Back to Journals » Therapeutics and Clinical Risk Management » Volume 11
Serous macular detachment, yellow macular deposits, and prominent middle limiting membrane in multiple myeloma
Authors Dogan B, Erol MK, Toslak D, Turgut Coban D, Bulut M, Cengiz A, Sogutlu Sari E
Received 13 December 2014
Accepted for publication 11 March 2015
Published 30 April 2015 Volume 2015:11 Pages 683—689
DOI https://doi.org/10.2147/TCRM.S79232
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Garry Walsh
Berna Dogan,1 Muhammet Kazim Erol,1 Devrim Toslak,1 Deniz Turgut Coban,1 Mehmet Bulut,1 Ayse Cengiz,1 Esin Sogutlu Sari2
1Antalya Training and Research Hospital, Eye Clinic, Antalya, Turkey; 2Balikesir University Medicine Faculty, Eye Clinic, Balikesir, Turkey
Abstract: Bone marrow-derived multiple myeloma is a type of plasma cell tumor that may be associated with ocular complications. A 52-year-old male patient was admitted to our eye clinic with the complaint of sudden visual loss and a visual acuity of 20/50 in the right eye and 20/800 in the left eye. Fundus examination revealed common flame-shaped hemorrhages, venous dilatation and tortuosity, Roth spots, serous macular detachment, and yellow macular deposits in both eyes. Evaluation with fundus fluorescein angiography, fundus autofluorescence, and spectral-domain optical coherence tomography resulted in suspicion of hyperviscosity retinopathy and referral to the hematology clinic. After hematology consultation confirmed a diagnosis of multiple myeloma, chemotherapy and plasmapheresis were initiated. Four months after presentation, best-corrected visual acuity was 20/20 in both eyes and improvement in hyperviscosity retinopathy, serous macular detachment, and yellow macular deposits was observed.
Keywords: serous macular detachment, yellow macular deposit, prominent middle limiting membrane, multiple myeloma
Introduction
Multiple myeloma is a malignant proliferation of plasma cells originating from a single clone. The characteristic signs of myeloma include bone pain, hypercalcemia, anemia, renal failure, and hyperviscosity syndrome.1
The retinal findings in hyperviscosity syndrome are common retinal hemorrhages, microaneurysms, venous dilatation and tortuosity, and white-centered hemorrhages.2 Serous macular detachment has been reported in a few cases in patients with multiple myeloma.3–5 Ho et al first reported serous macular detachment with or without subretinal precipitates or fundus signs of serum hyperviscosity in one patient with multiple myeloma, one patient with Waldenstrom’s macroglobulinemia, and one patient with benign polyclonal gammopathy.3 Rusu et al reported observation of associated serous detachment in four patients, three with multiple myeloma, and one with light-chain deposition disease.5
Here, we describe a rare case that presented as sudden vision loss and was diagnosed as multiple myeloma in which hyperviscosity retinopathy, serous macular detachment, yellow macular deposits, prominent middle limiting membrane (MLM), and Roth spots (white-centered hemorrhages) at the outer plexiform layer were evaluated by spectral-domain optical coherence tomography (SD-OCT), enhanced depth imaging OCT (EDI-OCT), fundus fluorescein angiography (FFA), and fundus autofluorescence.
Case report
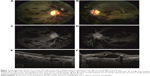
A 52-year-old male patient was admitted to our eye clinic with the complaint of sudden visual loss. Best-corrected visual acuity (BCVA) was 20/50 in the right eye and 20/800 in the left eye. Intraocular pressure, anterior segment examination, and the pupillary light reflex were normal. Fundus examination in both eyes revealed scattered flame-shaped hemorrhages, Roth spots, venous dilatation and tortuosity, and serous macular detachment (Figure 1A and B). FFA revealed scattered microaneurysms and hypofluorescent areas but no macula leakage (Figure 1C and D). SD-OCT revealed a central macular thickness of 329 μm in the right eye and 705 μm in the left eye, the presence of serous macular detachment (Figure 1E and F), small cystic spaces within the outer plexiform layer, and prominent MLM (Figure 1F). EDI-OCT revealed a choroidal thickness of 347 μm in the right eye and 421 μm in the left eye.
The patient was referred to the hematology clinic. The mean blood test results were hemoglobin (HGB) level 4.3 g/dL, hematocrit (HCT) level 13.3%, white blood cell count (WBC) 2.9×103/mm3, platelet (PLT) count 23.0×103/mm3, C-reactive protein (CRP) level 12.5 mg/L, immunoglobulin G (IgG) level 4,310 mg/dL, kappa light chain level 3.52 mg/dL, lambda light chain level 58 mg/dL, and micrototal protein 467.6 mg/dL. Serum and urine immunofixation electrophoresis revealed the presence of monoclonal IgG lambda and monoclonal lambda bands. After hematology consultation confirmed a diagnosis of multiple myeloma, chemotherapy and plasmapheresis were initiated.
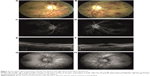
One month after presentation, BCVA was 20/32 in the right eye and 20/100 in the left eye. Fundus examination revealed a reduction in retinal hemorrhages and Roth spots and the presence of bilateral oval-to-round, yellow-to-orange deposits in the center of the macula (Figure 2A and B). FFA showed a decrease in microaneurysms and no macula leakage in both eyes (Figure 2C and D). SD-OCT revealed a central macula thickness of 299 μm in the right eye and 448 μm in the left eye, a decrease in serous macular detachment, and the presence of subretinal hyperreflective deposits in both eyes (Figure 2E and F). The yellow deposits observed in both eyes appeared hyperautofluorescent on fundus autofluorescence (Figure 2G and H). EDI-OCT revealed choroidal thickness of 298 μm in the right eye and 383 μm in the left eye.
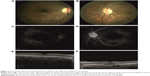
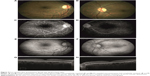
Two months after presentation, BCVA was 20/20 in the right eye and 20/32 in the left eye. Fundus examination revealed a complete reduction in retinal hemorrhages and Roth spots, a decrease in yellow macular deposits (Figure 3A and B), and complete resolution of microaneurysms in both eyes (Figure 3C and D). SD-OCT revealed central macular thickness of 268 μm in the right eye and 340 μm in the left eye, minimal serous macular detachment in the left eye, a decrease in subretinal hyperreflective deposits, and the complete disappearance of cysts in the outer plexiform layer (Figure 3E and F). EDI-OCT revealed choroidal thickness of 297 μm in the right eye and 399 μm in the left eye. Four months after presentation, BCVA was 20/20 in both eyes. Fundus examination revealed retinal hemorrhages (Figure 4A and B) and FFA revealed that microaneurysms (Figure 4C and D) had completely resolved. Fundus autofluorescence revealed that the appearance of hyperautofluorescent yellow deposits had decreased in the macula (Figure 4E and F). SD-OCT revealed central macular thickness to be 271 μm in the right eye and 231 μm in the left eye, and complete resolution of serous macular detachment in both eyes. EDI-OCT revealed choroidal thickness to be 301 μm in the right eye and 350 μm in the left eye. The final blood tests results were HGB level 10.9 g/dL, HCT level 30.9%, WBC 5.0×103/mm3, CRP level 3.45 mg/L, PLT count 130.0×103/mm3, and IgG level 4,310 mg/dL. The patient was followed up by both the hematology and eye clinics.
Conclusion
Multiple myeloma is a hematologic malignancy caused by infiltration of bone marrow by bone marrow-derived plasma cells followed by the production of monoclonal Ig by these cells.1 Symptomatic hyperviscosity is observed in 2%–6% of patients with multiple myeloma.6
Serous macular detachment with or without yellow deposits has been reported in a number of immunogammopathies.3 Multiple etiologies are associated with acquired yellow deposits, including conventional drusen, subretinal drusenoid deposits, cuticular drusen, some retinal dystrophies, tractional maculopathies,7 and central serous chorioretinopathy. Yellow macular deposits are usually hyperautofluorescent and relatively homogeneous, depending on their etiology. These deposits consist of debris containing lipofuscin, melanolipofuscin granules in macrophages, and extracellular material derived from photoreceptor outer segment discs that accumulate in the subretinal space from retinal pigment epithelium (RPE) dysfunction.8 The angiographically “silent macula” has been recognized as a hallmark of immunogammopathy-induced serous macular detachment. While angiographic leakage is usually not visible in patients with immunogammopathy-induced serous macular detachment,3 many other causes of serous macular detachment, such as central serous retinopathy, exudative age-related macular degeneration, hypertensive vascular disease, and infectious and inflammatory choroidopathy, demonstrate angiographic leakage. Ig accumulation in the subretinal space increases the oncotic pressure, while fluid accumulation in the subretinal area separates the RPE from the photoreceptors. This process leads outer segments containing fluorophores (A2E) to shed and accumulate in the subretinal space, resulting in the hyperautofluorescent signal present on fundus autofluorescence.5
Using SD-OCT in a patient with multiple myeloma, Priluck et al were the first to detect Roth spots in the outer plexiform layer of the retina.9 In our study, one of the very few studies that detected Roth spots by SD-OCT, SD-OCT revealed the presence of cysts in the outer plexiform layer at presentation. The SD-OCT also identified prominent MLM, which has not been reported before in multiple myeloma. Based on our literature review, this is the first description of prominent MLM as visualized by SD-OCT. A prominent MLM sign is a useful indicator of acute ischemic retinal damage.10 MLM has been identified by histologists but in actuality are not true membranes, they are only junctional systems. The inner third of the outer plexiform layer has a linear density where synaptic and desmosomal connections occur between the photoreceptor inner fibers and the processes of the bipolar cells. This linear density, which is also apparent with SD-OCT, is the junctional system that has been called the MLM, which tends to restrict the passage of fluids and exudates. The inner portion of the outer plexiform layer is also thought to landmark the outermost territory that normal retinal circulation can supply.11 A “prominent MLM sign” (a novel OCT finding for detecting retinal ischemia) has value especially for detecting vague acute ischemic changes.10 A prominent MLM in OCT was defined as a hyperreflective line located in the inner part of the outer plexiform layer.12 Electron microscopic study of experimental embolization of retinal vessels showed marked cytoplasmic swelling of bipolar cells sparing the rod and cone pedicles.13 Chu et al believe that the cytoplasmic swelling of these synaptic portions of the outer plexiform layer results in a hyperreflective line. The SD-OCT shows clearer distinction between ischemic and perfused areas. Detection of in vivo ischemic changes in occlusive retinal vascular disease may provide objective information in addition to fundus finding or invasive angiography.10 To date, two cases of serous retinal detachment with subretinal deposits have been reported in patients with multiple myeloma.3,4 Rusu et al reported acquired vitelliform macular detachment in three multiple myeloma patients.5 In our case, hyperviscosity retinopathy, prominent MLM, yellow macular deposits with serous macular detachment, and cysts in the outer plexiform layer were observed. FFA, SD-OCT, and fundus autofluorescence revealed rapid improvement in the retinal findings, visual acuity, and systemic symptoms with early diagnosis and treatment of multiple myeloma.
Eye symptoms and signs may be the earliest indications of multiple myeloma. If systemic treatment of multiple myeloma is not initiated early, retinal disease progresses and damage to pigment epithelium and likely to photoreceptor cells occurs with subsequent visual loss. Moreover, multiple myeloma and Waldenstrom’s macroglobulinemia should be considered in the differential diagnosis of pseudovitelliform lesions, prominent MLM, and serous macular detachment.5,14 In all patients diagnosed with multiple myeloma, routine ophthalmologic examination should be performed to detect ocular complications that may be the first signs of systemic disease.
Disclosure
The authors report no conflicts of interest in this work.
References
Knapp AJ, Gartner S, Henkind P. Multiple myeloma and its ocular manifestations. Surv Ophthalmol. 1987;31:343–351. | ||
Carr RE, Henkind P. Retinal findings associated with serum hyperviscosity. Am J Ophthalmol. 1963;56:23–31. | ||
Ho AC, Benson WE, Wong J. Unusual immunogammopathy maculopathy. Ophthalmology. 2000;107(6):1099–1103. | ||
Khan JM, McBain V, Santiago C, Lois N. Bilateral “vitelliform-like” macular lesions in a patient with multiple myeloma. BMJ Case Rep. 2010;2010:3049. | ||
Rusu IM, Mrejen S, Engelbert M, et al. Immunogammopathies and acquired vitelliform detachments: a report of four cases. Am J Ophthalmol. 2014;157:648–657. | ||
Mehta J, Singhal S. Hyperviscosity syndrome in plasma cell dyscrasias. Semin Thromb Hemost. 2003;29:467–471. | ||
Dupas B, Tadayoni R, Eriginay A, Massin P, Gaudric A. Subfoveal deposits secondary to idiopathic epiretinal membranes. Ophthalmology. 2009;116(9):1794–1798. | ||
Freund KB, Laud K, Lima LH. Acquired vitelliform lesions: correlation of clinical findings and multiple imaging analysis. Retina. 2011;31(1):13–25. | ||
Priluck JC, Chalam KV, Grover S. Spectral-domain optical coherence tomography of Roth spots in multiple myeloma. Eye. 2012;26(12):1588–1589. | ||
Chu YK, Hong YT, Byeon SH, Kwon OW. In vivo detection of acute ischemic damages in retinal arterial occlusion with optical coherence tomography: a “prominent middle limiting membrane sign”. Retina. 2013;33(10):2110–2117. | ||
Fine BS, Zimmerman LE. Muller’s cells and the “middle limiting membrane” of the human retina. An electron microscopic study. Invest Ophthalmol. 1962;1:304–326. | ||
Byeon SH, Kim M, Kwon OW. Ischemia. In: Bernardes R, Cunha-Vaz J, editors. Optical Coherence Tomography: A Clinical and Technical Update. Berlin/Heidelberg: Springer; 2012:23–40. | ||
Kroll AJ. Experimental central retinal artery occlusion. Arch Ophthalmol. 1968;79:453–469. | ||
Baker PS, Garg SJ, Fineman MS, et al. Serous macular detachment in Waldenström macroglobulinemia: a report of four cases. Am J Ophthalmol. 2013;155:448–455. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.