Back to Journals » Neuropsychiatric Disease and Treatment » Volume 11
Serum 25-hydroxyvitamin D predicts cognitive performance in adults
Authors Darwish H, Zeinoun P, Ghusn H, Khoury B, Tamim H, Khoury S
Received 20 April 2015
Accepted for publication 2 July 2015
Published 25 August 2015 Volume 2015:11 Pages 2217—2223
DOI https://doi.org/10.2147/NDT.S87014
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Roger Pinder
Hala Darwish,1 Pia Zeinoun,2 Husam Ghusn,3,4 Brigitte Khoury,2 Hani Tamim,5 Samia J Khoury6
1Hariri School of Nursing, Faculty of Medicine, American University of Beirut, Beirut, Lebanon; 2Psychiatry Department, Faculty of Medicine, American University of Beirut, Beirut, Lebanon; 3Internal Medicine Department, Faculty of Medicine, American University of Beirut, Beirut, Lebanon; 4Geriatrics Department, Ain Wazein Hospital, El Chouf, Lebanon; 5Clinical Research Institute, Faculty of Medicine, American University of Beirut, Beirut, Lebanon; 6Neurology Department, Faculty of Medicine, American University of Beirut, Beirut, Lebanon
Background: Vitamin D is an endogenous hormone known to regulate calcium levels in the body and plays a role in cognitive performance. Studies have shown an association between vitamin D deficiency and cognitive impairment in older adults. Lebanon has a high 25-hydroxyvitamin D (25(OH)D) deficiency prevalence across all age groups.
Methods: In this cross-sectional study, we explored the cognitive performance and serum 25(OH)D levels using an electrochemoluminescent immunoassay in 254 older (>60 years) as well as younger (30–60 years) adults. Subjects’ characteristics, including age, years of education, wearing of veil, alcohol consumption, smoking, and physical exercise, were collected. Participants were screened for depression prior to cognitive screening using the Montreal Cognitive Assessment Arabic version. Visuospatial memory was tested using the Rey Complex Figure Test and Recognition Trial, and speed of processing was assessed using the Symbol Digit Modalities test.
Results: Pearson’s correlation and stepwise linear regression analyses showed that a low vitamin D level was associated with greater risk of cognitive impairment in older as well as younger adults.
Conclusion: These findings suggest that correction of vitamin D needs to be explored as an intervention to prevent cognitive impairment. Prospective longitudinal studies are needed to ascertain the effect of such interventions.
Keywords: cognitive function, aging, neuro-psychological function
Introduction
Vitamin D is an endogenous hormone that regulates calcium levels in the body and maintains bone mineral density and muscle mass and function.1,2 Vitamin D is metabolized by the action of the ultra violet (UV) sun rays, liver, and kidney, from its inactive cholecalciferol form into the steroid and active forms 25-hydroxyvitamin D (25(OH)D) and 1,25-dihydroxyvitamin D. Human vitamin D levels are most commonly measured by 25(OH)D serum concentration. Vitamin D receptors are present in the brain, neurons, and glial cells, indicating additional functions. These brain receptors are present in both humans and animals, and play a role in cognitive function.3,4
Older adults with higher serum vitamin D have shown better cognitive performance compared with those with lower vitamin D levels. Similarly, vitamin D deficiency in older adults with or without mild Alzheimer’s disease has been correlated with decreased cognitive performance.5,6 In more recent studies, older adults with low vitamin D level showed more cognitive impairment than did those with sufficient levels.7 The majority of studies that showed association between vitamin D deficiency and cognitive impairment have been conducted on older adults, although a few studies have targeted younger adults.8–11 In this study, we examine the role of vitamin D on cognitive function in adults aged 30–60 and >60 years of age.
Vitamin D deficiency or insufficiency is prevalent in most countries; it is now considered a pandemic.12 Despite being a sunny country, Lebanon has a high prevalence of 25(OH)D deficiency in adults and older adults: 55% of older women and 25% of older men have deficient vitamin D levels.1
In this study, we hypothesized that cognitive impairment will be associated with low vitamin D levels in older (>60 years) as well as younger adults (30–60 years).
Methods
This was a cross-sectional study of 254 adults (30 and above years) recruited from the American University of Beirut-Medical Center (AUBMC) outpatient clinics, and elderly nursing homes in the greater Beirut and outside Beirut region over a 2-year period (2010–2012). Only 22 of 97 older adults were recruited from the nursing homes – the majority were recruited from the community.
Subjects with history of neurological disorders, traumatic brain injury, psychiatric disorders, cognitive impairment, and history of alcohol and/or drug abuse/dependence were excluded from the study. Men who consumed more than 15 drinks/week and women who consumed more than eight drinks/week were considered excessive alcohol consumers13 and were excluded from the study. Individuals who, during the 3 months prior to screening, were on antidepressants, mood stabilizers, or medications known to affect cognitive performance were excluded as well.
Prior to testing, informed consent was secured from all the participants. Then, health, anxiety, and depression screening questionnaires were administered to further screen for eligibility, followed by cognitive tests. The same psychologist administered the tests, and then a 5 mL blood sample was collected to test for serum 25(OH)D. This study received ethics approval from the Institutional Review Board of the American University of Beirut.
The Brief Risk Factor Surveillance System (BRFSS)14 is a screening questionnaire that inquires about the participant’s current health status, medical history, physical activity (weekly frequency, type, and duration of activity), smoking habits (current and previous smoking habits), age, years of education, and alcohol consumption (weekly and monthly). Age, smoking, and excessive alcohol consumption have been associated with impaired cognitive performance.15 However, increased years of education and physical activity have been positively correlated with cognitive performance.16,17
The Hopkins Symptoms Checklist-25 (HSCL-25)-Arabic was administered to screen for anxiety and depression that may affect cognitive performance negatively.18 This is a 25-item tool that has been translated into Arabic and is widely used due to its brevity, reliability, and validity.19–22
The Montreal Cognitive Assessment (MoCA) is designed to screen for cognitive impairment in adults. It requires approximately 10 minutes to administer. The MoCA-Arabic version has demonstrated 92.3% sensitivity and 85.7% specificity for detecting mild cognitive impairment in older adults in Egypt.23 In our sample, the test showed high internal consistency (α=0.83) and excellent test–retest reliability (α=0.92) in a subsample of healthy adults (n=100).
The Rey Complex Figure Test and Recognition Trial (RCFT) measures visual spatial memory and is extensively used in neuropsychological testing24,25 on adults. The 45-minute administration requires the participant to copy a complex shape (time to copy is measured) and immediately redraw it after 3 minutes (immediate recall [IR]), and 30 minutes from copying (delayed recall [DR]). Lastly, the participant is required to recognize parts of the initial figure from 20 shapes (recognition). The brief instructions were translated to Arabic, reviewed by the first two authors, and piloted on ten participants. This test does not require translation. An RCFT subsample (n=50) was rescored by the first two authors, and a 0.90 interrater reliability was found.
The Symbol Digit Modalities Test (SDMT) is a speed of processing test. It requires 10 minutes from administration to scoring.26 The SDMT requires minimal instructions, which were delivered in Arabic after being translated and reviewed by the first two authors.
Serum 25(OH)D was measured in the Endocrinology Department core laboratory at AUBMC, using a protein-binding assay (DiaSorin RIA; DiaSorin, Saluggia, Italy), with intra- and interassay variation coefficients less than 13% at a serum concentration of 47 ng/mL.27 The study laboratory is a participant in the international Vitamin D External Quality Assessment Scheme (DEQAS). This assay measures both 25- OHD2 and 25-OHD3. The intra-assay coefficient of variance is 5%±9%, based on 124 specimens run as separate samples in the assay, with a mean 25-OHD level of 19±5 ng/mL. Blood for the studies was stored as serum at −20°C in the Endocrinology Department core laboratory, and samples from each subject were assayed in duplicate.
The predictor variables – 25(OH)D, sex, age, years of education, physical activity, sun exposure, use of vitamin supplements, smoking, and alcohol consumption – were analyzed using descriptive measures. Independent sample t-tests and Pearson’s r correlations were performed between the predictor and outcome variables, MoCA, RCFT, and SDMT. A stepwise linear regression analysis was conducted to examine the relationship between predictor variables and the outcome variables.
Results
A total of 380 subjects were screened for eligibility; of these, 90 subjects were excluded before being enrolled for not meeting eligibility criteria or not showing up for an appointment. Of the 290 enrolled subjects, 19 were excluded after further screening because they did not meet eligibility criteria or had motor or sensory impairment. In addition, ten participants were found to have more than a moderate level of anxiety and depression (greater than 3.2 and 3.3 respectively) and were referred to the AUBMC Psychiatry Department for management, while seven refused to continue because they found the tests to be too time-consuming and chose to withdraw from the study.
Of the 254, 61.8% were adults aged 30–60 years old (with mean ± standard deviation of 44.31±8.72 years) and 38.2% were older than 60 years old (71.26±0.77 years). Their mean HSCL-25 anxiety and depression scores were 1.49±0.40 and 1.80±0.52 respectively. The majority were female (68.9%), among whom 83.1% did not wear a veil. The majority (67%) had received 12 or more years of education (Figure 1). Approximately 26% were smoking at the time of the study, and 40% were ever smokers, while 26.7% drank a moderate amount of alcohol. The majority (76.4%) had low 25(OH)D <30 ng/mL (25.50±12.67 ng/dL). The mean 25(OH)D level in veiled women (n=41) was lower than that in the rest (n=134) of the sample (16.02±7.73 ng/dL vs 27.11±14.15 ng/dL) (P<0.0001). The difference in 25(OH)D level between males and females was not statistically significant (21.25±9.59 ng/dL vs 24.51±13.75 ng/dL, respectively) (P=0.057) (Table 1).
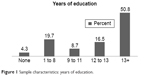  | Figure 1 Sample characteristics: years of education. |
A total of 75 older adults came from their homes to the AUBMC or Ain Wa Zein Hospital for testing and returned to their homes. Only 22 were residents of a nursing home. The research team, including the geriatrician coauthor on this study, screened the 22 nursing home residents carefully. The characteristics of the 22 older adults were compared with those of the rest of the population, and there was no statistical difference in terms of sex, vitamin D level, or cognitive performance. The 25(OH)D of older adults in nursing homes did not differ from that in the community subjects (20.26±6.83 ng/dL vs 23.46±11.00 ng/dL, respectively) (P=0.516).
After removing outliers, 51.6% of the sample scored lower than the cutoff score of 26 for cognitive impairment on the MoCA, after adjusting for years of education. It was noted in the factor analysis that the verbal fluency test had extremely low scores. Therefore, we hypothesized that lower education contributed to lower verbal fluency (X2=23.854, df=1, P<0.0001). After adjusting for verbal fluency scores, 40.3% scored lower than the cutoff score.
On the different RCFT subsections, 35% to 40% of the sample scored lower than the fifth percentile. Only 133 completed the SDMT; of those, 43.6% scored moderately low or very low after adjusting for age and years of education. There was no difference in cognitive performance between males and females on any of the tests. However, in the older adults subgroup, males performed better than females on the MoCA (23.06±4.22 vs 18.89±5.83, respectively) (P=0.01) and the RCFT-IR (11.23±4.22 vs 8.53±5.46, respectively) (P=0.029). Of those who scored low on the MoCA, 22% were males and 59.7% females.
As a whole, the mean years of education did not differ based on sex; however, it was significantly higher in elderly males than in females (P=0.002). Older adults scored lower than did young adults on the MoCA (20.23±5.7 vs 26.04±3.23, respectively) (P<0.0001), on the RCFT-IR (9.43±5.56 vs 15.47±6.31, respectively (P<0.0001), on the RCFT-DR (9.01±5.62 vs 15.47±6.54, respectively) (P<0.0001), and on the SDMT (29.92±13.37 vs 49.68±13.99, respectively) (P<0.0001). The cognitive performance of the older adults in nursing homes was not different than those in the community.
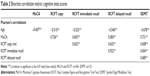
To ensure that the participants’ performance was consistent, Pearson’s bivariate correlation was performed. The results showed that the different measures correlated sufficiently high to indicate that they did not measure the same cognitive domains, but they were not repetitive (Table 2).
The bivariate correlations between age, years of education, physical activity, and alcohol, known confounding variables of cognitive performance,16,17,28 and the cognitive tests scores were examined. The Pearson’s r correlation was statistically significant and negative between age and the different cognitive tests scores (r=−0.435, P<0.01). Years of education correlated with better performance on all cognitive tests; MoCA, RCFT (IR, DR, and copy), and SDMT (r>0.5, P<0.01), and physical activity was associated with better cognitive performance on the MoCA, and RCFT-IR and -DR.
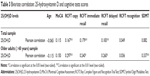
Overall, 25(OH)D level correlated positively with the RCFT-IR and -DR (r=0.179 and r=0.183, respectively; P<0.01). But in the older adults sample, 25(OH)D level showed a positive and statistically significant correlation with the SDMT as well as the RCFT-IR and -DR (r=0.337, r=0.240, and r=0.260, respectively; P<0.01) (Table 3). As expected, age, years of education, and alcohol were positive predictors of the MoCA, RCFT, and SDMT. Interestingly, 25(OH)D predicted RCFT-IR and the SDMT but not the MoCA, after controlling for age and years of education and alcohol (Table 4).
There was a significant relationship between years of education and 25(OH)D levels; those with ≥12 years of education had significantly higher 25(OH)D levels than did those with <12 years of education (25.04±13.62 ng/dL vs 20.32±9.8 ng/dL, P<0.005; respectively) (r=0.175 P=0.005). We attributed this difference to the intake of vitamin D supplements, since the correlation disappeared after removing subjects taking supplements from the analysis (Table 4).
Discussion
In this study, we explored the relationship of 25(OH)D levels and cognitive performance in younger and older adults. Our data showed a high prevalence of 25(OH)D deficiency that was significantly associated with poor cognitive performance in younger and older adults. Interestingly, we found that a moderate amount of alcohol consumption was associated with better cognitive performance.
The positive association between low 25(OH)D and cognitive impairment has been previously reported.11,29–32 In this study, we found that this association was significant in younger adults as early as 30 years of age. This observation suggests that early correction of vitamin D levels may control one of the factors associated with cognitive impairment and delay the onset of age-related cognitive impairment. Longitudinal studies examining the effect of early vitamin D supplementation on cognitive performance over time is therefore needed.
The high prevalence of hypovitaminosis D found in this study is in line with previous results.33 Genetic and environmental factors could be behind this widespread low vitamin D in this population.34 The significant association between age, years of education, and cognitive performance is well known.35 When controlling for age and years of education, 25(OH)D was a significant predictor of cognitive performance, in line with the previous studies in older adults.36
In this study, we could not demonstrate a correlation between 25(OH)D and the MoCA scores in either the whole group or the older adult subgroup. Moreover, 25(OH)D did not predict the MoCA score after controlling for age and education, and adjusting for verbal fluency. Given that the other cognitive tests coherently showed a correlation between 25(OH)D and cognitive performance, this suggests that the cutoffs and items of the MoCA are not appropriate for the population being tested. The MoCA scores may not account for education. The majority, across all age groups, were unable to complete the verbal fluency section. It is possible that the letter “f” used in the verbal fluency section of the MoCA-Arabic in Lebanon is not representative of verbal fluency due to low frequency of words in Lebanese spoken dialect. The MoCA is has many verbal sections, therefore requires cultural adaptability, which is not the case for the SDMT and the RCFT. It may be too broad as a measure of cognitive performance compared with the RCFT that targets one cognitive function, namely visuospatial memory. In this study, we showed that the SDMT is a sensitive screening measure of cognitive performance, and the RCFT a good measure for in-depth evaluation.
The main advantage of using the RCFT and SDMT is that neither requires translation. However, these tests have not been used before in Lebanon or in other Arab countries, therefore, we lack the normative values of this population. Nonetheless, the visuospatial performance and speed of processing of older adults by age group is in line with previous studies of the same age and similar years of education.37,38
Wearing the veil has been suggested as a cause of low vitamin D in women in Middle-Eastern countries. In this study, although the majority of women did not wear the veil, the vitamin D level was lower in women who wore the veil than in men and women who did not wear a veil, in line with findings of previous study.39
In our sample, the ratio of women to men was higher among older and younger adults – this may suggest that women are more likely to volunteer in research studies.
Our data revealed an association with alcohol intake; moderate alcohol intake was a positive predictor of cognitive performance. Recent studies have shown that moderate alcohol consumption is associated with smaller decline in cognitive impairment.28,40 Only 26.7% of our sample consumed any alcohol. Nonetheless, the positive alcohol consumption effect could be demonstrated. This association between alcohol and cognitive performance needs to be explored further in a larger sample.
In summary, this study showed that a low vitamin D level is associated with greater risk of cognitive impairment in older as well as younger adults. Moderate alcohol consumption improved cognitive performance. These findings suggest that there are areas of possible intervention to prevent cognitive impairment. Prospective longitudinal studies are needed to ascertain the effect of such interventions.
Acknowledgment
The authors thank Ms Maha Habre, Knar Sagherian, and Mr Walid Yassine for their assistance and technical support. This work was funded by the American University of Beirut University Research Board and Medical Practice Plan.
Disclosure
The authors report no conflicts of interest in this work.
References
Arabi A, Baddoura R, Awada H, Salamoun M, Ayoub G, El-Hajj Fuleihan G. Hypovitaminosis D osteopathy: is it mediated through PTH, lean mass, or is it a direct effect? Bone. 2006;39(2):268–275. | ||
Dawson-Hughes B. Serum 25-hydroxyvitamin D and functional outcomes in the elderly. Am J Clin Nutr. 2008;88(2):537S–540S. | ||
Sutherland MK, Somerville MJ, Yoong LK, Bergeron C, Haussler MR, McLachlan DR. Reduction of vitamin D hormone receptor mRNA levels in Alzheimer as compared to Huntington hippocampus: correlation with calbindin-28k mRNA levels. Brain Res Mol Brain Res. 1992;13(3):239–250. | ||
Langub MC, Herman JP, Malluche HH, Koszewski NJ. Evidence of functional vitamin D receptors in rat hippocampus. Neuroscience. 2001;104(1):49–56. | ||
Llewellyn DJ, Lang IA, Langa KM, Melzer D. Vitamin D and cognitive impairment in the elderly U.S. population. J Gerontol A Biol Sci Med Sci. 2011;66(1):59–65. | ||
Littlejohns TJ, Henley WE, Lang IA, et al. Vitamin D and the risk of dementia and Alzheimer disease. Neurology. 2014;83(10):920–928. | ||
Granic A, Hill TR, Kirkwood TB, et al. Serum 25-hydroxyvitamin D and cognitive decline in the very old: the Newcastle 85+ Study. Eur J Neurol. 2015;22(1):106–15, e6. | ||
Toffanello ED, Coin A, Perissinotto E, et al. Vitamin D deficiency predicts cognitive decline in older men and women: The Pro. V.A. Study. Neurology. 2014;83(24):2292–2298. | ||
Nagel G, Herbolsheimer F, Riepe M, et al; ActiFE Study Group. Serum vitamin D concentrations and cognitive function in a population-based study among older adults in south Germany. J Alzheimers Dis. 2015;45(4):1119–1126. | ||
Maddock J, Cavadino A, Power C, Hyppönen E. 25-hydroxyvitamin D, APOE ε4 genotype and cognitive function: findings from the 1958 British birth cohort. Eur J Clin Nutr. 2015;69(4):505–508. | ||
Lee DM, Tajar A, Ulubaev A, et al; EMAS study group. Association between 25-hydroxyvitamin D levels and cognitive performance in middle-aged and older European men. J Neurol Neurosurg Psychiatry. 2009;80(7):722–729. | ||
Holick MF, Chen TC. Vitamin D deficiency: a worldwide problem with health consequences. Am J Clin Nutr. 2008;87(4):1080S–1086S. | ||
US Department of Agriculture; US Department of Health and Human Services. Dietary Guidelines for Americans, 2010. 7th ed. Washington, DC: US Government Printing Office; 2010. | ||
Centers for Disease Control and Prevention (CDC). Behavioral Risk Factor Surveillance System Survey Questionnaire. Atlanta, Georgia: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention; 2009. Available from http://www.cdc.gov/brfss/questionnaires/index.htm. | ||
Sabia S, Elbaz A, Britton A, et al. Alcohol consumption and cognitive decline in early old age. Neurology. 2014;82(4):332–339. | ||
Schneeweis N, Skirbekk V, Winter-Ebmer R. Does education improve cognitive performance four decades after school completion? Demography. 2014;51(2):619–643. | ||
Snowden M, Steinman L, Mochan K, et al. Effect of exercise on cognitive performance in community-dwelling older adults: review of intervention trials and recommendations for public health practice and research. J Am Geriatr Soc. 2011;59(4):704–716. | ||
Evans VC, Iverson GL, Yatham LN, Lam RW. The relationship between neurocognitive and psychosocial functioning in major depressive disorder: a systematic review. J Clin Psychiatry. 2014;75(12):1359–1370. | ||
Mahfoud Z, Kobeissi L, Peters TJ, Araya R, Ghantous Z, Khoury B. The Arabic validation of Hopkins Symptoms Checklist-25 against MINI in a disadvantaged suburb of Beirut, Lebanon. The International Journal of Educational and Psychological Assessment (TIJEPA). 2013;13(1):17–33. | ||
Mahfoud Z, Abdulrahim S, Taha MB, et al. Validation of the Arab Youth Mental Health scale as a screening tool for depression/anxiety in Lebanese children. Child Adolesc Psychiatry Ment Health. 2011;5:9. | ||
Derogatis LR, Lipman RS, Rickels K, Uhlenhuth EH, Covi L. The Hopkins Symptom Checklist (HSCL): a self-report symptom inventory. Behav Sci. 1974;19(1):1–15. | ||
Kleijn WC, Hovens JE, Rodenburg JJ. Posttraumatic stress symptoms in refugees: assessments with the Harvard Trauma Questionnaire and the Hopkins symptom Checklist-25 in different languages. Psychol Rep. 2001;88(2):527–532. | ||
Rahman TT, El Gaafary MM. Montreal Cognitive Assessment Arabic version: reliability and validity prevalence of mild cognitive impairment among elderly attending geriatric clubs in Cairo. Geriatr Gerontol Int. 2009;9(1):54–61. | ||
Fastenau PS, Denburg NL, Hufford BJ. Adult norms for the Rey-Osterrieth Complex Figure Test and for supplemental recognition and matching trials from the Extended Complex Figure Test. Clin Neuropsychol. 1999;13(1):30–47. | ||
Schwarz L, Penna S, Novack T. Factors contributing to performance on the Rey Complex Figure Test in individuals with traumatic brain injury. Clin Neuropsychol. 2009;23(2):255–267. | ||
Cores EV, Vanotti S, Eizaguirre B, et al. The effect of culture on two information-processing speed tests. Appl Neuropsychol Adult. 2015;22(4):241–245. | ||
Maalouf J, Nabulsi M, Vieth R, et al. Short- and long-term safety of weekly high-dose vitamin D3 supplementation in school children. J Clin Endocrinol Metab. 2008;93(7):2693–2701. | ||
Davis BJ, Vidal JS, Garcia M, et al. The alcohol paradox: light-to-moderate alcohol consumption, cognitive function, and brain volume. J Gerontol A Biol Sci Med Sci. 2014;69(12):1528–1535. | ||
Brouwer-Brolsma EM, de Groot LC. Vitamin D and cognition in older adults: an update of recent findings. Curr Opin Clin Nutr Metab Care. 2015;18(1):11–16. | ||
Etgen T, Sander D, Bickel H, Sander K, Förstl H. Vitamin D deficiency, cognitive impairment and dementia: a systematic review and meta-analysis. Dement Geriatr Cogn Disord. 2012;33(5):297–305. | ||
Wilson VK, Houston DK, Kilpatrick L, et al; Health, Aging and Body Composition Study. Relationship between 25-hydroxyvitamin D and cognitive function in older adults: the Health, Aging and Body Composition Study. J Am Geriatr Soc. 2014;62(4):636–641. | ||
Perna L, Mons U, Kliegel M, Brenner H. Serum 25-hydroxyvitamin D and cognitive decline: a longitudinal study among non-demented older adults. Dement Geriatr Cogn Disord. 2014;38(3–4):254–263. | ||
Hoteit M, Al-Shaar L, Yazbeck C, Bou Sleiman M, Ghalayini T, Fuleihan Gel-H. Hypovitaminosis D in a sunny country: time trends, predictors, and implications for practice guidelines. Metabolism. 2014;63(7):968–978. | ||
El-Hajj Fuleihan G, Bouillon R, Clarke B, et al. Serum 25-hydroxyvitamin D levels: variability, knowledge gaps, and the concept of a desirable range. J Bone Miner Res. 2015;30(7):1119–1133. | ||
Subirana-Mirete J, Bruna O, Virgili C, Signo S, Palma C. Processing speed in the aging process: screening criteria for the Spanish Quick Test of Cognitive Speed. Percept Mot Skills. 2014;119(2):417–429. | ||
Annweiler C, Maby E, Meyerber M, Beauchet O. Hypovitaminosis D and executive dysfunction in older adults with memory complaint: a memory clinic-based study. Dement Geriatr Cogn Disord. 2014;37(5–6):286–293. | ||
Caffarra P, Vezzadini G, Dieci F, Zonato F, Venneri A. Rey-Osterrieth complex figure: normative values in an Italian population sample. Neurol Sci. 2002;22:443–447. | ||
Nocentini U, Giordano A, Di Vincenzo S, Panella M, Pasqualetti P. The Symbol Digit Modalities Test - Oral version: Italian normative data. Functional Neurology. 2006;21(2):93–96. | ||
Nabulsi M, Mahfoud Z, Maalouf J, Arabi A, Fuleihan GE. Impact of maternal veiling during pregnancy and socioeconomic status on offspring’s musculoskeletal health. Osteoporos Int. 2008;19(3):295–302. | ||
Ganguli M, Vander Bilt J, Saxton JA, Shen C, Dodge HH. Alcohol consumption and cognitive function in late life: a longitudinal community study. Neurology. 2005;65(8):1210–1217. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.