Back to Journals » Patient Preference and Adherence » Volume 12
Short-term effect of a smart nebulizing device on adherence to inhaled corticosteroid therapy in Asthma Predictive Index-positive wheezing children
Authors Zhou Y, Lu Y, Zhu H, Zhang Y, Li Y, Yu Q
Received 16 January 2018
Accepted for publication 18 March 2018
Published 18 May 2018 Volume 2018:12 Pages 861—868
DOI https://doi.org/10.2147/PPA.S162744
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Naifeng Liu
Yuan Zhou,1,* Yanming Lu,1,* Haojin Zhu,2 Yanhan Zhang,1 Yaqin Li,1 Qing Yu1
1Department of Pediatrics, South Campus, Renji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai, 201112, People’s Republic of China; 2School of Electronic Information and Electrical Engineering, Shanghai Jiao Tong University, Shanghai, 201112, People’s Republic of China
*These authors contributed equally to this work
Objective: To explore the effect of a smart nebulizing device on the rate of adherence to inhaled corticosteroid (ICS) in children with positive Asthma Predictive Index.
Methods: In total, 65 children with positive Asthma Predictive Index and under the age of 5 years who visited our hospital from October 2015 through October 2016, were randomly assigned to receive conventional nebulization or smart nebulization. The smart nebulizer was connected to smart phones via an App. The following information was collected: rate of adherence to ICS, frequency of emergency visits or hospitalizations, application of antibiotics or oral steroids, and wheezing progression or improvement.
Results: The rate of adherence to ICS was 86.67% (26/30), 76.67% (23/30), and 67.33% (20/30) in the smart nebulization group, and 62.86% (22/35), 51.42% (18/35), and 40.00% (14/35) in the conventional nebulization group after 4-, 8-, and 12-week therapy, respectively. There were significant differences between the 2 groups at all of the time points (P<0.05). Both day- and night-time wheezing scores were significantly lower in the smart nebulization group than those of the conventional nebulization group after 4-, 8-, and 12-week therapy (P<0.05). The frequency of emergency visits, comorbidity of respiratory infection, antibiotics or systemic steroid usage, and therapeutic cost for additional treatment during the 12-week study period, was significantly lower in the smart nebulization group than that in the conventional nebulization group (P<0.05).
Conclusion: A smart electronic nebulization device could significantly improve the rate of adherence to ICS in children under the age of 5 years, and thus could significantly reduce the frequency of emergency visits and respiratory infections as well as the usage of antibiotics or systemic steroids.
Keywords: smart nebulizer, adherence, inhaled corticosteroid, wheezing
Introduction
Wheezing is one of the most common symptoms in children <2 years of age, especially in infants 1–6 months old.1 Wheezing has tended to increase in recent years in China. Recurrent episodes of wheezing may develop into asthma, which is a chronic airway disease characterized by wheezing, breathlessness, chest tightness, and coughing.2,3 An epidemiologic study in China indicated that the prevalence of asthma in childhood has significantly increased in the past decade, predominantly in preschool-age children.4 Consistently, worldwide epidemiologic studies reported that 48.5% children had at least 1 episode of wheezing and breathlessness before 1 year of age, and that 30% of the preschool-age children who have wheezing episodes will develop asthma in the future.5 The prevalence of wheezing among 6–7 year old children ranged from 2.4% in Jodhpur (India) to 37.6% in Costa Rica.3 In addition, it has been reported that 80% of asthmatic children experienced wheezing episodes at age of 6 years or younger, and 50% of them had wheezing episodes when they were younger than 3 years.6 Furthermore, the most recent estimation indicated that while approximately 50% of young children experience at least 1 acute episode of wheezing before 6 years of age, less than 10%–15% of these children will develop asthma at school age (6–12 years old) even though 30%–40% of these young children may have recurrent wheezing during school age.7,8
Diagnosis and treatment of young children with recurrent wheezing is complex, because most of these children do not have persistent asthma with regular symptoms such as wheezing. Therefore, the Asthma Predictive Index (API) has been used to predict the risk of asthma development in children aged 5 years or younger,9–11 and API-positive children are recommended to receive long-term daily inhaled corticosteroid (ICS).12,13 In this regard, it is predicted that 77% of children will have asthma between age of 6 and 13 years if they were API positive at the age of 3, while only 3% of the children will have asthma if they were API negative at the age of 3.10 In addition, the lung function of the children with positive API was worse than that of the children with negative API.14 Therefore, in the current study, API was used to identify children with wheezing and those who would potentially have asthma in the future, and API-positive children under the age of 5 years were enrolled into this study.
The most recently published Chinese version of “Guideline for Diagnosis and Optimal Management of Asthma in Children (2016)” suggested that ICS is recommended as standard therapy for API-positive children in order to prevent and control asthma development in later life.15 ICS could effectively control airway inflammation and hyperreactivity and improve symptoms such as wheezing, lung function, and quality of life in asthmatic children.13,16,17 However, daily adherence to ICS was low in children at young age because the majority of children did not take their inhalers as prescribed even if they or their parents reported they did.18 In this context, it has been reported that adherence to long-term ICS in asthmatic children varied from 22% to 63%, and adherence to ICS remained mostly less than 50%.19 The current study was, therefore, designed to study the influence of smart nebulizing devices on the adherence rate to ICS in children under 5 years of age with positive API.
Materials and methods
Patients
Totally, 65 children who visited Renji Hospital, Shanghai Jiaotong University, from October 2015 through October 2016 were enrolled into this study. The protocol of this study was approved by the Ethics Committee of Renji Hospital, Shanghai Jiaotong University. A signed consent form was obtained from parents who were willing to have their children to enroll into this study. The inclusion criteria were as follows: 1) children with wheezing; 2) boys or girls under the age of 5 years; 3) children with positive API who came to Renji Hospital with asthma exacerbation for the first time; 4) parents having signed consent forms and agreed to provide information during the 12-week study period; 5) children with wheezing episodes that were not caused by congestive heart disease, airway deformity, or occlusive bronchitis.
Methods
Smart nebulization
This was carried out by assembling a smart electronic device (red framed in Figure 1) to a nebulizer, which automatically controlled the status of nebulization and recorded desired information. This smart electronic device was also wirelessly connected to smart phone through BLE4.0 software, through which the pediatrician reminded the children to take the nebulization if they forgot to do so. In contrast, the conventional nebulizer does not have such a remind function.
  | Figure 1 Photographs of an electronic smart device and nebulizer. |
Smart App for the parents
The App was available in both iOS and Android operation systems. The App was used by parents to instantly communicate with a pediatrician or their child. Through the smartphone App, parents obtained data on time, duration, and frequency of nebulization.
Website for pediatricians
The jQuery Datatables (SpryMedia Ltd, Dunfermline, Scotland) and Bootstrap were used by pediatricians to monitor children’s progress, adherence to ICS therapy, and to have real-time communication with children’s parents when necessary.
Data exchange between the smart electronic device and smart phone
BLE4.0 software was used for connection between the smart electronic device and smart phones, including iPhone 4S or later version iPhones (Apple Inc, Cupertino, CA, USA) as well as Android 4.3 (Google, Mountain View, CA, USA) or later version phones with the BLE4.0 system. With this system, the battery life lasted over 1 year without interruption of communication between the smart electronic device and phones.
Research design
Asthmatic children’s medical history and physical details were obtained. Of the 65 enrolled children, 35 children were treated with conventional nebulization and 30 children were treated with smart nebulization. The Omron Nebulizer (Model: NE-C900) (Omron Healthcare, Hoofddorp, the Netherlands) or Pari Medical Nebulizer (Model: JuniorBOY SX) (PARI GmbH, Starnberg, Germany) was used for delivery of drugs following the Guideline of Chinese Pediatric Association on ICS.20 Specifically, during the 1st week of enrollment, budesonide suspension (AstraZeneca, Cambridge, UK) 0.5–1.0 mg each time, plus terbutaline sulfate solution for nebulization (AstraZeneca) 0.25–0.5 mg each time, and Aloe Vera Inhalation solution (Boehringer-Ingelheim, Ingelheim am Rhein, Germany) 250–500 μg each time were used. From the 2nd week through the 12th week, budesonide suspension was used as ICS. The parents of children enrolled into this study were educated in the correct use of the nebulizers. During the total 12-week study period, either the children were seen by pediatricians or the children’s parents were interviewed by pediatricians through telephone call every 2 weeks. The following medications were permitted to be prescribed to the children by pediatricians during the 12-week study period: bronchial dilator, oral steroid, antibiotics, oral antihistamine drug, or leukotriene antagonists.
Statistical analysis
Continuous variables with normal distribution were expressed by either mean ± SD or median and interquartile range. Student’s t-test was used for comparison between 2 groups and Wilcoxon 2 samples test was used for nonnormal distribution data analysis. Discrete variables were expressed by frequency (%), and χ2 test or Fisher exact probability test was used. Cochran–Mantel–Haenszel test was used for comparison of the symptomatic score, drug usage, and severity of wheezing during the treatment. SAS9.3 statistical analysis software (SAS Institute, Cary, NC, USA) was used to conduct all the analyses. P<0.05 was considered as significant.
Results
Demographic and baseline characteristics of the participants
As shown in the Table 1, a total of 65 children with wheezing were enrolled in this study. Of them, 44 were boys and 21 were girls. Their mean age was 37.91±11.83 months. They were randomly assigned to the conventional nebulization group (35 cases) and the smart nebulization group (30 cases). There was no significant difference between the 2 groups in gender ratio, age, and baseline characteristics (P>0.05).
  | Table 1 General characteristics of the children before treatment |
Comparison of ICS adherence rate
The 4-, 8-, and 12-week treatment adherence rates were 86.67% (26/30), 76.67% (23/30), and 67.33% (20/30), respectively, in the smart nebulization group and 62.86% (22/35), 51.42% (18/35), and 40.00% (14/35), respectively, in the conventional nebulization group. There were significant differences between the 2 groups at any of the time points (P<0.05, Table 2).
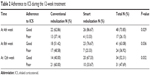  | Table 2 Adherence to ICS during the 12-week treatment |
Comparison of day- and night-time symptom scores
As shown in Table 3, both day- and night-time asthma-like symptom scores were significantly lower in the smart nebulization group than in the conventional nebulization group at 4, 8, and 12 weeks posttherapy (P<0.05).
  | Table 3 Scoring of the symptoms during the 12-week treatment |
Comparison of additional drug usage and therapy cost
During the 12-week study period, in addition to ICS nebulization therapy, some children received additional medication including antibiotics and oral steroid. As shown in Table 4, the frequency of emergency visit, comorbidity of respiratory infection, antibiotics or systemic steroid usage, and therapeutic cost for these additional treatments was significantly lower in the smart nebulization group than in the conventional nebulization (P<0.05).
  | Table 4 Comparison on additional drug usage and cost during the 12-week treatment |
However, usage of additional medicine for symptom relief and limitation of activities were not significantly different between the 2 groups during the 12-week treatment (Table 5, P>0.05).
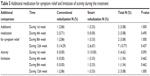  | Table 5 Additional medication for symptom relief and limitation of activity during the treatment |
Discussion
The current study compared the therapeutic effect of conventional nebulization and smart nebulization in the treatment of asthmatic children under the age of 5 years. It was found that children in the smart nebulization group more readily followed the treatment procedure (better compliance and adherence to inhaled corticosteroid), had a significantly lower symptomatic score, and a significantly lower frequency of emergency visits, comorbidity with respiratory infection, usage of antibiotics or systemic steroid usage, and cost for the additional treatments compared with children treated with conventional nebulization.
Asthma is the most prevalent chronic disease in children, and the majority of asthma or wheezing starts before the age of 5 years. While the management of these children is complicated, nebulization of steroid or bronchodilators is the most common method for delivery of drugs to the small airways. Efficient delivery of medication through an inhaler requires not only a delivery device, but also the coordination between inhalation and actuation on the patient’s inspiratory flow as well as compliance of the patients to the physician’s instruction. Currently, methods of assessing adherence to ICS include daily diaries, self-report questionnaire, prescription filling record, physician’s subjective assessment, dose meter, remaining medication amount check, and electronic monitoring device. Of these methods, an electronic monitoring device provides an objective method in assessing adherence to a therapy.21 The method of prescription filling record does not tell if the child takes the medicine as ordered, while method of daily diaries and self-report questionnaire largely depend on the children’s or the parents’ subjective assessment, and thus they lack validity;22 especially, under the influence of the “Hawthorne Effect,” overestimation of drug adherence is common in these assessing methods.23,24 In this regard, it has been reported that an objectively assessed adherence rate of 25% was reported as 85% by the parents subjectively.25 Thus, it is necessary to explore alternative methods to objectively evaluate the long-term adherence to ICS.
Chan et al26 reported that an electronic monitoring and reminding device could significantly improve the adherence to ICS in 220 cases of 6–12-year-old asthmatic children. In the current study, a smart nebulizer was used to remind the children to inhale corticosteroid on time, and by using the smart nebulizer the pediatricians were able to have real-time communication with the children through a smart phone App. As a result of this smart device application, adherence to ICS was significantly improved compared to the group using conventional nebulizing device. In addition, symptomatic score, frequency of emergency visit, comorbidity of respiratory infection, antibiotics or systemic steroid usage, and cost for additional treatment were significantly lower in children with smart nebulization than children with conventional nebulization.
Adherence to ICS decreased as a function of time. In this context, it has been reported that adherence to ICS at 4 months was 60.4%, which dropped to 49.8% after 1 year of therapy.27 Similarly, adherence to ICS in the current study was also reduced as a function of time, which was regardless of using a conventional nebulizer or a smart nebulizer, suggesting education of the children or their parents may be crucial to maintain a high adherence rate in addition to equipment modernization.
While a smart nebulization device could significantly improve the therapeutic effect, the cost of the device should also be considered in clinical application. Currently, the cost of a smart nebulizer is still fairly high, and most devices are still in the clinical research stage although the cost of long-term therapy in asthma may be reduced with significant improvement of adherence rate.26,28 In the current study, not only the health care cost, but also frequency of respiratory infection and emergency visit, and usage of antibiotics or systemic steroids were significantly reduced in children treated with smart nebulization compared with that of children treated with conventional nebulization. However, there were no differences between the groups in frequency and severity of recurrent wheezing or frequency of hospitalization during the 12-week treatment period. These findings suggested that clinical application of smart nebulizers in asthmatic children remains to be further improved, including inhaling methods,29 and to be further investigated in larger populations.
While smart devices with electronic monitoring are considered as “gold standard” in measuring adherence to or compliance with a therapy, smart electronic devices are not without problems. Smart devices can fail to record or download data due to a variety of reasons such as battery failure.23,30 It has been reported that 20% (380 out of 2,360) of failure in data download from smart monitoring devices occurred due to malfunction of monitor or battery.30 In the current study, however, the smart nebulizers functioned well throughout the study period, although occasional failure in connection to smart phone App or low battery occurred.
Conclusion
Taken together, the current study compared the outcomes of conventional nebulizer and smart nebulizer in the management of asthmatic children under the age of 5 years. Children treated with smart nebulizer had not only a higher rate of adherence to ICS, but also a significantly lower symptomatic score, a significantly lower frequency of emergency visits and respiratory infection, reduced usage of antibiotics or systemic steroid, and lower cost for additional treatment compared with children treated with conventional nebulizer. These findings suggested that smart nebulizers are superior to the conventional nebulizers in controlling asthmatic symptoms by increasing adherence to ICS therapy.
Ethics approval and consent to participate
Ethical approval for this study was given by the Ethics Committee of Renji Hospital, Shanghai Jiaotong University. Written informed consent was obtained from the parent(s) or guardian(s) of all study participants.
Acknowledgment
This study was supported by the Medical and Industrial Cross Research Fund of Shanghai Jiao Tong University (YG2016MS57).
Disclosure
The authors report no conflicts of interest in this work.
References
Ebisawa M, Terada A, Sato K, et al. Intermittent and episode-driven use of pranlukast to reduce the frequency of wheezing in atopic children: a randomized, double-blind, placebo-controlled trial. World Allergy Organ J. 2015;8(1):11. | ||
Braman SS. The global burden of asthma. Chest. 2006;130(Suppl 1):4S–12S. | ||
Lai CK, Beasley R, Crane J, et al. Global variation in the prevalence and severity of asthma symptoms: phase three of the International Study of Asthma and Allergies in Childhood (ISAAC). Thorax. 2009;64(6):476–483. | ||
National Cooperative Group on Childhood Asthma; Institute of Environmental Health and Related Product Safety, Chinese Center for Disease Control and Prevention; Chinese Center for Disease Control and Prevention. Third nationwide survey of childhood asthma in urban areas of China. Chin J Pediatr. 2013;51(10):729–735. | ||
Ferreira IC, Wandalsen NF. [Prevalence and severity of wheezing in the first year of life in the city of Santo Andre, Brazil]. Rev Paul Pediatr. 2014;32(3):164–170. Portuguese. | ||
Martinez FD. Development of wheezing disorders and asthma in preschool children. Pediatrics. 2002;109(Suppl 2):362–367. | ||
Beigelman A, Bacharier LB. Management of preschool recurrent wheezing and asthma: a phenotype-based approach. Curr Opin Allergy Clin Immunol. 2017;17(2):131–138. | ||
Sbihi H, Koehoorn M, Tamburic L, Brauer M. Asthma trajectories in a population-based birth cohort. Impacts of air pollution and greenness. Am J Respir Crit Care Med. 2017;195(5):607–613. | ||
Castro-Rodriguez JA, Holberg CJ, Wright AL, Martinez FD. A clinical index to define risk of asthma in young children with recurrent wheezing. Am J Respir Crit Care Med. 2000;162(4 Pt 1):1403–1406. | ||
Castro-Rodriguez JA. The Asthma Predictive Index: a very useful tool for predicting asthma in young children. J Allergy Clin Immunol. 2010;126(2):212–216. | ||
Taussig LM, Wright AL, Holberg CJ, Halonen M, Morgan WJ, Martinez FD. Tucson Children’s Respiratory Study: 1980 to present. J Allergy Clin Immunol. 2003;111(4):661–675; quiz 676. | ||
National Asthma Education and Prevention Program. Expert Panel Report 3: guidelines for the diagnosis and management of asthma: full report 2007. 2007; Available from: http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.pdf. Accessed April 20, 2018. | ||
Abrams EM, Szefler SJ, Becker AB. Does inhaled steroid therapy help emerging asthma in early childhood? Lancet Respir Med. 2017;5(10):827–834. | ||
Dinh-Xuan AT, Annesi-Maesano I, Berger P, et al. Contribution of exhaled nitric oxide measurement in airway inflammation assessment in asthma. A position paper from the French Speaking Respiratory Society. Rev Mal Respir. 2015;32(2):193–215. | ||
Subspecialty Group of Respiratory Diseases, Society of Pediatrics, Chinese Medical Association; Editorial Board, Chinese Journal of Pediatrics. [Guideline for the diagnosis and optimal management of asthma in children (2016)]. Chin J Pediatr. 2016;54(3):167–181. Chinese. | ||
Visness CM, London SJ, Daniels JL, et al. Association of childhood obesity with atopic and nonatopic asthma: results from the National Health and Nutrition Examination Survey 1999–2006. J Asthma. 2010;47(7):822–829. | ||
Baraldi E, Rossi GA, Boner AL; Beclomethasone, Salbutamol Treatment for Children Study Group. Budesonide in preschool-age children with recurrent wheezing. N Engl J Med. 2012;366(6):570–571; author reply 571. | ||
Bender B, Wamboldt FS, O’Connor SL, et al. Measurement of children’s asthma medication adherence by self report, mother report, canister weight, and Doser CT. Ann Allergy Asthma Immunol. 2000;85(5):416–421. | ||
Barnes CB, Ulrik CS. Asthma and adherence to inhaled corticosteroids: current status and future perspectives. Respir Care. 2015;60(3):455–468. | ||
Shen K, Deng L, Li Y. Experts consensus on the application of inhaled corticosteroid in children. J Clin Pediatr. 2014;6:504–511. | ||
Chan AH, Stewart AW, Foster JM, Mitchell EA, Camargo CA Jr, Harrison J. Factors associated with medication adherence in school-aged children with asthma. ERJ Open Res. 2016;2(1):pii:00087-2015. | ||
Morton RW, Everard ML, Elphick HE. Adherence in childhood asthma: the elephant in the room. Arch Dis Child. 2014;99(10):949–953. | ||
Lindsay JT, Heaney LG. Non-adherence in difficult asthma and advances in detection. Expert Rev Respir Med. 2013;7(6):607–614. | ||
Klok T, Kaptein AA, Brand PLP. Non-adherence in children with asthma reviewed: The need for improvement of asthma care and medical education. Pediatr Allergy Immunol. 2015;26(3):197–205. | ||
Otsuki M, Eakin MN, Rand CS, et al. Adherence feedback to improve asthma outcomes among inner-city children: a randomized trial. Pediatrics. 2009;124(6):1513–1521. | ||
Chan AH, Stewart AW, Harrison J, Camargo CA Jr, Black PN, Mitchell EA. The effect of an electronic monitoring device with audiovisual reminder function on adherence to inhaled corticosteroids and school attendance in children with asthma: a randomised controlled trial. Lancet Respir Med. 2015;3(3):210–219. | ||
Jentzsch NS, Camargos P, Sarinho ES, Bousquet J. Adherence rate to beclomethasone dipropionate and the level of asthma control. Respir Med. 2012;106(3):338–343. | ||
Chan AH, Reddel HK, Apter A, Eakin M, Riekert K, Foster JM. Adherence monitoring and e-health: how clinicians and researchers can use technology to promote inhaler adherence for asthma. J Allergy Clin Immunol Pract. 2013;1(5):446–454. | ||
Melani AS, Bonavia M, Cilenti V, et al. Inhaler mishandling remains common in real life and is associated with reduced disease control. Respir Med. 2011;105(6):930–938. | ||
Zeiger RS, Mauger D, Bacharier LB, et al. Daily or intermittent budesonide in preschool children with recurrent wheezing. N Engl J Med. 2011;365(21):1990–2001. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
