Back to Journals » International Journal of Nanomedicine » Volume 15
Selenium Nanoparticles Pre-Treatment Reverse Behavioral, Oxidative Damage, Neuronal Loss and Neurochemical Alterations in Pentylenetetrazole-Induced Epileptic Seizures in Mice
Authors Yuan X, Fu Z, Ji P, Guo L, Al-Ghamdy AO, Alkandiri A , Habotta OA , Abdel Moneim AE , Kassab RB
Received 21 April 2020
Accepted for publication 30 July 2020
Published 24 August 2020 Volume 2020:15 Pages 6339—6353
DOI https://doi.org/10.2147/IJN.S259134
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Linlin Sun
Xiaona Yuan,1 Zhenshuai Fu,2 Pengfei Ji,3 Lubo Guo,4 Ali O Al-Ghamdy,5 Ali Alkandiri,4,6 Ola A Habotta,7 Ahmed E Abdel Moneim,8 Rami B Kassab8
1Department of Emergency, The First Affiliated Hospital of Zhengzhou University, Zhengzhou City, Henan Province 450000, People’s Republic of China; 2Department of ICU, Sunshine Union Hospital, Weifang City, Shandong Province 261000, People’s Republic of China; 3Department of Ophthalmology, Zhengzhou Second Hospital, Zhengzhou City, Henan Province 450000, People’s Republic of China; 4Department of Pharmacy, Central Hospital Affiliated to Shandong First Medical University, Jinan City, Shandong Province 250013, People’s Republic of China; 5Biology Department, Faculty of Science and Arts, Al Baha University, Almakhwah, Saudi Arabia; 6Laboratory Technology Department, College of Technological Studies, Safat 13092, Kuwait; 7Forensic Medicine and Toxicology Department, Faculty of Veterinary Medicine, Mansoura University, Mansoura, Egypt; 8Department of Zoology and Entomology, Faculty of Science, Helwan University, Helwan 11795, Egypt
Correspondence: Lubo Guo
Department of Pharmacy, Central Hospital Affiliated to Shandong First Medical University, Jinan City, Shandong Province 250013, People’s Republic of China
Tel +86 18331125851
Email [email protected]
Introduction: Epilepsy is a chronic neurological condition characterized by behavioral, molecular, and neurochemical alterations. Current antiepileptic drugs are associated with various adverse impacts. The main goal of the current study is to investigate the possible anticonvulsant effect of selenium nanoparticles (SeNPs) against pentylenetetrazole (PTZ)-mediated epileptic seizures in mice hippocampus. Sodium valproate (VPA) was used as a standard anti-epileptic drug.
Methods: Mice were assigned into five groups (n=15): control, SeNPs (5 mg/kg, orally), PTZ (60 mg/kg, intraperitoneally), SeNPs+PTZ and VPA (200 mg/kg)+PTZ. All groups were treated for 10 days.
Results: PTZ injection triggered a state of oxidative stress in the hippocampal tissue as represented by the elevated lipoperoxidation, heat shock protein 70 level, and nitric oxide formation while decreased glutathione level and antioxidant enzymes activity. Additionally, the blotting analysis showed downregulation of nuclear factor erythroid 2-related factor 2 (Nrf2) and heme oxygenase-1 (HO-1) in the epileptic mice. A state of neuroinflammation was recorded following the developed seizures represented by the increased pro-inflammatory cytokines. Moreover, neuronal apoptosis was recorded following the development of epileptic convulsions. At the neurochemical level, acetylcholinesterase activity and monoamines content were decreased in the epileptic mice, accompanied by high glutamate and low GABA levels in the hippocampal tissue. However, SeNP supplementation was found to delay the onset and decreased the duration of tonic, myoclonic, and generalized seizures following PTZ injection. Moreover, SeNPs were found to provide neuroprotection through preventing the development of oxidative challenge via the upregulation of Nrf2 and HO-1, inhibiting the inflammatory response and apoptotic cascade. Additionally, SeNPs reversed the changes in the activity and levels of neuromodulators following the development of epileptic seizures.
Conclusion: The obtained results suggest that SeNPs could be used as a promising anticonvulsant drug due to its potent antioxidant, anti-inflammatory, and neuromodulatory activities.
Keywords: epilepsy, selenium nanoparticles, oxidative stress, neuroinflammation and apoptosis, monoamines, GABA and glutamate
Introduction
Epilepsy is a chronic neurodegenerative condition that affects about 0.5% to 1% of the total world population, featured with repeated and unprovoked seizures. Epileptic seizures are associated with socioeconomic, psychological, and behavioral changes due to neuronal hyper electrical activity and synchronous and continuous abnormal discharges.1,2 The development of seizures may be correlated with several factors including cerebrovascular disorders, trauma, cancers, oxygen deficiency, and infection. However, the precise causative factors are still unknown.3 Although the underlying cellular and molecular mechanisms involved in epileptogenesis are unclear, it is hypothesized that oxidative insult, neuroinflammation, neuronal apoptosis, and neurotransmission impairments lead to aberrant neural injury that mediates the onset of epileptic seizures.4 Experiments conducted in animal models suggest the overproduction of reactive oxygen/nitrosative species (ROS/RNS) and inactivation of endogenous antioxidant molecules in the brain tissue following the progression of epileptic seizures.5,6 In addition, the inflammatory response including activation of glial cells and secretion of pro-inflammatory cytokines has been assumed to enhance apoptotic events resulting in neuronal loss which play a crucial role in the development of seizures.7,8 Moreover, the dysregulation of the monoaminergic, amino acidergic and cholinergic transmission has been demonstrated during the epileptic seizures.9,10
Current antiepileptic drugs are associated with numerous adverse impacts such as memory deficits, fatigue, tremors, gastrointestinal symptoms, osteoporosis, depression, dizziness and nausea11 thus creating an urgent need to formulate more efficient anticonvulsant drugs with minimal side effects. Advances in nanotechnology led to the development of different nanoparticle formulations that recently used as therapeutic agents against various health problems.12,13 The effect of nanoparticles on tissues depends on their characteristics including size, shape, dose, concentration and type of nanoparticle, type of tissue, and period of exposure.14 Due to their nanoform proportions and other useful surface reactivity, nanoparticles are widely employed in various biomedical applications. Experimental results indicated that selenium nanoparticles (SeNPs) were more effectively absorbed by the chickens in comparison with Se(IV).15 However, the same experiment showed that both SeNPs and Se(IV) were equally absorbed, distributed, metabolized, and excreted. The minor size of SeNPs allowed them to cross membrane barriers and accumulate in tissues, resulting in more reactivity.16 Metal nanoparticles, including SeNPs, have been recently used in the prevention and/or treatment of neurodegenerative disorders due to their high bioavailability, low adverse effects, and promising therapeutic activity.17
Selenium is a fundamental micronutrient that is incorporated in the structure of the active center of selenoproteins as selenocysteine moiety. Selenium is necessary for various physiological functions such as regulating thyroid hormones production, redox homeostasis, and immune response.18 The deficiency of selenium and selenoproteins was associated with permanent brain injury.19 Previous studies demonstrated the neuroprotective activity of SeNPs against stroke murine model. The authors reported that these particles were transferred to the brain through transferrin receptor-mediated endocytosis and were found to inhibit the inflammatory response and enhanced hippocampal neuronal survival.20 Additionally, SeNPs showed high antioxidant properties and was found to enhance the selenoproteins-based endogenous antioxidant system.21 Moreover, SeNPs were found to enhance recovery of Alzheimer’s disease through suppression of amyloid-β accumulation, in vitro.22 Furthermore, therapeutic nanotechnology-based interventions can inhibit oxidative stress, neuroinflammation, and neuronal apoptosis may provide neuroprotection23 and thus prevent the development of epileptic seizures.24 Here, we aimed to investigate the potential neuroprotective and anticonvulsant activity of SeNPs against pentylenetetrazole (PTZ)-induced epileptic seizures through estimating the oxidative damage, neuroinflammation, neuronal apoptosis, cholinergic, as well as monoaminergic and amino acidergic transmission in the hippocampal tissue of mice.
Materials and Methods
Pentylenetetrazole (PTZ) was purchased from Sigma Chemical Co. (St. Louis, MO, USA). SeNPs, with a particle size <100 nm, a 99.9% trace metals basis and Stock No: NS6130-01-171 and CAS: 7782–49-2, was purchased from Nanoshel (Wilmington, DE, USA). According to the provider, the prepared SeNPs were characterized using Transmission Electron Microscopy (TEM), Dynamic Light Scattering (DLS), and Zeta Potential Measurements (ZPMs) to guarantee consistent materials. For an additional characterization, the particle size distribution was estimated by a Zetasizer (Figure 1). SeNPs in the administered doses were distributed more uniformly by sonication for 10 minutes prior to administration, to be taken by systemic circulation. All used solvents and other chemicals used in the study were of analytical grade.
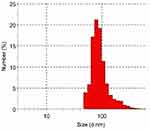 |
Figure 1 The histogram of size distribution measured by a Malvern Zetasizer. |
Experimental Protocol
Sixty adult male Swiss mice weighing 25–32 g each were purchased from VACSERA, Egypt. Mice were given free access to water and commercial pelleted rodent feed ad libitum. The mice were maintained in the animal lab facility of the Zoology Department at Helwan University, Cairo, Egypt under standard laboratory conditions at a temperature of 22–25°C and a 12 h artificial light/dark cycle.
The animals were treated according to the criteria of Investigations and Ethics for Laboratory Animal Care at the Zoology Department, Faculty of Science, Helwan University (approval No.HU2019/Z/RK0419-04). To explore the possible neuroprotective efficiency of SeNPs in epileptic mouse mode mediated by PTZ, mice were allocated randomly into five groups (n = 15) after 7 days of acclimatization. The five groups were divided as follows:
Control group (Cont): Mice were introduced to normal saline (0.9% NaCl) daily for 10 days. On the 10th day, they were injected intraperitoneally (i.p.) with saline 1 h after the oral administration of saline.
Selenium nanoparticles treated group (SeNPs): This group received oral administration of SeNPs (0.5 mg/kg) according to Dkhil et al25 daily for 10 days. On the 10th day, they were injected intraperitoneally (i.p.) with saline 1 h after the oral administration of SeNPs.
PTZ-injected group (PTZ): Mice were introduced to normal saline daily for 10 days. On the 10th day, these animals received a single i.p. dose of PTZ (60 mg/kg) according to Abdel-Rahman et al,10 1 h after the oral administration of saline.
SeNPs + PTZ-treated group (SeNPs + PTZ): This group received daily oral administration of SeNPs (0.5 mg/kg) and were injected with a single dose of PTZ (60 mg/kg) on the 10th day.
Sodium valproate (VPA)+ PTZ-treated group (VPA + PTZ): Mice administered orally with VPA (200 mg/Kg) according to Arafa et al9 and injected with a single dose of PTZ (60 mg/kg) on the 10th day.
In this investigation, both PTZ and SeNPs were dissolved in normal saline. Mice were euthanized 24 h after the last treatment. The hippocampus was immediately separated and washed with isotonic saline. To evaluate biochemical markers, the hippocampal tissue of seven mice was homogenized in ice-cold 10mM phosphate buffer (pH 7.4) to produce a 10% (w/v) homogenate. Meanwhile, to estimate the monoamines and amino acids neurotransmitters, Hippocampal tissue was homogenized in 75% methanolic HPLC (10% w/v), then centrifuged at 4000 rpm for 10 minutes. For histopathological examination, the hippocampal tissue of three mice was fixed in 10% neutral buffered formalin and the CA1 hippocampal region was selected to examine the histopathological changes in different experimental groups. In addition, the protein level in the hippocampal tissue was estimated using the method described by Lowry et al.26
PTZ-Induced Seizures
PTZ (60 mg/kg) was i.p. injected to induce epileptic convulsions in mice. SeNPs administration, at a dose of (0.5 mg/kg, orally), was administered 30minprior to PTZ injection. The seizure index was observed and recorded carefully for 40 min after PTZ injection according to the modified Racine scale27 as follows:
Phase 0: no response; Phase 1: ear and facial twitching; Phase 2: myoclonic jerks without rearing; Phase 3: myoclonic jerks, rearing; Phase 4: turning over onto side position, tonic-clonic seizures; Phase 5: turning over onto back position, generalized tonic-clonic seizures.
- Latency: the time between PTZ injection and the onset of seizures.28
- Duration: the time interval from the onset to termination of seizures or death of the animal.
- Percent of death: the number of mice that died after PTZ injection among the mice of a particular group.
Estimation of Oxidative Stress Indices
The hippocampal level of lipoperoxidation in terms of malondialdehyde (MDA) was assessed using the thiobarbituric acid method. The thiobarbituric acid reactive substances were measured at 535 nm and then presented in terms of MDA produced, as illustrated by Ohkawa et al.29 Heat shock protein 70 (Hsp70) was quantified using ELISA kit sourced from LifeSpanBioSciences, USA with Catalogue number: LS-F9139. Hippocampal nitric oxide (NO) level was determined by using the Griess reagent (sulfanilic acid and N-(1-naphthyl)ethylenediamine), and the formed azo dye was evaluated at 540 nm.30 The hippocampal glutathione (GSH) level was demonstrated using Ellman’s reagent, and the developed yellow chromagen was estimated at 412 nm.31
Estimation of Antioxidant Enzyme Activities
Glutathione Reductase (GR)
The activity of GR in hippocampal tissue was investigated based on the protocol illustrated by Factor et al.32 Briefly, 20 µL of hippocampal homogenate was added to oxidized glutathione (0.44 mM), EDTA (0.30 M), in phosphate buffer (0.1 M) at pH 7.0. The reaction was developed by pipetting NADPH (0.036 M). The oxidation rate of NADPH was followed by the reduction in absorbance at 340nm with duration. One unit of the enzyme was known as the quantity of enzyme required to oxidize 1 µmol of NADPH per minute.
Glutathione Peroxidase (GPx)
Based on the procedures described by Paglia and Valentine,33 the hippocampal activity of GPx was assayed. In brief, hippocampal supernatant (200 µL) was added to 1 mL phosphate buffer (75 mM) at pH 7.0, 10 mL glutathione (150 mM), 10 mL glutathione reductase (340 U/mL), 30 mL (25 mM EDTA), 30 mL NADPH (5 mM), 10 mL Triton X-100 (20%), and 50 µL H2O2 (7.5mM). The oxidation of NADPH to NADP+ was examined at 340 nm for 3 minutes. One unit of GPx activity was presented as the amount of GSH (nanomoles) oxidized/minute/milligram of protein (U/mg protein).
Catalase (CAT) Activity
CAT activity in the hippocampal supernatant was estimated according to the method illustrated by Aebi.34 In order to evaluate the CAT activity, the enzymatic reaction mixture (total volume 1 mL) contained 50 mM potassium phosphate (pH 7.0), 19 mM H2O2, and 50 µL of homogenate supernatant was employed. The molar attenuation coefficient of H2O2 was examined by a UV-VIS spectrophotometer at 240 nm. One unit of catalase activity was determined as the level of enzyme decomposing H2O2 (1 µmol)/minute/milligram of hippocampal tissue protein (U/mg protein).
Superoxide Dismutase (SOD) Activity
The determination of Hippocampal SOD activity was performed following Misra and Fridovich’s technique.35 This method depended on the capacity of epinephrine oxidation at pH 10.2 resulting in the formation of adrenochrome and superoxide radicals (O2.−). The suppression of SOD activity was investigated based on reducing the absorbance at 480 nm.
Estimation of Pro-Inflammation Cytokines
Hippocampal level of interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α) were measured using enzyme-linked immunosorbent assay kits sourced from ThermoFisher Scientific (Catalogue number ERIL1B) and R&D Systems (Catalogue number RTA00) following the manufacturers’ protocol.
Estimation of Apoptotic Proteins
The apoptotic proteins in the hippocampal tissue were estimated using enzyme-linked immunosorbent assay kits; Bax (BioVision, Inc., catalog number E4513) and Bcl-2 (BioVision, Inc., catalog number CSB-E08854r), whereas caspase 3 activity was measured by using a colorimetric kit (Sigma-Aldrich, CASP3C-1KT).
Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR) Analysis
Total hippocampal RNA was isolated using the standard TRIzol®method (Invitrogen, Carlsbad, CA, USA). RNA was reverse transcribed to cDNA. The primer sequences employed to estimate Gpx1, Gsr, Sod2, and Cat gene expressions are listed in Table 1 according to Abdel Moneim.36 Power SYBR® Green Master Mix was used for RT-qPCR analysis, which was performed in triplicate. The RT-qPCR cycling conditions were 10 min at 95°C followed by 40 cycles involving denaturation at 94°C for 10 s, annealing at 60°C for 30 s, and extension at 72°C for 20 s. Gene expression in the experimented groups is expressed as a fold change in expression relative to that of the control group. Glyceraldehyde-3-phosphate dehydrogenase was used as the standard gene; its expression remained unaltered throughout the experiment.
 |
Table 1 Primer Sequences of Genes Analyzed in Real-Time PCR |
Determination of Acetylcholinesterase (AChE)
The activity of AChE in the hippocampal homogenate was assessed based on the procedure described by Elman et al.37 The produced thiocholine by the action of AChE creates 5,5′-dithiobis (2-nitrobenzoic acid) which further reduced to thionitrobenzoic acid, a yellow-colored anion. The level of thionitrobenzoic acid yellow color was quantified at 412 nm and was relative to the activity of AChE in the hippocampal tissue.
Estimation of Monoamines and Free Amino Acids Neurotransmitters
The HPLC system consisted of a quaternary pump; a column oven, a Rheodine injector, 20 μL loop, and a UV variable wavelength detector. The reports and chromatogram were taken from the data acquisition program (ChemStation). The hippocampal supernatant was extracted from the trace elements and lipids by the use of solid-phase extraction CHROMABOND column NH2 phase cat. No. 730031. The sample was then injected directly into an AQUA column 150 mm 5 μ C18 (Phenomenex, USA) using the following settings: mobile phase 20 mM potassium phosphate, pH 2.5, flow rate 1.5 mL/min, UV 190 nm. Dopamine (DA), norepinephrine (NE) and serotonin (5-HT) were separated after 12 minutes. The resulting chromatogram identified each monoamine position and concentration from the sample as compared with that of the standard (Sigma Chemical Co., St. Louis, MO, USA), and finally, the estimation of the concentration of each monoamine as μg per gram brain tissue was quantified according to Pagel et al.38 Meanwhile, the levels of glutamate and γ-aminobutyric acid (GABA) were estimated by the precolumn PITC derivatization technique illustrated by Heinrikson and Meredith.39
Western Blotting Analyses
Protein extraction from the hippocampal tissue and blotting analyses were performed as previously illustrated.40 Nuclear factor erythroid 2-related factor 2 (Nrf2) (MAB3925, 1:1000; R&D System), heme oxygenase-1 (HO-1) (sc-390991, 1:1000; Santa Cruz Biotechnology, Santa Cruz, CA, USA), and β-actin (MAB8929, 1:2000; R&D System, Minneapolis, MN, USA) were used as a primary antibody, whereas goat anti-mouse IgG (sc-2039, 1:5000; Santa Cruz Biotechnology, Santa Cruz, CA, USA) conjugated with horseradish peroxidase (HRP) was used a secondary antibody. The protein bands were visualized using a chemiluminescence HRP substrate (Bio-Rad, Hercules, CA, USA). The blot intensity was then quantified using Image J software referenced to β-actin.
Histopathological Investigation
The hippocampal tissue was kept for 24 h at room temperature in 10% neutral-buffered formalin as a fixative, then dehydrated, paraffinized, and sectioned (4–5μm). Sections were stained with hematoxylin and eosin for light microscopy analysis. Light microscope photomicrographs were captured using a Nikon microscope (Eclipse E200-LED, Tokyo, Japan) at an original magnification of × 400. The severity of the brain injury was graded using the numerical method that described by Al-Quraishy et al41 with five grades of severity of injury specified as follows: 1 = minimal (<1%); 2 = slight (1%–25%); 3 = moderate (26%–50%); 4 = moderate/severe (51%–75%); and 5 = severe (76%–100%).
Statistical Analysis
Data are illustrated as means ± standard error of the mean (SEM). The recorded results from the performed biochemical measurements were examined by one-way analysis of variance and post hoc Duncan’s test, while Mann Whitney U and Student’s t-tests were applied to the analysis of behavioral scores using the statistical package program (SPSS version 14.0); P values <0.05 represent statistical significance.
Results
PTZ-Induced Epileptic Seizures
According to Racine scale, PTZ injection (60 mg/kg) enhanced the development of tonic, myoclonic and generalized seizures in all treated mice. Pre-administration of SeNPs at a dose of 5 mg/kg for 14 consecutive days delayed significantly (P<0.05) the onset and decreased the duration of these convulsions; reflecting its anticonvulsant effect against PTZ-induced seizures (Table 2).
 |
Table 2 The Effect of Selenium Nanoparticles (SeNPs) on Behavioral Alterations Induced Following Pentylenetetrazole (PTZ) Injection in Mice |
Effect of SeNPs on Redox Status in the Hippocampal Tissue Following PTZ Injection
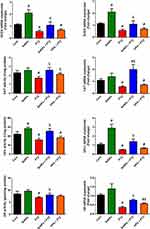
To evaluate the redox status in the hippocampal tissue, levels of pro-oxidants and antioxidant enzymes were evaluated. PTZ injection was found to provoke a state of oxidative stress as characterized by the increased neuronal lipoperoxidation in terms of MDA, Hsp70 and NO levels, and the decreased GSH level. This was accompanied by a decrease in the activity and mRNA expression of antioxidant enzymes (GPx, GR, SOD, and CAT) with respect to their control levels. SeNPs administered group exhibited a significant increase in the activity and mRNA expression of GPx and SOD. However, mice treated with SeNPs before PTZ suppressed significantly (P<0.05) the oxidative damage through decreasing MDA production, Hsp70, and NO levels and enhancing the activity and mRNA expression of endogenous antioxidant molecules in the hippocampal tissue as compared to the epileptic group; confirming the antioxidant impact of SeNPs against oxidative insults mediated following PTZ injection (Figures. 2 and 3).
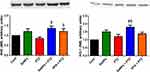
In order to assess the molecular antioxidant mechanism provided by SeNPs in the hippocampal tissue of epileptic mice, Nrf2 and HO-1 expression was experimented using the Western blotting technique. PTZ injection was associated with down-regulation of Nrf2 and HO-1 as compared to their expression in the control group. Notably, SeNPs supplementation enhanced the expression of these antioxidant regulators as compared to PTZ-treated mice; which may explain its antioxidant capacity against oxidative stress developed following the onset of epileptic seizures (Figure 4).
The Anti-Inflammatory Activity of SeNPs in the Hippocampal Tissue Following PTZ Injection
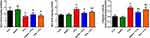
To elucidate the potential anti-inflammatory effect of SeNPs against PTZ-induced neuroinflammation, the level of TNF-α and IL-1β were determined. The epileptic group recorded a significant (P<0.05) elevation in the measured inflammatory cytokines when compared to the control group. No statistical change was observed in these pro-inflammatory mediators in the treated group with SeNPs. However, SeNPs inhibited significantly the inflammatory response developed upon PTZ injection, indicating the anti-inflammatory effect of SeNPs against PTZ-mediated neuroinflammation (Figure 5).
SeNPs Inhibits Neuronal Loss in the Hippocampal Tissue Following PTZ Injection
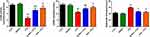
PTZ injection was found to enhance neuronal apoptosis in the examined brain tissue as presented by the increased pro-apoptotic proteins and their mRNA expression including Bax and caspase-3, and the decreased anti-apoptotic proteins and its mRNA expression, Bcl2. SeNPs alone did not affect the levels and expression of the examined apoptotic markers. Meanwhile, pre-administration with SeNPs was found to protect the brain tissue through inhibiting the neuronal apoptosis triggered following PTZ injection (Figure 6).
Effect of SeNPs Administration on Cholinergic, Monoamines and Amino Acids Neurotransmitters in the Hippocampal Tissue Following PTZ Injection
PTZ-injected mice showed a significant (P<0.05) decrease in DA, NE, 5-HT, GABA levels, and AChE activity in the hippocampal tissue, accompanied by a significant increase in the glutamate content as compared to the control group. In addition, the levels of these neurotransmitters remained unchanged when compared to their corresponding level in SeNPs supplemented group. Meanwhile, SeNPs administration prior to PTZ protected the examined neurotransmitters in the hippocampal tissue when compared to PTZ-treated group; reflecting its neuroprotective effect against PTZ-induced neurochemical alterations (Figures. 7 and 8).
Effect of SeNPs Administration on Histopathological Changes in the CA1 Hippocampal Tissue Following PTZ Injection
Neither vehicle nor SeNPs administration affected the morphology of CA1 hippocampal tissue, in both control and SeNPs-treated alone groups, where the neurons displayed normal structure with intact shapes, and vesicular nuclei (Figure 9A and B). Conversely, the PTZ-injected mice exhibited frequent neuronal damages and increased the number of dark neurons in CA1 (Figure 9C). However, the neurons in the SeNPs+ PTZ and VPA+PTZ groups showed near-normal morphology compared to the PTZ-injected mice (Figure 9D and E) (Supplementary data: Table S1).
Discussion
Application of metal nanotechnology in medicine is a promising approach in terms of improving delivery, bioavailability, and release of drugs to the target tissue.42 In the current study, we explored the potential neuroprotective and anticonvulsant activity of SeNPs against PTZ-mediated epileptic seizures. PTZ is widely employed to induce epileptic experimental model and its associated behavioral, molecular, and neurochemical alterations in order to screen novel potential antiepileptic drugs. PTZ is known to enhance the development of seizures by blocking the benzodiazepine recognition sites in GABAA receptors.43
Our findings showed that a single convulsive dose of PTZ potentiated a state of oxidative stress in the hippocampal tissue as represented by the increased neuronal lipoperoxidation level in terms of MDA, Hsp70 level and NO production, while GSH content was decreased. This was accompanied by a decrease in antioxidant enzymes and their mRNA expression. Brain tissue is liable to the progression of oxidative damage due to its high unsaturated fatty acids constituent, high oxygen demand and low antioxidant content.44 Neuronal hyperexcitability and epileptogenesis are associated with oxidative stress result from mitochondrial and endoplasmic reticulum dysfunction leading to free radicals overproduction that exhausts and depletes the neuronal antioxidant molecules.45,46 Lipoperoxidation results from the interaction of hydroxyl radicals with unsaturated fatty acids in the neuronal membrane resulted in the formation of lipid peroxide, hydroxide radicals and MDA.47,48 Heat shock proteins (HSPs) including Hsp70are playing cytoprotective activities in response to pathological conditions including oxidative stress through their chaperoning activities.49 It has been shown that neuronal cells are able to produce HSPs. Accumulative evidences reported the relationship between the development of epileptic seizures and the upregulation of Hsp70 in animal models50 and in humans.51 The authors described the upregulation of Hsp70 as a protective effect potentiated to protect neurons against hyperexcitability coupled with the development of epileptic seizures. The excessive secretion of NO has been reported following epileptic convulsions. This elevation may be attributed due to over-expression of nitric oxide synthase (NOS) that responsible for NO formation. The deleterious effect of the increased NO came from its interaction with superoxide anions to form peroxynitrite radicals that are associated with various neurological consequences.52 The antioxidant system including GSH, GPx, GR, SOD, and CAT protected against free radicals accumulation in the cell.23 Depletion of GSH content and antioxidant enzymes activity in brain tissue is associated with epileptic seizures in human and animal models due to the excessive generation of ROS and this decrease was shown to increase with repetitive seizures.24,53,54
On the other hand, SeNPs administration prior to PTZ was found to protect neuronal oxidative injury through inhibiting lipoperoxidation by-product (MDA), decreasing Hsp70 and NO formation accompanied by increasing GSH level. Additionally, SeNPs activated and upregulated mRNA expression of GPx, GR, SOD, and CAT in the hippocampus. Selenium is incorporated in the structure of selenoproteins and selenoenzymes that are able to quench ROS and consequently suppress the development of oxidative damage.55 SeNPs are characterized by small size and large surface area. This structure allows more selenium atoms to scavenge more free radicals. Therefore, SeNPs have more powerful scavenging activity and able to enhance endogenous antioxidant proteins.56 SeNPs inhibited the neuronal oxidative damage in Alzheimer’s disease through inhibiting ROS production and increasing the endogenous antioxidant molecules including GPx, SOD, and CAT.57 In another experimental model, SeNPs were found to inhibit lipid peroxidation product in the renal tissue, suggesting its ability to protect the membrane structure and integrity in response to glycerol injection-induced acute kidney injury.58 The authors also recorded an increase in GSH content and, level and mRNA expression of its based enzymes (GPx and GR) along with SOD and CAT. Selenium administration is known to quench ROS and RNS in different experimental studies. It has been demonstrated that the epileptic mice treated with selenium showed a decrease in NO generation due to the inhibition of NOS. Cao et al59 reported that dietary administration of selenium reversed the increased NO in the cerebral tissue following aluminum exposure. The authors attributed this reduction to the ability of selenium to down-regulate NOS.60 Selenium-enriched probiotics were found to inhibit oxidative stress following heat stress through stimulating the activity of antioxidant molecules along with Hsp70.61
Neuroinflammation is tightly linked with epileptic seizures. In the current study, a marked increase in the production of pro-inflammatory cytokines was obvious in the hippocampal tissue upon PTZ injection. Previous reports have demonstrated the development of inflammatory response in epileptic model.7,62 The authors attributed this effect to the disturbance in the oxidative status and the overproduction of ROS which activates nuclear factor kappa B (NF-κB) resulting in the elevation in pro-inflammatory cytokines. Meanwhile, the SeNPs administration suppressed the inflammation in the hippocampal tissue. It was reported that selenium protected the brain tissue in different neurodegenerative models against the activation of microglial inflammatory responses and the over release of pro-inflammatory cytokines through inhibiting the expression of NF-κB.63
Neuronal loss is a characteristic feature following epileptic seizures. PTZ injection was found to trigger neuronal apoptosis in the hippocampal tissue as represented by the increased Bax and caspase-3 levels and the decreased Bcl2 level. The obtained results are in line with previous reports.24,64 The authors recorded a decrease in Bcl2, the anti-apoptotic protein associated with an increase in the pro-apoptotic protein (Bax) upon epileptic convulsions induced by PTZ. Previous reports have shown a link between the incidence of oxidative stress and inflammation and the development of neuronal apoptosis. The authors attributed the neuronal cell death to the overproduction of ROS following mitochondrial dysfunction.65 On the other hand, SeNPs supplementation inhibited the hippocampal neurons loss through increasing Bcl2 and decreasing Bax levels. The anti-apoptotic effect of selenium application and its nano-sized particles has been demonstrated in different experimental protocols through enhancing the anti-apoptotic markers and suppressing the pro-apoptotic markers.25,66
In the current study, PTZ was found to induce epileptic seizures via decreasing AChE activity, NE, DA, 5-HT, glycine and GABA content, and increasing glutamate level in the hippocampal tissue. Alterations in the cholinergic, monoaminergic and amino acidergic transmission have been reported upon epileptic seizures.10 Crosstalk has been recorded between the neurochemical alterations and the imbalance between pro-oxidants and antioxidants following epileptic seizures.67 Inhibition of AChE activity is associated with the accumulation of acetylcholine resulting in the development of neurological consequences including convulsions and tremors.68 Monoamines are known to play an essential role in the development of epileptogenesis. The deficiency of monoamines was found to promote neuronal hyperexcitability in human and animal studies.10,69 The decrease in 5-HT content has been linked with a decrease in its synaptosomal uptake, inhibition of tryptophan hydroxylase activity and the decrease of tryptophan level in epileptic model.70,71 Meanwhile, the reduced DA level in epileptic patients has been attributed to the increased monoamine oxidase activity and the decreased reuptake.72 Furthermore, NE is a neuromodulator act as an anticonvulsant agent and its decrease in epileptic patients is due to the downregulation of α1 receptors’ density in the brain tissue.73 The decrease may be also due to the decrease in Dopamine-β-hydroxylase, the rate-limiting enzyme in NE synthesis.74
GABA is the predominant inhibitory amino acid neurotransmitter in the neuronal tissue and its reduction was associated with the development of seizures that also coupled with the blockade of GABA receptors and the inhibition of chloride ions influx resulting in hyperexcitability and neuronal death.75 Glutamate represents the main excitatory amino acid neurotransmitter in the mammalian brain. The excessive release of glutamate over activates glutamatergic receptors including NMDARs that enhance the development of seizure.76 Moreover, the neurochemical alteration following the progression of seizures may be responsible for the recorded behavioral changes.
Enhancing the activity of AChE and monoaminergic transmission is associated with inhibition of epileptogenesis.10 In the current study, SeNPs administration increased AChE activity and DA, NE and 5-HT content in the hippocampal tissue; indicating its anticonvulsant impact against PTZ-induced epileptic seizures. Selenium depletion is associated with the development of neurodegenerative diseases including epileptic seizures, while its supplementation decreases the risk of seizure progression.19 SeNPs was found to protect against cadmium-mediated neuronal impairments by modulating levels of AChE activity and monoaminergic transmission, along with increasing GABA content and decreasing glutamate level in the hippocampus that may be due to its potent antioxidant activity.77 Selenium supplementation increased neurotransmitters level through inhibiting monoamine oxidase (MAO) activity.78–80 SeNPs administration increased NE and 5-HT in the brain tissue following acrylamide intoxication, authors attributed this effect to the inhibition of MAO activity which may be through its ROS scavenging capacity.79 Moreover, selenium administration prevented dopamine loss and degeneration in Parkinson’s disease model.81 Additionally, the anticonvulsant activity of SeNPs in the present study was extended to enhance the GABAergic transmission and suppressing glutamatergic transmission in the hippocampus. Solovyev82 suggested that selenoproteins play a role in the development and functioning of GABAergic neurons. Selenium was found to protect against neuronal hyperexcitability results from excessive production of glutamate.83 In the same context, Ma et al84 showed that selenium blocked neuronal loss and protected the hippocampal HT22 nerve cells against glutamate-induced mitochondrial dysfunction.
Conclusion
In the current work, PTZ injection-induced behavioral changes coupled with oxidative stress in the hippocampal tissue as indicated by the increased neuronal lipoperoxidation and nitric oxide formation, accompanied by a decrease in the level and mRNA expression of the antioxidant enzymes. Additionally, a state of neuroinflammation was recorded represented by the elevated pro-inflammatory cytokines (TNF-α and IL-1β). Neuronal apoptosis was also observed as indicated by the increased pro-apoptotic markers and the decreased anti-apoptotic marker. Moreover, a disturbance in acetylcholinesterase activity, monoaminergic neurotransmission, and imbalance between GABA and glutamate were also recorded. However, SeNPs supplementation reversed the behavioral changes, oxidative damage, histopathological, neurochemical alterations following PTZ-induced epileptic seizures, suggesting that SeNPs may use as an anticonvulsant agent (Figure 10).
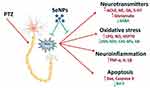 |
Figure 10 Schematic diagram shows the protective role of selenium nanoparticles (SeNPs) against epileptic model induced by pentylenetetrazole (PTZ). |
Disclosure
The authors report no conflict of interests in this work.
References
1. Engelborghs S, D’Hooge R, Paul D. Pathophysiology of epilepsy. Acta neurologicaBelgica. 2001;100:201–213.
2. Jennum P, Sabers A, Christensen J, Ibsen R, Kjellberg J. Socioeconomic outcome of epilepsy surgery: a controlled national study. Seizure. 2016;42:52–56. doi:10.1016/j.seizure.2016.09.016
3. Liu S, Yu W, Lu Y. The causes of new-onset epilepsy and seizures in the elderly. Neuropsychiatr Dis Treat. 2016;12:1425–1434. doi:10.2147/NDT.S107905
4. Mao XY, Zhou HH, Jin WL. Redox-related neuronal death and crosstalk as drug targets: focus on epilepsy. Front Neurosci. 2019;13:512. doi:10.3389/fnins.2019.00512
5. da Fonseca DV, da Silva Maia Bezerra Filho C, Lima TC, de Almeida RN, de Sousa DP. Anticonvulsant essential oils and their relationship with oxidative stress in epilepsy. Biomolecules. 2019;9:12. doi:10.3390/biom9120835
6. Ryan K, Liang LP, Rivard C, Patel M. Temporal and spatial increase of reactive nitrogen species in the kainate model of temporal lobe epilepsy. Neurobiol Dis. 2014;64:8–15. doi:10.1016/j.nbd.2013.12.006
7. Attia GM, Elmansy RA, Elsaed WM. Neuroprotective effect of nilotinib on pentylenetetrazol-induced epilepsy in adult rat hippocampus: involvement of oxidative stress, autophagy, inflammation, and apoptosis. Folia Neuropathologica. 2019;57(2):146–160. doi:10.5114/fn.2019.84423
8. Hashemian M, Anissian D, Ghasemi-Kasman M, et al. Curcumin-loaded chitosan-alginate-STPP nanoparticles ameliorate memory deficits and reduce glial activation in pentylenetetrazol-induced kindling model of epilepsy. Prog Neuropsychopharmacol Biol Psychiatry. 2017;79(Pt B):462–471. doi:10.1016/j.pnpbp.2017.07.025
9. Arafa NM, Abdel-Rahman M, El-khadragy MF, Kassab RB. Evaluation of the possible epileptogenic activity of ciprofloxacin: the role of Nigella sativa on amino acids neurotransmitters. Neurochem Res. 2013;38(1):174–185. doi:10.1007/s11064-012-0905-z
10. Abdel-Rahman M, Arafa N, El-Khadragy M, Kassab R. The neuroprotective role of Nigella sativa extract on ciprofloxacin and pentylenetetrazole treated rats. Afr J Pharm Pharmacol. 2013;7:660–1670. doi:10.5897/AJPP12.897
11. de Kinderen RJ, Evers SM, Rinkens R, et al. Side-effects of antiepileptic drugs: the economic burden. Seizure. 2014;23(3):184–190. doi:10.1016/j.seizure.2013.11.009
12. Gupta J, Fatima MT, Islam Z, Khan RH, Uversky VN, Salahuddin P. Nanoparticle formulations in the diagnosis and therapy of Alzheimer’s disease. Int J Biol Macromol. 2019;130:515–526. doi:10.1016/j.ijbiomac.2019.02.156
13. Anissian D, Ghasemi-Kasman M, Khalili-Fomeshi M, et al. Piperine-loaded chitosan-STPP nanoparticles reduce neuronal loss and astrocytes activation in chemical kindling model of epilepsy. Int J Biol Macromol. 2018;107(Pt A):973–983. doi:10.1016/j.ijbiomac.2017.09.073
14. Rawat M, Yadukrishnan P, Kumar N. Mechanisms of action of nanoparticles in living systems. In: Microbial Biotechnology in Environmental Monitoring and Cleanup. IGI Global; 2018:220–236.
15. Loeschner K, Hadrup N, Hansen M, et al. Absorption, distribution, metabolism and excretion of selenium following oral administration of elemental selenium nanoparticles or selenite in rats. Metallomics. 2014;6(2):330–337. doi:10.1039/c3mt00309d
16. Alajmi RA, Al-Megrin WA, Metwally D, et al. Anti-Toxoplasma activity of silver nanoparticles green synthesized with Phoenix dactylifera and Ziziphus spina-christi extracts which inhibits inflammation through liver regulation of cytokines in Balb/c mice. Biosci Rep. 2019;39(5):5. doi:10.1042/BSR20190379
17. Huo X, Zhang Y, Jin X, Li Y, Zhang L. A novel synthesis of selenium nanoparticles encapsulated PLGA nanospheres with curcumin molecules for the inhibition of amyloid beta aggregation in Alzheimer’s disease. J Photochem Photobiol B. 2019;190:98–102. doi:10.1016/j.jphotobiol.2018.11.008
18. Al-Quraishy S, Dkhil MA, Abdel Moneim AE. Anti-hyperglycemic activity of selenium nanoparticles in streptozotocin-induced diabetic rats. Int J Nanomedicine. 2015;10:6741–6756. doi:10.2147/IJN.S91377
19. Pillai R, Uyehara-Lock JH, Bellinger FP. Selenium and selenoprotein function in brain disorders. IUBMB Life. 2014;66(4):229–239. doi:10.1002/iub.1262
20. Amani H, Habibey R, Shokri F, et al. Selenium nanoparticles for targeted stroke therapy through modulation of inflammatory and metabolic signaling. Sci Rep. 2019;9(1):6044. doi:10.1038/s41598-019-42633-9
21. Wadhwani SA, Shedbalkar UU, Singh R, Chopade BA. Biogenic selenium nanoparticles: current status and future prospects. Appl Microbiol Biotechnol. 2016;100(6):2555–2566. doi:10.1007/s00253-016-7300-7
22. Zhang J, Zhou X, Yu Q, et al. Epigallocatechin-3-gallate (EGCG)-stabilized selenium nanoparticles coated with Tet-1 peptide to reduce amyloid-β aggregation and cytotoxicity. ACS Appl Mater Interfaces. 2014;6(11):8475–8487. doi:10.1021/am501341u
23. Abdelfattah MS, Badr SEA, Lotfy SA, et al. Rutin and selenium co-administration reverse 3-nitropropionic acid-induced neurochemical and molecular impairments in a mouse model of Huntington’s disease. Neurotox Res. 2019.
24. El-Missiry MA, Othman AI, Amer MA, Sedki M, Ali SM, El-Sherbiny IM. Nanoformulated ellagic acid ameliorates pentylenetetrazol-induced experimental epileptic seizures by modulating oxidative stress, inflammatory cytokines and apoptosis in the brains of male mice. Metab Brain Dis. 2019.
25. Dkhil MA, Zrieq R, Al-Quraishy S, Abdel Moneim AE. Selenium nanoparticles attenuate oxidative stress and testicular damage in streptozotocin-induced diabetic rats. Molecules. 2016;21:11. doi:10.3390/molecules21111517
26. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193(1):265–275.
27. Kim YS, Kim SH, Shin J, et al. Luteolin suppresses cancer cell proliferation by targeting vaccinia-related kinase 1. PLoS One. 2014;9(10):e109655. doi:10.1371/journal.pone.0109655
28. Shi F, Zhao P, Li X, Pan H, Ma S, Ding L. Cytotoxicity of luteolin in primary rat hepatocytes: the role of CYP3A-mediated ortho-benzoquinone metabolite formation and glutathione depletion. J Appl Toxicol. 2015;35(11):1372–1380. doi:10.1002/jat.3106
29. Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979;95(2):351–358. doi:10.1016/0003-2697(79)90738-3
30. Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem. 1982;126(1):131–138. doi:10.1016/0003-2697(82)90118-X
31. Ellman GL. Tissue sulfhydryl groups. Arch BiochemBiophys. 1959;82(1):70–77. doi:10.1016/0003-9861(59)90090-6
32. Factor VM, Kiss A, Woitach JT, Wirth PJ, Thorgeirsson SS. Disruption of redox homeostasis in the transforming growth factor-alpha/c-myc transgenic mouse model of accelerated hepatocarcinogenesis. J Biol Chem. 1998;273(25):15846–15853. doi:10.1074/jbc.273.25.15846
33. Paglia DE, Valentine WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med. 1967;70(1):158–169.
34. Aebi H. Catalase in vitro. Methods Enzymol. 1984;105:121–126.
35. Misra HP, Fridovich I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem. 1972;247(10):3170–3175.
36. Abdel Moneim AE. Indigofera oblongifolia prevents lead acetate-induced hepatotoxicity, oxidative stress, fibrosis and apoptosis in rats. PLoS One. 2016;11(7):e0158965. doi:10.1371/journal.pone.0158965
37. Ellman GL, Courtney KD, Andres V
38. Pagel P, Blome J, Wolf HU. High-performance liquid chromatographic separation and measurement of various biogenic compounds possibly involved in the pathomechanism of Parkinson’s disease. J Chromatogr B Biomed Sci Appl. 2000;746(2):297–304. doi:10.1016/S0378-4347(00)00348-0
39. Heinrikson RL, Meredith SC. Amino acid analysis by reverse-phase high-performance liquid chromatography: precolumn derivatization with phenylisothiocyanate. Anal Biochem. 1984;136(1):65–74. doi:10.1016/0003-2697(84)90307-5
40. Almeer RS, AlBasher GI, Alarifi S, Alkahtani S, Ali D, Abdel Moneim AE. Royal jelly attenuates cadmium-induced nephrotoxicity in male mice. Sci Rep. 2019;9(1):5825. doi:10.1038/s41598-019-42368-7
41. Al-Quraishy S, Dkhil MA, Abdel-Gaber R, Zrieq R, Hafez TA, Mubaraki MA, Abdel Moneim AE. Myristica fragrans seed extract reverses scopolamine-induced cortical injury via stimulation of HO-1 expression in male rats. Environ Sci Pollut Res. 2020;27(11):12395–12404. doi:10.1007/s11356-020-07686-8
42. Hasan A, Morshed M, Memic A, Hassan S, Webster TJ, Marei HE. Nanoparticles in tissue engineering: applications, challenges and prospects. Int J Nanomedicine. 2018;13:5637-5655. doi:10.2147/IJN.S153758
43. Samokhina E, Samokhin A. Neuropathological profile of the pentylenetetrazol (PTZ) kindling model. Int J Neurosci. 2018;128(11):1086–1096. doi:10.1080/00207454.2018.1481064
44. Al Omairi NE, Radwan OK, Alzahrani YA, Kassab RB. Neuroprotective efficiency of Mangiferaindica leaves extract on cadmium-induced cortical damage in rats. Metab Brain Dis. 2018. doi:10.1007/s11011-018-0222-6
45. Zhu X, Dong J, Han B, et al. Neuronal nitric oxide synthase contributes to PTZ kindling epilepsy-induced hippocampal endoplasmic reticulum stress and oxidative damage. Front Cell Neurosci. 2017;11:377. doi:10.3389/fncel.2017.00377
46. Zsurka G, Kunz W. Mitochondrial dysfunction and seizures: the neuronal energy crisis. Lancet Neurol. 2015;14:956–966. doi:10.1016/S1474-4422(15)00148-9
47. Uchida K, Shiraishi M, Naito Y, Torii Y, Nakamura Y, Osawa T. Activation of stress signaling pathways by the end product of lipid peroxidation. 4-hydroxy-2-nonenal is a potential inducer of intracellular peroxide production. J Biol Chem. 1999;274(4):2234–2242. doi:10.1074/jbc.274.4.2234
48. Kassab RB, Lokman MS, Essawy EA. Neurochemical alterations following the exposure to di-n-butyl phthalate in rats. Metab Brain Dis. 2019;34(1):235–244. doi:10.1007/s11011-018-0341-0
49. Ikwegbue PC, Masamba P, Oyinloye BE, Kappo AP. Roles of heat shock proteins in apoptosis, oxidative stress, human inflammatory diseases, and cancer. Pharmaceuticals. 2018;11(1):2. doi:10.3390/ph11010002
50. Hussein AM, Adel M, El-Mesery M, Abbas KM, Ali AN, Abulseoud OA. L-carnitine modulates epileptic seizures in pentylenetetrazole-kindled rats via suppression of apoptosis and autophagy and upregulation of hsp70. Brain Sci. 2018;8(3):45. doi:10.3390/brainsci8030045
51. Dericioglu N, Soylemezoglu F, Gursoy-Ozdemir Y, Akalan N, Saygi S, Dalkara T. Cell death and survival mechanisms are concomitantly active in the hippocampus of patients with mesial temporal sclerosis. Neuroscience. 2013;237:56–65. doi:10.1016/j.neuroscience.2013.01.050
52. Banach M, Piskorska B, Czuczwar SJ, Borowicz KK. Nitric oxide, epileptic seizures, and action of antiepileptic drugs. CNS Neurol Disord Drug Targets. 2011;10(7):808–819. doi:10.2174/187152711798072347
53. Mueller SG, Trabesinger AH, Boesiger P, Wieser HG. Brain glutathione levels in patients with epilepsy measured by in vivo (1)H-MRS. Neurology. 2001;57(8):1422–1427. doi:10.1212/WNL.57.8.1422
54. Lee SH, Choi BY, Kho AR, et al. Protective effects of protocatechuic acid on seizure-induced neuronal death. Int J Mol Sci. 2018;19:1.
55. Ahmed HH, Abd El-Maksoud MD, Abdel Moneim AE, Aglan HA. Pre-clinical study for the antidiabetic potential of selenium nanoparticles. Biol Trace Elem Res. 2016.
56. Shalby AB, El-Maksoud MDA, Moneim AEA, Ahmed HH. Antifibrotic candidates of Selenium nanoparticles and selenium in the experimental model. J Appl Pharm Sci. 2017;7(09):191–198.
57. Naziroglu M, Muhamad S, Pecze L. Nanoparticles as potential clinical therapeutic agents in Alzheimer’s disease: focus on selenium nanoparticles. Expert Rev Clin Pharmacol. 2017;10(7):773–782. doi:10.1080/17512433.2017.1324781
58. AlBasher G, Alfarraj S, Alarifi S, et al. Nephroprotective role of selenium nanoparticles against glycerol-induced acute kidney injury in rats. Biol Trace Elem Res. 2019.
59. Cao C, Li X, Qin L, et al. High selenium yeast mitigates aluminum-induced cerebral inflammation by increasing oxidative stress and blocking NO production. Biometals. 2018;31(5):835–843. doi:10.1007/s10534-018-0128-0
60. Tawfik KM, Moustafa YM, El-Azab MF. Neuroprotective mechanisms of sildenafil and selenium in PTZ-kindling model: implications in epilepsy. Eur J Pharmacol. 2018;833:131–144. doi:10.1016/j.ejphar.2018.05.035
61. Malyar RM, Li H, Liu D, et al. Selenium/Zinc-enriched probiotics improve serum enzyme activity, antioxidant ability, inflammatory factors and related gene expression of Wistar rats inflated under heat stress. Life Sci. 2020;248:117464. doi:10.1016/j.lfs.2020.117464
62. Huang WY, Lin S, Chen HY, et al. NADPH oxidases as potential pharmacological targets against increased seizure susceptibility after systemic inflammation. J Neuroinflammation. 2018;15(1):140. doi:10.1186/s12974-018-1186-5
63. Demirci K, Naziroglu M, Ovey IS, Balaban H. Selenium attenuates apoptosis, inflammation and oxidative stress in the blood and brain of aged rats with scopolamine-induced dementia. Metab Brain Dis. 2017;32(2):321–329. doi:10.1007/s11011-016-9903-1
64. Singh N, Saha L, Kumari P, et al. Effect of dimethyl fumarate on neuroinflammation and apoptosis in pentylenetetrazol kindling model in rats. Brain Res Bull. 2019;144:233–245. doi:10.1016/j.brainresbull.2018.11.013
65. Yousef S, Omar A, Fahad A, et al. The neuroprotective role of coenzyme Q10 against lead acetate-induced neurotoxicity is mediated by antioxidant, anti-inflammatory and anti-apoptotic activities. Int J Environ Res Public Health. 2019;16(16):2895. doi:10.3390/ijerph16162895
66. Long M, Yang S, Wang Y, et al. The protective effect of selenium on chronic zearalenone-induced reproductive system damage in male mice. Molecules. 2016;21:12. doi:10.3390/molecules21121687
67. Pauletti A, Terrone G, Shekh-Ahmad T, et al. Targeting oxidative stress improves disease outcomes in a rat model of acquired epilepsy. Brain. 2019;142(7):e39. doi:10.1093/brain/awz130
68. de Sales Santos IM, Feitosa CM, de Freitas RM. Pilocarpine-induced seizures produce alterations on choline acetyltransferase and acetylcholinesterase activities and deficit memory in rats. Cell Mol Neurobiol. 2010;30(4):569–575. doi:10.1007/s10571-009-9481-4
69. Kurian MA, Gissen P, Smith M, Heales S
70. Statnick MA, Dailey JW, Jobe PC, Browning RA. Abnormalities in brain serotonin concentration, high-affinity uptake, and tryptophan hydroxylase activity in severe-seizure genetically epilepsy-prone rats. Epilepsia. 1996;37(4):311–321. doi:10.1111/j.1528-1157.1996.tb00565.x
71. Tchekalarova J, Pechlivanova D, Atanasova T, Markova P, Lozanov V, Stoynev A. Diurnal variations in depression-like behavior of Wistar and spontaneously hypertensive rats in the kainate model of temporal lobe epilepsy. Epilepsy Behav. 2011;20(2):277–285. doi:10.1016/j.yebeh.2010.12.021
72. Rezaei M, Sadeghian A, Roohi N, Shojaei A, Mirnajafi-Zadeh J. Epilepsy and dopaminergic system. Physiol Pharmacol. 2017;21:1–14.
73. Briere R, Sherwin AL, Robitaille Y, Olivier A, Quesney LF, Reader TA. Alpha-1 adrenoceptors are decreased in human epileptic foci. Ann Neurol. 1986;19(1):26–30. doi:10.1002/ana.410190106
74. Giorgi F, Pizzanelli C, Biagioni F, Murri L, Fornai F. The role of norepinephrine in epilepsy: from the bench to the bedside. Neurosci Biobehav Rev. 2004;28:507–524. doi:10.1016/j.neubiorev.2004.06.008
75. Cavalcante TMB, De Melo JMAJ, Lopes LB, et al. Ivabradine possesses anticonvulsant and neuroprotective action in mice. Biomed Pharmacother/Biomedecine&pharmacotherapie. 2019;109:2499–2512. doi:10.1016/j.biopha.2018.11.096
76. Barker-Haliski M, White HS. Glutamatergic mechanisms associated with seizures and epilepsy. Cold Spring Harb Perspect Med. 2015;5(8):a022863. doi:10.1101/cshperspect.a022863
77. Al Kahtani MA. Effect of both selenium and biosynthesized nanoselenium particles on cadmium-induced neurotoxicity in albino rats. Hum Exp Toxicol. 2019;960327119880589.
78. Ansari MA, Ahmad AS, Ahmad M, et al. Selenium protects cerebral ischemia in rat brain mitochondria. Biol Trace Elem Res. 2004;101(1):73–86. doi:10.1385/BTER:101:1:73
79. Abozaid O, El-sonbaty S, Ezz W. Effect of Acrylamide on neurotransmitters and acetyl-cholinesterase activity in the brain of rats: therapeutic effect of ferulic acid and selenium nanoparticles. 2017.
80. Tang Y-L, Wang S-W, Lin S-M. Both inorganic and organic selenium supplements can decrease brain monoamine oxidase B enzyme activity in adult rats. Br J Nutri. 2008;100(3):660–665. doi:10.1017/S0007114508911594
81. Zafar KS, Siddiqui A, Sayeed I, Ahmad M, Salim S, Islam F. Dose-dependent protective effect of selenium in rat model of Parkinson’s disease: neurobehavioral and neurochemical evidences. J Neurochem. 2001;57(8):438–446. doi:10.1046/j.1471-4159.2003.01531.x
82. Solovyev ND. Importance of selenium and selenoprotein for brain function: from antioxidant protection to neuronal signalling. J Inorg Biochem. 2015;153:1–12. doi:10.1016/j.jinorgbio.2015.09.003
83. Savaskan NE, Brauer AU, Kuhbacher M, et al. Selenium deficiency increases susceptibility to glutamate-induced excitotoxicity. FASEB J. 2003;17(1):112–114. doi:10.1096/fj.02-0067fje
84. Ma YM, Ibeanu G, Wang LY, et al. Selenium suppresses glutamate-induced cell death and prevents mitochondrial morphological dynamic alterations in hippocampal HT22 neuronal cells. BMC Neurosci. 2017;18(1):15. doi:10.1186/s12868-017-0337-4
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.