás JP, Gonzalez-Christen J, Morones-Ramírez JR. Antibacterial activity of combinatorial treatments composed of transition-metal/antibiotics against Mycobacterium tuberculosis. Sci Rep. 2019;9(1):1–6. doi:10.1038/s41598-019-42049-5
21. Maher D, Raviglione M. Global epidemiology of tuberculosis. Clin Chest Med. 2005;26(2):167–182. doi:10.1016/j.ccm.2005.02.009
22. Seung et al. 2015. Multidrug-resistant tuberculosis and extensively drug-resistant tuberculosis.pdf. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4561400/pdf/cshperspectmed-TUB-a017863.pdf.
23. Seung KJ, Keshavjee S, Rich ML. Multidrug-resistant tuberculosis and extensively drug-resistant tuberculosis. Cold Spring Harb Perspect Med. 2015;5:9. doi:10.1101/cshperspect.a017863
24. World Health Organization 2007 - Global tuberculosis control surveillance, plannin.pdf. Available from: https://apps.who.int/iris/bitstream/handle/10665/144567/9241563141_eng.pdf?sequence=1.
25. Chung-Delgado K, Guillen-Bravo S, Revilla-Montag A, Bernabe-Ortiz A. Mortality among MDR-TB cases: comparison with drug-susceptible tuberculosis and associated factors. PLoS One. 2015;10:3. doi:10/f7b7v6
26. Shin SS, Furin JJ, Alcántara F, Bayona J, Sánchez E, Mitnick CD. Long-term follow-up for multidrug-resistant tuberculosis. Emerg Infect Dis. 2006;12(4):687–688. doi:10.3201/eid1204.041256
27. Bernabé-Ortiz A. Factores asociados a supervivencia en pacientes con tuberculosis en Lima, Perú [Factors associated with survival of patients with tuberculosis in Lima, Peru]. Rev Chil Infectologia Organo of Soc Chil Infectologia. 2008;25(2):104–107. Spanish.
28. Ginsberg AM, Spigelman M. Challenges in tuberculosis drug research and development. Nat Med. 2007;13(3):290–294. doi:10.1038/nm0307-290
29. Takiff H, Guerrero E. Current prospects for the fluoroquinolones as first-line tuberculosis therapy. Antimicrob Agents Chemother. 2011;55(12):5421–5429. doi:10.1128/AAC.00695-11
30. Ramachandran G, Swaminathan S. Safety and tolerability profile of second-line anti-tuberculosis medications. Drug Saf. 2015;38(3):253–269. doi:10.1007/s40264-015-0267-y
31. Marra F, Marra CA, Bruchet N, et al. Adverse drug reactions associated with first-line anti-tuberculosis drug regimens. Available from: https://www.ingentaconnect.com/content/iuatld/ijtld/2007/00000011/00000008/art00007. August 2007.
32. Natarajan S, Subramanian P. Adverse drug reactions to second line anti tuberculosis drugs: a prospective study in Mumbai, India. Eur Respir J. 2013;42(Suppl57).
33. Sotgiu G, Centis R, D’ambrosio L, Migliori GB. Tuberculosis treatment and drug regimens. Cold Spring Harb Perspect Med. 2015;5(5):a017822–a017822. doi:10.1101/cshperspect.a017822
34. Crofton SJ, Chaulet P, Maher D, et al. Guidelines for the Management of Drug-Resistant Tuberculosis. Vol No. WHO/TB/96.210 (Rev. 1). Geneva: World Health Organization; 1997.
35. O’Brien RJ, Spigelman M. New drugs for tuberculosis: current status and future prospects. Clin Chest Med. 2005;26(2):327–340. doi:10.1016/j.ccm.2005.02.013
36. Ernst JD. Macrophage receptors for Mycobacterium tuberculosis. Infect Immun. 1998;66(4):1277–1281. doi:10.1128/IAI.66.4.1277-1281.1998
37. Porvaznik I, Solovič I, Mokrý J. Non-tuberculous mycobacteria: classification, diagnostics, and therapy. Adv Exp Med Biol. 2017;944:19–25. doi:10.1007/978-3-319-44488-8_45
38. Azadi D, Motallebirad T, Ghaffari K, Shojaei H. Mycobacteriosis and tuberculosis: laboratory diagnosis. Open Microbiol J. 2018;12:41–58. doi:10.2174/1874285801812010041
39. Lesley R, Ramakrishnan L. Insights into early mycobacterial pathogenesis from the zebrafish. Curr Opin Microbiol. 2008;11(3):277–283. doi:10.1016/j.mib.2008.05.013
40. Zachary JF, McGavin MD. Pathologic Basis of Veterinary Disease Expert Consult. Elsevier Health Sciences; 2016.
41. Smith I. Mycobacterium tuberculosis pathogenesis and molecular determinants of virulence. Clin Microbiol Rev. 2003;16(3):463–496. doi:10.1128/CMR.16.3.463-496.2003
42. Echeverria-Valencia G, Flores-Villalva S, Espitia CI. Virulence factors and pathogenicity of Mycobacterium. In: Ribón W editor. Mycobacterium - Research and Development. InTech; 2018:231–255. doi:10.5772/intechopen.72027
43. Torrelles JB, Sieling PA, Zhang N, et al. Isolation of a distinct Mycobacterium tuberculosis mannose-capped lipoarabinomannan isoform responsible for recognition by CD1b-restricted T cells. Glycobiology. 2012;22(8):1118–1127. doi:10.1093/glycob/cws078
44. Fratti RA, Chua J, Vergne I, Deretic V. Mycobacterium tuberculosis glycosylated phosphatidylinositol causes phagosome maturation arrest. Proc Natl Acad Sci. 2003;100(9):5437–5442. doi:10.1073/pnas.0737613100
45. Hmama Z. Quantitative analysis of phagolysosome fusion in intact cells: inhibition by mycobacterial lipoarabinomannan and rescue by an 1,25-dihydroxyvitamin D3-phosphoinositide 3-kinase pathway. J Cell Sci. 2004;117(10):2131–2140. doi:10.1242/jcs.01072
46. Chan J, Xuedong F, Shirley WH, Patrick B, Bloom B. Lipoarabinomannan, a possible virulence factor involved in persistence of Mycobacterium tuberculosis within macrophages. Infect Immun. 1991;59(5):1755–1761. doi:10.1128/IAI.59.5.1755-1761.1991
47. Shabaana AK, Kulangara K, Semac I, et al. Mycobacterial lipoarabinomannans modulate cytokine production in human T helper cells by interfering with raft/microdomain signalling. CMLS Cell Mol Life Sci. 2005;62(2):179–187. doi:10.1007/s00018-004-4404-5
48. Geijtenbeek TBH, van Vliet SJ, Koppel EA, et al. Mycobacteria target DC-SIGN to suppress dendritic cell function. J Exp Med. 2003;197(1):7–17. doi:10.1084/jem.20021229
49. Dao et al. 2004 Mycobacterium tuberculosis lipomannan induces apoptosis and interleukin-12.pdf. Available from: https://iai.asm.org/content/72/4/2067.full.pdf.
50. Quesniaux VJ, Nicolle DM, Torres D, et al. Toll-Like Receptor 2 (TLR2)-dependent-positive and TLR2-independent-negative regulation of proinflammatory cytokines by mycobacterial lipomannans. J Immunol. 2004;172(7):4425–4434. doi:10.4049/jimmunol.172.7.4425
51. Indrigo J. Cord factor trehalose 6,6ʹ-dimycolate (TDM) mediates trafficking events during mycobacterial infection of murine macrophages. Microbiology. 2003;149(8):2049–2059. doi:10.1099/mic.0.26226-0
52. Axelrod S, Oschkinat H, Enders J, et al. Delay of phagosome maturation by a mycobacterial lipid is reversed by nitric oxide. Cell Microbiol. 2008;10(7):1530–1545. doi:10.1111/j.1462-5822.2008.01147.x
53. Welsh KJ, Abbott AN, Hwang S-A, et al. A role for tumour necrosis factor-, complement C5 and interleukin-6 in the initiation and development of the mycobacterial cord factor trehalose 6,6ʹ-dimycolate induced granulomatous response. Microbiology. 2008;154(6):1813–1824. doi:10.1099/mic.0.2008/016923-0
54. Perez RL, Roman J, Roser S, et al. Cytokine message and protein expression during lung granuloma formation and resolution induced by the mycobacterial cord factor trehalose-6,6′-dimycolate. J Interferon Cytokine Res. 2000;20(9):795–804. doi:10.1089/10799900050151067
55. Laneelle G, Tocanne J-F. Evidence for penetration in liposomes and in mitochondrial membranes of a fluorescent analogue of cord factor. Eur J Biochem. 1980;109(1):177–182. doi:10.1111/j.1432-1033.1980.tb04782.x
56. Kato M. Site II-specific inhibition of mitochondrial oxidative phosphorylation by trehalose-6,6′-dimycolate (cord factor) of Mycobacterium tuberculosis. Arch Biochem Biophys. 1970;140(2):379–390. doi:10.1016/0003-9861(70)90079-2
57. Ozeki Y, Kaneda K, Fujiwara N, Morimoto M, Oka S, Yano I. In vivo induction of apoptosis in the thymus by administration of mycobacterial cord factor (trehalose 6,6’-dimycolate).. Infect Immun. 1997;65(5):1793–1799. doi:10.1128/IAI.65.5.1793-1799.1997
58. Gilleron M, Ronet C, Mempel M, Monsarrat B, Gachelin G, Puzo G. Acylation state of the phosphatidylinositol mannosides from mycobacterium bovis bacillus calmette guérin and ability to induce granuloma and recruit natural killer T cells. J Biol Chem. 2001;276(37):34896–34904. doi:10.1074/jbc.M103908200
59. Court N, Rose S, Bourigault M-L, et al. Mycobacterial PIMs inhibit host inflammatory responses through CD14-Dependent and CD14-Independent mechanisms. PLoS One. 2011;6(9):e24631. doi:10.1371/journal.pone.0024631
60. Cambier CJ, Takaki KK, Larson RP, et al. Mycobacteria manipulate macrophage recruitment through coordinated use of membrane lipids. Nature. 2014;505(7482):218–222. doi:10.1038/nature12799
61. Rousseau C, Winter N, Pivert E, et al. Production of phthiocerol dimycocerosates protects Mycobacterium tuberculosis from the cidal activity of reactive nitrogen intermediates produced by macrophages and modulates the early immune response to infection. Cell Microbiol. 2004;6(3):277–287. doi:10.1046/j.1462-5822.2004.00368.x
62. Astarie-Dequeker C, Le Guyader L, Malaga W, et al. Phthiocerol dimycocerosates of M. tuberculosis participate in macrophage invasion by inducing changes in the organization of plasma membrane lipids. PLoS Pathog. 2009;5(2):e1000289. doi:10.1371/journal.ppat.1000289
63. Augenstreich J, Arbues A, Simeone R, et al. ESX-1 and phthiocerol dimycocerosates of Mycobacterium tuberculosis act in concert to cause phagosomal rupture and host cell apoptosis. Cell Microbiol. 2017;19(7):e12726. doi:10.1111/cmi.12726
64. Saint-Joanis B, Demangel C, Jackson M, et al. Inactivation of Rv2525c, a substrate of the Twin Arginine Translocation (Tat) System of Mycobacterium tuberculosis, increases -lactam susceptibility and virulence. J Bacteriol. 2006;188(18):6669–6679. doi:10.1128/JB.00631-06
65. Bhuwan M, Arora N, Sharma A, et al. Interaction of Mycobacterium tuberculosis virulence factor RipA with chaperone MoxR1 is required for transport through the TAT secretion system. mBio. 2016;7(2):e02259–15. doi:10.1128/mBio.02259-15
66. Berthet F. Attenuation of virulence by disruption of the Mycobacterium tuberculosis erp Gene. Science. 1998;282(5389):759–762. doi:10.1126/science.282.5389.759
67. Skjot RLV, Oettinger T, Rosenkrands I, et al. Comparative evaluation of low-molecular-mass proteins from mycobacterium tuberculosis identifies members of the ESAT-6 family as immunodominant T-cell antigens. Infect Immun. 2000;68(1):214–220. doi:10.1128/IAI.68.1.214-220.2000
68. Elhay MJ, Oettinger T, Andersen P. Delayed-type hypersensitivity responses to ESAT-6 and MPT64 from Mycobacterium tuberculosis in the Guinea Pig.:3. Infect Immun. 1998;66:3454–3456. doi:10.1128/IAI.66.7.3454-3456.1998
69. Ganguly N, Giang PH, Gupta C, et al. Mycobacterium tuberculosis secretory proteins CFP-10, ESAT-6 and the CFP10: ESAT6complex inhibit lipopolysaccharide-induced NF-κB transactivation by downregulation of reactive oxidative species (ROS) production. Immunol Cell Biol. 2008;86(1):98–106. doi:10.1038/sj.icb.7100117
70. Pathak SK, Basu S, Basu KK, et al. Direct extracellular interaction between the early secreted antigen ESAT-6 of Mycobacterium tuberculosis and TLR2 inhibits TLR signaling in macrophages. Nat Immunol. 2007;8(6):610–618. doi:10.1038/ni1468
71. Derrick SC, Morris SL. The ESAT6 protein of Mycobacterium tuberculosis induces apoptosis of macrophages by activating caspase expression. Cell Microbiol. 2007;9(6):1547–1555. doi:10.1111/j.1462-5822.2007.00892.x
72. Smith J, Manoranjan J, Pan M, et al. Evidence for pore formation in host cell membranes by ESX-1-secreted ESAT-6 and its role in Mycobacterium marinum escape from the vacuole. Infect Immun. 2008;76(12):5478–5487. doi:10.1128/IAI.00614-08
73. Kinhikar AG, Verma I, Chandra D, et al. Potential role for ESAT6 in dissemination of Maf tuberculosis via human lung epithelial cells. Mol Microbiol. 2010;75(1):92–106. doi:10.1111/j.1365-2958.2009.06959.x
74. van der Wel N, Hava D, Houben D, et al. M. tuberculosis and M. leprae translocate from the phagolysosome to the cytosol in myeloid cells. Cell. 2007;129(7):1287–1298. doi:10.1016/j.cell.2007.05.059
75. Reed MB, Domenech P, Manca C, et al. A glycolipid of hypervirulent tuberculosis strains that inhibits the innate immune response. Nature. 2004;431(7004):84–87. doi:10.1038/nature02837
76. Alexander KA, Sanderson CE, Larsen MH, Robbe-austerman S, Williams MC, Palmer MV. Emerging tuberculosis pathogen hijacks social communication behavior in the group-living banded mongoose (Mungos mungo). mBio. 2016;7:3. doi:10.1128/mBio.00281-16
77. Henderson RA, Watkins SC, Flynn JL. Activation of human dendritic cells following infection with Mycobacterium tuberculosis. J Immunol. 1997;159(2):635–643.
78. Ganbat D, Seehase S, Richter E, et al. Mycobacteria infect different cell types in the human lung and cause species dependent cellular changes in infected cells. BMC Pulm Med. 2016:16. doi:10.1186/s12890-016-0185-5.
79. Divangahi M, Mostowy S, Coulombe F, et al. NOD2-deficient mice have impaired resistance to mycobacterium tuberculosis infection through defective innate and adaptive immunity. J Immunol. 2008;181(10):7157–7165. doi:10.4049/jimmunol.181.10.7157
80. Flannagan RS, Cosío G, Grinstein S. Antimicrobial mechanisms of phagocytes and bacterial evasion strategies. Nat Rev Microbiol. 2009;7(5):355–366. doi:10.1038/nrmicro2128
81. Philips JA, Ernst JD. Tuberculosis pathogenesis and Immunity. Annu Rev Pathol Mech Dis. 2012;7(1):353–384. doi:10.1146/annurev-pathol-011811-132458
82. Schnettger L, Rodgers A, Repnik U, et al. A Rab20-dependent membrane trafficking pathway controls M. tuberculosis replication by regulating phagosome spaciousness and integrity. Cell Host Microbe. 2017;21(5):619–628.e5. doi:10.1021/es1034188
83. Tăbăran A-F, Cornel C. Macrophages targeted drug delivery as a key therapy in infectious disease. Biotechnol Mol Biol Nanomedicine. 2014;2:1.
84. Sturgill-Koszycki S, Schlesinger PH, Chakraborty P, et al. Lack of acidification in Mycobacterium phagosomes produced by exclusion of the vesicular proton-ATPase. Science. 1994;263(5147):678–681. doi:10.1126/science.8303277
85. Stanley LR, Vinay K, Abul K, Ramzi S, Nelson F. Robbins & Cotran Pathologic Basis of Disease. Saunders/Elsevier; 2010.
86. Ferrari G, Langen H, Naito M, Pieters J. A coat protein on phagosomes involved in the intracellular survival of mycobacteria. Cell. 1999;97(4):435–447. doi:10.1016/S0092-8674(00)80754-0
87. Hmama Z, Sendide K, Talal A, Garcia R, Dobos K, Reiner NE. Quantitative analysis of phagolysosome fusion in intact cells: inhibition by mycobacterial lipoarabinomannan and rescue by an 1α, 25-dihydroxyvitamin D3–phosphoinositide 3-kinase pathway. J Cell Sci. 2004;117(10):2131–2140. doi:10.1242/jcs.01072
88. Pabst M, Gross J, Brozna J, Goren M. Inhibition of macrophage priming by sulfatide from Mycobacterium tuberculosis. J Immunol. 1988;140(2):634–640.
89. Vergne I, Chua J, Deretic V. Tuberculosis toxin blocking phagosome maturation inhibits a novel Ca2+/calmodulin-PI3K hVPS34 cascade. J Exp Med. 2003;198(4):653–659. doi:10.1084/jem.20030527
90. Clemens DL, Horwitz MA. The Mycobacterium tuberculosis phagosome interacts with early endosomes and is accessible to exogenously administered transferrin. J Exp Med. 1996;184(4):1349–1355. doi:10.1084/jem.184.4.1349
91. Lyadova IV, Panteleev AV. Th1 and Th17 cells in tuberculosis: protection, pathology, and biomarkers. Mediators Inflamm. 2015;2015:1–13. doi:10.1155/2015/854507
92. Ashenafi S, Aderaye G, Bekele A, et al. Progression of clinical tuberculosis is associated with a Th2 immune response signature in combination with elevated levels of SOCS3. Clin Immunol. 2014;151(2):84–99. doi:10.1016/j.clim.2014.01.010
93. Infante-Duarto C, Kamradt T Thl/Th2 balance in infection.:22.
94. Mohareer K, Asalla S, Banerjee S. Cell death at the cross roads of host-pathogen interaction in Mycobacterium tuberculosis infection. Tuberculosis. 2018;113:99–121. doi:10.1016/j.tube.2018.09.007
95. Lerner TR, Borel S, Greenwood DJ, et al. Mycobacterium tuberculosis replicates within necrotic human macrophages. J Cell Biol. 2017;216(3):583–594. doi:10.1161/CIRCRESAHA.117.311401
96. Dallenga T, Repnik U, Corleis B, et al. M. tuberculosis-induced necrosis of infected neutrophils promotes bacterial growth following phagocytosis by macrophages. Cell Host Microbe. 2017;22(4):519–530.e3. doi:10.1016/j.chom.2017.09.003
97. Queval CJ, Brosch R, Simeone R. The macrophage: a disputed fortress in the battle against mycobacterium tuberculosis. Front Microbiol. 2017;8. doi:10.3389/fmicb.2017.02284
98. Shanmugasundaram T, Radhakrishnan M, Gopikrishnan V, Pazhanimurugan R, Balagurunathan R. A study of the bactericidal, anti-biofouling, cytotoxic and antioxidant properties of actinobacterially synthesised silver nanoparticles. Colloids Surf B Biointerfaces. 2013;111:680–687. doi:10.1016/j.colsurfb.2013.06.045
99. Aderibigbe BA. Metal-based nanoparticles for the treatment of infectious diseases. Mol Basel Switz. 2017;22:8. doi:10.3390/molecules22081370
100. Fathima JB, Pugazhendhi A, Venis R. Synthesis and characterization of ZrO2 nanoparticles-antimicrobial activity and their prospective role in dental care. Microb Pathog. 2017;110:245–251. doi:10.1016/j.micpath.2017.06.039
101. Shanmuganathan R, LewisOscar F, Shanmugam S, et al. Core/shell nanoparticles: synthesis, investigation of antimicrobial potential and photocatalytic degradation of Rhodamine B. J Photochem Photobiol B. 2020;202:111729. doi:10.1016/j.jphotobiol.2019.111729
102. Pugazhendhi A, Kumar SS, Manikandan M, Saravanan M. Photocatalytic properties and antimicrobial efficacy of Fe doped CuO nanoparticles against the pathogenic bacteria and fungi. Microb Pathog. 2018;122:84–89. doi:10.1016/j.micpath.2018.06.016
103. Vasantharaj S, Sathiyavimal S, Senthilkumar P, LewisOscar F, Pugazhendhi A. Biosynthesis of iron oxide nanoparticles using leaf extract of Ruellia tuberosa: antimicrobial properties and their applications in photocatalytic degradation. J Photochem Photobiol B. 2019;192:74–82. doi:10.1016/j.jphotobiol.2018.12.025
104. Reznickova A, Slavikova N, Kolska Z, et al. PEGylated gold nanoparticles: stability, cytotoxicity and antibacterial activity. Colloids Surf Physicochem Eng Asp. 2019;560:26–34. doi:10.1016/j.colsurfa.2018.09.083
105. Mocan L, Matea C, Tabaran FA, et al. Selective in vitro photothermal nano-therapy of MRSA infections mediated by IgG conjugated gold nanoparticles. Sci Rep. 2016:6. doi:10.1038/srep39466.
106. Jeyarani S, Vinita NM, Puja P, et al. Biomimetic gold nanoparticles for its cytotoxicity and biocompatibility evidenced by fluorescence-based assays in cancer (MDA-MB-231) and non-cancerous (HEK-293) cells. J Photochem Photobiol B. 2020;202:111715. doi:10.1016/j.jphotobiol.2019.111715
107. Chellapandian C, Ramkumar B, Puja P, Shanmuganathan R, Pugazhendhi A, Kumar P. Gold nanoparticles using red seaweed Gracilaria verrucosa: green synthesis, characterization and biocompatibility studies. Process Biochem. 2019;80:58–63. doi:10.1016/j.procbio.2019.02.009
108. Pugazhendhi A, Prabhu R, Muruganantham K, Shanmuganathan R, Natarajan S. Anticancer, antimicrobial and photocatalytic activities of green synthesized magnesium oxide nanoparticles (MgONPs) using aqueous extract of Sargassum wightii. J Photochem Photobiol B. 2019;190:86–97. doi:10.1016/j.jphotobiol.2018.11.014
109. Hariharan D, Thangamuniyandi P, Jegatha Christy A, et al. Enhanced photocatalysis and anticancer activity of green hydrothermal synthesized Ag@TiO2 nanoparticles. J Photochem Photobiol B. 2020;202:111636. doi:10.1016/j.jphotobiol.2019.111636
110. Hariharan D, Thangamuniyandi P, Selvakumar P, et al. Green approach synthesis of Pd@TiO2 nanoparticles: characterization, visible light active picric acid degradation and anticancer activity. Process Biochem. 2019;87:83–88. doi:10.1016/j.procbio.2019.09.024
111. Ramkumar VS, Pugazhendhi A, Gopalakrishnan K, et al. Biofabrication and characterization of silver nanoparticles using aqueous extract of seaweed Enteromorpha compressa and its biomedical properties. Biotechnol Rep. 2017;14:1–7. doi:10.1016/j.btre.2017.02.001
112. Madhubala V, Pugazhendhi A, Thirunavukarasu K. Cytotoxic and immunomodulatory effects of the low concentration of titanium dioxide nanoparticles (TiO2 NPs) on human cell lines - An in vitro study. Process Biochem. 2019;86:186–195. doi:10.1016/j.procbio.2019.08.004
113. Fathima JB, Pugazhendhi A, Oves M, Venis R. Synthesis of eco-friendly copper nanoparticles for augmentation of catalytic degradation of organic dyes. J Mol Liq. 2018;260:1–8. doi:10.1016/j.molliq.2018.03.033
114. Vasantharaj S, Sathiyavimal S, Saravanan M, et al. Synthesis of ecofriendly copper oxide nanoparticles for fabrication over textile fabrics: characterization of antibacterial activity and dye degradation potential. J Photochem Photobiol B. 2019;191:143–149. doi:10.1016/j.jphotobiol.2018.12.026
115. Saratale RG, Ghodake GS, Shinde SK, et al. Photocatalytic activity of CuO/Cu(OH)2 nanostructures in the degradation of Reactive Green 19A and textile effluent, phytotoxicity studies and their biogenic properties (antibacterial and anticancer). J Environ Manage. 2018;223:1086–1097. doi:10.1016/j.jenvman.2018.04.072
116. Varadavenkatesan T, Lyubchik E, Pai S, Pugazhendhi A, Vinayagam R, Selvaraj R. Photocatalytic degradation of Rhodamine B by zinc oxide nanoparticles synthesized using the leaf extract of Cyanometra ramiflora. J Photochem Photobiol B. 2019;199:111621. doi:10.1016/j.jphotobiol.2019.111621
117. Shanmuganathan R, Edison TNJI, LewisOscar F, Kumar P, Shanmugam S, Pugazhendhi A. Chitosan nanopolymers: an overview of drug delivery against cancer. Int J Biol Macromol. 2019;130:727–736. doi:10.1016/j.ijbiomac.2019.02.060
118. Sanpui P, Chattopadhyay A, Ghosh SS. Induction of apoptosis in cancer cells at low silver nanoparticle concentrations using chitosan nanocarrier. ACS Appl Mater Interfaces. 2011;3(2):218–228. doi:10.1021/am100840c
119. Suganya M, Gnanamangai BM, Govindasamy C, et al. Mitochondrial dysfunction mediated apoptosis of HT-29 cells through CS-PAC-AgNPs and investigation of genotoxic effects in zebra (Danio rerio) fish model for drug delivery. Saudi J Biol Sci. 2019;26(4):767–776. doi:10.1016/j.sjbs.2019.03.007
120. Russell AD, Hugo WB. 7 Antimicrobial activity and action of silver. In: Progress in Medicinal Chemistry. Vol. 31. Elsevier;1994:
121. Silva JP, Appelberg R, Gama FM. Antimicrobial peptides as novel anti-tuberculosis therapeutics. Biotechnol Adv. 2016;34(5):924–940. doi:10.1016/j.biotechadv.2016.05.007
122. Khan F, Khan MM, Kim Y-M. Recent progress and future perspectives of antibiofilm drugs immobilized on nanomaterials. Curr Pharm Biotechnol. 2018;19(8):631–643. doi:10.2174/1389201019666180828090052
123. Raffi M, Mehrwan S, Bhatti TM, et al. Investigations into the antibacterial behavior of copper nanoparticles against Escherichia coli. Ann Microbiol. 2010;60(1):75–80. doi:10.1007/s13213-010-0015-6
124. Pham DTN, Khan F, Phan TTV, et al. Biofilm inhibition, modulation of virulence and motility properties by FeOOH nanoparticle in Pseudomonas aeruginosa. Braz J Microbiol. 2019;50(3):791–805. doi:10.1007/s42770-019-00108-z
125. Khan F, Manivasagan P, Lee J-W, Pham DTN, Oh J, Kim Y-M. Fucoidan-stabilized gold nanoparticle-mediated biofilm inhibition, attenuation of virulence and motility properties in Pseudomonas aeruginosa PAO1. Mar Drugs. 2019;17:4. doi:10.3390/md17040208
126. Javaid A, Oloketuyi SF, Khan MM, Khan F. Diversity of bacterial synthesis of silver nanoparticles. BioNanoScience. 2018;8(1):43–59. doi:10.1007/s12668-017-0496-x
127. Mocan T, Matea CT, Pop T, et al. Carbon nanotubes as anti-bacterial agents. Cell Mol Life Sci CMLS. 2017;74(19):3467–3479. doi:10.1007/s00018-017-2532-y
128. Khan F, Pham DTN, Oloketuyi SF, Manivasagan P, Oh J, Kim Y-M. Chitosan and their derivatives: antibiofilm drugs against pathogenic bacteria. Colloids Surf B Biointerfaces. 2020;185:110627. doi:10.1016/j.colsurfb.2019.110627
129. Khan F, Manivasagan P, Pham DTN, Oh J, Kim S-K, Kim Y-M. Antibiofilm and antivirulence properties of chitosan-polypyrrole nanocomposites to Pseudomonas aeruginosa. Microb Pathog. 2019;128:363–373. doi:10.1016/j.micpath.2019.01.033
130. Manivasagan P, Khan F, Hoang G, et al. Thiol chitosan-wrapped gold nanoshells for near-infrared laser-induced photothermal destruction of antibiotic-resistant bacteria. Carbohydr Polym. 2019;225:115228. doi:10.1016/j.carbpol.2019.115228
131. Zazo H, Colino CI, Lanao JM. Current applications of nanoparticles in infectious diseases. J Control Release off J Control Release Soc. 2016;224:86–102. doi:10.1016/j.jconrel.2016.01.008
132. Costa-gouveia J, Aínsa JA, Brodin P, Lucía A. How can nanoparticles contribute to antituberculosis therapy? Drug Discov Today. 2017;22(3):600–607. doi:10.1016/j.drudis.2017.01.011
133. Kreytsberg GN, Gracheva IE, Kibrik BS, Golikov IV. Antituberculous effect of silver nanoparticles. J Phys Conf Ser. 2011;291:012030. doi:10.1088/1742-6596/291/1/012030
134. Song B, Zhang C, Zeng G, Gong J, Chang Y, Jiang Y. Antibacterial properties and mechanism of graphene oxide-silver nanocomposites as bactericidal agents for water disinfection. Arch Biochem Biophys. 2016;604:167–176. doi:10.1016/j.abb.2016.04.018
135. Liu C, Guo J, Yan X, et al. Antimicrobial nanomaterials against biofilms: an alternative strategy. Environ Rev. 2016;25(2):225–244. doi:10.1139/er-2016-0046
136. Franci G, Falanga A, Galdiero S, et al. Silver nanoparticles as potential antibacterial agents. Mol Basel Switz. 2015;20(5):8856–8874. doi:10.3390/molecules20058856
137. Dakal TC, Kumar A, Majumdar RS, Yadav V. Mechanistic basis of antimicrobial actions of silver nanoparticles. Front Microbiol. 2016;7:1831. doi:10.3389/fmicb.2016.01831
138. Morones JR, Elechiguerra JL, Camacho A, et al. The bactericidal effect of silver nanoparticles. Nanotechnology. 2005;16(10):2346. doi:10.1088/0957-4484/16/10/059
139. Kumar DA, Palanichamy V, Roopan SM. Green synthesis of silver nanoparticles using Alternanthera dentata leaf extract at room temperature and their antimicrobial activity. Spectrochim Acta A Mol Biomol Spectrosc. 2014;127:168–171. doi:10.1016/j.saa.2014.02.058
140. Jain J, Arora S, Rajwade JM, Omray P, Khandelwal S, Paknikar KM. Silver nanoparticles in therapeutics: development of an antimicrobial gel formulation for topical use. Mol Pharm. 2009;6(5):1388–1401. doi:10.1021/mp900056g
141. Le Ouay B, Stellacci F. Antibacterial activity of silver nanoparticles: a surface science insight. Nano Today. 2015;10(3):339–354. doi:10.1016/j.nantod.2015.04.002
142. Huh AJ, Kwon YJ. “Nanoantibiotics”: a new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. J Control Release off J Control Release Soc. 2011;156(2):128–145. doi:10.1016/j.jconrel.2011.07.002
143. Kim JS, Kuk E, Yu KN, et al. Antimicrobial effects of silver nanoparticles. Nanomedicine Nanotechnol Biol Med. 2007;3(1):95–101. doi:10.1016/j.nano.2006.12.001
144. Al-Sharqi A, Apun K, Vincent M, Kanakaraju D, Bilung LM. Enhancement of the antibacterial efficiency of silver nanoparticles against gram-positive and gram-negative bacteria using blue laser light. Int J Photoenergy. doi:10.1155/2019/2528490
145. Velayati AA, Farnia P, Ibrahim TA, et al. Differences in cell wall thickness between resistant and nonresistant strains of Mycobacterium tuberculosis: using transmission electron microscopy. Chemotherapy. 2009;55(5):303–307. doi:10.1159/000226425
146. Mai-Prochnow A, Clauson M, Hong J, Murphy AB. Gram positive and Gram negative bacteria differ in their sensitivity to cold plasma. Sci Rep. 2016;6. doi:10.1038/srep38610
147. Hett EC, Rubin EJ. Bacterial growth and cell division: a mycobacterial perspective. Microbiol Mol Biol Rev MMBR. 2008;72(1):126–156. doi:10.1128/MMBR.00028-07
148. Saravanan M, Barik SK, MubarakAli D, Prakash P, Pugazhendhi A. Synthesis of silver nanoparticles from Bacillus brevis (NCIM 2533) and their antibacterial activity against pathogenic bacteria. Microb Pathog. 2018;116:221–226. doi:10.1016/j.micpath.2018.01.038
149. Seth D, Sarkar A, Mitra D. Nanomedicine to counter syndemic tuberculosis and HIV infection: current knowledge and state of art. Nanosci Nanoeng. 2014;9.
150. Abdel-Aziz MM, Elella MHA, Mohamed RR. Green synthesis of quaternized chitosan/silver nanocomposites for targeting mycobacterium tuberculosis and lung carcinoma cells (A-549). Int J Biol Macromol. 2019. doi:10/ggdd6n
151. El Badawy AM, Silva RG, Morris B, Scheckel KG, Suidan MT, Tolaymat TM. Surface charge-dependent toxicity of silver nanoparticles. Environ Sci Technol. 2011;45(1):283–287. doi:10/cc29h5
152. Sondi I, Salopek-Sondi B. Silver nanoparticles as antimicrobial agent: a case study on E. coli as a model for Gram-negative bacteria. J Colloid Interface Sci. 2004;275(1):177–182. doi:10.1016/j.jcis.2004.02.012
153. Cho K-H, Park J-E, Osaka T, Park S-G. The study of antimicrobial activity and preservative effects of nanosilver ingredient. Electrochim Acta. 2005;51(5):956–960. doi:10.1016/j.electacta.2005.04.071
154. McQuillan JS, Groenaga Infante H, Stokes E, Shaw AM. Silver nanoparticle enhanced silver ion stress response in Escherichia coli K12. Nanotoxicology. 2012;6(8):857–866. doi:10.3109/17435390.2011.626532
155. Orlov IA, Sankova TP, Babich PS, et al. New silver nanoparticles induce apoptosis-like process in E. coli and interfere with mammalian copper metabolism. Int J Nanomedicine. doi:10.2147/IJN.S117745
156. Rodgers FG, Tzianabos AO, Elliott TSJ. The effect of antibiotics that inhibit cell-wall, protein, and DNA synthesis on the growth and morphology of Legionella pneumophila. J Med Microbiol. 1990;31(1):37–44. doi:10.1099/00222615-31-1-37
157. van der Wal A, Norde W, Zehnder AJB, Lyklema J. Determination of the total charge in the cell walls of Gram-positive bacteria. Colloids Surf B Biointerfaces. 1997;9(1–2):81–100. doi:10.1016/S0927-7765(96)01340-9
158. Shaik M, Albalawi G, Khan S, et al. “Miswak” based green synthesis of silver nanoparticles: evaluation and comparison of their microbicidal activities with the chemical synthesis. Molecules. 2016;21(11):1478. doi:10.3390/molecules21111478
159. Ashraf S, Akhtar N, Ghauri M, Rajoka M, Khalid ZM, Hussain I. Polyhexamethylene biguanide functionalized cationic silver nanoparticles for enhanced antimicrobial activity. Nanoscale Res Lett. 2012;7(1):267. doi:10.1186/1556-276X-7-267
160. Ivask A, ElBadawy A, Kaweeteerawat C, et al. Toxicity mechanisms in escherichia coli vary for silver nanoparticles and differ from ionic silver. ACS Nano. 2014;8(1):374–386. doi:10/f5qqhz
161. Kvítek L, Panáček A, Soukupová J, et al. Effect of surfactants and polymers on stability and antibacterial activity of silver nanoparticles (NPs). J Phys Chem C. 2008;112(15):5825–5834. doi:10.1021/jp711616v
162. Yin R, Agrawal T, Khan U, et al. Antimicrobial photodynamic inactivation in nanomedicine: small light strides against bad bugs. Nanomed. 2015;10(15):2379–2404. doi:10.2217/nnm.15.67
163. Yun’an Qing LC, Li R, Liu G, et al. Potential antibacterial mechanism of silver nanoparticles and the optimization of orthopedic implants by advanced modification technologies. Int J Nanomedicine. 2018;13:3311. doi:10.2147/IJN.S165125
164. Bonnet M, Massard C, Veisseire P, Camares O, Awitor KO. Environmental toxicity and antimicrobial efficiency of titanium dioxide nanoparticles in suspension. J Biomater Nanobiotech. 2015;6(3):213–224. doi:10.4236/jbnb.2015.63020
165. Parikh AN, Gillmor SD, Beers JD, Beardmore KM, Cutts RW, Swanson BI. Characterization of chain molecular assemblies in long-chain, layered silver thiolates: a joint infrared spectroscopy and X-ray diffraction study. J Phys Chem B. 1999;103(15):2850–2861. doi:10.1021/jp983938b
166. Dobias J, Bernier-Latmani R. Silver release from silver nanoparticles in natural waters. Environ Sci Technol. 2013;47(9):4140–4146. doi:10.1021/es304023p
167. Park E-J, Yi J, Kim Y, Choi K, Park K. Silver nanoparticles induce cytotoxicity by a Trojan-horse type mechanism. Toxicol in Vitro. 2010;24(3):872–878. doi:10.1016/j.tiv.2009.12.001
168. He D, Miller CJ, Waite TD. Fenton-like zero-valent silver nanoparticle-mediated hydroxyl radical production. J Catal. 2014;317:198–205. doi:10.1016/j.jcat.2014.06.016
169. Sisubalan N, Ramkumar VS, Pugazhendhi A, et al. ROS-mediated cytotoxic activity of ZnO and CeO2 nanoparticles synthesized using the Rubia cordifolia L. leaf extract on MG-63 human osteosarcoma cell lines. Environ Sci Pollut Res. 2018;25(11):10482–10492. doi:10.1007/s11356-017-0003-5
170. Knaapen AM, Borm PJA, Albrecht C, Schins RPF. Inhaled particles and lung cancer. Part A: mechanisms. Int J Cancer. 2004;109(6):799–809. doi:10.1002/ijc.11708
171. Samuel MS, Jose S, Selvarajan E, Mathimani T, Pugazhendhi A. Biosynthesized silver nanoparticles using Bacillus amyloliquefaciens; application for cytotoxicity effect on A549 cell line and photocatalytic degradation of p-nitrophenol. J Photochem Photobiol B. 2020;202:111642. doi:10.1016/j.jphotobiol.2019.111642
172. Speight JG, editor. Redox Transformations. In: Reaction Mechanisms in Environmental Engineering. Elsevier; 2018:231–267. doi:10.1016/B978-0-12-804422-3.00007-9
173. Forrester SJ, Kikuchi DS, Hernandes MS, Xu Q, Griendling KK. Reactive oxygen species in metabolic and inflammatory signaling. Circ Res. 2018;122(6):877–902. doi:10.1161/CIRCRESAHA.117.311401
174. Zielinska E, Tukaj C, Radomski MW, Inkielewicz-stepniak I. Molecular mechanism of silver nanoparticles-induced human osteoblast cell death: protective effect of inducible nitric oxide synthase inhibitor. PLoS One. 2016;11(10):e0164137. doi:10/f9rvdx
175. Bressan E, Ferroni L, Gardin C, et al. Silver nanoparticles and mitochondrial interaction. Int J Dent. 2013;2013:1–8. doi:10.1155/2013/312747
176. Maurer LL, Meyer JN. A systematic review of evidence for silver nanoparticle-induced mitochondrial toxicity. Environ Sci Nano. 2016;3(2):311–322. doi:10.1039/C5EN00187K
177. Kim S, Choi JE, Choi J, et al. Oxidative stress-dependent toxicity of silver nanoparticles in human hepatoma cells. Toxicol in Vitro. 2009;23(6):1076–1084. doi:10.1016/j.tiv.2009.06.001
178. Miyayama T, Matsuoka M. Involvement of lysosomal dysfunction in silver nanoparticle-induced cellular damage in A549 human lung alveolar epithelial cells. J Occup Med Toxicol. 2016;11(1):1. doi:10.1186/s12995-016-0090-0
179. Park MVDZ, Neigh AM, Vermeulen JP, et al. The effect of particle size on the cytotoxicity, inflammation, developmental toxicity and genotoxicity of silver nanoparticles. Biomaterials. 2011;32(36):9810–9817. doi:10.1016/j.biomaterials.2011.08.085
180. Singh R, Nawale L, Arkile M, et al. Phytogenic silver, gold, and bimetallic nanoparticles as novel antitubercular agents. Int J Nanomedicine. 2016;11:1889.
181. Xiu Z, Zhang Q, Puppala HL, Colvin VL, Alvarez PJJ. Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Lett. 2012;12(8):4271–4275. doi:10.1021/nl301934w
182. Behra R, Sigg L, Clift MJD, et al. Bioavailability of silver nanoparticles and ions: from a chemical and biochemical perspective. J R Soc Interface. 2013;10:87. doi:10.1098/rsif.2013.0396
183. Saratale RG, Karuppusamy I, Saratale GD, et al. A comprehensive review on green nanomaterials using biological systems: recent perception and their future applications. Colloids Surf B Biointerfaces. 2018;170:20–35. doi:10.1016/j.colsurfb.2018.05.045
184. Saratale RG, Saratale GD, Shin HS, et al. New insights on the green synthesis of metallic nanoparticles using plant and waste biomaterials: current knowledge, their agricultural and environmental applications. Environ Sci Pollut Res. 2018;25(11):10164–10183. doi:10.1007/s11356-017-9912-6
185. Kote JR, Kadam AS, Patil SS, Mane RS. Green functionalized silver nanoparticles with significantly enhanced antimycobactericidal and cytotoxicity performances of asparagus racemosus Linn. Int J New Technol Sci. 2016;3(2):15.
186. Roy A, Bulut O, Some S, Mandal AK, Yilmaz MD. Green synthesis of silver nanoparticles: biomolecule-nanoparticle organizations targeting antimicrobial activity. RSC Adv. 2019;9(5):2673–2702. doi:10.1039/C8RA08982E
187. Kerry RG, Gouda S, Sil B, et al. Cure of tuberculosis using nanotechnology: an overview. J Microbiol. 2018;56(5):287–299. doi:10.1007/s12275-018-7414-y
188. Arokiyaraj S, Arasu MV, Vincent S, et al. Rapid green synthesis of silver nanoparticles from Chrysanthemum indicum L and its antibacterial and cytotoxic effects: an in vitro study. Int J Nanomedicine. doi:10.2147/IJN.S53546
189. Ahmed S, Ahmad M, Swami BL, Ikram S. A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: a green expertise. J Adv Res. 2016;7(1):17–28. doi:10.1016/j.jare.2015.02.007
190. Shaik M, Khan M, Kuniyil M, et al. Plant-extract-assisted green synthesis of silver nanoparticles using origanum vulgare L. Extract and their microbicidal activities. Sustainability. 2018;10(4):913. doi:10.3390/su10040913
191. Pugazhendhi A, Prabakar D, Jacob JM, Karuppusamy I, Saratale RG. Synthesis and characterization of silver nanoparticles using Gelidium amansii and its antimicrobial property against various pathogenic bacteria. Microb Pathog. 2018;114:41–45. doi:10.1016/j.micpath.2017.11.013
192. Oves M, Aslam M, Rauf MA, et al. Antimicrobial and anticancer activities of silver nanoparticles synthesized from the root hair extract of Phoenix dactylifera. Mater Sci Eng C. 2018;89:429–443. doi:10.1016/j.msec.2018.03.035
193. Singh R, Nawale LU, Arkile M, et al. Chemical and biological metal nanoparticles as antimycobacterial agents: a comparative study. Int J Antimicrob Agents. 2015;46(2):183–188. doi:10.1016/j.ijantimicag.2015.03.014
194. Rónavári A, Kovács D, Igaz N, et al. Biological activity of green-synthesized silver nanoparticles depends on the applied natural extracts: a comprehensive study. Int J Nanomedicine. 2017;12:871–883. doi:10.2147/IJN.S122842
195. Rai M, Ingle AP, Pandit R, et al. Broadening the spectrum of small-molecule antibacterials by metallic nanoparticles to overcome microbial resistance. Int J Pharm. 2017;532(1):139–148. doi:10.1016/j.ijpharm.2017.08.127
196. Baptista PV, McCusker MP, Carvalho A, et al. Nano-strategies to fight multidrug resistant bacteria—“a battle of the titans. Front Microbiol. 2018;9:1441. doi:10.3389/fmicb.2018.01441
197. Djafari J, Marinho C, Santos T, et al. New synthesis of gold- and silver-based nano-tetracycline composites. ChemistryOpen. 2016;5(3):206–212. doi:10.1002/open.201600016
198. Deng H, McShan D, Zhang Y, et al. Mechanistic study of the synergistic antibacterial activity of combined silver nanoparticles and common antibiotics. Environ Sci Technol. 2016;50(16):8840–8848. doi:10.1021/acs.est.6b00998
199. Birla SS, Tiwari VV, Gade AK, Ingle AP, Yadav AP, Rai MK. Fabrication of silver nanoparticles by Phoma glomerata and its combined effect against Escherichia coli, Pseudomonas aeruginosa and Staphylococcus aureus. Lett Appl Microbiol. 2009;48(2):173–179. doi:10.1111/j.1472-765X.2008.02510.x
200. Brown AN, Smith K, Samuels TA, Lu J, Obare SO, Scott ME. Nanoparticles functionalized with ampicillin destroy multiple-antibiotic-resistant isolates of pseudomonas aeruginosa and enterobacter aerogenes and methicillin-resistant Staphylococcus aureus. Appl Environ Microbiol. 2012;78(8):2768–2774. doi:10.1128/AEM.06513-11
201. Saratale GD, Saratale RG, Benelli G, et al. Anti-diabetic potential of silver nanoparticles synthesized with argyreia nervosa leaf extract high synergistic antibacterial activity with standard antibiotics against foodborne bacteria. J Clust Sci. 2017;28(3):1709–1727. doi:10.1007/s10876-017-1179-z
202. Shanmuganathan R, MubarakAli D, Prabakar D, et al. An enhancement of antimicrobial efficacy of biogenic and ceftriaxone-conjugated silver nanoparticles: green approach. Environ Sci Pollut Res. 2018;25(11):10362–10370. doi:10.1007/s11356-017-9367-9
203. Hwang I-S, Hwang JH, Choi H, Kim K-J, Lee DG. Synergistic effects between silver nanoparticles and antibiotics and the mechanisms involved. J Med Microbiol. 2012;61(Pt_12):1719–1726. doi:10.1099/jmm.0.047100-0
204. Farooq U, Ahmad T, Khan A, et al. Rifampicin conjugated silver nanoparticles: a new arena for development of antibiofilm potential against methicillin resistant Staphylococcus aureus and Klebsiella pneumoniae. Int J Nanomedicine. 2019;14:3983–3993. doi:10.2147/IJN.S198194
205. Li P, Li J, Wu C, Wu Q, Li J. Synergistic antibacterial effects of β-lactam antibiotic combined with silver nanoparticles. Nanotechnology. 2005;16(9):1912–1917. doi:10.1088/0957-4484/16/9/082
206. Arakha M, Jha S. Effects of photocatalytic nanoparticle interfaces on biological membranes and biomacromolecules. 2017. doi:10/ggdp7k
207. Nasiruddin M, Neyaz M, Das S. Nanotechnology-based approach in tuberculosis treatment. Tuberc Res Treat. 2017;2017. doi:10.1155/2017/4920209.
208. Singh R, Nawale L, Arkile M, et al. Phytogenic silver, gold, and bimetallic nanoparticles as novel antitubercular agents. Int J Nanomedicine. 2016;11:1889–1897. doi:10.2147/IJN.S102488
209. Song HY, Ko KK, Oh IH, Lee BT. Fabrication of silver nanoparticles and their antimicrobial mechanisms. Eur Cell Mater. 2006;11(suppl 1):58.
210. Jaryal N, Kaur H. Plumbago auriculata leaf extract-mediated AgNPs and its activities as antioxidant, anti-TB and dye degrading agents. J Biomater Sci Polym Ed. 2017;28(16):1847–1858. doi:10.1080/09205063.2017.1354673
211. Paarakh PM. Anti-tubercular activity of silver nanoparticle synthesized from the fruits of coriandrum sativum linn. World J Pharm Pharm Sci. 2017;1720–1727. doi:10/ggdkzz
212. Raja A, Salique SM, Gajalakshmi P, James A. Antibacterial and hemolytic activity of green silver nanoparticles from Catharanthus roseus. Int J Pharm Sci Nanotechnol. 2016;9(1):7.
213. Kote JR, Mulani RM, Kadam AS, Solankar BM. Anti-Mycobacterial Activity of Nanoparticles from Psidium Guajava L. 2014:5.
214. Daniel SCGK, Banu BN, Harshiny M, et al. Ipomea carnea -based silver nanoparticle synthesis for antibacterial activity against selected human pathogens. J Exp Nanosci. 2014;9(2):197–209. doi:10.1080/17458080.2011.654274
215. Banu A. Biosynthesis of monodispersed silver nanoparticles and their activity against Mycobacterium tuberculosis. J Nanomedicine Biotherapeutic Discov. 2013;03:01. doi:10.4172/2155-983X.1000110
216. Agarwal P, Mehta A, Kachhwaha S, Kothari SL. Green synthesis of silver nanoparticles and their activity against Mycobacterium tuberculosis. Adv Sci Eng Med. 2013;5(7):709–714. doi:10.1166/asem.2013.1307
217. Heidary M, Zaker Bostanabad S, Amini SM, et al. The anti-mycobacterial activity of Ag, ZnO, And Ag- ZnO nanoparticles against MDR- and XDR-Mycobacterium tuberculosis. Infect Drug Resist. 2019;12:3425–3435. doi:10.2147/IDR.S221408
218. Ellis T, Chiappi M, García-trenco A, et al. Multimetallic microparticles increase the potency of rifampicin against intracellular Mycobacterium tuberculosis. ACS Nano. 2018;12(6):5228–5240. doi:10.1021/acsnano.7b08264
219. Jafari A, Mosavari N, Movahedzadeh F, et al. Bactericidal impact of Ag, ZnO and mixed AgZnO colloidal nanoparticles on HRv Mycobacterium tuberculosis phagocytized by THP-1 cell lines. Microb Pathog. 2017;110:335–344. doi:10.1016/j.micpath.2017.07.010
220. Jena P, Mohanty S, Mallick R, Jacob B, Sonawane A. Toxicity and antibacterial assessment of chitosancoated silver nanoparticles on human pathogens and macrophage cells. Int J Nanomedicine. 2012;7:1805. doi:10.2147/IJN.S30631
221. Selim A, Elhaig MM, Taha SA, Nasr EA. Antibacterial activity of silver nanoparticles against field and reference strains of Mycobacterium tuberculosis, Mycobacterium bovis and multiple-drug-resistant tuberculosis strains. Rev Sci Tech OIE. 2018;37(3):823–830. doi:10.20506/rst.37.3.2888
222. Sivaraj A, Kumar V, Sunder R, Parthasarathy K, Kasivelu G. Commercial yeast extracts mediated green synthesis of silver chloride nanoparticles and their anti-mycobacterial activity. J Clust Sci. 2019. doi:10/ggdgtw
223. Kim J, Pitts B, Stewart PS, Camper A, Yoon J. Comparison of the antimicrobial effects of chlorine, silver ion, and tobramycin on biofilm. Antimicrob Agents Chemother. 2008;52(4):1446–1453. doi:10.1128/AAC.00054-07
224. Patel S. Biogenic silver nanoparticles as potential agent against mycobacterium tuberculosis. Int J Res Appl Sci Eng Technol. 2018;6(1):505–511. doi:10.22214/ijraset.2018.1075
225. Seth D, Choudhury SR, Pradhan S, et al. Nature-inspired novel drug design paradigm using nanosilver: efficacy on multi-drug-resistant clinical isolates of tuberculosis. Curr Microbiol. 2011;62(3):715–726. doi:10.1007/s00284-010-9770-7
226. Punjabi K, Mehta S, Chavan R, Chitalia V, Deogharkar D, Deshpande S. Efficiency of biosynthesized silver and zinc nanoparticles against multi-drug resistant pathogens. Front Microbiol. 2018;9. doi:10/gfdvdk
227. Sun F, Oh S, Kim J, et al. Enhanced internalization of macromolecular drugs into mycobacterium smegmatis with the assistance of silver nanoparticles. J Microbiol Biotechnol. 2017;27(8):1483–1490. doi:10.4014/jmb.1612.12041
228. Padmaa MP. Green synthesis of silver nanoparticles using fruits of coriandrum sativum linn and its antioxidant activity. J Nat Prod Resour. 2015;1(1):19–22.
229. Donnellan S, Tran L, Johnston H, McLuckie J, Stevenson K, Stone V. A rapid screening assay for identifying mycobacteria targeted nanoparticle antibiotics. Nanotoxicology. 2016;10(6):761–769. doi:10.3109/17435390.2015.1124468
230. Jafari AR, Mosavi T, Mosavari N, et al. Mixed metal oxide nanoparticles inhibit growth of Mycobacterium tuberculosis into THP-1 cells. Int J Mycobacteriology. 2016;5:S181–S183. doi:10.1016/j.ijmyco.2016.09.011
231. Islam MS, Larimer C, Ojha A, Nettleship I. Antimycobacterial efficacy of silver nanoparticles as deposited on porous membrane filters. Mater Sci Eng C. 2013;33(8):4575–4581. doi:10.1016/j.msec.2013.07.013
232. Mohanty S, Mishra S, Jena P, Jacob B, Sarkar B, Sonawane A. An investigation on the antibacterial, cytotoxic, and antibiofilm efficacy of starch-stabilized silver nanoparticles. Nanomedicine Nanotechnol Biol Med. 2012;8(6):916–924. doi:10.1016/j.nano.2011.11.007
233. Zhou Y, Kong Y, Kundu S, Cirillo JD, Liang H. Antibacterial activities of gold and silver nanoparticles against Escherichia coli and bacillus Calmette-Guérin. J Nanobiotechnology. 2012;10(1):19. doi:10.1186/1477-3155-10-19
234. Martinez-Gutierrez F, Olive PL, Banuelos A, et al. Synthesis, characterization, and evaluation of antimicrobial and cytotoxic effect of silver and titanium nanoparticles. Nanomedicine Nanotechnol Biol Med. 2010;6(5):681–688. doi:10.1016/j.nano.2010.02.001
235. Varghese MV, Dhumal RS, Patil SS, Paradkar AR, Khanna PK. Synthesis and in-vitro antimycobacterial studies of cysteine capped silver nano-particles. Synth React Inorg Met-Org Nano-Met Chem. 2009;39(9):554–558. doi:10.1080/15533170903327869
236. Uraskulova BB, Gyusan AO. The clinical and bacteriological study of the effectiveness of the application of silver nanoparticle for the treatment of tuberculosis. Vestn Otorinolaringol. 2017;82(3):54–57. doi:10.17116/otorino201782354-57
237. Gmoshinski IV, Shumakova AA, Shipelin VA, Maltsev G, Khotimchenko SA. Influence of orally introduced silver nanoparticles on content of essential and toxic trace elements in organism. Nanotechnologies Russ. 2016;11(9–10):646–652. doi:10.1134/S1995078016050074
238. Zakharov AV, Khokhlov AL, Kibrik BS. Effectiveness of combination of isoniazid and silver nanoparticles in the treatment of experimental tuberculosis. Tuberc Lung Dis. 2017;95(6):51–58. doi:10.21292/2075-1230-2017-95-6-51-58
239. Zakharov AV, Khokhlov A. The results of experimental studies of the use of silver nanoparticles in tuberculosis drug-resistant pathogen. Med News North Cauc. 2019;14:1. doi:10/ggdjnk
240. Pugazhendhi A, Edison TNJI, Karuppusamy I, Kathirvel B. Inorganic nanoparticles: a potential cancer therapy for human welfare. Int J Pharm. 2018;539(1):104–111. doi:10.1016/j.ijpharm.2018.01.034
241. Srinivasan M, Venkatesan M, Arumugam V, et al. Green synthesis and characterization of titanium dioxide nanoparticles (TiO2 NPs) using Sesbania grandiflora and evaluation of toxicity in zebrafish embryos. Process Biochem. 2019;80:197–202. doi:10.1016/j.procbio.2019.02.010
242. Vazquez-Muñoz R, Borrego B, Juárez-moreno K, et al. Toxicity of silver nanoparticles in biological systems: does the complexity of biological systems matter? Toxicol Lett. 2017;276:11–20. doi:10.1016/j.toxlet.2017.05.007
243. Tarannum N, Divya K, Gautam Y. Facile green synthesis and applications of silver nanoparticles: a state-of-the-art review. RSC Adv. 2019;9(60):34926–34948. doi:10.1039/C9RA04164H
244. Dey Bhowmik A, Bandyopadhyay A, Chattopadhyay A. Cytotoxic and mutagenic effects of green silver nanoparticles in cancer and normal cells: a brief review. The Nucleus. 2019;62(3):277–285. doi:10.1007/s13237-019-00293-0
245. Saravanan M, Arokiyaraj S, Lakshmi T, Pugazhendhi A. Synthesis of silver nanoparticles from Phenerochaete chrysosporium (MTCC-787) and their antibacterial activity against human pathogenic bacteria. Microb Pathog. 2018;117:68–72. doi:10.1016/j.micpath.2018.02.008
246. Yuan D, He H, Wu Y, Fan J, Cao Y. Physiologically based pharmacokinetic modeling of nanoparticles. J Pharm Sci. 2019;108(1):58–72. doi:10.1016/j.xphs.2018.10.037
247. Ehlers S, Schaible UE. The granuloma in tuberculosis: dynamics of a host–pathogen collusion. Front Immunol. 2013;3. doi:10.3389/fimmu.2012.00411.
248. Sarkar S, Leo BF, Carranza C, et al. Modulation of human macrophage responses to mycobacterium tuberculosis by silver nanoparticles of different size and surface modification. PLoS One. 2015;10(11):e0143077. doi:10.1371/journal.pone.0143077
249. De Jong WH, Van Der Ven LTM, Sleijffers A, et al. Systemic and immunotoxicity of silver nanoparticles in an intravenous 28 days repeated dose toxicity study in rats. Biomaterials. 2013;34(33):8333–8343. doi:10.1016/j.biomaterials.2013.06.048
250. Müller L, Steiner SK, Rodriguez-lorenzo L, Petri-fink A, Rothen-rutishauser B, Latzin P. Exposure to silver nanoparticles affects viability and function of natural killer cells, mostly via the release of ions. Cell Biol Toxicol. 2018;34(3):167–176. doi:10.1007/s10565-017-9403-z
251. Alsaleh NB, Minarchick VC, Mendoza RP, Sharma B, Podila R, Brown JM. Silver nanoparticle immunomodulatory potential in absence of direct cytotoxicity in RAW 264.7 macrophages and MPRO 2.1 neutrophils. J Immunotoxicol. 2019;16(1):63–73. doi:10.1080/1547691X.2019.1588928
252. Yen H, Hsu S, Tsai C. Cytotoxicity and immunological response of gold and silver nanoparticles of different sizes. Small. 2009;5(13):1553–1561. doi:10.1002/smll.200900126
253. Li T, Albee B, Alemayehu M, et al. Comparative toxicity study of Ag, Au, and Ag–Au bimetallic nanoparticles on Daphnia magna. Anal Bioanal Chem. 2010;398(2):689–700. doi:10.1007/s00216-010-3915-1
254. Suganthy N, Sri Ramkumar V, Pugazhendhi A, Benelli G, Archunan G. Biogenic synthesis of gold nanoparticles from Terminalia arjuna bark extract: assessment of safety aspects and neuroprotective potential via antioxidant, anticholinesterase, and antiamyloidogenic effects. Environ Sci Pollut Res. 2018;25(11):10418–10433. doi:10.1007/s11356-017-9789-4
255. Larimer C, Islam MS, Ojha A, Nettleship I. Mutation of environmental mycobacteria to resist silver nanoparticles also confers resistance to a common antibiotic. BioMetals. 2014;27(4):695–702. doi:10.1007/s10534-014-9761-4
256. Panáček A, Kvítek L, Smékalová M, et al. Bacterial resistance to silver nanoparticles and how to overcome it. Nat Nanotechnol. 2018;13(1):65–71. doi:10.1038/s41565-017-0013-y
257. Muller M. Bacterial silver resistance gained by cooperative interspecies redox behavior. Antimicrob Agents Chemother. 2018;62:8. doi:10.1128/AAC.00672-18
258. Silver S. Bacterial silver resistance: molecular biology and uses and misuses of silver compounds. FEMS Microbiol Rev. 2003;27(2–3):341–353. doi:10.1016/S0168-6445(03)00047-0
259. Mijnendonckx K, Ali MM, Provoost A, et al. Spontaneous mutation in the AgrRS two-component regulatory system of Cupriavidus metallidurans results in enhanced silver resistance. Metallomics. 2019;11(11):1912–1924. doi:10.1039/C9MT00123A
260. McDonnell G, Russell AD. Antiseptics and disinfectants: activity, action, and resistance. Clin Microbiol Rev. 1999;12(1):147–179. doi:10.1128/CMR.12.1.147
261. Percival SL, Bowler PG, Russell D. Bacterial resistance to silver in wound care. J Hosp Infect. 2005;60(1):1–7. doi:10.1016/j.jhin.2004.11.014
262. Woods EJ, Cochrane CA, Percival SL. Prevalence of silver resistance genes in bacteria isolated from human and horse wounds. Vet Microbiol. 2009;138(3–4):325–329. doi:10.1016/j.vetmic.2009.03.023
263. Loh JV, Percival SL, Woods EJ, Williams NJ, Cochrane CA. Silver resistance in MRSA isolated from wound and nasal sources in humans and animals. Int Wound J. 2009;6(1):32–38. doi:10.1111/j.1742-481X.2008.00563.x
264. Finley PJ, Norton R, Austin C, Mitchell A, Zank S, Durham P. Unprecedented silver resistance in clinically isolated enterobacteriaceae: major implications for burn and wound management. Antimicrob Agents Chemother. 2015;59(8):4734–4741. doi:10.1128/AAC.00026-15
265. Percival SL, Woods E, Nutekpor M, Bowler P, Radford A, Cochrane C. Prevalence of silver resistance in bacteria isolated from diabetic foot ulcers and efficacy of silver-containing wound dressings. Ostomy Wound Manage. 2008;54(3):30–40.
266. Davis IJ, Richards H, Mullany P. Isolation of silver- and antibiotic-resistant Enterobacter cloacae from teeth. Oral Microbiol Immunol. 2005;20(3):191–194. doi:10.1111/j.1399-302X.2005.00218.x
267. Summers AO, Wireman J, Vimy MJ, et al. Mercury released from dental “silver” fillings provokes an increase in mercury- and antibiotic-resistant bacteria in oral and intestinal floras of primates. Antimicrob Agents Chemother. 1993;37(4):825–834. doi:10.1128/AAC.37.4.825
268. Haefeli C, Franklin C, Hardy K. Plasmid-determined silver resistance in Pseudomonas stutzeri isolated from a silver mine. J Bacteriol. 1984;158(1):389–392. doi:10.1128/JB.158.1.389-392.1984
269. Hanczvikkel A, Víg A, Tóth Á. Survival capability of healthcare-associated, multidrug-resistant bacteria on untreated and on antimicrobial textiles. J Ind Text. 2019;48(7):1113–1135. doi:10.1177/1528083718754901
270. Chapman JS. Disinfectant resistance mechanisms, cross-resistance, and co-resistance. Int Biodeterior Biodegrad. 2003;51(4):271–276. doi:10.1016/S0964-8305(03)00044-1
271. Rodgers MR, Blackstone BJ, Reyes AL, Covert TC. Colonisation of point of use water filters by silver resistant non-tuberculous mycobacteria. J Clin Pathol. 1999;52(8):629. doi:10.1136/jcp.52.8.629a
272. Li XZ, Nikaido H, Williams KE. Silver-resistant mutants of Escherichia coli display active efflux of Ag+ and are deficient in porins. J Bacteriol. 1997;179(19):6127–6132. doi:10.1128/JB.179.19.6127-6132.1997
273. Gupta A, Matsui K, Lo J-F, Silver S. Molecular basis for resistance to silver cations in Salmonella. Nat Med. 1999;5(2):183–188. doi:10.1038/5545
274. Li W, Zhang H, Assaraf YG, et al. Overcoming ABC transporter-mediated multidrug resistance: molecular mechanisms and novel therapeutic drug strategies. Drug Resist Updat Rev Comment Antimicrob Anticancer Chemother. 2016;27:14–29. doi:10.1016/j.drup.2016.05.001
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.