Back to Journals » Journal of Blood Medicine » Volume 6
Seroprevalence of human T-cell lymphoma/leukemia virus type-1 (HTLV-1) antibodies among blood donors at Enugu, Nigeria
Authors Okoye A, Ibegbulam OG, Onoh R, Ugwu NI, Anigbo CS, Nonyelu CE
Received 5 April 2014
Accepted for publication 24 May 2014
Published 19 January 2015 Volume 2015:6 Pages 31—36
DOI https://doi.org/10.2147/JBM.S65556
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Augustine Ejike Okoye,1 Obike Godswill Ibegbulam,2 Robinson Chukwudi Onoh,3 Ngozi Immaculata Ugwu,1 Chukwudi Simon Anigbo,2 Charles Emeka Nonyelu2
1Department of Haematology and Immunology, Federal Teaching Hospital Abakaliki, Ebonyi State, Nigeria; 2Department of Haematology and Immunology, University of Nigeria Teaching Hospital (UNTH) Ituku-Ozalla, Enugu State, Nigeria; 3Department of Obstetrics and Gynaecology, Federal Teaching Hospital Abakaliki, Ebonyi State, Nigeria
Background: Human T-cell lymphotrophic/leukemia virus (HTLV-1) is a retrovirus implicated in transfusion-transmitted infection.
Objective: The objective of this study was to determine the seroprevalence of HTLV-1 antibodies among blood donors at the University of Nigeria Teaching Hospital, Enugu, Eastern Nigeria.
Methods: A cross-sectional study was carried out on consented participants over 4 months. A total of 300 blood donors were recruited consecutively from the blood bank. The serum of the collected 5 mL of blood obtained from each participant was stored at -20°C until required for analysis. The serum samples were then analyzed for antibodies to HTLV-1 using a one-step incubation double-antigen sandwich ELISA (enzyme-linked immunosorbent assay) kit. Participants' demographic characteristics and degree of exposure to the risk factors associated with HTLV-1 infection were captured using a questionnaire. Statistical analysis of results was done using SPSS version 17.
Results: Of the 300 blood donors, 288 (96%) were male, while 12 (4%) were female. The average age of the blood donors was 26.85±8.52 years. The age group with the highest representation among the blood donors were those aged between 21 and 25 years. Only 22.3% of the blood donors were above 30 years. None of the 300 screened blood donors tested positive to HTLV-1 antibodies. Hence, the seroprevalence of HTLV-1 infection among blood donors was 0%. Of the blood donors, 5% had history of previous sexually transmitted disease, while 34.7% used condoms during sexual intercourse.
Conclusion: The seroprevalence obtained in this study cannot statistically support the justification of routine screening of blood donors for HTLV-1 infection. More prospective and multicentered studies are required to determine the infectivity of HTLV-1 in blood donors in Nigeria.
Keywords: retrovirus, transfusion, blood-borne infection, screening, Africa
Introduction
Human T-cell lymphotrophic/leukemia virus type 1 (HTLV-1) is one of the reemerging blood-borne infections. It is a member of the delta-retroviruses, which include HTLV-II, bovine leukemia virus and simian T-cell leukemia virus.1–3 It is an oncogenic human RNA virus that has a heterogeneous or restricted distribution with high endemicity in certain geographic areas and ethnic groups.1,4,5
Over 20 million persons are infected globally.5–7 Several studies, have reported high prevalence of HTLV-1 infection in Africa especially in Guinea, Nigeria, Cameroon, Democratic republic of Congo, and Ivory Coast.5,6 The seroprevalence of HTLV-1 antibodies in Africa ranges from 0.5% to 33% in the general population.1,6 Europe and the United States have very low seroprevalence rates of less than 1%.6
HTLV-1 is transmitted via infected lymphocytes by three main routes; namely; vertical transmission (transplacental and breast feeding), parenterally (via blood transfusion, needle sharing, and accidental pricks), and sexually (both heterosexual and homosexual).8,9 Its mode of transmission is similar to that of human immunodeficiency virus (HIV); hence, several European countries like France, United Kingdom, Holland, Sweden, and Netherlands have incorporated routine screening of donors for HTLV-1 antibodies in their blood-transfusion safety protocols.3,10,11
In Nigeria and most parts of Africa, routine screening and diagnosis of HTLV-1 infection among blood donors is rarely practiced, despite the fact that they constitute one of the areas of high to moderate endemicity.5
Fleming et al12 reported an overall prevalence of 3.7% in Zaria, Northern Nigeria. Analo et al13 reported a prevalence of 0.7% among blood donors in Lagos, south-western Nigeria.
In Norway, a seroprevalence of more than eight in 100,000 blood donors was used as the basis for routine screening of blood donors for HTLV-1 infection.14,15 However, Nigeria is yet to establish its own critical seroprevalence for HTLV-1 infection. In every endemic population, there is an increase of HTLV-1 seroprevalence with age, especially among women.16,17
HTLV-1 has been implicated in the pathogenesis of lymphoproliferative malignancy of CD4+ cells, known as adult T-cell lymphoma (ATL), and a chronic neurological disease, tropical spastic paralysis/HTLV-1-associated myelopathy.13,16 HTLV-1 infection has also been associated with a number of inflammatory diseases such as pediatric infectious dermatitis, uveitis, and some cases of myositis.18–21
Nigerian policy on blood transfusion emphasizes screening for the basic transfusion-transmitted infections like HIV, hepatitis B virus (HBV), hepatitis C virus (HCV), and syphilis, without HTLV-1 in nearly all the centers. The consequences of HTLV-1 infection are grave and the prognosis very poor.
Blood donors were chosen as the study group because they constitute a major source of contracting, spreading, and maintenance of HTLV-1 infection in the population. Hence prophylactic measures targeted against them will have a great strategic impact on curtailing the spread of HTLV-1. While prevalence studies on HTLV-1 infection have been documented in the North (Zaria), South-West (Ibadan), and South-South (Calabar), none have been published in South-East Nigeria. Most of the published HTLV-1 prevalence studies were conducted in the 1980s and 1990s, hence the need for more recent studies.
Findings from this study are expected to serve as a baseline for further research and as a reference material for comparative studies of the seroprevalences of HTLV-1 infection in different geopolitical zones of Nigeria.
Considering the importance of blood transfusion in clinical practice and the awareness of malignant diseases, this study is undertaken to shed more light on the magnitude of this reemerging transmissible viral infection by determining the seroprevalence of anti-HTLV-1 antibodies in blood donors in Enugu, Eastern Nigeria.
We therefore aim to determine the seroprevalence of anti-HTLV-1 antibody in blood donors and to identify the sociodemographic correlates of HTLV-1 infection, as well as to justify the inclusion of HTLV-1 antibody screening in blood transfusion safety protocols.
Materials and methods
A cross-sectional study was carried out among qualified blood donors negative for HIV, HBV, HCV, and syphilis at the University of Nigeria Teaching Hospital (UNTH), Ituku-Ozalla, Enugu State, Eastern Nigeria. The majority of the donors at the blood bank were family “replacement” donors.
Sample collection was done over a period of 4 months, and the minimum sample size required for this study was obtained using the following formula:
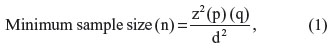
where z is the area under the curve =1.96, p is the prevalence of HTLV-1 antibodies in blood donors13 (7%), q=1 - p, and d is the standard error margin =0.05. Therefore,
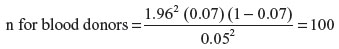
Statistical credence was given to the study by increasing the sample size to 300 blood donors. The recruited blood donors were clinically qualified and negative for routinely screened blood-borne infection markers.
Informed consent was obtained, and the participants were enrolled consecutively. This study was approved by the joint UNN (University of Nigeria, Nsukka)/UNTH Research Ethics Committee. Demographic data were obtained from each participant using a questionnaire. The inclusion criteria were blood donors who had given an informed consent and also satisfied all the requirements for blood donation.
Blood (5 mL) was collected from the antecubital fossa of each participant after cleaning with methylated spirits. The blood was transferred into a sterile plain bottle and allowed to clot. The serum was separated into another sterile plain bottle and stored immediately at −20°C, on the same day of collection, until the required sample size was obtained. Sample collection lasted for 4 months, from July to October 2010. The entire collected specimen was screened for anti-HTLV-1 antibody using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (HTLV 1 + 2 ELISA Kit, lot number 8196-12; Diagnostic Automation, Inc., Calabasas, CA, USA) based on one-step incubation, double-antigen sandwich principle. The sample that tested positive to HTLV-1 antibodies was then subjected to Western blotting (WB) to confirm that indeed it was HTLV-1 infection.
The ELISA was used to screen all the sera collected from the participants. The ELISA kit uses polystyrene microwell strips pre-coated with recombinant HTLV antigen. Criteria for reactive, nonreactive, and intermediate results were clearly stated in the manufacturer’s quality control ranges for the validation of results.
The manufacturer’s quality control range for validation of results are the optical density (OD) value of the blank well, which contains only chromogen and stop solution, and should be less than 0.080 at 450 nm. Also, the OD value of the positive control should be equal or greater than 0.800 at 450 nm after blanking, and the OD value of the negative control should be equal to or less than 0.100 at 450 nm after blanking.
Each microplate was considered separately when the results of the assay were calculated and interpreted. The results were calculated by relating each sample’s OD value to the cutoff value (CO) of the plate. Calculation of the CO was as follows:
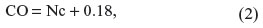
where Nc is the mean absorbance value for the three negative controls.
Negative results are samples with OD less than the CO, which indicates that no HTLV antibodies were detected. That is, S/CO <1, where S is the individual absorbance (OD) of each sample. Positive results are samples with OD greater than the CO, which indicates that HTLV antibodies were probably present. If the sample remained reactive after retesting in duplicate, then the sample should be considered positive for antibodies to HTLV. That is, S/CO ≥1. Samples with OD to CO ratio between 0.9 and 1.1 were considered borderline.
The WB technique is one way of confirming a serum as positive for HTLV-1 antibodies as well as separating HTLV-1 from HTLV-2 infections. This is imperative because the ELISA kit used in this study incorporated both HTLV-1 and HTLV-2 antigens on the microwell. WB is able to detect antibodies against the complex mixture of antigens found in HTLV infection. It is a highly specific technique.
There are criteria for confirmation of HTLV-1 infection by WB, but the Public Health Service Working Group criteria for confirmation of seropositivity is the most popular.
In view of the difficulty procuring the WB, the test was proposed to be done in the United Kingdom by Dr Emmanuel Nna of Safety Biomedical Ltd, Innovation Center, Crainfield, University Teaching Park, Bedfordshire. The kit used for the confirmation of the positive sample in this study was purchased from Thermo Fisher (Hampshire, UK). Electroblotting was to be done with Invitrogen™ 1-blot machine (Thermo Fisher). Monoclonal antibody of IgG1 isotype (MAB 811) specific for HTLV-1 core protein P19 was used to confirm that indeed the reactive sample was as a result of HTLV-1 infection.
The data collected were subjected to analysis by generation of frequency distribution data for the different variable responses. These generated data were displayed in tables. Data were processed using the Statistical Package for Social Science (SPSS) version 17.
Results
A total of 300 blood samples were screened for anti-HTLV antibodies. Most of the participants were male (288, 96%), and the majority of them were aged less than 30 years (244, 74.7%). The mean age of the donors was 26.85±8.52 years. Most of the subjects (270, 90%) were Igbos. The majority of the blood donors were single (230, 76.7%) and had secondary-school education or less (243, 80.9%). The most common accommodation system among blood donors (207, 69%) were two rooms or less. Half of the blood donors (150, 50%) were either students or unemployed (Table 1).
  | Table 1 Sociodemographic variables of participants |
Table 2 showed previous history of blood donation and transfusion. A total of 285 (95%) blood donors had donated blood previously, and 289 (96.3%) blood donors had not received blood transfusion.
  | Table 2 Frequency of blood donation and transfusion history |
The history of intravenous drug use and use of condoms is shown in Table 3. Approximately 88% had not injected themselves or shared instruments. Among the blood donors, 62.7% used condoms.
  | Table 3 History of intravenous drug use and use of condoms |
The majority of the blood donors (280, 93.3%) had not been treated or diagnosed with sexually transmitted diseases. Of the blood donors, 58% had just one sex partner. The other sex-related risk behaviors associated with transmission of HTLV-1 infection are represented in Table 4.
  | Table 4 History of sexual practices and STDs |
Of the participants, 300 were blood donors, and none of them were seropositive. Therefore, the prevalence of HTLV-1 among the blood donors in this study was zero (0%). The very low prevalence of HTLV-1 infection in the study population makes it difficult to statistically analyze its relationship with sociodemographic factors and subjects’ risk behaviors.
Discussions
Blood donors are sources of dissemination of HTLV-1 infection. Since HTLV-1 is blood borne, a blood donor, who tested negative for routinely screened transfusion-transmitted infections but is infected with HTLV-1 can transmit the virus to a recipient if not screened and excluded from donation.
This study group constitute a critical and strategic source of spreading HTLV-1 infection in the community. Preventive measures targeted at them, especially in countries with high prevalence will have a significant impact in the reduction of the spread of this deadly blood-borne virus.
In Nigeria, little or no attention has been paid to this viral infection since studies on HTLV-1 infection and its seroprevalence still remain few. The few available studies were done in Ibadan, Zaria, and Calabar more than a decade ago.12,22 This study appears to be the first of its kind in Eastern Nigeria. The seroprevalence of HTLV-1 infection among blood donors in this study was 0%. This value was lower than previous Nigerian reports.12,22
None of the 300 recruited blood donors tested positive to HTLV-1 infection in this study. The 0% prevalence rate, which is lower than the earlier Nigerian studies, can be criticized in that it may not be representative of the actual population prevalence on account of this being hospital-based data coupled with the fact that most participants were replacement blood donors. Also the distribution of HTLV-1 in the population may be restricted or heterogeneous, and thus cases of HTLV-1 infection may have been missed. The strength of this study, however, was that unlike some of the previous studies, the target population were blood donors who had been certified fit to donate blood for the blood-banking system.
A review of previous studies on the seroprevalences of HTLV-1 infection among blood donors in Nigeria showed a consistent decline. In 1983, Williams et al22 reported a seroprevalence of 7.0% among blood donors in Ibadan. Fleming et al12 in 1984 and 1986 reported prevalence of 3.7% in the general population and 0.7% among blood donors in Zaria and Calabar, respectively. Analo et al13 in 1998 reported a prevalence of 0.7%. This progressive decline in prevalence rate could be because of the improvement in the blood-donor screening for transfusion-transmitted infections since the advent of HIV and HCV. The low prevalence obtained in this study is comparable to the finding in Europe where HTLV-1 infection is not endemic. For instance, the prevalence of HTLV-1 infection among blood donors was 0.03% in the USA,14 0.011% in France,14 0.005% in Britain,15 and 0.002% in Norway.15 The finding of this study calls to question the assertion that HTLV-1 is endemic in tropical Africa, at least in Eastern Nigeria. However a multicenter population-based study would have to be conducted to determine the veracity of this finding.
Though some studies have shown that HTLV-1 infection is endemic in Africa,6,16 low prevalence rates have been recorded among the general population in various African countries. For instance, South Africa has a prevalence of 0%,13 Zimbabwe 0.11%,13 Senegal 1.2%,13 Namibia 1%,13 Mozambique 0.7%,17 and Congo 0.7%.17 In Europe, it ranges from 0% to 0.02%.6,16 Japan has a range of 0.08%–8.0%.14 In addition, genomic sequencing of HTLV-1 suggests that the virus originated from the Indo-Malay region and eventually spread to Africa, Japan, and Europe.12 This may help justify that indeed HTLV-1 infection is not endemic in many African countries.
It is also known that high prevalence of HTLV-1 infection has a direct influence on the incidence of ATL. For instance, in Japan with a prevalence of 3%, ATL accounts for 43% of all cases of Non-Hodgkin’s lymphoma.13 In Kyushu district of Japan, the prevalence is 15% and ATL accounts for 75% of Non-Hodgkin’s lymphoma.13 Despite the claim that HTLV-1 infection is endemic in Africa, there is little or no evidence to prove an increasing incidence of ATL in Nigeria. However, one cannot rule out under-diagnosis as a result of inadequate diagnostic tools and poor awareness.
Previous investigators have found that certain sociodemographic factors influence the seroprevalence of HTLV-1 infection. These factors include increasing age, sex, geographical location, marital status, poor socioeconomic status, recurrent sexually transmitted infection, and female sex.6 However, the low seroprevalence rates of 0% obtained among blood donors is not high enough to justify a statistical correlation between these risk factors and their effect on HTLV-1 infection. Such correlation may be possible if the sample size is increased.
Finally, this study recorded a low seroprevalence of HTLV-1 infection, but multicenter and large prospective studies are needed to conclusively establish the true seroprevalance of HTLV-1 among blood donors in Nigeria.
Conclusion
The seroprevalence of HTLV-1 infection among the blood donors who participated in this study was 0%. The low seroprevalence of HTLV-1 infection obtained in this study made it statistically difficult to establish a correlation between the effect of various sociodemographic variables and the prevalence of HTLV-1 infection.
Recommendations
Prospective and multicentered research on the seroprevalence of HTLV-1 infection among blood donors should be encouraged to help justify the need for routine screening for HTLV-1 infection for blood donors. A standard and simultaneous national epidemiological survey should be carried out in all the geopolitical zones of Nigeria to establish the true seroprevalence of HTLV-1 infection and its pattern of distribution. The reference data obtained should be periodically reviewed and updated. A national research center should be set up and adequately funded to coordinate and possibly subsidize the cost of important capital-intensive research. Payments and procurement of materials and kits required for such research should be facilitated by the national research center.
Acknowledgments
We wish to acknowledge the management of UNTH Enugu for providing the enabling environment for this study, and our gratitude goes to the departmental consultants and resident doctors who supported us in the course of this research. We are also grateful to Dr Emmanuel Nna of Safety Biomedical Ltd, Innovation Center, Crainfield, University Teaching Park, Bedfordshire.
Disclosure
The authors report no conflicts of interest in this work.
References
Plancouline S, Gessain A, Tortevoye P, Matsuda F, Abel L. A major susceptibility locus for HTLV-1 infection in childhood, maps to chromosome 6q27. Hum Mol Genet. 2006;15(22):3306–3312. | |
Poiez BJ, Ruscetti FW, Gazdar AF, Bunn PA, Gallo RC. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc Natl Acad Sci U S A. 1980;77:7415–7419. | |
Carlos T, Berta R, Antonio A, Estrella C, Rafael B, Garcia J. Clinical impact of HTLV-1 infection in Spain: implications for public health and mandatory screening. J AIDS. 2002;30(3):366–368. | |
Pagliuca A, Pawson R, Mufti GJ. HTLV-1 screening in Britain. BMJ. 1995;311:1313–1314. | |
Forbi JC, Odetunde AB. Human T-cell lymphotropic virus in a population of pregnant women and commercial sex workers in South Western Nigeria. Afr Health Sci. 2007;7(3):129–132. | |
Feigin RD, Cherry JD, editors. Textbook of Paediatric Infectious Diseases. 5th ed. Philadelphia: WB Sanders Company; 2004:2433–2434. | |
Seiki M, Hattori S, Hirayama Y, Yoshida M. Human adult T-cell leukemia virus: complete nucleotide sequence of the provirus genome integrated in leukemia cell DNA. Proc Natl Acad Sci U S A. 1983;80:3618–3622. | |
Donati M, Seyedzadeh H, Leung T, Blott M. Prevalence of antibody to human T-cell leukaemia/lymphoma virus in women attending antenatal clinic in Southeast London: retrospective study. BMJ. 2000;320(7227): 91–93. | |
Takezaki T, Tajima K, Ito M, et al. Short-term breast-feeding may reduce the risk of vertical transmission of HTLV-I. The Tsushima ATL Study Group. Leukemia. 1997;11(Suppl 3):60–62. | |
Soriano V, Gutierrez M, Vallejo A, Aguilera A, Pujol E. Epidemiology of HTLV-1 infection in Spain. Int J Epidemiol. 1996;8(1):443–448. | |
Courouce A, Phillonel J, Lemaire JM, Maniez M, Brunet JB. Seroepidemiology of HTLV-1/11 in universal screening of blood donations in France. AIDS. 1993;7:841–847. | |
Fleming AF, Yamamoto N, Bhusnurmath SR, Maharajan R, Schneider J, Hunsmann G. Antibodies to ATLV (HTLV) in Nigeria blood donors and patients with chronic lymphocytic leukaemia or lymphoma. Lancet. 1983;2(8345):334–335. | |
Analo HI, Akanmu AS, Akinsete I, Njoku OS, Okany CC. Seroprevalence of HTLV-1 and HIV infection in blood donors and patients with lymphoid malignancies. Cent Afri J Med. 1998;44(5):130–134. | |
Chaudhari CN, Shah T, Misra RN. Prevalence of human T-cell leukaemia virus amongst blood donors. Med J Armed Forces India. 2009;65:38–40. | |
Stigum H, Magnus P, Samdal HH, Nord E. Human T-cell lymphotropic virus testing of blood donors in Norway: a cost-effective model. Int J Epidemiol. 2000;29:1076–1084. | |
Kazanji M, Gessain A. Human T-cell lymphotropic virus types I and II (HTLV-I/II) in French Guiana: clinical and molecular epidemiology. Cad Saude Publica. 2003;19(5):1227–1240. | |
Kaplan JE, Khabbaz RF. The epidemiology of human T-lymphotropic virus type 1 and 11. Rev Med Virol. 1993;3:137–148. | |
Etenna SL, Caron M, Besson G, et al. New insights into prevalence, genetic diversity and proviral load of human T-cell leukaemia virus types 1 and 2, in pregnant women in Gabon in equatorial central Africa. J Clin Microbiol. 2008;48(11):3607–3614. | |
Ferreira OC, Planelles JD, Rusenblatt JD. Human T-cell leukaemia viruses: epidemiology, biology and pathogenesis. Blood Rev. 1997;11:91–104. | |
Uchiyama T, Yodoi J, Sagawa K, Takatsuki K, Uchino H. Adult T-cell leukemia: clinical and hematologic features of 16 cases. Blood. 1977;50:481–492. | |
Matsuoka M. Human T-cell leukemia virus type I (HTLV-I) infection and the onset of adult T-cell leukemia (ATL). Retrovirology. 2005;2:27. | |
Williams CK, Johnson AO, Blattner WA. Human T-cell leukaemia virus in Africa: possible roles in health and disease. IARC Sci Publ. 1984;(63):712–726. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
