Back to Journals » Journal of Blood Medicine » Volume 5
Seroprevalence of human T-lymphotropic virus antibodies among patients with lymphoid malignancies at a tertiary center in Lagos, Nigeria
Authors Akinbami A, Durojaiye I, Dosunmu A, John-Olabode S , Adediran A, Oshinaike O, Uche E , Dada A, Odesanya M, Okunoye O
Received 18 May 2014
Accepted for publication 27 June 2014
Published 5 September 2014 Volume 2014:5 Pages 169—174
DOI https://doi.org/10.2147/JBM.S67912
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Akinsegun Akinbami,1 Idris Durojaiye,2 Adedoyin Dosunmu,1 Sarah John-Olabode,3 Adewumi Adediran,4 Olajumoke Oshinaike,5 Ebele Uche,1 Akinola Dada,5 Mojeed Odesanya,6 Olaitan Okunoye7
1Department of Haematology and Blood Transfusion, College of Medicine, Lagos State University, Lagos, Nigeria; 2Department of Haematology and Blood Transfusion, Lagos State University Teaching Hospital, Ikeja, Lagos, Nigeria; 3Department of Haematology and Immunology, Ben Carson School of Medicine, Babcock University, Ilisan-Remo, Ogun State, Nigeria; 4Department of Haematology and Blood Transfusion, Faculty of Clinical Sciences, College of Medicine, University of Lagos, Lagos, Nigeria; 5Department of Medicine, College of Medicine, Lagos State University, Lagos, Nigeria; 6Oak Hospitals, Ikorodu, Nigeria; 7Department of Medicine, University of Port Harcourt, Rivers, Nigeria
Background: There is a significant association of human T-lymphotropic viruses (HTLV) with lymphoid malignancies. HTLV causes a lymphoproliferative malignancy of CD4-activated cells called adult T-cell leukemia/lymphoma (ATL) and a chronic myelopathy called tropical spastic paraparesis/HTLV-1-associated myelopathy (TSP/HAM). This study aims to determine the prevalence of HTLV among patients with lymphoid malignancies at a tertiary center in Lagos.
Methods: A cross-sectional study was carried out at the hematology clinic of the Lagos State University Teaching Hospital. After obtaining consent, approximately 5 mL of venous blood was collected from each subject. The serum was separated and stored at –20°C. Sera were assayed for HTLV by an enzyme-linked immunoassay (ELISA) for the determination of antibodies to HTLV-1 and -2. Western blot confirmatory testing was done on reactive samples. All patients were also screened for human immunodeficiency virus (HIV), hepatitis B surface antigen (HBsAg) and hepatitis C virus (HCV) by rapid kits.
Results: A total of 39 patients with lymphoid malignancies were enrolled, consisting of 24 (61.5%) with solid malignancies, while 15 (38.5%) had leukemia. Only two patients (5.1%) with lymphoid malignancies were reactive on the ELISA test. On confirmatory testing with Western blot, two patients (5.1%) with lymphoid malignancies were also positive for HTLV. All patients were HIV negative, but four were positive to HBsAg and HCV. There was no association between history of previous blood transfusion and positivity to HTLV (P=0.544).
Conclusion: A prevalence of 5.1% of HTLV among patients with lymphoid malignancies was found in this study, and previous history of blood transfusion was not found to be a significant cause of HTLV infection.
Keywords: HTLV, lymphoid malignancies, ATL, ELISA, TSP/HAM
Introduction
The human T-lymphotropic viruses, type 1 (HTLV-1) and type 2 (HTLV-2), were the first human retroviruses discovered.1,2 They are single-stranded RNA retroviruses of the so-called C type originally described by Gallo’s group at the National Cancer Institute in 1980 and 1982, respectively.2,3 HTLV-1, the first human oncoretrovirus to be discovered,1 causes a lymphoproliferative malignancy of CD4-activated cells called adult T-cell leukemia/lymphoma (ATL) and a chronic myelopathy called tropical spastic paraparesis/HTLV-1 associated myelopathy (TSP/HAM).4 There is also a significant association between HTLV-1 with lymphoid malignancies.5
Infections
Infections of HTLV-1/2 are lifelong, with an asymptomatic carrier state.3 Over 20 million people are infected with HTLV-1/2 globally, with varying levels of seroprevalence reported in almost every region of the world.6 These retroviruses are found in foci of micro-endemicity, particularly in southern Japan,7 equatorial Africa,8,9 and parts of the Americas, including the Caribbean basin,10 and the Southeastern US.10 The frequency of antibodies in symptom-free adults throughout Sub-Saharan Africa has been reported to be from 3%–4%.11,12
Transmission
Transmission of HTLV-1 occurs from mother to child,13,14 by sexual contact,15 blood transfusion,16,17 and by sharing contaminated needles.16,18 Mother-to-child transmission occurs primarily by breast-feeding through ingestion of infected milk-borne lymphocytes.19 In HTLV-1-endemic areas, approximately 25% of breast-fed infants born to HTLV-1-seropositive mothers acquire infection.19 The transmission efficiency is dependent on the duration of breast-feeding and the presence of maternal antibodies to HTLV-1.20,21 The time of infant seroconversion typically ranges from 1–3 years of age.19,21 Intrauterine or perinatal transmission of HTLV-1 does occur, but it appears to be less frequent than transmission by breast-feeding; approximately 5% of children born to infected mothers but not breast-fed acquire infection.20
Sexual transmission of HTLV-1 is bidirectional.15,22 However, the frequency of HTLV-1 transmission is much higher from male to female than from female to male.22,23 The presence of genital ulcers increases the risk of virus transmission.23
Transmission of HTLV-1 by blood transfusion occurs with transfusion of cellular blood products (whole blood, red blood cells, and platelets) but not with the plasma fraction or plasma derivatives from HTLV-1-infected blood.17 Seroconversion rates of 44%–63% have been reported in recipients of HTLV-1-infected cellular components in HTLV-1 endemic areas.16,17 The probability of transmission by whole blood or packed red blood cells appears to diminish with greater duration of product storage; this finding has been ascribed to a depletion of infected cells, presumably T-lymphocytes.17,24 Sharing blood-contaminated needles is the likely mode of transmission among intravenous drug users (IDUs).25
Blood transfusion is a common occurrence amongst patients with lymphoid malignancies, whilst HTLV are transmitted through blood transfusion, screening for antibodies and discarding seropositive units should efficiently interrupt this transmission. Concern about HTLV-1 transmission through blood transfusion has led to the introduction of routine blood-donor screening for antibodies to HTLV-1 in developed countries.26,27 The decision to extend universal screening of blood donations to all industrialized countries with a low prevalence is a matter of debate and it has been suggested that the decision should be made country by country.28
The study aims to determine the prevalence of HTLV-1 among patients with lymphoid malignancies and ascertain the relationship between HTLV-1 infection, and previous history of blood transfusion.
Materials and methods
Patients
A cross-sectional study was carried out at the haematology clinic and medical wards of the Lagos State University Teaching Hospital, between February and May 2012. Routine donor blood screening in Lagos does not include testing for HTLV, because Lagos is not considered an endemic area for HTLV. Ethical clearance for this study was obtained from the Health Research and Ethics Committee of Lagos State University Teaching Hospital.
Adult patients with lymphoid malignancies including Hodgkin lymphoma, non-Hodgkin lymphoma, chronic lymphocytic leukemia, acute lymphoblastic leukemia and multiple myeloma were included in this study. Hemato-oncology patients with non-lymphoid malignancies were excluded from this study. Sample size was determined using the Yamane (Sloven)29 formula:
where n is the minimum sample size required, N is the population size; e is the degree of accuracy desired (ie, confidence interval) expressed as a decimal (±5% = ±0.05). Hematology department records of patients with lymphoid malignancies showed an average of 40 old and new patients were seen over a 3-month period. The minimum sample size can be calculated using N=40, and is shown below:
obtaining a minimum sample size of 36 patients with lymphoid malignancies.
Specimen collection and processing
After obtaining consent, approximately 5 mL of venous blood was collected from each subject and placed into a sterile bottle. The blood was allowed to clot completely before centrifugation. The serum was separated within 6 hours and stored in sterile cryovials at −20°C.
Serum samples were assayed for HTLV-1 using the HTLV-1 and -2 Ab ULTRA version (Diagnostic Bioprobes srl, Milan, Italy), an enzyme-linked immunoassay (ELISA) for the determination of antibodies to HTLV-1 and -2 in serum and plasma. Western blot confirmatory testing was done on reactive samples. Caliberation was performed according to manufacturers’ instruction.
Western blot confirmatory testing was carried out on reactive samples using the HTLV BLOT 2.4 (MP Diagnostics, Santa Ana, CA, USA), a qualitative ELISA for the in vitro detection of antibodies to HTLV-1 and HTLV-2 in human serum or plasma. It is intended for use as a supplemental antibody assay for characterizing human serum or plasma specimens found repeatedly reactive to HTLV-1/2 using screening procedures like the ELISA.
The sera of patients with lymphoid malignancies were also tested for the presence of antibodies to the human immunodeficiency virus (HIV) with the Alere Determine™ HIV-1/2 test kit, hepatitis B surface antigen (HBsAg) with the Micropoint™HBsAg Gold rapid screen test, and hepatitis C virus (HCV) with DiaSpot HCV test kit. They are rapid screening test kits for the qualitative detection of the respective antibodies in whole blood, plasma or serum.
Data analysis and presentation
The data were recorded in a compatible computer and analyzed with Epi-Info version 3.5.3 software. The mean, median, standard deviation and other statistical parameters were generated as necessary for continuous data. Tests of statistical significance between variables included chi-square analysis and Fisher’s exact test for discrete data. The level of significance was set at P<0.05.
Results
A total 39 patients with lymphoid malignancies were enrolled. Among the patients, 21 (53.8%) were males and 18 (46.2%) were females (Table 1). The mean age of the patients was 51.9±17.8 years. The mean age of the male patients was 51.3±19.2 years, and of the female patients was 52.6±16.6 years. Of the 39 patients with lymphoid malignancies, six (15.4%) were single, 27 (69.2%) were married, one (2.6%) was divorced or separated, while five (12.8%) were widowed (Table 2).
  | Table 1 Age and sex distribution of the subjects |
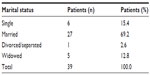  | Table 2 Marital status of the subjects |
Out of the 39 patients with lymphoid malignancies, 24 (61.5%) had solid malignancies while 15 (38.5%) had leukemias (Table 3). Of the 24 patients with solid malignancies, 18 (75%) had non-Hodgkin lymphoma, four (16.7%) had multiple myeloma, and two (8.3%) had Hodgkin lymphoma. Of the patients with leukemias, 13 (86.7%) had chronic lymphocytic leukemia, while (13.3%) had acute lymphoblastic leukemia.
  | Table 3 Diagnoses of patients with lymphoid malignancies |
Only two (5.1%) patients with lymphoid malignancies were reactive with an ELISA test. On confirmatory testing with Western blot, two (5.1%) patients with lymphoid malignancies were also positive for HTLV-1 (Table 4). One of the patients was a case of non-Hodgkin’s lymphoma, while the other was a case of chronic lymphocytic leukemia (Table 5).
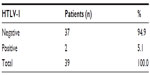  | Table 4 HTLV-1 status of the subjects |
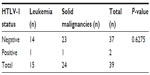  | Table 5 HTLV-1 status of the patients with lymphoid malignancies |
Of the 24 patients with solid malignancies, one (4.2%) was positive for HTLV-1, while 23 (95.8%) were negative for HTLV-1. Of the 15 patients with leukemia, one (6.7%) was positive for HTLV-1, while 14 (93.3%) were positive for HTLV-1 (Table 5).
Among the 39 patients with lymphoid malignancies 13 (33.3%) had history of previous blood transfusion, and 26 (66.7%) had no history of previous blood transfusion. Of the 13 patients with history of previous blood transfusion, none (0.0%) were positive for HTLV-1, while two (7.7%) of the 26 patients with no history of previous blood transfusion were HTLV-1 positive. Therefore there was no association between history of previous blood transfusion and positivity to HTLV-1 (P=0.544). Of the 13 patients with history of previous blood transfusion, two (15.4%) were HBsAg positive, while 11 (84.6%) were HBsAg negative. Among the 26 patients without history of previous blood transfusion, two (7.7%) were HBsAg positive, while 24 (92.3%) were HBsAg negative. There was no relationship between history of previous blood transfusion and HBsAg status (P=0.4073).
Of the 13 patients with history of previous blood transfusion, one (7.7%) was positive for HCV, while 12 (92.3%) were negative for HCV. Among the 26 patients without history of previous blood transfusion, one (3.8%) was positive for HCV, while 25 (96.2%) were negative for HCV. There was no relationship between history of previous blood transfusion and HCV status (P=0.5614).
All 39 patients with lymphoid malignancies were HIV negative. Among the 15 patients with leukemia, two (13.3%) were positive for HBsAg, while 13 (86.7%) were negative for HBsAg. Of the 24 patients with solid malignancies, two (8.3%) were positive for HBsAg, while 22 (91.7%) were negative for HBsAg (Table 6). There was no difference in HBsAg status between the patients with leukemia and the patients with solid malignancies (P=0.5017).
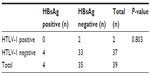  | Table 6 HTLV-1 and HBsAg status of patients with lymphoid malignancies |
Among the 15 patients with leukemia, one (6.7%) was positive for HCV, while 14 (93.3%) were negative for HCV. Of the 24 patients with solid malignancies, one (4.2%) was positive for HCV, while 23 (95.8%) were negative for HCV (Table 7). There was no difference in HCV status between the patients with leukemia and the patients with solid malignancies (P=0.6275).
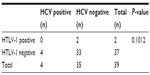  | Table 7 HTLV-1 and HCV status of patients with lymphoid malignancies |
Among the patients with lymphoid malignancies two (7.4%) of the 27 married patients were HTLV-1 positive. None of the single, divorced/separated or widowed patients were HTLV-1 positive. There was no association between marital status and HTLV-1 positivity (P=0.324).
Among the patients with lymphoid malignancies, two (5.1%) were HTLV-1 positive. One was aged 52 years old, and the other was 61 years old (P=1.000). Of the patients with lymphoid malignancies, two of the 21 males (9.5%) were positive for HTLV-1. No female was positive for HTLV-1. Sex differences were not statistically significant in this study (P=0.490).
Discussion
A seroprevalence of 5.1% of HTLV was obtained among patients with lymphoid malignancies. This could be compared with 0.5% seroprevalence of HTLV in the general population in Lagos, who had no lymphoid malignancies.29 Based on these results, this study suggests a significant association between HTLV with lymphoid malignancies. The seroprevalence of HTLV obtained in this study is higher than that found among similar patients in Lagos by Analo et al in 1998, in which none of the patients with chronic lymphocytic leukemia and non-Hodgkin’s lymphoma were found to be HTLV-1 positive.31 However, it is lower than the seroprevalence of 18.8% reported by Rastin et al in Iran.32 Geographical differences and more sensitive methods (ie, polymerase chain reaction [PCR] and nested reverse transcription [RT]-PCR) used in the Iranian study could account for a higher prevalence obtained.
In 2004, Adedayo and Shehu found much higher seroprevalence rates among patients with lymphoid malignancies in Dominica: 50% of Hodgkin lymphoma, 44.4% of non-Hodgkin lymphoma, and 37.5% of unclassified lymphomas were positive for HTLV-1.5
History of previous blood transfusion appeared to have no impact on HTLV-1 status in this study as the two patients that were positive for HTLV-1 among the patients with lymphoid malignancies had no past history of previous blood transfusion. Furthermore, none of the 13 patients with history of previous blood transfusion was positive for HTLV-1. None of the blood donors had been previously transfused. Although it was not statistically significant, Murphy et al reported past history of transfusion as an important risk factor for HTLV-1 seropositivity.32 The absence of positivity for HTLV-1 among the 13 patients that had been transfused in the past may be due to the fact that most transfusions in our center are administered with blood that had been stored in the refrigerator, which reduces the risk of transmission of HTLV-1 within days, due to death of HTLV-1 infected lymphocytes.34
History of previous blood transfusion among patients with lymphoid malignancies did not appear to have any impact on infection with other transfusion transmissible viruses as all patients were HIV negative. Seropositivity to HBsAg and HCV was also not significant.
Several studies have demonstrated higher HTLV-1 prevalence with increasing age,35,36 in this study, seropositivity to HTLV-1 tended to occur in older age groups as both HTLV-1 positive patients were aged 52 and 61 years old, although the mean age of study population was 51.9±17.8 years.
Two patients who tested positively for HTLV-1 were males. This is not in conformity with studies that have shown the frequency of HTLV-1 transmission to be much higher from male-to-female than from female-to-male.22,23 This is most likely due to the higher proportion of males compared with females in this study. This is another limitation of the study, as the sex of the participants was skewed unintentionally in favor of the male sex. However, sex differences were not statistically significant in this study (P=0.490).
In conclusion, a prevalence of 5.1% of HTLV was obtained among patients with lymphoid malignancies in this study, and a previous history of blood transfusion was not found to be a significant cause of HTLV-1 infection.
Disclosure
The authors have no conflicts of interest in this work.
References
Poiesz BJ, Ruscetti FW, Gazdar AF, Bunn PA, Minna JD, Gallo RC. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc Natl Acad Sci U S A. 1980;77(12):7415–7419. | |
Kalyanaraman VS, Sarngadharan MG, Robert-Guroff M, Miyoshi I, Golde D, Gallo RC. A new subtype of human T-cell leukemia virus (HTLV-II) associated with a T-cell variant of hairy cell leukemia. Science. 1982;218:571–573. | |
Fischer HE. Human T-Lymphotropic Virus Types I and II: Screening and Seroprevalence in Blood Donors. Current issues in Transfusion Medicine. 1995;3(4). | |
Gessain A, Barin F, Vernant JC, et al. Antibodies to human T-lymphotropic virus type-I in patients with tropical spastic paraparesis. Lancet. 1985;2:407–410. | |
Adedayo OA, Shehu SM. Human T-cell lymphotropic virus type 1 (HTLV-1) and lymphoid malignancies in Dominica: A seroprevalence study. Am J Hematol. 2004;77:336–339. | |
Williams CKO, Alabi GO, Junaid TA, et al. Human T cell leukaemia virus associated lymphoproliferative disease: report of two cases in Nigeria. Br Med J. 1984;288:1495–1496. | |
Clark JW, Robert-Guroff M, Ikehara O, Henzan E, Blattner WA. Human T-cell leukaemia-lymphoma virus type I and adult T-cell leukaemia-lymphoma in Okinawa. Cancer Research. 1985;45:2849–2852. | |
Wolfe ND, Heneine W, Carr JK, et al. Emergence of unique primate T-lymphotropic viruses among Central African bushmeat hunters. Proc Natl Acad Sci U S A. 2005;102:7994–7999. | |
Sarkodie F, Adarkwa M, Adu-Sarkodie Y, Candotti D, Acheampong JW, Allain JP. Screening for viral markers in volunteer and replacement blood donors in West Africa. Vox Sang. 2001;80:142–147. | |
Blattner WA, Kalyanaraman VS, Robert-Guroff M, et al. The human type C retrovirus, HTLV, in blacks from the Caribbean region, and relationship to adult T-cell leukaemia/lymphoma. Int J Cancer. 1982;30:257–264. | |
Hunsmann G, Bayer H, Schneider J, et al. Antibodies to ATLV/HTLV-1n Africa. Med Microbiol Immunol. 1984;173:167–170. | |
Fleming AF, Yamamoto N, Bhusnurmath SR, Maharajan R, Schneider J, Hunsmann G. Antibodies to ATLV (HTLV) in Nigerian blood donors and patients with chronic lymphatic leukaemia or lymphoma. Lancet. 1983;11:334–335. | |
Hino S, Yamaguchi K, Katamine S, et al. Mother-to-child transmission of human T-cell leukaemia virus type-I. Jpn J Cancer Res (Gann). 1985;76:474–80. | |
Ureta-Vidal A, Angelin-Duclos C, Tortevoye P, et al. Mother-to-child transmission of human T-cell-leukaemia/lymphoma virus type I: Implication of high antiviral antibody titer and high proviral load in carrier mothers. Int J Cancer. 1999;82(6):832–836. | |
Roucoux DF, Wang B, Smith D, et al. A prospective study of sexual transmission of human T lymphotropic virus (HTLV)-I and HTLV-2, J Infect Dis. 2005;191:1490–1497. | |
Okochi K, Sato H, Hinuma Y. A retrospective study on transmission of adult T cell leukaemia virus by blood transfusion: seroconversion in recipients. Vox Sanguinis. 1984;46:245–253. | |
Manns A, Wilks RJ, Murphy EL, et al. A prospective study of transmission by transfusion of HTLV-1 and risk factors associated with seroconversion. Int J Cancer. 1992;51:886–891. | |
Khabbaz RF, Onorato IM, Cannon RO, et al. Seroprevalence of HTLV-1 and HTLV-2 among intravenous drug users and persons in clinics for sexually transmitted diseases. N Engl J Med. 1992;326:375–380. | |
Kusuhara K, Sonoda S, Takahashi K, Tokugawa K, Fukushige J, Ueda K. Mother-to-child transmission of human T-cell leukaemia virus type-I (HTLV-1): a fifteen-year follow-up study in Okinawa, Japan. Int J Cancer. 1997;40:755–777. | |
Takahashi K, Takezaki T, Oki T, et al. Inhibitory effect of maternal antibody on mother-to-child transmission of human T-lymphotropic virus type I. Int J Cancer. 1991;49:673–677. | |
Nyambi P, Ville Y, Louwagie J, et al. Mother-to-child transmission of human T-cell lymphotropic virus types I and II (HTLV-1/2) in Gabon: a prospective follow-up of 4 years. J Acquir Immune Defic Syndr Hum Retrovirol. 1996;12:187–192. | |
Kajiyama W, Kashiwagi S, Ikematsu H, Hayashi J, Nomura H, Okochi K. Intrafamilial transmission of adult T cell leukaemia virus. J Infect Dis. 1986;154:851–857. | |
Murphy EL, Figueroa JP, Gibbs WN, et al. Sexual transmission of human T-lymphotropic virus type I (HTLV-1). Ann Intern Med. 1989;111:555–560. | |
Donegan E, Transfusion Safety Study (TSS) Group. Comparison of HTLV-1/2 with HIV-1 transmission by component type and shelf storage before administration. Transfusion. 1989;29:38S. | |
Feigal E, Murphy E, Vranizan K, et al. Human T-cell lymphotropic virus types I and II in intravenous drug users in San Francisco: risk factors associated with seropositivity. J Infect Dis. 1991;164:36–42. | |
Stigum H, Magnus P, Samdal HH, Nord E. Human T-cell lymphotrophic virus testing of blood donors in Norway: a cost–effect model. Int J Epidemiol. 2000;29:1076–1084. | |
Centers for Disease Control and Prevention (CDC). Licensure of screening test for antibody to HTLV-1. Morbidity and Mortality Weekly Report. 1988;37:736–747. | |
Anonymous. HTLV-1 – A screen too many? (Editorial). Lancet. 1990;336:1161. | |
Durojaiye I, Akinbami A, Dosunmu A, et al. Seroprevalence of human T lymphotropic virus antibodies among healthy blood donors at a tertiary centre in Lagos, Nigeria. The Pan African Medical Journal. 2014;17:301. | |
Analo HI, Akanmu AS, Akinsete I, Njoku OS, Okany CC. Seroprevalence of HTLV-1 and HIV infection in blood donors and patients with lymphoid malignancies. Central African Journal of Medicine. 1998;44(5):130–134. | |
Rastin M, Khoee AR, Tabasi N, et al. Evaluation of HTLV-I and HCV Prevalence In Non-Hodgkin’s lymphoma. Iran J Basic Med Sci. 2013;16:242–246. | |
Murphy EL, Wilks R, Hanchard B, et al. A case-control study of risk factors for seropositivity to human T-lymphotropic virus type I (HTLV-1) in Jamaica. Int J Epidemiol. 1996;25:1083–1089. | |
Donegan E, Busch MP, Galleshaw JA, Shaw GM, Mosley JW. Transfusion of blood components from a donor with human T-lymphotropic virus type II (HTLV-2) infection. The Transfusion Safety Study Group. Ann Intern Med. 1990;113:555–556. | |
Olaleye OD, Bernstein L, Sheng Z. Type specific immune response to HTLV-1/2 infections in Nigeria. American Journal of Tropical Medicine and Hygiene. 1994;50:479–486. | |
Olaleye DO, Omotade OO, Sheng Z, Adeyemo AA, Odaibo GN. HTLV types I and II infections in Mother-Child pairs in Nigeria. Journal of Tropical Pediatrics. 1999;45:66–70. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
