Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
Sensitive electrochemical immunosensor based on three-dimensional nanostructure gold electrode
Authors Zhong G, Lan R, Zhang W, Fu F, Sun Y, Peng H, Chen T, Cai Y, Liu A , Lin J, Lin X, Bai F
Received 22 October 2014
Accepted for publication 14 January 2015
Published 19 March 2015 Volume 2015:10(1) Pages 2219—2228
DOI https://doi.org/10.2147/IJN.S76200
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Carlos Rinaldi
Guangxian Zhong,1,2,* Ruilong Lan,3,* Wenxin Zhang,1,4 Feihuan Fu,5 Yiming Sun,1,4 Huaping Peng,1,4 Tianbin Chen,3 Yishan Cai,6 Ailin Liu,1,4 Jianhua Lin,2 Xinhua Lin1,4
1Department of Pharmaceutical Analysis, Faculty of Pharmacy, Fujian Medical University, 2Department of Orthopaedics, 3The Centralab, First Affiliated Hospital of Fujian Medical University, 4Nano Medical Technology Research Institute, Fujian Medical University, Fuzhou, 5Department of Endocrinology, The County Hospital of Anxi, Anxi, 6Fujian International Travel Healthcare Center, Fujian Entry-Exit Inspection and Quarantine Bureau, Fuzhou, People’s Republic of China
*These authors contributed equally to this work
Abstract: A sensitive electrochemical immunosensor was developed for detection of alpha-fetoprotein (AFP) based on a three-dimensional nanostructure gold electrode using a facile, rapid, “green” square-wave oxidation-reduction cycle technique. The resulting three-dimensional gold nanocomposites were characterized by scanning electron microscopy and cyclic voltammetry. A “sandwich-type” detection strategy using an electrochemical immunosensor was employed. Under optimal conditions, a good linear relationship between the current response signal and the AFP concentrations was observed in the range of 10–50 ng/mL with a detection limit of 3 pg/mL. This new immunosensor showed a fast amperometric response and high sensitivity and selectivity. It was successfully used to determine AFP in a human serum sample with a relative standard deviation of <5% (n=5). The proposed immunosensor represents a significant step toward practical application in clinical diagnosis and monitoring of prognosis.
Keywords: electrochemical immunosensors, three-dimensional nanostructure gold electrode, square-wave oxidation-reduction cycle, alpha-fetoprotein
Introduction
Immunoassays based on specific recognition between an antigen and an antibody have been used successfully in a number of fields, including medical detection, processing quality control, and environmental monitoring.1–3 Traditional immunoassay methods have included the radioimmunoassay, enzyme-linked immunosorbent assay, and electrochemiluminescence immunoassay. In spite of their good sensitivity, the radioimmunoassay significantly increases the radioactivity hazard for laboratory workers and the enzyme-linked immunosorbent assay is tedious and time-consuming. Newer methods, including surface plasmon resonance,4 quartz crystal microbalance,5 and optical6 and electrochemical methods,7 have attracted considerable interest for immunoassays due to their rapid recognition and simple fabrication. Among these methods, the electrochemical immunoassay has received much attention in view of its high sensitivity and low cost.7,8
The crucial step in construction of an electrochemical immunosensor is efficient immobilization of biomolecules onto the electrode surface.9 New opportunities for fabrication of highly sensitive electrochemical immunosensors have been opened up by the advent of nanomaterials and nanotechnologies.10 Advances in biomolecular detection have included use of nanomaterials, such as nanotubes, nanoparticles, and nanowires, as a medium of signal amplification.11–13 In recent years, due to its interesting properties, including a large surface area, superior conductivity, biocompatibility, and high stability, nanoporous gold has attracted considerable attention. Several methods, including electrochemical deposition,14 electrochemical erosion,15 “direct freezing”,16 a voltage-induced dimension change method,17 a “dealloying process”,18–20 template synthesis,21,22 and repetitive square-wave oxidation- reduction cycle (SWORC)23 were been used to prepare a three-dimensional gold (3D-Au) nanostructure. Due to the time-saving, easy, and controllable preparation of nanoporous gold, SWORC is very attractive among these methods.24,25
Alpha-fetoprotein (AFP) is normally synthesized by the liver, yolk sac, and gastrointestinal tract in the human fetus. Plasma levels of AFP decrease rapidly after birth and reach normal adult levels by the age of 8–12 months. Elevated serum AFP levels in adults are significant in the diagnosis of ataxia-telangiectasia syndrome, hereditary tyrosinemia, cirrhosis, alcoholic hepatitis, hepatocellular carcinoma, and viral hepatitis, and are also useful for monitoring the effectiveness of surgical and chemotherapeutic management of hepatomas and germ cell neoplasms.8 AFP testing is currently performed using the radioimmunoassay and enzyme-linked immunosorbent assay.26,27 However, there are some limitations, such as radiation hazards and a long analysis time. Therefore, it is important to explore new routes for detection of AFP.
In the present study, a facile electrochemical immunosensor based on a 3D-Au electrode was developed for detection of AFP. The 3D-Au electrode was prepared by a simple, rapid, and “green” SWORC approach. The 3D-Au electrode was then used as a substrate for construction of a “sandwich-type” AFP immunosensor. This new immunosensor showed a fast amperometric response, high sensitivity, and selectivity, and was used to assay of AFP in a real serum sample with a satisfactory result. The proposed immunosensor represents a significant step towards practical application in clinical diagnosis and prognostication.
Materials and methods
Chemicals and materials
3-mercaptopropionic acid, nitric acid, sulfuric acid, potassium permanganate, ethanol, N-hydroxysuccinimide, and 1-ethyl-3-(3-dimethylamino propyl) carbodiimide hydrochloride (EDC) were provided by Sinopharm Chemical Reagent Co Ltd (Shanghai, People’s Republic of China). Bovine serum albumin was sourced from Sigma-Aldrich (St Louis, MO, USA). 3,3′,5,5′-tetramethylbenzidine (TMB) substrate (K-blue low activity substrate) was purchased from Neogen (Lansing, MI, USA). Human serum samples were obtained from the First Affiliated Hospital of Fujian Medical University. All chemicals were of analytical reagent grade and were used without further purification. All solutions were prepared with Milli Q water (18 MΩ·cm resistivity) from a Millipore system (Billerica, MA, USA).
Mouse anti-AFP monoclonal antibody (8.556 mg/mL) and horseradish peroxidase (HRP)-conjugated mouse anti-AFP monoclonal antibody (13.34 mg/mL), AFP standard solution, and cancer antigen 125, 153, and 199 standard solutions were from Zhengzhou Biocell Biotechnology Co Ltd (Zhengzhou, People’s Republic of China). The antibody dilution was 10 mM phosphate-buffered saline (PBS) containing 0.08 M NaCl (pH 7.4). The antibodies were aliquoted and stored at −20°C. The purified antibodies were diluted upon use in PBS suitable for the immobilizing and capture reactions.
Apparatus
All electrochemical measurements were performed on a CHI 760D Electrochemical Workstation (CH Instruments, Austin, TX, USA). Electrochemical experiments were carried out with a conventional three-electrode system comprising a 3D-Au working electrode, an Ag/AgCl (with saturated KCl) reference electrode, and a platinum wire auxiliary electrode. Cyclic voltammetry was carried out at a scan rate of 100 mV per second. Amperometric detection was performed at a fixed potential of 100 mV. Steady-state currents were recorded within 100 seconds. Scanning electron microscopic images were obtained using an S-4800 instrument (Hitachi, Tokyo, Japan). Atomic force microscopic images were acquired using an SPM-Multimode 8 system (Bruker, Santa Barbara, CA, USA) with Nanoscope Analysis version 1.40.0.0 software. All the images presented in this paper were acquired using ScanAsyst mode under ambient conditions.
Preparation of 3D-Au electrode
The method used to prepare the 3D-Au electrode was the same as that described in our previous reports.24,25 Briefly, before each experiment, a gold electrode (2 mm in diameter) was polished with 1.0, 0.3, and 0.05 μm alumina powder. The gold electrode was then cleaned ultrasonically sequentially and thoroughly in ethanol and deionized water. The nanoporous gold electrode was electrochemically cleaned in fresh 0.5 M H2SO4 solution by potential scanning between −0.35 V and 1.5 V until a cyclic reproducible voltammogram was obtained to remove any possible impurities on the surface28 and the electrode was rinsed with doubly distilled water, then the electroabsorption charge of the O adatoms between the rough gold electrode and the flat gold electrode was checked.
Fabrication of immunosensor
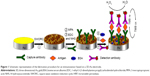
The cleaned 3D-Au electrode was first immersed in an aqueous solution of 5 mM 3-mercaptopropionic acid for 24 hours at room temperature. After thorough rinsing with deionized water to remove the physically adsorbed 3-mercaptopropionic acid, the nanoporous gold electrode was immersed in 10 mM PBS (pH 7.4) with 20 mg/mL of EDC and 10 mg/mL of N-hydroxysuccinimide for 40 minutes. The activated 3D-Au electrode was thoroughly rinsed with 10 mM PBS (pH 7.4). Next, 5 μL of capture antibody (mouse monoclonal antibody directed against AFP, 0.5 mg/mL solution) was dropped onto the electrode surface and incubated overnight to yield sensing interfaces. A blocking treatment aimed at preventing nonspecific responses during incubation of the sample was carried out by casting 10 μL of 5% (v/w) bovine serum albumin at room temperature for 1 hour, followed by careful washing in 10 mM PBS (pH 7.4). Thereafter, 5 μL of AFP antigen solution at different concentrations were added onto the electrode surface and incubated for 1 hour, and the modified electrode was then rinsed extensively to remove unbound antigen molecules. Next, 5 μL of detection antibody (HRP-conjugated mouse monoclonal antibody directed against AFP, 0.02 mg/mL) was dropped onto the modified electrode surface and incubated for 1 hour, after which the electrode was washed and ready for measurement. A schematic illustration of the stepwise preparation of the immunosensor is shown in Figure 1.
Electrochemical measurement
Electrochemical measurements, including cyclic voltammetry and amperometric i-t curve studies, were performed using a commercial TMB substrate solution containing a certain percentage of H2O2. Cyclic voltammetry was carried out at a scan rate of 100 mV per second. Amperometric detection was performed at 100 mV. Steady-state current was usually reached within 100 seconds. The data for the condition, optimization, and calibration curves was the average of three measurements.
Results and discussion
Preparation of 3D-Au electrode
Figure 2A and B shows scanning electron microscopic images of the same gold electrode before (Figure 2A) and after (Figure 2B) pretreatment with SWORC at a frequency of 8,000 Hz for 5 minutes. As shown in Figure 2B, the 3D-Au electrode surface was divided into two layers. The bottom layer was a brain-like nanoporous structure revealing the presence of small grains and channels about 120 nm in width, and the upper layer was a cotton wool-like nanostructure. The brain-like structure shown in the scanning electron microscopic images had been reported previously as being characteristic of the gold overlayers resulting from the electroreduction process.29 The atomic force microscopic images (Figure 2C and D) also revealed a difference between the surface structure of flat gold and that of 3D-Au. The surface of 3D-Au showed a rounded cap columnar-type structure composed of many grains. It should be noted that some large grains (120–150 nm) were formed by small subunits (40–60 nm). Some channels surrounded by a grain-like structure can also be observed in Figure 2D, and the width of channels was about 120 nm.
The 3D-Au electrode was electrochemically characterized in 0.5 M H2SO4 at a scan rate of 100 mV per seconds (Figure 2E, curve b). For comparison with the nanoporous gold electrode, the voltammogram for a bare flat gold electrode with the same geometric surface area is also presented (Figure 2E, curve a). The real surface area of the 3D-Au electrode was approximately 31 times that of the flat gold electrode, assuming that the reduction of the monolayer of gold oxide requires 386 μC/cm2, and the roughness factor (ratio of the real surface area to the geometric surface area) is about 114.21
Electrochemical responses of immunosensor based on 3D-Au electrode
Figure 3 shows the i-t curve for the capture antibody or capture antibody/antigen/detection antibody sandwich-modified biosensor at different electrodes in TMB solution. As shown in Figure 3, curve b was the i-t curve for the capture antibody-modified 3D-Au electrode after the antigen and detection antibody were immobilized on the surface of the 3D-Au electrode by immunoreaction. The current value (curve d) increased significantly, and the difference in current value between curve d and curve b was 3.07 μA. This indicates that the sandwich structure was formed upon immunoreaction, and the HRP-conjugated detection antibody was bound onto the 3D-Au electrode surface to produce the current signal. Notably, because of the site of HRP redox, which was shielded within insulating peptide backbones, the site of HRP redox did not directly exchange electrons with the electrode.30,31 Therefore, small redox molecule TMB can easily diffuse in and out of the redox site of HRP as an electron shuttle, and coupling with the redox reaction of TMB, the catalytic reduction of H2O2 occurs on the electrode.30,31 Thus, the HRP modified on the surface of 3D-Au electrode could efficiently catalyze reduction reactions of H2O2 with the redox reaction of TMB, leading to significant amplification of current.31,32 However, due to the absence of the detection antibody, this catalytic reduction current on the capture antibody-modified 3D-Au electrode was not obvious. The phenomenon at the flat gold electrode surface (Figure 3, curves a and c) was also similar, but the current signals decreased significantly and the difference in current value between the two curves was only 0.63 μA. Compared with the flat gold electrode, the current difference of the 3D-Au electrode was about 4.9 times higher, demonstrating that the 3D-Au-based biosensor could significantly improve the electrocatalytic activity.
Optimization of experimental conditions
The concentration of solutions used for incubation of the capture antibody and detection antibody dramatically influenced the performance of the immunosensors. On the basis of our preliminary experiments, we set three concentrations of the solutions used for incubation of both antibodies by evaluating its effect on the blank signal. The concentration levels explored were 0.1, 0.5, and 1.0 mg/mL for the capture antibody and 0.01, 0.05, and 0.1 mg/mL for the detection antibody. As shown in Figure S1A, when the concentration of the detection antibody was fixed at 0.02 mg/mL and 0.05 mg/mL, respectively, the blank signal decreased significantly with increasing concentration of the solution used for incubation of the capture antibody, whereas an opposite trend was observed for the detection antibody when concentrations of the capture antibody were fixed at 0.5 mg/mL and 1.0 mg/mL, respectively. This phenomenon could be explained by the fact that a high concentration of the capture antibody solution allowed complete and regular functionalization of the nanoporous gold substrate, thus preventing nonspecific binding of detection antibody. Conversely, an excessive concentration of the solution used for incubation of the detection antibody could promote its nonspecific binding. Further experiments carried out by incubating the sensors with blank solution and with 10 ng/mL of AFP solution indicated that the signal arising from the AFP solution did not significantly decrease, even when the concentration of the detection solution was reduced to 0.02 mg/mL. On the basis of these results, we fixed the concentrations of the incubating solution at 0.5 and 0.02 mg/mL for the capture and detection antibodies, respectively.
The effects of incubation temperature and incubation time on the current value for antibody and antigen reaction were also studied, as shown in Figure S1B and C, respectively. The results indicate that the optimal reaction temperature was 37°C and the optimal reaction time was 60 minutes. As a result, an incubation temperature of 37°C, a reaction time of 60 minutes, and incubating solution concentrations of 0.5 mg/mL for the capture antibody and 0.02 mg/mL for the detection antibody were selected for immunoassay of the AFP antigen.
Specificity of immunosensor
Specificity is an important parameter that influences the performance of an immunosensor in practical application. To evaluate the specificity of the presented immunosensor toward the target AFP analyte, the system was challenged with other biomarkers, including cancer antigens 153, 199, and 125. Figure 4 showed the histograms corresponding to the current intensity arising from immunoreaction of the AFP antibody with different proteins. As indicated in Figure 4, the amperometric current intensity for the target AFP increased markedly after immunoreaction with AFP antigen as compared with the other antigens and the blank solution. Meanwhile, the presence of sample matrix components did not obviously change the current intensity of the developed immunosensor toward the target AFP. The results clearly indicate the high specificity of the developed immunosensor.
Sensitivity of fabricated immunosensor
In order to examine the sensitivity of the 3D-Au-based biosensor, amperometric signals were measured before and after immunoreaction with the AFP antigen at different concentrations. Figure 5A shows an increase in the currents with increasing AFP antigen concentration in the range of 1–50 ng/mL. Under optimal conditions, a good linear relationship was observed between the current signal and the logarithmic value of the antigen concentration (shown in Figure 5B) in the range of 10–50 ng/mL. The linear regression equation was I(μA) =0.9421+0.4138lgCAFP(ng mL−1) with a correlation coefficient 0.9950. The lowest detection limit of 3 pg/mL AFP antigen could be estimated using 3σ (where σ is the standard deviation of the blank solution, n=5). According to the linear equation, the concentration of AFP antigen could be detected quantitatively, and lower serum AFP levels could also be detected by appropriate dilution with pH 7.4 PBS.
To further elucidate the advantage of the developed electrochemical immunosensor, the sensitivity and linear range of the immunosensor were compared with other AFP electrochemical immunosensors.22,33–38 As shown in Table 1, the sensitivity and linear range of the electrochemical immunosensor are comparable. Moreover, preparation of the 3D-Au electrode is more simple, more economical, and greener than the other nanocomposites.
  | Table 1 Comparison of different electrochemical immunosensors |
Precision, reproducibility, and stability of AFP immunosensor
The precision of the electrochemical immunosensors was evaluated by calculating the intra-assay and interassay variation coefficients. The intra-assay precision of the immunosensor was evaluated by assaying one AFP level for three repeated measurements, whereas the interassay precision was estimated by measuring one AFP level with three immunosensors prepared independently under the same experimental conditions. The intra-assay and interassay variation coefficients obtained from 10 ng/mL AFP were 4.56% and 6.84%, respectively, indicating acceptable precision and reproducibility. Further, the stability of the electrochemical immunosensor was evaluated. The immunosensors were stored at 4°C. The amperometric currents were measured every 3–5 days using the same immunosensors. The currents were 97.3%, 94.9%, and 91.8% of the initial current at days 10, 20, and 30. The immunosensor was regenerated by rinsing with 0.1 M glycine-HCl (pH 2.2) stripping buffer for 3 minutes to dissociate the antigen-antibody complex.39 The renewed immunosensor could restore 96% of the initial value after five assay runs, indicating high reusability and stability.
Application of immunosensor for human AFP levels
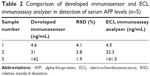
The AFP levels obtained in a human serum sample using the proposed immunosensor are shown in Table 2, which compares the results with those obtained from the Roche electrochemiluminescence immunoassay analyzer performed by the First Affiliated Hospital of Fujian Medical University. As shown in Table 2, the results obtained with the immunosensor are in good agreement with those measured by the electrochemiluminescence immunoassay analyzer. The relative standard deviation less than 5% in five experiments were in the acceptable range. Thus, this method has potential application for detection of AFP levels in clinical samples.
Conclusion
In this work, an electrochemical biosensor was developed based on a 3D-Au electrode, and was prepared by a simple, economical, and controllable approach known as SWORC. Due to the superior conductivity and large surface area of the 3D-Au electrode, the developed immunosensor showed excellent sensitivity and selectivity for detection of AFP antigens. Compared with other electrochemical immunosensors,22,33–38 the selectivity, sensitivity, and linear range of the developed immunosensor is competitive. Further, the procedure used to prepare the 3D-Au electrode is more simple, economical, and greener than the alternative nanocomposites. This new immunosensor was also used to assay AFP levels in a human serum sample and satisfactory results were obtained. This proposed method is stable, versatile, and highly sensitive, and should have wide application in the diagnosis and monitoring of cancers or other immunoassay for non-tumor biomarkers such as immunoglobulin, glycosylated hemoglobin, and troponin.
Acknowledgments
The authors gratefully acknowledge the financial support of the National High Technology and Development of China (2012AA022604), the National Natural Science Foundation of China (21275028, 81301520), the Natural Science Foundation of Fujian Province (2014J05088), the rolling subsidy scheme of Outstanding Youth Science Fund Project of Fujian province (2014J07009), the Scientific Research Major Program of Fujian Medical University (09ZD013), the Research Fund for the Doctoral Program of Higher Education of China (20123518110001), the Fujian Provincial Important Science and Technology Foundation (2011R1007-2) sponsored by the Medical Elite Cultivation Program of Fujian, People’s Republic of China (2013-ZQN-JC-5), the Science and Technology Plan Project, General Administration of Quality Supervision (2014IK060), the Foundation of Fujian Provincial Department of Education (JA11110, JA12130), and the Key Clinical Specialty Discipline Construction Program of Fujian, People’s Republic of China.
Disclosure
The authors report no conflicts of interest in this work.
References
Liu G, Wang S, Liu J, Song D. An electrochemical immunosensor based on chemical assembly of vertically aligned carbon nanotubes on carbon substrates for direct detection of the pesticide endosulfan in environmental water. Anal Chem. 2012;84(9):3921–3928. | ||
Liu J, Zanardi S, Powers S, Suman M. Development and practical application in the cereal food industry of a rapid and quantitative lateral flow immunoassay for deoxynivalenol. Food Control. 2012;26(1):88–91. | ||
Al-Khafaji QAM, Harris M, Tombelli S, et al. An electrochemical immunoassay for HER2 detection. Electroanalysis. 2012;24(4):735–742. | ||
Uludag Y, Tothill IE. Cancer biomarker detection in serum samples using surface plasmon resonance and quartz crystal microbalance sensors with nanoparticle signal amplification. Anal Chem. 2012;84(14):5898–5904. | ||
Kim YJ, Rahman MM, Lee JJ. Ultrasensitive and label-free detection of annexin A3 based on quartz crystal microbalance. Sens Actuators B Chem. 2013;177:172–177. | ||
Wenger B, Kugelbrey K, Gao H, Sigrist H, Voirin G. Au-labeled antibodies to enhance the sensitivity of a refractometric immunoassay: detection of cocaine. Biosens Bioelectron. 2012;34(1):94–99. | ||
Zhang Y, Dai WJ, Liu F, et al. Ultrasensitive electrochemiluminescent immunosensor based on dual signal amplification strategy of gold nanoparticles-dotted graphene composites and CdTe quantum dots coated silica nanoparticles. Anal Bioanal Chem. 2013;405(14):4921–4929. | ||
Liu L, Xing Y, Zhang H, Liu R, Liu H, Xia N. Amplified voltammetric detection of glycoproteins using 4-mercaptophenylboronic acid/biotin-modified multifunctional gold nanoparticles as labels. Int J Nanomedicine. 2014;9(1):2619–2626. | ||
Dempsey E, Diamond D, Collier A. Development of a biosensor for endocrine disrupting compounds based on tyrosinase entrapped within a poly(thionine) film. Biosens Bioelectron. 2004;20(2):367–377. | ||
Hahm J, Lieber CM. Direct ultrasensitive electrical detection of DNA and DNA sequence variations using nanowire nanosensors. Nano Lett. 2004;4(1):51–54. | ||
Sánchez S, Pumera M, Fàbregas E. Carbon nanotube/polysulfone screen-printed electrochemical immunosensor. Biosens Bioelectron. 2007;23(3):332–340. | ||
Rahman MA, Son JI, Won MS, Shim YB. Gold nanoparticles doped conducting polymer nanorod electrodes: ferrocene catalyzed aptamer-based thrombin immunosensor. Anal Chem. 2009;81(16):6604–6611. | ||
Bangar MA, Shirale DJ, Chen W, Myung NV, Mulchandani A. Single conducting polymer nanowire chemiresistive label-free immunosensor for cancer biomarker. Anal Chem. 2009;81(6):2168–2175. | ||
Li F, Han XP, Liu SF. Development of an electrochemical DNA biosensor with a high sensitivity of fM by dendritic gold nanostructure modified electrode. Biosens Bioelectron. 2011;26(5):2619–2625. | ||
Deng YP, Huang W, Chen X, Li ZL. Facile fabrication of nanoporous gold film electrodes. Electrochem Commun. 2008;10(5):810–813. | ||
Zhang HF, Hussain I, Brust M, Butler MF, Rannard SP, Cooper AI. Aligned two- and three-dimensional structures by directional freezing of polymers and nanoparticles. Nat Mater. 2005;4(10):787–793. | ||
Weissmüller J, Viswanath RN, Kramer D, Zimmer P, Würschum R, Gleiter H. Charge-induced reversible strain in a metal. Science. 2003;300(5617):312–315. | ||
Zielasek V, Jürgens B, Schulz C, et al. Gold catalysts: nanoporous gold foams. Angew Chem Int Ed. 2006;45(48):8241–8244. | ||
Lang XY, Guo H, Chen LY, et al. Novel nanoporous Au-Pd alloy with high catalytic activity and excellent electrochemical stability. J Phys Chem C. 2010;114(6):2600–2603. | ||
Hu KC, Lan DX, Li XM, Zhang S. Electrochemical DNA biosensor based on nanoporous gold electrode and multifunctional encoded DNA-Au bio bar codes. Anal Chem. 2008;80(23):9124–9130. | ||
Gao W, Xia XH, Xu JJ, Chen HY. Three-dimensionally ordered macroporous gold structure as an efficient matrix for solid-state electrochemiluminescence of Ru(bpy)32+/TPA system with high sensitivity. J Phys Chem C. 2007;111(13):12213–12219. | ||
Li Y, Song YY, Yang C, Xia XH. Hydrogen bubble dynamic template synthesis of porous gold for nonenzymatic electrochemical detection of glucose. Electrochem Commun. 2007;9(5):981–988. | ||
Gamero M, Pariente F, Lorenzo E, Alonso C. Nanostructured rough gold electrodes for the development of lactate oxidase-based biosensors. Biosens Bioelectron. 2010;25(9):2038–2044. | ||
Zhong GX, Liu AL, Chen XH, et al. Electrochemical biosensor based on nanoporous gold electrode for detection of PML/RARα fusion gene. Biosens Bioelectron. 2011;26(9):3812–3817. | ||
Zhong GX, Liu AL, Xu XW, et al. Detection of femtomolar level osteosarcoma-related gene via a chronocoulometric DNA biosensor based on nanostructure gold electrode. Int J Nanomedicine. 2012;7:527–536. | ||
Shim JH, Yoon DL, Han S, et al. Is serum alpha-fetoprotein useful for predicting recurrence and mortality specific to hepatocellular carcinoma after hepatectomy? A test based on propensity scores and competing risks analysis. Ann Surg Oncol. 2012;19(12):3687–3696. | ||
Ho HK, Pok S, Streit S, et al. Fibroblast growth factor receptor 4 regulates proliferation, anti-apoptosis and alpha-fetoprotein secretion during hepatocellular carcinoma progression and represents a potential target for therapeutic intervention. J Hepatol. 2009;50(1):118–127. | ||
Fan CH, Plaxco KW, Heeger AJ. Electrochemical interrogation of conformational changes as a reagentless method for the sequence-specific detection of DNA. Proc Nat Acad Sci U S A. 2003;100(16): 9134–9137. | ||
Vazquez L, Bartolome A, Baro AM. STM-SEM combination study on the electrochemical growth mechanism and structure of gold overlayers: a quantitative approach to electrochemical metal surface roughening. Surf Sci. 1989;215(1–2):171–189. | ||
Liu G, Wan Y, Gau V, et al. An enzyme-based e-DNA sensor for sequence-specific detection of femtomolar DNA targets. J Am Chem Soc. 2008;130(21):6820–6825. | ||
Fanjul-Bolado P, Gonzalez-Garcia MB, Costa-Garcia A. Amperometric detection in TMB/HRP-based assays. Anal Bioanal Chem. 2005;382(2):297–302. | ||
Das A, Hecht MH. Peroxidase activity of de novo heme proteins immobilized on electrodes. J Inorg Biochem. 2007;101(11–12):1820–1826. | ||
Yang S, Jia WZ, Qian QY, Zhou YG, Xia XH. Simple approach for efficient encapsulation of enzyme in silica matrix with retained bioactivity. Anal Chem. 2009;81(9):3478–3484. | ||
Wu YM, Xu WJ, Wang Y, Yuan YL, Yuan R. Silver-graphene oxide nanocomposites as redox probes for electrochemical determination of α-1-fetoprotein. Electrochim Acta. 2013;88:135–140. | ||
Park JY, Lee YS, Kim BH, Park SM. Label-free detection of antibody-antigen interactions on (R)-lipo-diaza-18-crown-6 self-assembled monolayer modified gold electrodes. Anal Chem. 2008;80(13):4986–4993. | ||
Huang KJ, Li J, Wu YY, Liu YM. Amperometric immunobiosensor for α-fetoprotein using Au nanoparticles/chitosan/TiO(2)-graphene composite based platform. Bioelectrochemistry. 2013;90:18–23. | ||
Liu S, Zhang J, Tu W, Bao J, Dai Z. Using ruthenium polypyridyl functionalized ZnO mesocrystals and gold nanoparticle dotted graphene composite for biological recognition and electrochemiluminescence biosensing. Nanoscale. 2014;6(4):2419–2425. | ||
Zhao L, Li S, He J, Tian G, Wei Q, Li H. Enzyme-free electrochemical immunosensor configured with Au-Pd nanocrystals and N-doped graphene sheets for sensitive detection of AFP. Biosens Bioelectron. 2013;49:222–225. | ||
Lin J, Wei Z, Zhang H, Shao M. Sensitive immunosensor for the label-free determination of tumor marker based on carbon nanotubes/mesoporous silica and graphene modified electrode. Biosens Bioelectron. 2013;41:342–347. |
Supplementary material
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.