Back to Journals » Clinical Interventions in Aging » Volume 15
Short-Term Effect of Self-Selected Training Intensity on Ambulatory Blood Pressure in Hypertensive Older Women: A Randomized Controlled Trial
Authors Sócrates J , Browne RAV , Macêdo GAD , Araújo MBF, Paulo-Pereira R , Cabral LLP , Lucena BEB , Farias-Junior LF , Costa EC
Received 2 May 2020
Accepted for publication 4 July 2020
Published 21 August 2020 Volume 2020:15 Pages 1449—1460
DOI https://doi.org/10.2147/CIA.S260134
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Walker
Júlio Sócrates,1 Rodrigo Alberto Vieira Browne,1 Geovani Araújo Dantas Macêdo,2 Maria Beatriz Fonseca Araújo,3 Ronildo Paulo-Pereira,3 Ludmila Lucena Pereira Cabral,1 Bruno Erick Barros Lucena,1 Luiz Fernando Farias-Junior,4 Eduardo Caldas Costa1– 3
1Graduate Program in Health Sciences, Federal University of Rio Grande do Norte, Natal, Brazil; 2Graduate Program in Physical Education, Federal University of Rio Grande do Norte, Natal, Brazil; 3Department of Physical Education, Federal University of Rio Grande do Norte, Natal, Brazil; 4Graduate Program in Psychobiology, Federal University of Rio Grande do Norte, Natal, Brazil
Correspondence: Eduardo Caldas Costa
Department of Physical Education, Federal University of Rio Grande do Norte, BR 101, Lagoa Nova, Natal, RN 59078-970, Brazil
Tel +55 84 3215 3436
Fax +55 84 3215 3451
Email [email protected]
Purpose: To investigate the short-term effect of self-selected training intensity (SSTI) on ambulatory blood pressure (BP) in hypertensive older women.
Participants and Methods: This is a randomized, single-blind, two-arm, parallel-group controlled trial that included 40 medicated hypertensive older women (64.4± 3.6 years; resting systolic 118± 19 and diastolic BP 68± 9 mmHg). SSTI intervention was performed three times per week, 30– 50 minutes per session (n=20). The control group participated in health education meetings once per week (n=20). Ambulatory BP (primary outcome) and six-minute walking test performance (secondary outcome) were assessed at baseline and following 8 weeks of intervention. Heart rate (HR), rating of perceived exertion (RPE, 6– 20), and affective valence (ie, feeling scale, − 5/+5) were recorded during all SSTI sessions. Intention-to-treat and per-protocol analyses were used for data analyses.
Results: Fifteen participants from the SSTI group and 17 from the control group completed the study. No differences in ambulatory BP (24-h, awake, and asleep) were observed between SSTI and control groups (intention-to-treat and per-protocol analyses; p> 0.05). The SSTI group showed a greater six-minute walking test performance than the control group in the intention-to-treat and per-protocol analyses (p< 0.05). The participants exercised at 52± 10% of HR reserve reported an RPE of 11± 1 and an affective valence of 3.4± 1.1 over the 8-week period.
Conclusion: SSTI is a feasible approach to induce a more active lifestyle and increase health-related fitness in hypertensive older women, although it does not improve BP control over a short-term period.
Keywords: exercise, hypertension, aging
Introduction
Hypertension is a major risk factor for premature mortality worldwide, being mainly associated with cardiovascular diseases.1 In the last four decades, the number of adults with hypertension increased from 594 million in 1975 to 1.1 billion in 2015. The increase of hypertension occurred mainly in low-income and middle-income countries.2 In addition, the hypertension care cascade (ie, hypertension awareness, treatment, and control) was worse in low- and middle-income than in high-income countries.3 It should be noted that the prevalence of hypertension is higher in older adults,1 especially in older women.4,5 Therefore, improvement in the hypertension care cascade is needed, especially at the blood pressure (BP) control level, in order to attenuate the risk of premature cardiovascular events and mortality.
Physical exercise is a cornerstone of non-pharmacological approaches to reduce BP and improve hypertension control.6,7 A recent network meta-analysis showed that the systolic BP-lowering effect of exercise training appears to be similar to antihypertensive medications among individuals with hypertension.8 Despite the well-known antihypertensive effect of regular exercise training, the SABE Survey (Health, Welfare and Aging Survey), which is a home-focused epidemiological study in Brazil, revealed that 71.4% of hypertensive older adults are inactive; ie, <150 minutes per week of moderate physical activity, and <75 minutes per week of vigorous physical activity.9 Thus, effort should be made to increase the participation and adherence of hypertensive older adults in regular physical exercise.
The traditional exercise prescription recommended by clinical hypertension guidelines have focused on the FITT principle (Frequency, Intensity, Time, and Type),6,7 using heart rate- (HR) and oxygen uptake-based (VO2) approaches for exercise intensity prescription and monitoring. However, these methods are not accessible or easy to understand for most hypertensive older adults. It has been argued that self-selected exercise intensity may be an interesting option to increase participation and adherence to regular physical exercise,10 mainly due to its psychological advantages such as higher perceived autonomy, self-efficacy, affective response, and lower perceived exertion.11–13 Self-selected exercise intensity is characterized when individuals choose their own exercise intensity according to their preferences.10 Older adults typically self-select an exercise intensity close to their ventilatory threshold,10,14 which is within the recommended range proposed for hypertensive patients. For example, the American College of Sports Medicine recommends an exercise intensity between 40% and 59% of VO2 reserve or HR reserve for hypertensive individuals.7
Two previous studies from our group showed that hypertensive older women self-select an exercise intensity between 50% and 60% of HR reserve and report the exercise sessions as “light” and “pleasant”.15,16 In addition, Costa et al16 showed that a single 30-minute self-selected exercise intensity session elicited a reduction in ambulatory systolic (BP) in inactive hypertensive older women. The systolic post-exercise hypotension (PEH) of ~6 mmHg lasted up to a six-hour period and contributed to a reduction in the mean values of systolic BP in the 20-hour and awake periods. These results suggest that the self-selected exercise intensity has the potential to induce a BP-lowering effect following a regular exercise training period.17 However, to the best of our knowledge, there is a scarcity of studies investigating the chronic effect of a self-selected training intensity (SSTI) intervention on BP and fitness-related outcomes in hypertensive older adults. Therefore, this study investigated the short-term chronic effect of a SSTI intervention on ambulatory BP (primary outcome) and six-minute walking test performance (secondary outcome) in inactive hypertensive older women. It was hypothesized that 8 weeks of SSTI would reduce ambulatory BP and improve the six-minute walking test performance in these individuals.
Participants and Methods
Trial Design
This is a randomized, single-blind, two-arm, parallel-group controlled trial designed to investigate the short-term chronic effect of a SSTI intervention on ambulatory BP in hypertensive older women. This trial is reported in accordance with the CONSORT statement guidelines.17 All procedures related to this study were conducted at the Department of Physical Education and at the University Hospital Onofre Lopes from the Federal University of Rio Grande do Norte, Natal, RN, Brazil, between September 2018 and November 2019. All participants were informed about the study procedures and provided written informed consent. The study was carried out in line with the Declaration of Helsinki and was approved by the Institutional Ethics Committee (protocol 2.840.999/2018). The study was also registered in the Brazilian Clinical Trials Registry (http://ensaiosclinicos.gov.br/; ID: RBR-3h5394).
Participants
A total of 40 community-dwelling hypertensive older women were included in this study (Figure 1). All participants were recruited from Natal, RN, Brazil, by advertisements on radio, flyers, and e-flyers in social media (WhatsApp®, Instagram® and Facebook®). The inclusion criteria for the participants were: i) aged 60–75 years; ii) previous diagnosis of hypertension according to the Brazilian guidelines on hypertension;18 iii) currently taking antihypertensive medication(s); iv) body mass index (BMI) <35 kg/m2; v) being inactive; ie, less than 600 MET/min/week of physical activities19 and <7500 steps per day;20 vi) no prior history of major cardiovascular disease or cardiovascular events (eg, acute myocardial infarction, stroke, coronary artery disease, arrhythmias, or peripheral vascular disease); vii) no osteomioarticular injury which limited the ability to perform exercise. The exclusion criteria were: i) having pain or injury which limits their ability to exercise during the study; ii) changes in antihypertensive medication(s) during the study.
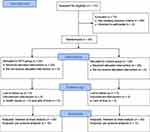 |
Figure 1 Study flowchart. |
Procedures
All participants were screened at their first visit using the Physical Activity Readiness Questionnaire,21 medical history, medication use, and the initial physical activity level assessment using an adapted version of the Minnesota Leisure Time Activities Questionnaire.19 A clinical examination provided body weight (kg), height (m), resting HR (bpm), and resting BP (mmHg) measurements. BMI was calculated as weight (kg) divided by height (m) squared. Resting HR was measured after 10 minutes of rest in a supine position in a calm and controlled room (24–26°C) using an HR monitor (Polar Electro®, Kempele, Finland). Resting BP was measured in a seated position using an oscillometric device (Omron® HEM-780-E, Kyoto, Japan) in triplicate with 1-minute intervals between each measure. The average value of the last two measures was recorded. In the second visit, the participants performed the maximal graded exercise test with a cardiologist in order to exclude symptoms and electrocardiographical alterations suggestive of heart disease. In the third visit, the ambulatory BP of the participants during a 24-hour period was assessed. The participants then returned to the laboratory on the next day to remove the ambulatory BP monitoring device (CardioMAPA, Cardios®, Brazil) and to perform the six-minute walking test. Afterward, participants were fitted with a wrist pedometer (Omron® HJ-321, Kyoto, Japan) to confirm their inactive physical activity level as assessed by the number of steps accumulated per day over a 1-week period. The participants returned to the laboratory 1 week later to remove the wrist pedometer and were randomly allocated to the SSTI or control group.
Physical Activity Level
An adapted version of the Minnesota Leisure Time Activities Questionnaire19 was used to identify inactive participants in the screening visit. Afterward, those identified as inactive (ie, <600 MET/min/week) were selected for the pedometer-measured physical activity level assessment in order to confirm their inactive status. Pedometers (Omron®, HJ-321 Tri-Axis Alvita, USA) were individually adjusted for the participants based on their stride length, weight and height according to the manufacturer’s instructions. A total number of steps per day less than 7500 was used as a criterion to determine participants as inactive.20 The participants returned to the laboratory 1 week after the fourth screening visit to remove the wrist pedometer. Only participants classified as inactive in the Minnesota Leisure Time Activities Questionnaire19 and who showed less than 7500 steps per day in the pedometer-measured physical activity level assessment were included in the study.
Maximal Graded Exercise Test
The participants performed a maximal graded exercise test on a motorized treadmill (Centurion 200, Micromed®, Brazil) to determine their HRmax and to exclude symptoms and electrocardiographical alterations suggestive of heart disease. Participants were continuously monitored with 12-lead electrocardiography by a cardiologist during the maximal graded exercise test and during a five-minute recovery period. An incremental ramp protocol was performed by the participants starting at a speed of 2.5 km/h and 4% grade with continuous increments to reach 5.5 km/h and 14% grade at 10 minutes. The BP was measured every 2 minutes during and immediately following the end of the maximal graded exercise test by the auscultatory method. RPE was recorded every minute using Borg’s RPE scale (6–20).22 The graded exercise test was considered maximal when the participant reached an RPE > 17 and HR within ± 10 bpm of age-predicted HRmax (220-age).23 For participants using a β-blocker, only RPE was used as a criterion of volitional exhaustion. Participants performed the maximal graded exercise test under their regular antihypertensive medication.
Six-Minute Walking Test
The six-minute walking test (6MWT) was conducted according to the procedures established by Rikli and Jones24 and was used as a proxy of cardiorespiratory fitness.25 A previous study showed that the walking distance covered in the 6MWT predicted maximal oxygen uptake (R2 = 0.76) obtained from a maximal cardiopulmonary test, which is a “gold-standard” method to assess cardiorespiratory fitness.26 Therefore, we used the 6MWT as a proxy of cardiorespiratory fitness due to its feasibility and potential to provide valuable information in clinical practice.25
Ambulatory Blood Pressure Monitoring
Participants wore an ambulatory BP device (CardioMAPA, Cardios®, Brazil) on their non-dominant arm according to the Brazilian guidelines for ambulatory BP monitoring27 over a 24-hour period. The device was programmed to measure BP every 15 minutes during their awake and every 30 minutes during their sleeping periods. A minimum of 16 and eight BP measurements during awake and sleeping periods, respectively, had to be successfully recorded to be included in the analysis. Participants came back to the laboratory the following morning after each session so that the BP information could be downloaded to a computer. Data was recorded during the awake and sleeping periods defined according to each participant. The average values for each period were considered for data analysis (ie, 24-hour, awake, and asleep). In addition, BP load was calculated during the 24-hour period. This index is defined as the percentage of readings that exceeds the normal BP values in both awake and asleep periods. Systolic BP ≥ 135 and diastolic BP ≥ 85 mmHg were considered abnormal in the awake period, while systolic BP ≥ 120 and diastolic BP ≥ 70 mmHg were considered abnormal in the asleep period. All procedures related to the ambulatory BP monitoring were explained to the participants at the health screening visit based on the Brazilian guidelines for ambulatory BP monitoring.27
Self-Selected Training Intensity Intervention
The SSTI occurred three times per week over 8 weeks. The duration of the SSTI sessions was increased from week one (30 minutes) to week five (50 minutes); ie, 5 minutes per week. The SSTI sessions had 50 minutes duration from week five to week eight. All SSTI sessions were performed in the afternoon between 4:00 and 6:00 p.m. on a standard outdoor 400 m track. Participants’ BP was measured immediately prior to each SSTI session. They were allowed to exercise if their BP was lower than 160/105 mmHg.23 Participants were instructed to regulate their own pace before the SSTI sessions using the following statement and following the same procedures of previous studies15,16 from our group: We would like that you exercise for [30, 35, 40, 45, or 50 minutes] at a pace of your own choosing. You can freely change the pace at any time during the exercise. Participants performed a stretching routing and then warmed up for 5 minutes to adjust their pace before starting the SSTI session. Participants then cooled down afterwards for 5 minutes by walking at a slow pace. All participants were fitted with a HR monitor (Polar Electro®, Kempele, Finland) to monitor HR responses during SSTI sessions. In addition, whole-body perceived exertion using Borg’s RPE scale (6–20)22 and affective valence using the Feeling Scale (−5/+5)28 were reported by participants every 10 minutes during the SSTI sessions over the eight-week period of intervention. All participants were individually instructed about the use of the Borg’s RPE scale and Feeling Scale prior to beginning the SSTI intervention. RPE was defined as subjective intensity of effort, strain, and/or fatigue felt during exercise.22 Low and high perceptual anchors for Borg’s RPE scale were established during the maximal graded exercise test. A rating of 6 (low anchor, “very, very light”) was assigned to the lowest exercise intensity, while a rating of 20 (high anchor, “very, very hard”) was assigned to the highest exercise intensity. The Feeling Scale is an 11-point bipolar scale ranging from +5 to −5. This scale presents verbal anchors: −5= very bad; −3= bad; −1= fairly bad; 0= neutral; +1 fairly good; +3= good; and +5= very good. The participants received standard instructions explaining the Feeling Scale according to Hardy and Rejeski.28 All SSTI sessions were performed individually in order to avoid interference on the self-selected pace, RPE and affective valence.
Control Group
The control group participated in group-based health education meetings once per week over the 8 weeks of intervention at the Department of Physical Education, Federal University of Rio Grande do Norte. Every education meeting had 60 minutes of duration and was conducted by two researchers (LLPC, MBFA). The topics of the education meetings covered basic knowledge about self-care in aging, healthy and unhealthy behaviors (eg hydration, sleep, alcohol intake, sedentary time), and prevention and management of non-communicable diseases (eg, hypertension, type 2 diabetes, signals and symptoms of cardiovascular events). Moreover, group dynamics, ludic activities and board games were also conducted during the meetings. The comparator of the SSTI intervention was health education because this is a usual care approach in public primary care settings in Brazil for hypertensive adults, in addition to the pharmacological treatment.
Sample Size and Randomization
Previous meta-analyses showed that a decrease of 5 mmHg in systolic BP provides a clinically meaningful risk reduction in cardiovascular diseases and mortality.29,30 A meta-analysis conducted by Sosner et al31 showed that aerobic exercise training reduces 24-hour ambulatory BP by 4 mmHg; however, the magnitude of the BP-lowering effect is higher in individuals with pre- to established hypertension (ie, resting BP ≥ 130/85 mmHg). Based on these previous studies,29–31 a priori sample size calculation was conducted to provide sufficient power (1-β of 80% and alpha of 5%) for a reduction of 5 ± 5 mmHg in 24-hour ambulatory BP (primary outcome) in the SSTI group compared to the control group. A total of 34 participants were needed to fulfill the study power (G*Power software, version 3.1.9.2). Thus, considering a dropout rate of 20%, 40 participants were recruited for the study. After all baseline assessments, a computer-based simple randomization (www.graphpad.com) was carried out to determine the allocation of the participants to the SSTI (n = 20) or the control group (n = 20). This procedure was conducted by an independent researcher (ECC) not involved in the recruitment, allocation, assessment (baseline and following 8 weeks), and SSTI intervention or health education meetings. Afterward, the 40 sequential codes from the randomization list were included in 40 opaque and closed envelopes. Thus, allocation concealment was implemented. In addition, blinded researchers (RAVB, LFFJ, GADM) conducted all procedures related to the assessments at baseline and following 8 weeks of intervention.
Statistical Analysis
Data normality was tested using the Shapiro–Wilk test, skewness and kurtosis (score-z: −1.96 to +1.96). Results are expressed as mean ± SD and absolute and relative frequency. An independent t-test was used to compare the mean values of resting and ambulatory BP, 6MWT, pedometer-measured physical activity level, and anthropometric measures at baseline. The Friedman test was used to compare HR reserve, RPE and affective valence during the SSTI sessions over the 8 weeks of intervention. Intention-to-treat and per-protocol analyses were conducted for both primary and secondary outcomes. A generalized estimating equation (GEE) model was used to assess the group (2 factors: SSTI vs control) by time (2 factors: baseline vs eight weeks) interaction on primary (ambulatory BP) and secondary (6MWT performance) outcomes. All GEE models used the gamma distribution with an identity link based on the goodness of fit, except for the BP load, which used a tweedie distribution with identity link. The goodness of fit of the models was verified by the normal Q–Q plot and Akaike information criterion. A two-tailed p < 0.05 was considered statistically significant for all analyses. Data analysis was performed using IBM SPSS Statistics for Win/v.25.0 (IBM Corp., Armonk, NY). Cohen’s ds for comparisons between-groups and Cohen’s dz for comparisons within-group were used to calculate the effect size (ES).32 The following criteria for the interpretation of the ES were adopted: <0.50, small; 0.50 to 0.79, medium; and ≥0.80, large.33
Results
Eight participants dropped out of the study (ie, five from the SSTI group and three from the control group). Six participants reported a lack of time to continue the study, one participant had a renal calculi crisis not related to the study, and one was excluded because they reported having knee pain in the second SSTI session. No more adverse events occurred over the 8 weeks of the SSTI intervention. The attendance rate of the SSTI sessions was 91.7%. Table 1 shows the characteristics of the participants included in the study. The participants had 9.4 ± 5.9 years of hypertension diagnosis. Regarding the antihypertensive medication use, 45% (n = 18) used only one medication, 47% (n = 19) two medications, and 8% (n = 3) three or more medications. A total of 10 Caucasian, 8 Pardo, and 2 Black individuals were included in the SSTI group and 9 Caucasian, 7 Pardo, and 4 Black individuals were included in the control group. No differences were observed for the pedometer-measured physical activity level between the groups at baseline (4213 ± 1665 vs 4603 ± 1520 steps/day; p = 0.808; Cohen’s ds = 0.24).
 |
Table 1 Characteristics of the Self-Selected Training Intensity and Control Groups |
Table 2 shows the HR, RPE, and affective valence during the SSTI sessions over the 8 weeks of intervention. No differences were found over the 8 weeks of intervention in the HR, RPE and affective valence (p > 0.05). Participants exercised at an average of 52 ± 10% of HR reserve, reported an RPE of 11 ± 1 and an affective valence of 3.4 ± 1.1.
 |
Table 2 Heart Rate, Rating of Perceived Exertion and Affective Valence During the Self-Selected Training Intensity Sessions Over the Eight Weeks of Intervention |
Table 3 shows the values of ambulatory BP at baseline and following the 8 weeks of intervention in the SSTI and control groups. Both intention-to-treat (n = 40) and per-protocol (n = 32) analyses revealed no significant differences in systolic and diastolic 24-hour, awake, and asleep BP between the SSTI and control groups (p > 0.05). There was no difference in systolic 24-hour BP load in the SSTI compared to the control group, both in the intention-to-treat analysis (p = 0.079) and per-protocol analysis (p = 0.076).
 |
Table 3 Ambulatory BP at Baseline and Following Eight Weeks of Intervention in the Self-Selected Training Intensity and Control Groups |
Figure 2 shows the individual 24-hour BP pre-post changes of participants with uncontrolled (ie, systolic BP ≥ 130 mmHg; panel A) and controlled (ie, systolic BP < 130 mmHg; panel B) ambulatory BP levels at baseline based on per-protocol analysis. All participants from the SSTI group with uncontrolled BP levels showed a reduction in the 24-hour systolic BP (from ~2 to ~6 mmHg). Considering the five participants from the control group with uncontrolled BP levels, one did not change the 24-hour systolic BP, two showed an increase (~7 and ~14 mmHg), and two showed a decrease (~9 and ~3 mmHg). Regarding the participants from the SSTI group with controlled BP levels at baseline, 63.7% (n = 7) did not change or reduced (from ~6 to ~9 mmHg) the 24-hour systolic BP, and 36.3% (n = 4) showed an increase. For the control group, 41.7% (n = 5) did not change or reduced (from ~3 to ~8 mmHg) the 24-hour systolic BP and 58.3% (n = 7) showed an increase.
Table 4 shows the values of six-minute walking test performance at baseline and following the 8 weeks of intervention in the SSTI and control groups. Intention-to-treat and per-protocol analysis revealed a significantly greater performance in the SSTI compared to the control group (p < 0.05).
 |
Table 4 Six-Minute Walking Test Performance at Baseline and Following Eight Weeks of Intervention in the Self-Selected Training Intensity and Control Groups |
Discussion
The main findings of this study were: i) no changes in ambulatory BP occurred following 8 weeks of SSTI; ii) participants from the SSTI group improved their 6MWT performance; iii) the participants exercised at a moderate intensity (40–59% of HR reserve) over the eight-week intervention period, reported the SSTI sessions as “light” (RPE ~ 11) and “pleasant” (FS ~ 3.5), and showed an attendance rate >90%. Thus, our hypothesis that 8 weeks of SSTI would elicit a reduction on ambulatory BP was not confirmed. To the best of our knowledge, this is the first randomized controlled trial that has investigated the short-term chronic effect of a SSTI intervention on ambulatory BP in hypertensive older women.
Our research group has previously shown that a single 30-minute SSTI session reduced ~6 mmHg of ambulatory systolic BP in the first 6 hours post-exercise, which elicited a decrease in mean values of ambulatory BP in 20-hour and awake periods.16 Therefore, a SSTI session elicits an ambulatory PEH, which plays a role in attenuating the overload imposed on the cardiovascular system during daily activities in hypertensive individuals, and as a consequence can contribute to reduce the risk of acute cardiovascular events. The magnitude and duration of PEH elicited by a single exercise session provides insight into the future chronic BP-lowering effect following a regular exercise training period. It is argued that repeated PEH over an exercise training period contributes to a chronic reduction in BP.17 This can occur as a simple superimposition of a temporal summation of this acute effect or as a combination with morphological and functional adaptation over time,34 for example, in the hemodynamic determinants of the BP (ie, cardiac output and peripheral vascular resistance) and its regulatory mechanisms.
However, similarly to what occurs with PEH,17 it seems that factors such as the participants’ characteristics may influence the occurrence (or not) of a chronic BP-lowering effect and benefits on the mechanisms related to the hemodynamic determinants of the BP control. Considering both intention-to-treat and per-protocol analyses, no significant changes in ambulatory BP (ie, 24-hour, awake and asleep periods) occurred following 8 weeks of SSTI. Regarding the baseline BP levels of the participants, 70% had controlled 24-hour ambulatory BP levels (~115/66 mmHg) and 30% had uncontrolled BP levels (~143/78 mmHg). Previous meta-analyses clearly showed that the BP level pre-intervention influences the magnitude of BP-lowering effect, both for resting35 and ambulatory BP.31 Cornelissen et al35 and Sosner et al31 showed that the magnitude of a BP-lowering effect following an aerobic exercise training intervention is greater in individuals with higher BP levels compared to those with optimal BP levels. A recent network meta-analysis involving more than 10,000 participants in exercise training interventions confirmed these previous findings.8 Considering the individual responses of those who concluded the 8 weeks of SSTI, all participants with uncontrolled BP level in our study showed a reduction in the 24-hour systolic BP (from ~2 to ~6 mmHg). In addition, 63.7% (n = 7) of those with controlled BP level did not change or reduced their 24-hour systolic BP. This scenario was different for the control group, considering both participants with uncontrolled (40% increased BP; n = 2) and controlled (58.3% increased BP; n = 7) BP level. Our findings seem to support previous studies that have shown a more consistent BP-lowering effect following an exercise training intervention in individuals with higher BP levels.31,35 Thus, we do not rule out the possibility that a short-term SSTI-based intervention elicits better BP control in hypertensive older women with uncontrolled BP levels. Future randomized controlled trials, including only hypertensive older individuals with uncontrolled BP levels, should be conducted in order to confirm (or not) this assumption.
A reduction of 5 mmHg in BP unequivocally decreases the risk of cardiovascular diseases and mortality.29,30 However, it should be noted that a reduction in BP of a lower magnitude than 5 mmHg may be clinically meaningful. For example, a 2 mmHg BP reduction is sufficient to reduce the mortality risk by stroke in 6%, coronary heart disease in 4%, and all-cause mortality in 3%.36 As all participants from the SSTI group with uncontrolled BP had a reduction in 24-hour systolic BP of at least 2 mmHg, it seems reasonable to assume that these individuals improved their cardiovascular disease risk profile following the 8 weeks of the exercise training intervention. In addition, the participants accumulated 150 minutes per week of moderate-vigorous aerobic exercise from week five to eight of the SSTI, meeting the threshold recommended by the American College of Sports Medicine7 and most hypertension guidelines.37,38 A longitudinal study conducted by Brown et al39 involving 10,665 middle-aged and older adults with a mean follow-up of 8.6 years revealed that those who were inactive with controlled BP levels among medicated hypertensive individuals had a 42% higher risk of all-cause mortality than their active peers. Inactive individuals with uncontrolled BP levels had a 55% higher risk of all-cause mortality than the active individuals with controlled BP levels. Active individuals with uncontrolled BP levels showed a trend (p = 0.08) of 17% higher risk of all-cause mortality compared to their peers with controlled BP levels. These data reinforce the protective role of regular exercise against all-cause mortality in medicated hypertensive middle-aged and older individuals, independent of their BP status (ie, controlled or uncontrolled). The SSTI, which is a simple, low-cost, and feasible approach for “real world” settings, may be a viable way to induce an active lifestyle in hypertensive older individuals and potentially reduce their risk of mortality.
It was observed an increase of 6MWT performance (the secondary outcome of the present study) in the SSTI group compared to control group following 8 weeks of intervention. This result suggests an improvement in cardiorespiratory fitness and, as a consequence, a potential cardioprotective benefit in the participants from the SSTI group. Previous studies have shown a strong correlation between the distance covered in the 6MWT and the maximal oxygen uptake obtained from a maximal cardiopulmonary exercise, which is a “gold standard” method to determine cardiorespiratory fitness in different populations, including older adults with and without known cardiovascular disease.40–42 In the present study, the participants exercised within the moderate-intensity domain (ie, 40–59% of HR reserve) over the 8 weeks of SSTI. Moderate-intensity aerobic exercise is recommended to improve cardiorespiratory fitness in older adults,43 which seems to explain the improvements in the 6MWT performance observed in the SSTI group. Future studies should determine the effect of SSTI on cardiorespiratory fitness using a cardiopulmonary exercise test, given the clinical importance of this outcome for the cardiovascular health; ie, an increase of 1 metabolic equivalent (MET) in cardiorespiratory fitness is associated with a 10% to 20% decrease in mortality rates.25
The participants showed a consistent HR (52 ± 10% of HR reserve), RPE (11 ± 1), and affective valence (3.4 ± 1.1) during the exercise sessions over the 8 weeks of SSTI. Overall, these responses indicate that the inactive hypertensive older women exercised within a moderate intensity and reported the exercise sessions as “light” and “pleasant”. In addition, they showed a high attendance rate to the exercise sessions (91.7%). These findings confirm the results of two previous studies from our research group which showed similar HR, RPE, and affective valence patterns during a single16 and two SSTI sessions15 separated by a one-week interval. The consistent positive affective valence over the SSTI sessions may be partially attributed to the magnitude of the metabolic stress imposed on the organism of the participants. Considering the HR and RPE responses, the participants probably self-selected an exercise intensity close but below their ventilatory or lactate threshold or the onset of blood lactate accumulation, which is consistent with the dual-mode theory. This theory postulates that when the individuals exercise at a moderate intensity (below the ventilatory threshold) they report a positive affective valence.13 In addition, the outdoor environment also contributes to a more positive affective valence due to a more dissociative attentional focus during exercise.44 Taken together, SSTI outdoors seems to be a feasible approach to encourage a more active lifestyle in hypertensive older women. From a practical perspective, SSTI should be considered in primary care and clinical settings both for physical activity counseling interventions and structured exercise programs delivered to hypertensive older women.
This study has limitations that should be mentioned. First, most participants from our sample had well-controlled ambulatory BP levels, which limits understanding the effect of SSTI in hypertensive older women with uncontrolled BP levels. Second, although we instructed the participants to maintain the physical activity pattern in their daily routine, we did not objectively assess this aspect. Therefore, it is not possible to determine whether the SSTI group or control group changed their physical activity level over the 8 weeks of intervention. Third, the participants were not familiarized with the ambulatory BP device, which can have elicited an alert reaction to wearing this device for the first time.45
Conclusion
Eight weeks of SSTI does not reduce 24-hour ambulatory BP in medicated hypertensive older women. However, SSTI increases the 6MWT performance in these individuals, which suggests improvement in cardiorespiratory fitness and a potential cardioprotective effect. Hypertensive older women self-select an exercise intensity within the recommendations of the clinical hypertension guidelines (ie, moderate-vigorous intensity), report the exercise sessions as “light” and “pleasant” and show a high attendance rate to the SSTI intervention. Future studies should consider investigating the long-term effect of SSTI on ambulatory BP and other cardiovascular- and fitness-related outcomes in hypertensive older patients.
Data Sharing Statement
The authors share the original database regarding outcomes of this study in the public repository for undetermined time, in order to independent verification of research results. The database can be found in: http://dx.doi.org/10.17632/ym2wtvmd7g.1.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Whelton PK, Carey RM, Aronow WS, et al. ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: executive summary: a report of the American College of Cardiology/American Heart Association Task F. Hypertens (Dallas, Tex 1979). 2017;2018(71):1269–1324.
2. Zhou B, Bentham J, Di Cesare M, et al. Worldwide trends in blood pressure from 1975 to 2015: a pooled analysis of 1479 population-based measurement studies with 19·1 million participants. Lancet. 2017;389:37–55. doi:10.1016/S0140-6736(16)31919-5
3. Mills KT, Bundy JD, Kelly TN, et al. Global disparities of hypertension prevalence and control: a systematic analysis of population-based studies from 90 countries. Circulation. 2016;134:441–450. doi:10.1161/CIRCULATIONAHA.115.018912
4. Benjamin EJ, Virani SS, Callaway CW, et al. Heart disease and stroke statistics—2018 update: a report from the American Heart Association. Circulation. 2018;137. doi:10.1161/CIR.0000000000000558
5. Reichert FF, Azevedo MR, Breier A, et al. Physical activity and prevalence of hypertension in a population-based sample of Brazilian adults and elderly. Prev Med (Baltim). 2009;49:200–204. doi:10.1016/j.ypmed.2009.06.009
6. Weber MA, Schiffrin EL, White WB, et al. Clinical practice guidelines for the management of hypertension in the community: a statement by the American Society of Hypertension and the International Society of Hypertension. J Clin Hypertens (Greenwich). 2014;16:14–26. doi:10.1111/jch.12237
7. Pescatello LS, MacDonald HV, Lamberti L, et al. Exercise for hypertension: a prescription update integrating existing recommendations with emerging research. Curr Hypertens Rep. 2015;17:87. doi:10.1007/s11906-015-0600-y
8. Naci H, Salcher-Konrad M, Dias S, et al. How does exercise treatment compare with antihypertensive medications? A network meta-analysis of 391 randomised controlled trials assessing exercise and medication effects on systolic blood pressure. Br J Sports Med. 2019;53:859–869. doi:10.1136/bjsports-2018-099921
9. Bueno DR, de Fátima Nunes Marucci M, Gobbo LAet al. Expenditures of medicine use in hypertensive/diabetic elderly and physical activity and engagement in walking: cross sectional analysis of SABE survey. BMC Geriatr. 2017;17:70. doi:10.1186/s12877-017-0437-0
10. Ekkekakis P. Let them roam free? Physiological and psychological evidence for the potential of self-selected exercise intensity in public health. Sport Med. 2009;39:857–888. doi:10.2165/11315210-000000000-00000
11. Lind E, Joens-Matre RR, Ekkekakis P. What intensity of physical activity do previously sedentary middle-aged women select? Evidence of a coherent pattern from physiological, perceptual, and affective markers. Prev Med (Baltim). 2005;40:407–419. doi:10.1016/j.ypmed.2004.07.006
12. Ekkekakis P, Lind E. Exercise does not feel the same when you are overweight: the impact of self-selected and imposed intensity on affect and exertion. Int J Obes (Lond). 2006;30:652–660. doi:10.1038/sj.ijo.0803052
13. Ekkekakis P, Parfitt G, Petruzzello SJ. The pleasure and displeasure people feel when they exercise at different intensities decennial update and progress towards a tripartite rationale for exercise intensity prescription. Sport Med. 2011;41:641–671. doi:10.2165/11590680-000000000-00000
14. Smith AE, Eston R, Tempest GD, et al. Patterning of physiological and affective responses in older active adults during a maximal graded exercise test and self-selected exercise. Eur J Appl Physiol. 2015;115:1855–1866.
15. Sócrates J, Macêdo GAD, Costa IBB, et al. Self-selected exercise intensity for inactive hypertensive older women: a pilot study. Rev Bras Atividade Física Saúde. 2019;24:1–9. doi:10.12820/Rbafs.24e0084
16. Costa IBB, Schwade D, Macêdo GAD, et al. Acute antihypertensive effect of self-selected exercise intensity in older women with hypertension: a crossover trial. Clin Interv Aging. 2019;14:1–12. doi:10.2147/CIA.S207254
17. Brito LC, Fecchio RY, Peçanha T, et al. Postexercise hypotension as a clinical tool: a ‘single brick’ in the wall. J Am Soc Hypertens. 2018;12:e59–e64. doi:10.1016/j.jash.2018.10.006
18. Malachias MVB, Gomes MAM, Nobre F, et al. 7th Brazilian Guideline of Arterial Hypertension: chapter 2 - diagnosis and classification. Arq Bras Cardiol. 2016;107:7–13. doi:10.5935/abc.20160152
19. Pereira DS, Dias RC; Lustosa LPT and cultural adaptation of the MLTAQ in community-dwelling older people, et al.. Translation and cultural adaptation of the Minnesota Leisure Time Activities Questionnaire in community-dwelling older people. Geriatr Gerontol. 2011;5:57–65.
20. Tudor-Locke C, Hatano Y, Pangrazi RP, et al. Revisiting ‘how many steps are enough?’. Med Sci Sports Exerc. 2008;40:S537–43. doi:10.1249/MSS.0b013e31817c7133
21. Thomas S, Reading J, Shephard RJ. Revision of the Physical Activity Readiness Questionnaire (PAR-Q).. Can J Sport Sci. 1992;17(4):338–345.
22. Borg G. The Borg RPE Scale. In: Borg G. Borg’s Perceived Exertion and Pain Scales. Champaign, IL: Human Kinetics; 1998:29–38.
23. Riebe D, Ehrman JK, Liguori G, et al. ACSM’s Guidelines for Exercise Testing and Prescription.
24. Rikli RE, Jones CJ. Development and validation of a functional fitness test for community-residing older adults. J Aging Phys Act. 1999;7:129–161. doi:10.1123/japa.7.2.129
25. Ross R, Blair SN, Arena R, et al. Importance of assessing cardiorespiratory fitness in clinical practice: a case for fitness as a clinical vital sign: a scientific statement from the American Heart Association. Circulation. 2016;134:e653–e699. doi:10.1161/CIR.0000000000000461
26. Sperandio EF, Arantes RL, Matheus AC, et al. Intensity and physiological responses to the 6-minute walk test in middle-aged and older adults: a comparison with cardiopulmonary exercise testing. Brazilian J Med Biol Res. 2015;48:349–353. doi:10.1590/1414-431x20144235
27. Nobre F, Mion Júnior D, Gomes M, et al. 6th guideline for ambulatory blood pressure monitoring and 4th guideline for household blood pressure monitoring. Arq Bras Cardiol. 2018;110:1–29. doi:10.5935/abc.20180012
28. Hardy CJ, Rejeski WJ. Not what, but how one feels: the measurement of affect during exercise. J Sport Exerc Psychol. 1989;11:304–317. doi:10.1123/jsep.11.3.304
29. Bundy JD, Li C, Stuchlik P, et al. Systolic blood pressure reduction and risk of cardiovascular disease and mortality: a systematic review and network meta-analysis. JAMA Cardiol. 2017;2:775–781. doi:10.1001/jamacardio.2017.1421
30. Ettehad D, Emdin CA, Kiran A, et al. Blood pressure lowering for prevention of cardiovascular disease and death: a systematic review and meta-analysis. Lancet (London, England). 2016;387:957–967. doi:10.1016/S0140-6736(15)01225-8
31. Sosner P, Guiraud T, Gremeaux V, et al. The ambulatory hypotensive effect of aerobic training: a reappraisal through a meta-analysis of selected moderators. Scand J Med Sci Sports. 2017;27:327–341. doi:10.1111/sms.12661
32. Lakens D. Calculating and reporting effect sizes to facilitate cumulative science: a practical primer for t-tests and ANOVAs. Front Psychol. 2013;4. doi:10.3389/fpsyg.2013.00863.
33. Cohen J. Statistical Power Analysis for the Behavioral Sciences.
34. Lucas da Nobrega AC. The subacute effects of exercise: concept, characteristics, and clinical implications. Exerc Sport Sci Rev. 2005;33:84–87. doi:10.1097/00003677-200504000-00005
35. Cornelissen VA, Smart NA. Exercise training for blood pressure: a systematic review and meta-analysis. J Am Heart Assoc. 2013;2:e004473–e004473. doi:10.1161/JAHA.112.004473
36. Whelton PK, He J, Appel LJ, et al. Primary prevention of hypertension: clinical and public health advisory from The National High Blood Pressure Education Program. JAMA. 2002;288:1882–1888. doi:10.1001/jama.288.15.1882
37. Eckel RH, Jakicic JM, Ard JD, et al. AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2013;2014(129):S76–99.
38. Mancia G, Fagard R, Narkiewicz K, et al. ESH/ESC Guidelines for the management of arterial hypertension: the Task Force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens. 2013;2013(31):1281–1357. doi:10.1097/01.hjh.0000431740.32696.cc
39. Brown RE, Riddell MC, Macpherson AK, et al. The joint association of physical activity, blood-pressure control, and pharmacologic treatment of hypertension for all-cause mortality risk. Am J Hypertens. 2013;26:1005–1010. doi:10.1093/ajh/hpt063
40. Araújo CO, Makdisse MRP, Peres PAT, et al. Different patterns for the 6-minute walk test as a test to measure exercise ability in elderly with and without clinically evident cardiopathy. Arq Bras Cardiol. 2006;86:198–205. doi:10.1590/S0066-782X2006000300007
41. Roul G, Germain P, Bareiss P. Does the 6-minute walk test predict the prognosis in patients with NYHA class II or III chronic heart failure? Am Heart J. 1998;136:449–457. doi:10.1016/S0002-8703(98)70219-4
42. Zugck C, Krüger C, Dürr S, et al. Is the 6-minute walk test a reliable substitute for peak oxygen uptake in patients with dilated cardiomyopathy? Eur Heart J. 2000;21:540–549. doi:10.1053/euhj.1999.1861
43. Piercy KL, Troiano RP, Ballard RM, et al. The physical activity guidelines for Americans. JAMA. 2018;320:2020–2028.
44. Krinski K, Machado DGS, Lirani LS, et al. Let’s walk outdoors! self-paced walking outdoors improves future intention to exercise in women with obesity. J Sport Exerc Psychol. 2017;39:145–157. doi:10.1123/jsep.2016-0220
45. Ash G, Walker T, Olson K, et al. Reproducibility of ambulatory blood pressure changes from the initial values on two different days. Clinics. 2013;68:1509–1515. doi:10.6061/clinics/2013(12)06
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

