Back to Journals » Journal of Blood Medicine » Volume 11
Salvage Therapy for Hodgkin’s Lymphoma: A Review of Current Regimens and Outcomes
Authors Castagna L , Santoro A, Carlo-Stella C
Received 16 June 2020
Accepted for publication 6 October 2020
Published 27 October 2020 Volume 2020:11 Pages 389—403
DOI https://doi.org/10.2147/JBM.S250581
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Martin H Bluth
Luca Castagna,1 Armando Santoro,1,2 Carmelo Carlo-Stella1,2
1Humanitas Clinical and Research Center, IRCCS, Rozzano, Milan 20089, Italy; 2Humanitas University, Department of Biomedical Sciences, Pieve Emanuele, Milan 20090, Italy
Correspondence: Luca Castagna
Humanitas Clinical and Research Center, IRCCS, Via Manzoni 56, Rozzano, Milan 20089, Italy
Email [email protected]
Abstract: Relapse/refractory Hodgkin lymphoma patients are still a clinical concern. Indeed, despite more effective first-line chemotherapy regimens and better stratification of unresponsive patients by clinical factors and use of early PET, roughly one-third of such patients need salvage chemotherapy and consolidation with high-dose chemotherapy. In this paper, the authors review the different salvage treatments, with special emphasis on newer combinations with brentuximab vedotin or check point inhibitors. The overall response rate is constantly increasing, with a complete remission rate approaching 80%. Functional response evaluation by PET imaging is a strong predictive factor of longer survival, and more sophisticated tools, such as detection of circulating tumour DNA, are emerging to refine the disease-status assessment after treatment. Consolidation by high-dose chemotherapy is still considered the standard of care in chemosensitive patients, leading to a high fraction of patients towards long-term disease control. Maintenance therapy with BV is now approved, reducing disease relapse/progression. An increasing number of Hodgkin lymphoma patients will be cured after first- and second-line therapy, and long-term toxicity needs to be continuously assessed and avoided.
Keywords: Hodgkin lymphoma, refractory/relapsed disease, checkpoint inhibitors, brentuximab vedotin, high-dose chemotherapy
Introduction
The treatment of Hodgkin lymphoma (HL) is considered a paradigm of optimization of therapeutic resources and strategies to maximize responses while minimizing acute and mainly long-term toxicities.
The number of patients not cured by first-line therapy has changed over time due to the improvement of drug combinations employed in first-line treatment, either from the activity and safety profiles. An important point is to define the meaning of relapsed and refractory patients. In this review, patients were defined as refractory (Refr) when they did not show any response or progressed in the first 3 months after the end of first-line chemotherapy (CT); early relapsed (E-Rel) when relapse was diagnosed after 3 months and before 12 months after the end of first-line CT; late relapsed (L-Rel) when relapse occurred after 12 months.
Analysing data from several prospective, randomized studies, we can conclude that 2%-30% of HL patients (all stages together) required salvage chemotherapy (CT) because of a relapsed/refractory (R/R) status after first-line treatment. Two factors had an impact on the rate of R/R in patients: the disease or patient characteristics (early vs late stage, international prognostic score, age) and the intensity of initial treatment (BEACOPP vs ABVD). The prognosis of R/R HL patients varied based on both response and duration after first-line therapy. Indeed, in the era before the extensive use of PET to stage and evaluate the response, many papers identified some clinical factors affecting the prognosis of R/R patients, resulting in different clinical scores summarized in Table 1. The prognostic score from the German group (comprising stage IV, E-rel, and anaemia) was also validated in a prospective randomized trial.1 However, the advent of PET for treatment evaluation of HL patients changed the paradigm (Table 2) because most of the prognostic clinical factors were cancelled out by the PET results. In a seminal paper from MSKCC, Moskowitz et al reported that PET positivity and the presence of extranodal disease (END) at the time of relapse were the only prognostic factors affecting the outcome. Three groups of patients were identified as having significantly different 3-y event-free survival (EFS: PET negative without END, PET negative with END, and PET positive.2 In an international cooperative study, Brockelmann et al analysed more than 1000 patients, identifying 5 clinical risk factors and 4 prognostic groups (0 vs 1 vs 2 vs 3–5) with different survival after salvage treatment. Indeed, stage IV at relapse, bulky disease (>5 cm), time to relapse <3 months, inadequate response to salvage CT (defined as less than partial remission evaluated by CT scan or PET), and performance status >1 were significantly associated to survival after autologous stem cell transplantation.3
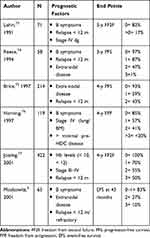 |
Table 1 Prognostic Factors Identified in the Pre-PET Era |
 |
Table 2 Studies Evaluating PET Predictive Significance in HL |
More recently, a review and meta-analysis of studies investigating the role of pre-HDC PET stated that there was moderate evidence that PET was valuable for predicting the outcome.4
Even if relapsed and refractory patients are grouped as unique, refractory patients, those progressing during first therapy or relapsing shortly after (<3 months from the end of treatment) represent a homogenous cohort from the prognostic point of view since they historically have a dismal prognosis with conventional treatment strategy, as reported in Table 3.
 |
Table 3 Clinical Results Obtained in Patients Refractory to First-Line CT |
Salvage Therapies
Conventional Salvage CT
To date, randomized studies have not been conducted to establish the best salvage regimen in R/R HL. In Table 4–6, we summarize the results of different salvage regimens. In Table 4, the main results of platinum-based salvage CT are reported. In all CT schemes but one, cis-platinum (CDDP) was used. Only one regimen included carboplatin instead of CDDP.5 The total dose of CDDP was 100 mg/m2 in all but one regimen, which used 75 mg/m2.6 Furthermore, in one study, DHAP was administered using a more intensive schedule every 16 days instead of every 21 days.7 An important point is the number of included patients with refractory HL, consisting of a biologically less sensitive disease with a lower response rate. Indeed, the included refractory patients ranged from 9% to 41%. Another point is the disease-response evaluation performed by CT scan, which can explain the quite low complete remission (CR) rate, namely, from 17% to 50%. Last, toxicity expressed as toxic death is not a concern with platinum-based regimens. The frequency of haematological toxicity varies from 9%6 to 100%.8
 |
Table 4 Platinum-Based Salvage CT Reported in the Literature |
 |
Table 5 Gemcitabine-Based Salvage CT |
 |
Table 6 Salvage CT Without Gemcitabine and Platine-Derived Compounds |
In Table 5 and 4 studies reported the outcomes using a gemcitabine-based CT regimen. In Bartlett’s study,9 liposomal doxorubicin was used in combination with gemcitabine and vinorelbine, while in the 2 other studies from the same group, ifosfamide10 or bendamustine11,12 were used in combination with the same 2 drugs. The overall response rate (ORR) was similar (from 69% to 83%), but the CR rate was higher with IGEV and BeGEV, even if only 51 patients were transplant-naïve in Bartlett’s study. The BeGEV study is unique because PET was used to evaluate the response at the end of treatment, probably explaining the high CR rate (73%). Overall, these regimens are well tolerated, and the toxicity is mostly haematological, with grade 3–4 neutropenia observed in 24–51% and thrombopenia in 14–16% of patients. Gemcitabine (1000 mg/m2) was used in combination with bendamustine (240 mg/m2 in 2 days) in a prospective Phase I/II study. Twenty-six heavily pretreated (46% relapsed after HDC, 69% after BV, and 15% after checkpoint inhibitors) patients were included, and the ORR and CR rate were 69% and 46%, respectively. Grade 3–4 adverse events comprised lymphopenia, thrombopenia, anaemia, and pneumonia.13 In Table 6, we report salvage CT not containing gemcitabine or CDDP. The ORR was similar to other regimens as well as the CR rate, evaluated by CT scan. Overall, even if the comparison between all these studies should be taken with caution, the ORR of CDDP-based and gemcitabine-based studies was similar, ranging from 67%14 to 89%.7 On the other hand, the difference in terms of the CR rate was more pronounced, ranging from 17%6 to 73%,11 but not all studies used PET scans to evaluate the response. The toxicity of all these schemes was good because they were mainly haematological. All CT regimens were able to mobilize stem cells in peripheral blood.
Some CT-based salvage regimens have been used in combination with biological drugs to improve the ORR and the CR rate (Table 7). Anti-CD20 monoclonal antibodies have been used in combination with conventional salvage CT based on the expression of the target antigen on Reed Stenberg cells, on the putative B-lineage of stem cell origin and on the presence of B-cells in the microenvironment. In a prospective Phase 2 study, Martinez et al employed the combination of ofatumumab plus ESHAP in 62 R/R HL patients. One-third of patients had Refr disease. Disease evaluation was performed by PET scan. The ORR was 73%, and the CR rate was 44%. Tolerance was good as well as the capability of CD34-positive cell mobilization.15
 |
Table 7 Salvage CT Associating CT and BV and Salvage Therapy with BV Alone |
BV Alone and BV-Based Regimens
One of the most relevant steps forward in the R/R HL treatment was the introduction of anti-CD30 monoclonal antibody conjugated to mono-methyl auristatin molecule (brentuximab vedotin, BV). Several studies have been published mostly in the setting of R/R patients (Table 7). BV was first used as a single drug and, more recently, in combination with conventional CT.
As a single drug, the ORR was encouraging, ranging from 60% to 75%, with CR rates of 17%-44%. It should be noted that in all these studies, most patients had relapsed after previous HDC (from 33% to 100%). Overall, the drug was well tolerated, with peripheral neuropathy as a peculiar side effect affecting 42% of patients (8% grade 3) and haematopoietic toxicity (mainly neutropenia 19%) as the second most frequent adverse event.16 In one study, the activity of BV as a single agent was exclusively evaluated in patients not previously treated by HDC. In this investigation, 37 patients were included, 65% were refractory to first-line CT, and the ORR was 68% (CR rate 35%).17 Non-responding patients were treated with conventional salvage CT, and overall, 86% proceeded to HDC. In a prospective Phase II study, Moskowitz et al treated 46 patients with BV alone, and the CR rate was 27%. The remaining patients, who were PET positive after BV, were treated with high-dose ICE, and the CR rate was 69%. Overall, 76% of the patients achieved CR.18
In combination schemes, BV has been associated mainly with bendamustine (benda).19–24 In all but one study, the doses of BV and bendamustine were the same (BV 1.8 mg/kg and bendamustine 180 mg/m2 in 2 days). In a phase I/II trial, different doses of BV (1.2 and 1.8 mg/kg) and benda (70, 80, 90 mg/m2) were tested, and the definitive dose level was associated with a regular dose of BV and 90 mg/m2 of benda.20 In only one study,22 the dose of bendamustine was higher (240 mg/m2 in 2 days). The tolerance was acceptable, and G3-4 toxicity consisted of neutropenia (23%-35%), lung infections (4%-14%) and peripheral neuropathy (2%-11%). With a higher dose of bendamustine, an increased incidence of CMV reactivation (20%) was observed. The ORR, evaluated by PET in all studies, was interesting, ranging from 71% to 100%. This result is of particular value if we consider that a high proportion of the patients were refractory to previous CT lines (from 50% to 100%). The association of BV plus conventional polychemotherapy (poly-CT) was reported in 3 studies. In the first paper,25 28 patients (refractory 43%) were treated with BV-IGEV as first or subsequent salvage therapy. Grade 3–4 haematopoietic toxicity (neutropenia) was observed in 96% of patients, and febrile neutropenia was observed in 57%. Peripheral neuropathy was rare (4%). The ORR was 96% (CR rate 70%). The second trial was a Phase I study combining BV with DHAP.26 Only 12 patients were included, and 3 dose levels of platinum and cytarabine were tested with a fixed dose of BV (1.8 mg/kg), and the full dose levels were considered feasible. The ORR was 92% (CR rate 92%). In the third phase I/II study, BV was used in combination with ESHAP.27 Here, the authors tested 3 dose levels of BV (0.9, 1.2, and 1.8 mg/kg) with a standard dose of ESHAP. In the phase II part of the study, the BV dose was 1.8 mg/kg. Grade 3–4 haematological toxicity was observed in 50% of patients; febrile neutropenia in 8% and CMV reactivation in 3%. The ORR was 91% (CR rate 70%). Finally, Michallet et al reported on 11 patients treated with the combination of BV plus GVD. The ORR was 100% (CR rate 72%), and the 1-y OS was 60%.28
In some studies, the ORR or CR rates were separately reported for Refr patients. For the BV-benda association, the differences in terms of ORR and CR rate were not significant between Refr and other categories of disease (92% vs 85% and 73% vs 64%, respectively.19,23,24 On the other hand, in the BV-ESHAP, the CR rate was lower for Refr patients than for the others (70% vs 37%, respectively).25 We do not have an immediate explanation for why disease characteristics (Refr vs Rel) matter only after BV-polyCT and not after BV-benda, but a potential synergistic effect between benda and BV could be a possible explanation. Furthermore, the disease response criteria differed in the various studies, as follows: Cheson 200729 in the LaCasce,19 O’Connor,20 and Garcia-Sanz27 studies; RECIL criteria30 in the Lannitto24 study; and Lugano criteria31 in the Broccoli23 and Abuelgasim25 studies. In Moskowitz’s study,18 PET negative was defined as DS 1 and 2.
The efficacy of BV alone was tested in patients not responding to first salvage therapy. The response rate in these poor prognosis patients was low, and the outcome was dismal. In the first study, Eyre et al treated 99 patients in a multicentre retrospective study, and the ORR and CR rate were 56% and 29%, respectively; 61% of patients received a transplant (15% HDC), achieving a much longer overall survival than those not transplanted.32 Picardi et al treated 20 patients with BV in combination with bendamustine in a small prospective study. The CR rate was 80%, and 70% of patients were consolidated by HDC, reaching a 2-y PFS of 97%.22 Several years ago, Villa et al treated 19 patients who were chemorefractory to first salvage CT (GDP) by mini-BEAM. The CR rate was 32%, but the 2-y OS and PFS rates were only 20% and 11%, respectively.33
Check-Point Inhibitors (CPIs)
The introduction of monoclonal antibodies against checkpoint molecules such as PD1 in the armamentarium of treatments for poor-prognosis HL has been a major step forward. HL cells are intrinsically susceptible to the inhibition of PD1 because all of them carry out a copy gain of chromosome 9p24.1, leading to overexpression of PD1 ligands (PDL1 and 2) on tumoural cells. The CPI sensitizes Reed Stenberg cells to the immunological attack masking PD1 and blocking the inhibitory effect on T-cells. In Table 8, we report the results published to date, using 2 different CPIs, nivolumab and pembrolizumab, as single agents in advanced HL patients. In the first group, 23 patients34 relapsing after HDC (78%) or BV (78%) and nivolumab (3 mg/m2, every 15 days) showed a high ORR, but the CR rate was low (17%). The toxicity profile of the drug was linked to the overreactivity of the immune system with autoimmune complications. In a second prospective phase II trial, 80 patients were treated with nivolumab, the ORR was 66%, and the CR rate was 9%. All patients had relapsed after HDC, and 54% were not responsive to BV. The median time to detect an objective response was 2.1 months (meaning 4 doses of drug), and the median duration of response was almost 8 months. The higher the expression of PDL1 on PAX5-positive cells (Reed Sternberg cells), the higher was the probability of achieving an objective response. Adverse events were recorded in 41% of patients, and the most frequent events were neutropenia and lipase increase.35 Armand et al published the results of a previous study after an extended follow-up with more patients. Three cohorts of patients (n= 243) were indeed included, and the results were analysed in these different patient types: relapsing after HDC, relapsing after HDC and BV, and BV-naïve. For the whole cohort, the ORR and CR rate were 69% and 16%, respectively. The ORR and CR rate were similar in the 3 cohorts of patients.36 The authors outlined that atypical patterns of response could explain why the time to next treatment was longer than progression-free survival. In a real-world analysis from the US, 53 patients were included; the ORR was 68%, and the CR rate was 45%. The patient characteristics were similar to those in prospective studies because the median number of previous CT lines was 4; 53% of patients had relapsed after HDC, and 19% had relapsed after allogeneic stem cell transplantation. An interesting point addressed in this analysis was the high efficacy of conventional therapy (ORR 70%) when patients failed CPI.37 In a French retrospective study comprising 78 patients and with a long follow-up (median 34 months), the ORR was 68% and the CR rate was 38%.38 Another retrospective study from Turkey reported on 82 heavily pretreated patients (5 median CT lines, 70% relapsed after HDC). The ORR was 64%, and the CR rate was 21%.39 Nivolumab was associated with BV in a prospective phase I/II study, and the interim results were recently published. Sixty-two patients were included; the ORR was 83%, and the CR rate was 61%. Moreover, 87%33 of patients proceeded to consolidation by HDC, and of these, 77% proceeded directly after BV-nivolumab. Patients not responding to BV-nivo were sensitive to conventional chemotherapy, and in these patients, the ORR was 80% and the CR rate was 40%. The tolerance was good, and grade 3–4 adverse events occurred in 31% of patients.40 Forty-two patients out of 60 received HDC after stem cell mobilization by growth factor (G-CSF) alone or by G-CSF plus chemotherapy or plerixafor at a median time from the end of the BV/nivo treatment period of 9 days. No significant toxicities were recorded after the HDC course.40
 |
Table 8 Results from Clinical Trials Using CPIs in R/R HL |
It is quite evident that the CR rate was higher in retrospective than in prospective studies. This difference was probably due to PET interpretation, which is particularly troublesome after CPI. Indeed, these difficulties have been taken into account in the adapted Lugano response criteria,31 where the definition of progressive disease was modified by introducing 3 different scenarios: immune response (IR) 1 (defined as a ≥ 50% increase in the sum of the products of the diameter in the first 12 weeks); IR2: <50% increase in the sum of the products of the diameter in the first 12 weeks with new lesions or ≥50% increase in the product of the perpendicular diameters of a lesion or set of lesions at any time during treatment; IR3: increase in FDG uptake without a concomitant increase in lesion size meeting the criteria for PD). In a Cochrane review, the data on the efficacy of nivolumab, which were derived from a few studies, were considered too sparse and limited to make a strong recommendation on its use in R/R HL.41 Chen et al used pembrolizumab (200 mg every 3 weeks) as CPI in 210 patients, subdivided into 3 cohorts: relapsing after HDC and BV; relapsing after CT and BV; relapsing after HDC. The ORR and CR rate were 69% and 22%, respectively. The response rate did not seem different between the 3 cohorts.42 The toxicity profile was also similar to that reported with nivolumab. In both prospective studies with nivolumab36 and pembrolizumab,42 one of the 3 cohorts included patients not eligible for HDC. In Armand’s study, the ORR in this cohort of patients (cohort A) was similar to that in the others, while the CR rate was higher (29% vs 13% and 12%). The 1-year OS was in line with that of the whole population. In this study, 9 patients in this cohort underwent allogeneic stem cell transplantation, but it was not reported if any patients received consolidation by HDC.36 In the second study, cohort 2 included patients ineligible for HDC and failing BV. The ORR and CR rate were superimposable on the other cohorts, as well as survival. In this study, only 4 patients, out of 210, received HDC.42 However, in Herrera’s study, R/R HL received the combination of BV plus nivolumab as first salvage therapy.
Finally, a third fully humanized CPI, sintilimab, was tested in China in a multicentre phase II study including 96 R/R HL patients. The ORR was 80%, and the CR rate was 31%. The drug was well tolerated. It should be noted that in this cohort, few patients were previously treated with BV and HDC.43
Predictive and Prognostic Factors of Response
Other than the clinical factors (Table 1), interim PET evaluation of the response during salvage therapy is considered to be a surrogate marker of chemosensitive disease and survival. This assumption is supported by the results of prospective randomized studies during first-line therapy in advanced disease, leading to an improvement in survival and a reduction in toxicity.44–46 In the context of R/R HL, the value of interim PET is well established as a strong factor predicting survival, usually after consolidation of the response by HDC.47–49 Less clear was the role of interim PET as a predictor of response. Our group evaluated R/R HL treated by conventional salvage CT (IGEV), where PET was performed after 2 courses (PET2). PET2-positive patients achieved less CR after 2 supplemental courses, and survival was significantly reduced.50 The prognostic value of interim PET during CPI therapy was analysed in 45 patients in a multicentre retrospective study. The first interim PET was performed after a median of 2 months. Responses were classified following the Lugano and LYRIC criteria, and complete metabolic response (CMR) was observed in 29% of the patients; partial MR, in 36%; no MR, in 9%; and progressive MR, in 27%. The survival of patients was correlated and significantly different based on the response obtained. It is interesting to note that there was a direct correlation between progressive disease defined by the Lugano criteria and the 3 categories of progression defined according to the LYRIC criteria. In other words, all patients classified as having immunological responses 1, 2, and 3 were progressive following PET evaluation.51 If these results will be confirmed in a large prospective study, early interim PET during CPI could be used as a prognostic marker of response and survival, helping to stratify patients for different treatments.
Recently, a more sophisticated and appealing molecular method was introduced to evaluate the sensitivity to treatment based on the tracking of circulating tumour DNA (ctDNA). Spina et al elegantly showed that ctDNA is representative of native tumoural DNA and contains different mutations. The most frequent mutation is located on the STAT6 gene (40%). In the setting of R/R HL, ctDNA contains either original mutations or other mutations not presented at diagnosis, confirming clonal evolution. In classical HL, the researchers identified a high number of mutations similar to that found in solid tumours. The performance of ctDNA to identify resistant clones was coupled to interim PET response, and a high log reduction of ctDNA was predictive of cure even in PET-positive patients.52
Serum biomarkers such as thymus and activation-regulated chemokine (TARC) and galectin-1, both highly expressed on Reed Sternberg cells, can be useful to monitor the response during first-line treatment.53–55 However, TARC was prospectively proven to be predictive of response in only one study where 109 R/R HL patients were treated with panobinostat.56
Outcome After HDC and Maintenance Therapy
HDC followed by autologous stem cell support is considered the standard for R/R HL patients responding to first salvage chemotherapy. This was well established by 2 randomized studies,57,58 where OS was almost 80% and 71%, event-free survival was 53%57 and freedom from treatment failure was 55%.58 In these studies, the conventional arm was represented by mini-BEAM and dexa-BEAM. A third randomized study compared the standard arm (salvage CT plus HDC) to an experimental intensified arm (salvage CT, cyclophosphamide, methotrexate, and etoposide). The OS and PFS were not different (87% vs 80% and 72% vs 67%, respectively), while the PFS was influenced by the factors included in the German score (stage IV, anaemia, and early or multiple relapse).1 In Table 4–7, we report the survival obtained in patients treated with salvage CT and HDC.
Recently, a supposed improvement in survival by adding a maintenance treatment after HDC has been reported. The AETHERA study randomized patients at high risk of relapse after HDC (relapsed or progressive less than 12 months from the end of frontline therapy; refractory HL or extranodal involvement at the time of relapse) to receive BV (up to 16 cycles) or placebo. This study showed that PFS was significantly enhanced with BV, with tolerable neurological toxicity. The five-year PFS in the BV arm was 59% compared to 41% in the placebo arm, and fewer patients in the BV arm required additional therapy (32% vs 54%).59 Recently, a prospective phase 2 study reported on 30 patients treated with HDC. Among the included patients, 87% had at least 1 criterion corresponding to the inclusion criteria of the AETHERA study. They received 8 courses of pembrolizumab starting from day +21. The 19-month PFS was 85%, and the tolerance was good, with grade 3–4 toxic complications of 30%.60
Allogeneic Stem Cell Transplantation in R/R HL
Although many patients can be cured by conventional chemoradiotherapy or autologous transplantation, some of them remain refractory or relapse. These refractory patients can finally receive allogeneic stem cell transplantation (allo-SCT). Historically, HL cells are not considered to be sensitive to immunocompetent cells, and this was well explained by an array of cellular abnormalities that transform HL cells in ghost cells to the immune system.61,62 The most interesting of this aberrant phenotype is the overexpression of PDL1 and PDL2 molecules, due to recurrent chromosome 9p24.1 amplification, which are targeted by CPI. The activity of CPI against HL cells changed the paradigm of resistance of HL to immune cells.
Allo-SCT is considered the prototype of adoptive immunotherapy, but at least in Europe, only 2–3% of HL patients received allo-SCT,63 probably because of supposed immunoresistance and high toxicity. In recent years, many advances have been made in the allo-SCT field, such as the introduction of reduced intensity or nonmyeloablative conditioning regimens (RIC and NMAC), improvement of infectious complication control, HLA typing resolution, graft-versus-host disease therapy and prophylaxis. Altogether, these improvements lead to lower nonrelapse mortality and better survival.64
Moving to the results obtained with allo-SCT in HL, we summarized the clinical results in Tables 9 and 10. In Table 9, we report the results obtained using so-called conventional donors, namely, matched related (MRD) and unrelated donors (MUD). It is evident that survival and toxicities were heterogeneous due to selection bias. Chemosensitive disease before allo-SCT ranged from 44% to 92%, the incidence of grade 2–4 acute GVHD ranged from 16% to 60%, chronic GVHD ranged from 14% to 73%, and OS and PFS ranged from 25% to 82% and from 20% to 54%, respectively. The relapse rate ranged from 15% to 81%, and the NRM ranged from 3% to 33%. Only 1 prospective study had been published to date,65 where 67% of patients were in CR or PR before allo-SCT from MRD or MUD. The incidence of acute and chronic GVHD was 32% and 44%, respectively, and the survival was disappointing (OS 43%, PFS 24%) due to the high relapse rate (59%). In recent years, an increasing number of patients have received allo-SCT from haploidentical donors. Different platforms have been developed, and most of them use unmanipulated stem cell sources. The most popular platform, at least in Europe and the US, was based on the use of post-transplantation cyclophosphamide (PT-Cy), as pioneered by the Baltimore group.66 In Table 10, we summarized the results obtained using unmanipulated stem cell support and PT-Cy. Disease before allo-SCT was considered chemosensitive in 46% to 93% of patients, the incidence of grade 2–4 acute GVHD was detected in 24% to 43% of patients, and chronic GVHD was detected in 9% to 35% of patients. The OS and PFS ranged from 58% to 81% and from 42% to 66%, respectively. The relapse rate ranged from 15% to 55%, and the NRM ranged from 4% to 21%. Even if it is difficult to compare the results after allo-SCT from conventional donors to those with haploidentical donors, it seems that haploidentical transplantation leads to a low relapse rate without a high NRM. This tendency was also reported in 2 recent papers.67,68
 |
Table 9 Clinical Results After Allo-SCT from MRD and MUD |
 |
Table 10 Clinical Results After Allo-SCT from Haploidentical Donors |
Considering that most, if not all R/R HL patients will receive CPI, it was important to look to the outcome in this special cohort of patients. Indeed, exposure to CPI was considered potentially dangerous, as initially reported, leading the FDA to release warning notes on the use of allo-SCT after CPI. Other authors reported a higher incidence of acute and chronic GVHD and NRM in this situation,69,70 in part due to a persistent high Tcons/Treg ratio after allo-SCT.69 However, this immune hyperreactivity induced by CPI could be better controlled using a different GVH prophylaxis. McCurdy et al reported that the use of PT-Cy could overcome the negative impact of CPI on GVHD, also outside the haploidentical context.71 Finally, our group retrospectively compared the impact of CPI before haploidentical transplantation using PT-Cy, suggesting that the incidence of GVHD was not different, but the relapse rate was lower with CPI.72 The efficacy of allo-SCT after CPI was reported by Armand et al analysing the data of the Check-Mate 205 study, in which 44 patients underwent allo-SCT after CPI. The survival rate was extremely promising, even if after a short follow-up, the 6-month PFS was 82% and the OS was 87%; the cumulative incidence of TRM and relapse was 7% and 13%, respectively.36 Overall, these data suggest that allo-SCT is feasible after CPI, in particular using PT-Cy prophylaxis, and that at least in patients not achieving complete remission, allo-SCT is effective. For patients achieving a CR, the indication to perform an allo-SCT should be carefully discussed with the patient, considering the consistent risk of relapse.
Conclusions
R/R HL patients still represent a clinical concern. Salvage CT in combination with BV or treatment with BV plus CPI is highly effective in inducing a PET-negative disease status. Almost 2/3 of patients can indeed achieve this status, which is predictive of survival after consolidation by high-dose chemotherapy plus autologous stem cell support (HDC). HDC remains the gold standard for these patients, even if this could be challenged by CPI. PET remains the most useful technique to evaluate the disease status before HDC, with strong predictive significance. However, new methods such as the detection of ctDNA could help to further define the disease status. Maintenance therapy after HDC has been approved for BV, while the use of CPI after HDC has begun to be evaluated with encouraging results.
Allo-SCT remains, in our opinion, an important therapeutic tool in the most advanced cohort of patients not responding to BV and CPI.
Disclosure
Armando Santoro reports advisory board for BMS, SERVIER, GILEAD, PFIZER, EISAI, BAYER, and MSD, consultancy for ARQULE and SANOFI, speaker’s bureau for TAKEDA, BMS, ROCHE, ABB-VIE, AMGEN, CELGENE, SERVIER, GILEAD, ASTRAZENECA, PFIZER, ARQULE, LILLY, SANDOZ, EISAI, NOVARTIS, BAYER, and MSD, during the conduct of the study. C. Carlo-Stella has received research support from ADC Therapeutics and Rhizen Pharmaceuticals; has served as consultant or advisor for Servier, Novartis, Genenta Science srl, ADC Therapeutics, Roche, Sanofi, Karyopharm; and has received honoraria for speaker engagements from Bristol-Myers Squibb, Merck Sharp & Dohme, Janssen Oncology, Astra-ZenecaLuca Castagna. The authors report no other potential conflicts of interest in this work.
References
1. Josting A, Müller H, Borchmann P, et al. Dose Intensity of Chemotherapy in Patients With Relapsed Hodgkin’s Lymphoma. J Clin Oncol. 2010;28(34):5074–5080. doi:10.1200/JCO.2010.30.5771
2. Moskowitz CH, Matasar MJ, Zelenetz AD, et al. Normalization of pre-ASCT, FDG-PET imaging with second-line, non–cross-resistant, chemotherapy programs improves event-free survival in patients with Hodgkin lymphoma. Blood. 2012;119(7):1665–1670. doi:10.1182/blood-2011-10-388058
3. Bröckelmann PJ, Müller H, Casasnovas O, et al. Risk factors and a prognostic score for survival after autologous stem-cell transplantation for relapsed or refractory Hodgkin lymphoma. Ann Oncol. 2017;28(6):1352–1358. doi:10.1093/annonc/mdx072
4. Adams HJA, Kwee TC. Prognostic value of pretransplant FDG-PET in refractory/relapsed Hodgkin lymphoma treated with autologous stem cell transplantation: systematic review and meta-analysis. Ann Hematol. 2016;95(5):695–706. doi:10.1007/s00277-016-2619-9
5. Moskowitz CH, Nimer SD, Zelenetz AD, et al. A 2-step comprehensive high-dose chemoradiotherapy second-line program for relapsed and refractory Hodgkin disease: analysis by intent to treat and development of a prognostic model. Blood. 2001;97(3):616–623. doi:10.1182/blood.V97.3.616
6. Baetz T, Belch A, Couban S, et al. Gemcitabine, dexamethasone and cisplatin is an active andnon-toxic chemotherapy regimen in relapsed or refractory Hodgkin’s disease: a phase II study by the National Cancer Institute of Canada Clinical Trials Group. Ann Oncol. 2003;14(12):1762–1767. doi:10.1093/annonc/mdg496
7. Josting A, Rudolph C, Reiser M, et al. Time-intensified dexamethasone/cisplatin/cytarabine:an effective salvage therapy with low toxicity in patients with relapsed and refractory Hodgkin’s disease. Ann Oncol. 2002;13(10):1628–1635. doi:10.1093/annonc/mdf221
8. Rodriguez J, Rodriguez MA, Fayad L, et al. ASHAP: a regimen for cytoreduction of refractory or recurrent Hodgkin’s disease. Blood. 1999;93(11):3632–3636. doi:10.1182/blood.V93.11.3632
9. Bartlett NL, Niedzwiecki D, Johnson JL, et al. Gemcitabine, vinorelbine, and pegylated liposomal doxorubicin (GVD), a salvage regimen in relapsed Hodgkin’s lymphoma: CALGB 59804. Ann Oncol. 2007;18(6):1071–1079. doi:10.1093/annonc/mdm090
10. Santoro A, Magagnoli M, Spina M, et al. Ifosfamide, gemcitabine, and vinorelbine: a new induction regimen for refractory and relapsed Hodgkin’s lymphoma. Haematologica. 2007;92(1):35–41. doi:10.3324/haematol.10661
11. Santoro A, Mazza R, Pulsoni A, et al. Bendamustine in Combination With Gemcitabine and Vinorelbine Is an Effective Regimen As Induction Chemotherapy Before Autologous Stem-Cell Transplantation for Relapsed or Refractory Hodgkin Lymphoma: final Results of a Multicenter Phase II Study. J Clin Oncol. 2016;34(27):3293–3299. doi:10.1200/JCO.2016.66.4466
12. Santoro A, Mazza R, Pulsoni A, et al. Five-year results of the BEGEV salvage regimen in relapsed/refractory classical Hodgkin lymphoma. Blood Advances. 2020;4(1):
13. Cohen JB, Wei L, Maddocks KJ, et al. Gemcitabine and bendamustine is a safe and effective salvage regimen for patients with recurrent/refractory Hodgkin lymphoma: results of a Phase 1/2 study. Cancer. 2020;126(6):1235–1242. doi:10.1002/cncr.32640
14. Labrador J, Cabrero-Calvo M, Pérez-López E, et al. ESHAP as salvage therapy for relapsed or refractory Hodgkin’s lymphoma. Ann Hematol. 2014;93(10):1745–1753. doi:10.1007/s00277-014-2114-0
15. Martínez C, Díaz-López A, Rodriguez-Calvillo M, et al. Phase II trial of ofatumumab plus ESHAP (O-ESHAP) as salvage treatment for patients with relapsed or refractory classical Hodgkin lymphoma after first-line chemotherapy. Br J Haematol. 2016;174(6):859–867. doi:10.1111/bjh.14133
16. Younes A, Gopal AK, Smith SE, et al. Results of a Pivotal Phase II Study of Brentuximab Vedotin for Patients With Relapsed or Refractory Hodgkin’s Lymphoma. J Clin Oncol. 2012;30(18):2183–2189. doi:10.1200/JCO.2011.38.0410
17. Chen R, Palmer JM, Martin P, et al. Results of a Multicenter Phase II Trial of Brentuximab Vedotin as Second-Line Therapy before Autologous Transplantation in Relapsed/Refractory Hodgkin Lymphoma. Biol Blood Marrow Transplant. 2015;21(12):2136–2140. doi:10.1016/j.bbmt.2015.07.018
18. Moskowitz AJ, Schöder H, Yahalom J, et al. PET-adapted sequential salvage therapy with brentuximab vedotin followed by augmented ifosamide, carboplatin, and etoposide for patients with relapsed and refractory Hodgkin’s lymphoma: a non-randomised, open-label, single-centre, phase 2 study. Lancet Oncol. 2015;16(3):284–292. doi:10.1016/S1470-2045(15)70013-6
19. LaCasce AS, Bociek RG, Sawas A, et al. Brentuximab vedotin plus bendamustine: a highly active first salvage regimen for relapsed or refractory Hodgkin lymphoma. Blood. 2018;132(1):40–48. doi:10.1182/blood-2017-11-815183
20. O’Connor OA, Lue JK, Sawas A, et al. Brentuximab vedotin plus bendamustine in relapsed or refractory Hodgkin’s lymphoma: an international, multicentre, single-arm, phase 1–2 trial. Lancet Oncol. 2018;19(2):257–266. doi:10.1016/S1470-2045(17)30912-9
21. Kalac M, Lue JK, Lichtenstein E, et al. Brentuximab vedotin and bendamustine produce high complete response rates in patients with chemotherapy refractory Hodgkin lymphoma. Br J Haematol. 2018;180(5):757–760. doi:10.1111/bjh.14449
22. Picardi M, Della Pepa R, Giordano C, et al. Brentuximab vedotin followed by bendamustine supercharge for refractory or relapsed Hodgkin lymphoma. Blood Advances. 2019;3(9):1546–1552. doi:10.1182/bloodadvances.2019000123
23. Broccoli A, Argnani L, Botto B, et al. First salvage treatment with bendamustine and brentuximab vedotin in Hodgkin lymphoma: a phase 2 study of the Fondazione Italiana Linfomi. Blood Cancer Journal. 2019;9(12):100. doi:10.1038/s41408-019-0265-x
24. Iannitto E, Romano A, Scalzulli PR, et al. Brentuximab vedotin in association with bendamustine in refractory or multiple relapsed Hodgkin lymphoma. A retrospective real-world study. Eur J Haematol. 2020:27. doi:10.1111/ejh.13400
25. Abuelgasim KA, Alzahrani M, Alsharhan Y, et al. Chemoimmunotherapy with brentuximab vedotin combined with ifosfamide, gemcitabine, and vinorelbine is highly active in relapsed or refractory classical Hodgkin lymphoma. Bone Marrow Transplantation. 2019;54(7):1168–1172. doi:10.1038/s41409-019-0454-z
26. Hagenbeek A, Mooij H, Zijlstra J, et al. Phase I dose-escalation study of brentuximab-vedotin combined with dexamethasone, high-dose cytarabine and cisplatin, as salvage treatment in relapsed/refractory classical Hodgkin lymphoma: the HOVON/LLPC Transplant BRaVE study. Haematologica. 2019;104(4):e151–e153. doi:10.3324/haematol.2018.196899
27. Garcia-Sanz R, Sureda A, de la Cruz F, et al. Brentuximab vedotin and ESHAP is highly effective as second-line therapy for Hodgkin lymphoma patients (long-term results of a trial by the Spanish GELTAMO Group). Ann Oncol. 2019;30(4):612–620. doi:10.1093/annonc/mdz009
28. Michallet A-S, Guillermin Y, Deau B, et al. Sequential combination of gemcitabine, vinorelbine, pegylated liposomal doxorubicin and brentuximab as a bridge regimen to transplant in relapsed or refractory Hodgkin lymphoma. Haematologica. 2015;100(7):e269–271. doi:10.3324/haematol.2015.124784
29. Cheson BD, Pfistner B, Juweid ME, et al. Revised response criteria for malignant lymphoma. J Clin Oncol. 2007;25(5):579–586. doi:10.1200/JCO.2006.09.2403
30. Younes A, Hilden P, Coiffier B, et al. International Working Group consensus response evaluation criteria in lymphoma (RECIL 2017). Ann Oncol. 2017;28(7):1436–1447. doi:10.1093/annonc/mdx097
31. Cheson BD, Fisher RI, Barrington SF, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014;32(27):3059–3068. doi:10.1200/JCO.2013.54.8800
32. Eyre TA, Phillips EH, Linton KM, et al. Results of a multicentre UK-wide retrospective study evaluating the efficacy of brentuximab vedotin in relapsed, refractory classical Hodgkin lymphoma in the transplant naive setting. Br J Haematol. 2017;179(3):471–479. doi:10.1111/bjh.14898
33. Villa D, Seshadri T, Puig N, et al. Second-line salvage chemotherapy for transplant-eligible patients with Hodgkin’s lymphoma resistant to platinum-containing first-line salvage chemotherapy. Haematologica. 2012;97(5):751–757. doi:10.3324/haematol.2011.047670
34. Ansell SM, Lesokhin AM, Borrello I, et al. PD-1 Blockade with Nivolumab in Relapsed or Refractory Hodgkin’s Lymphoma. N Engl J Med. 2015;372(4):311–319. doi:10.1056/NEJMoa1411087
35. Younes A, Santoro A, Shipp M, et al. Nivolumab for classical Hodgkin’s lymphoma after failure of both autologous stem-cell transplantation and brentuximab vedotin: a multicentre, multicohort, single-arm phase 2 trial. Lancet Oncol. 2016;17(9):1283–1294. doi:10.1016/S1470-2045(16)30167-X
36. Armand P, Engert A, Younes A, et al. Nivolumab for Relapsed/Refractory Classic Hodgkin Lymphoma After Failure of Autologous Hematopoietic Cell Transplantation: extended Follow-Up of the Multicohort Single-Arm Phase II CheckMate 205 Trial. J Clin Oncol. 2018;36(14):1428–1439. doi:10.1200/JCO.2017.76.0793
37. Bair SM, Strelec LE, Feldman TA, et al. Outcomes and Toxicities of Programmed Death-1 (PD-1) Inhibitors in Hodgkin Lymphoma Patients in the United States: A Real-World, Multicenter Retrospective Analysis. The Oncologist. 2019;24(7):955–962. doi:10.1634/theoncologist.2018-0538
38. Manson G, Mear JB, Herbaux C, et al. Long-term efficacy of anti-PD1 therapy in Hodgkin lymphoma with and without allogenic stem cell transplantation. Eur J Cancer. 2019;115:47–56.
39. Beköz H, Karadurmuş N, Paydaş S, et al. Nivolumab for relapsed or refractory Hodgkin lymphoma: real-life experience. Ann Oncol. 2017;28(10):2496–2502. doi:10.1093/annonc/mdx341
40. Herrera AF, Moskowitz AJ, Bartlett NL, et al. Interim results of brentuximab vedotin in combination with nivolumab in patients with relapsed or refractory Hodgkin lymphoma. Blood. 2018;131(11):1183–1194. doi:10.1182/blood-2017-10-811224
41. Goldkuhle M, Dimaki M, Gartlehner G, et al. Nivolumab for adults with Hodgkin’s lymphoma (a rapid review using the software RobotReviewer). Cochrane Database Syst Rev. 2018;7:CD012556. doi:10.1002/14651858.CD012556
42. Chen R, Zinzani PL, Lee HJ, et al. Pembrolizumab in relapsed or refractory Hodgkin lymphoma: 2-year follow-up of KEYNOTE-087. Blood. 2019;134(14):1144–1153. doi:10.1182/blood.2019000324
43. Shi Y, Su H, Song Y, et al. Safety and activity of sintilimab in patients with relapsed or refractory classical Hodgkin lymphoma (ORIENT-1): a multicentre, single-arm, phase 2 trial. Lancet Haematol. 2019;6(1):e12–e19. doi:10.1016/S2352-3026(18)30192-3
44. Johnson P, Federico M, Kirkwood A, et al. Adapted Treatment Guided by Interim PET-CT Scan in Advanced Hodgkin’s Lymphoma. N Engl J Med. 2016;374(25):2419–2429. doi:10.1056/NEJMoa1510093
45. Press OW, Li H, Schöder H, et al. US Intergroup Trial of Response-Adapted Therapy for Stage III to IV Hodgkin Lymphoma Using Early Interim Fluorodeoxyglucose–Positron Emission Tomography Imaging: southwest Oncology Group S0816. J Clin Oncol. 2016;34(17):2020–2027. doi:10.1200/JCO.2015.63.1119
46. Borchmann P, Goergen H, Kobe C, et al. PET-guided treatment in patients with advanced-stage Hodgkin’s lymphoma (HD18): final results of an open-label, international, randomised Phase 3 trial by the German Hodgkin Study Group. Lancet. 2017;390(10114):2790–2802. doi:10.1016/S0140-6736(17)32134-7
47. Schot BW, Zijlstra JM, Sluiter WJ, et al. Early FDG-PET assessment in combination with clinical risk scores determines prognosis in recurring lymphoma. Blood. 2007;109(2):486–491. doi:10.1182/blood-2005-11-006957
48. Filmont J-E, Gisselbrecht C, Cuenca X, et al. The impact of pre- and post-transplantation positron emission tomography using 18-fluorodeoxyglucose on poor-prognosis lymphoma patients undergoing autologous stem cell transplantation. Cancer. 2007;110(6):1361–1369. doi:10.1002/cncr.22911
49. Moskowitz AJ, Yahalom J, Kewalramani T, et al. Pretransplantation functional imaging predicts outcome following autologous stem cell transplantation for relapsed and refractory Hodgkin lymphoma. Blood. 2010;116(23):4934–4937. doi:10.1182/blood-2010-05-282756
50. Castagna L, Bramanti S, Balzarotti M, et al. Predictive value of early 18F-fluorodeoxyglucose positron emission tomography (FDG-PET) during salvage chemotherapy in relapsing/refractory Hodgkin lymphoma (HL) treated with high-dose chemotherapy. Br J Haematol. 2009;145(3):369–372. doi:10.1111/j.1365-2141.2009.07645.x
51. Chen A, Mokrane FZ, Schwartz L, et al. Early 18F-FDG PET/CT response predicts survival in Relapsed/Refractory Hodgkin Lymphoma treated with Nivolumab. J Nucl Med. 2019;119:232827. doi:10.2967/jnumed.119.232827
52. Spina V, Bruscaggin A, Cuccaro A, et al. Circulating tumor DNA reveals genetics, clonal evolution, and residual disease in classical Hodgkin lymphoma. Blood. 2018;131(22):2413–2425.
53. Plattel WJ, Visser L, Diepstra A, et al. Interim thymus and activation regulated chemokine versus interim 18 F-fluorodeoxyglucose positron-emission tomography in classical Hodgkin lymphoma response evaluation. Br J Haematol. 2020:27. doi:10.1111/bjh.16514
54. Venkataraman G, Mirza MK, Eichenauer DA, Diehl V. Current status of prognostication in classical Hodgkin lymphoma. Br J Haematol. 2014;165(3):287–299. doi:10.1111/bjh.12759
55. Kamper P, Ludvigsen M, Bendix K, et al. Proteomic analysis identifies galectin-1 as a predictive biomarker for relapsed/refractory disease in classical Hodgkin lymphoma. Blood. 2011;117(24):6638–6649. doi:10.1182/blood-2010-12-327346
56. Harrison SJ, Hsu AK, Neeson P, et al. Early thymus and activation-regulated chemokine (TARC) reduction and response following panobinostat treatment in patients with relapsed/refractory Hodgkin lymphoma following autologous stem cell transplant. Leuk Lymphoma. 2014;55(5):1053–1060. doi:10.3109/10428194.2013.820287
57. Linch DC, Winfield D, Goldstone AH, et al. Dose intensification with autologous bone-marrow transplantation in relapsed and resistant Hodgkin’s disease: results of a BNLI randomised trial. Lancet. 1993;341(8852):1051–1054.
58. Schmitz N, Pfistner B, Sextro M. Aggressive conventional chemotherapy compared with high-dose chemotherapy with autologous haemopoietic stem-cell transplantation for relapsed chemosensitive Hodgkin’s disease: a randomised trial. Lancet. 2002;359(9323):2065–2071. doi:10.1016/S0140-6736(02)08938-9
59. Moskowitz CH, Walewski J, Nademanee A, et al. Five-year PFS from the AETHERA trial of brentuximab vedotin for Hodgkin lymphoma at high risk of progression or relapse. Blood. 2018;132(25):2639–2642. doi:10.1182/blood-2018-07-861641
60. Armand P, Chen Y-B, Redd RA, et al. PD-1 blockade with pembrolizumab for classical Hodgkin lymphoma after autologous stem cell transplantation. Blood. 2019;134(1):22–29. doi:10.1182/blood.2019000215
61. Aldinucci D, Gloghini A, Pinto A, De Filippi R, Carbone A. The classical Hodgkin’s lymphoma microenvironment and its role in promoting tumour growth and immune escape. J Pathol. 2010;221(3):248–263. doi:10.1002/path.2711
62. Carbone A, Gloghini A, Castagna L, Santoro A, Carlo-Stella C. Primary refractory and early-relapsed Hodgkin’s lymphoma: strategies for therapeutic targeting based on the tumour microenvironment. J Pathol. 2015;237(1):4–13. doi:10.1002/path.4558
63. Passweg JR, Baldomero H, Chabannon C, et al. The EBMT activity survey on hematopoietic-cell transplantation and cellular therapy 2018: CAR-T’s come into focus. Bone Marrow Transplantation. 2020;55(8):1604–1613. doi:10.1038/s41409-020-0826-4
64. Gooley TA, Chien JW, Pergam SA, et al. Reduced mortality after allogeneic hematopoietic-cell transplantation. N Engl J Med. 2010;363(22):2091–2101. doi:10.1056/NEJMoa1004383
65. Sureda A, Canals C, Arranz R, et al. Allogeneic stem cell transplantation after reduced intensity conditioning in patients with relapsed or refractory Hodgkin’s lymphoma. Results of the HDR-ALLO study - a prospective clinical trial by the Grupo Español de Linfomas/Trasplante de Médula Osea (GEL/TAMO) and the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation. Haematologica. 2012;97:310–317.
66. Luznik L, O’Donnell PV, Symons HJ, et al. HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, posttransplantation cyclophosphamide. Biol Blood Marrow Transplant. 2008;14(6):641–650. doi:10.1016/j.bbmt.2008.03.005
67. Mariotti J, Devillier R, Bramanti S, et al. Peripheral Blood Stem Cells versus Bone Marrow for T Cell–Replete Haploidentical Transplantation with Post-Transplant Cyclophosphamide in Hodgkin Lymphoma. Biol Blood Marrow Transplant. 2019;25(9):1810–1817. doi:10.1016/j.bbmt.2019.05.017
68. Ahmed S, Kanakry JA, Ahn KW, et al. Lower Graft-versus-Host Disease and Relapse Risk in Post-Transplant Cyclophosphamide–Based Haploidentical versus Matched Sibling Donor Reduced-Intensity Conditioning Transplant for Hodgkin Lymphoma. Biol Blood Marrow Transplant. 2019;25(9):1859–1868. doi:10.1016/j.bbmt.2019.05.025
69. Merryman RW, Kim HT, Zinzani PL, et al. Safety and efficacy of allogeneic hematopoietic stem cell transplant after PD-1 blockade in relapsed/refractory lymphoma. Blood. 2017;129(10):1380–1388. doi:10.1182/blood-2016-09-738385
70. Schoch LK, Cooke KR, Wagner-Johnston ND, et al. Immune checkpoint inhibitors as a bridge to allogeneic transplantation with posttransplant cyclophosphamide. Blood Advances. 2018;2(17):2226–2229. doi:10.1182/bloodadvances.2018019208
71. De Philippis C, Legrand-Izadifar F, Bramanti S, et al. Checkpoint inhibition before haploidentical transplantation with posttransplant cyclophosphamide in Hodgkin lymphoma. Blood Advances. 2020;4(7):1242–1249. doi:10.1182/bloodadvances.2019001336
72. Robinson SP, Goldstone AH, Mackinnon S, et al. Chemoresistant or aggressive lymphoma predicts for a poor outcome following reduced-intensity allogeneic progenitor cell transplantation: an analysis from the Lymphoma Working Party of the European Group for Blood and Bone Marrow Transplantation. Blood. 2002;100(13):4310–4316. doi:10.1182/blood-2001-11-0107
73. Lohri A, Barnett M, Fairey RN, et al. Outcome of treatment of first relapse of Hodgkin’s disease after primary chemotherapy: identification of risk factors from the British Columbia experience 1970 to 1988. Blood. 1991;77(10):2292–2298. doi:10.1182/blood.V77.10.2292.2292
74. Reece DE, Connors JM, Spinelli JJ, et al. Intensive therapy with cyclophosphamide, carmustine, etoposide ± cisplatin, and autologous bone marrow transplantation for Hodgkin’s disease in first relapse after combination chemotherapy [see comments]. Blood. 1994;83(5):1193–1199. doi:10.1182/blood.V83.5.1193.1193
75. Brice P, Bouabdallah R, Moreau P, et al. Prognostic factors for survival after high-dose therapy and autologous stem cell transplantation for patients with relapsing Hodgkin’s disease: analysis of 280 patients from the French registry. Société Française de Greffe de Moëlle. Bone Marrow Transplantation. 1997;20(1):21–26. doi:10.1038/sj.bmt.1700838
76. Horning SJ, Chao NJ, Negrin RS, et al. High-Dose Therapy and Autologous Hematopoietic Progenitor Cell Transplantation for Recurrent or Refractory Hodgkin’s Disease: analysis of the Stanford University Results and Prognostic Indices. Blood. 1997;89(3):801–813. doi:10.1182/blood.V89.3.801
77. Josting A, Franklin J, May M, et al. New prognostic score based on treatment outcome of patients with relapsed Hodgkin’s lymphoma registered in the database of the German Hodgkin’s lymphoma study group. J Clin Oncol. 2002;20(1):221–230.
78. Spaepen K, Stroobants S, Verhoef G, Mortelmans L. Positron emission tomography with [18F]FDG for therapy response monitoring in lymphoma patients. Eur J Nucl Med Mol Imaging. 2003;30(Suppl S1):S97–105. doi:10.1007/s00259-003-1166-5
79. Jabbour E, Hosing C, Ayers G, et al. Pretransplant positive positron emission tomography/gallium scans predict poor outcome in patients with recurrent/refractory Hodgkin lymphoma. Cancer. 2007;109(12):2481–2489. doi:10.1002/cncr.22714
80. André M, Henry-Amar M, Pico JL, et al. Comparison of high-dose therapy and autologous stem-cell transplantation with conventional therapy for Hodgkin’s disease induction failure: a case-control study. Société Francaise de Greffe de Moelle. J Clin Oncol. 1999;17(1):222–229.
81. Sweetenham JW, Carella AM, Taghipour G, et al. High-dose therapy and autologous stem-cell transplantation for adult patients with Hodgkin’s disease who do not enter remission after induction chemotherapy: results in 175 patients reported to the European Group for Blood and Marrow Transplantation. Lymphoma Working Party. J Clin Oncol. 1999;17(10):3101–3109.
82. Josting A, Rueffer U, Franklin J, Sieber M, Diehl V, Engert A. Prognostic factors and treatment outcome in primary progressive Hodgkin lymphoma: a report from the German Hodgkin Lymphoma Study Group. Blood. 2000;96(4):1280–1286. doi:10.1182/blood.V96.4.1280
83. Constans M, Sureda A, Terol MJ, et al. Autologous stem cell transplantation for primary refractory Hodgkin’s disease: results and clinical variables affecting outcome. Ann Oncol. 2003;14(5):745–751. doi:10.1093/annonc/mdg206
84. Czyz J, Szydlo R, Knopinska-Posluszny W, et al. Treatment for primary refractory Hodgkin’s disease: a comparison of high-dose chemotherapy followed by ASCT with conventional therapy. Bone Marrow Transplantation. 2004;33(12):1225–1229. doi:10.1038/sj.bmt.1704508
85. Aparicio J, Segura A, Garcerá S, et al. ESHAP is an active regimen for relapsing Hodgkin’s disease. Ann Oncol. 1999;10(5):593–595. doi:10.1023/A:1026454831340
86. Fermé C, Bastion Y, Lepage E, et al. The MINE regimen as intensive salvage chemotherapy for relapsed and refractory Hodgkin’s disease. Ann Oncol. 1995;6(6):543–549. doi:10.1093/oxfordjournals.annonc.a059242
87. Proctor SJ, Jackson GH, Lennard A, et al. Strategic approach to the management of Hodgkin’s disease incorporating salvage therapy with high-dose ifosfamide, etoposide and epirubicin: a Northern Region Lymphoma Group study (UK). Ann Oncol. 2003;14(Suppl 1):i47–50. doi:10.1093/annonc/mdg710
88. Rothe A, Sasse S, Goergen H, et al. Brentuximab vedotin for relapsed or refractory CD30+ hematologic malignancies: the German Hodgkin Study Group experience. Blood. 2012;120(7):1470–1472. doi:10.1182/blood-2012-05-430918
89. Zinzani PL, Viviani S, Anastasia A, et al. Brentuximab vedotin in relapsed/refractory Hodgkin’s lymphoma: the Italian experience and results of its use in daily clinical practice outside clinical trials. Haematologica. 2013;98(8):1232–1236. doi:10.3324/haematol.2012.083048
90. Gibb A, Jones C, Bloor A, et al. Brentuximab vedotin in refractory CD30+ lymphomas: a bridge to allogeneic transplantation in approximately one quarter of patients treated on a Named Patient Programme at a single UK center. Haematologica. 2013;98(4):611–614. doi:10.3324/haematol.2012.069393
91. Perrot A, Monjanel H, Bouabdallah R, et al. Impact of post-brentuximab vedotin consolidation on relapsed/refractory CD30 + Hodgkin lymphomas: a large retrospective study on 240 patients enrolled in the French Named-Patient Program. Haematologica. 2016;101(4):466–473. doi:10.3324/haematol.2015.134213
92. Peggs KS, Hunter A, Chopra R, et al. Clinical evidence of a graft-versus-Hodgkin’s-lymphoma effect after reduced-intensity allogeneic transplantation. Lancet. 2005;365(9475):1934–1941.
93. Alvarez I, Sureda A, Caballero MD, et al. Nonmyeloablative stem cell transplantation is an effective therapy for refractory or relapsed hodgkin lymphoma: results of a spanish prospective cooperative protocol. Biol Blood Marrow Transplant. 2006;12(2):172–183.
94. Corradini P, Dodero A, Farina L, et al. Allogeneic stem cell transplantation following reduced-intensity conditioning can induce durable clinical and molecular remissions in relapsed lymphomas: pre-transplant disease status and histotype heavily influence outcome. Leukemia. 2007;21(11):2316–2323.
95. Anderlini P, Saliba R, Acholonu S, et al. Fludarabine-melphalan as a preparative regimen for reduced-intensity conditioning allogeneic stem cell transplantation in relapsed and refractory Hodgkin’s lymphoma: the updated M.D. Anderson Cancer Center experience. Haematologica. 2008;93(2):257–264.
96. Devetten MP, Hari PN, Carreras J, et al. Unrelated donor reduced-intensity allogeneic hematopoietic stem cell transplantation for relapsed and refractory Hodgkin lymphoma. Biol Blood Marrow Transplant. 2009;15(1):109–117.
97. Robinson SP, Sureda A, Canals C, et al. Reduced intensity conditioning allogeneic stem cell transplantation for Hodgkin’s lymphoma: identification of prognostic factors predicting outcome. Haematologica. 2009;94(2):230–238.
98. Sureda A, Canals C, Arranz R, et al. Allogeneic stem cell transplantation after reduced intensity conditioning in patients with relapsed or refractory Hodgkin’s lymphoma. Results of the HDR-ALLO study - a prospective clinical trial by the Grupo Español de Linfomas/Trasplante de Médula Osea (GEL/TAMO) and the Lymphoma Working Party of the European Group for Blood and Marrow Transplantation. Haematologica. 2012;97(2):310–317.
99. Kanate AS, Mussetti A, Kharfan-Dabaja MA, et al. Reduced-intensity transplantation for lymphomas using haploidentical related donors vs HLA-matched unrelated donors. Blood. 2016;127(7):938–947.
100. Ghosh N, Karmali R, Rocha V, et al. Reduced-Intensity Transplantation for Lymphomas Using Haploidentical Related Donors Versus HLA-Matched Sibling Donors: A Center for International Blood and Marrow Transplant Research Analysis. J Clin Oncol. 2016;34(26):3141–3149.
101. Anderlini P, Saliba RM, Ledesma C, et al. Gemcitabine, Fludarabine, and Melphalan for Reduced-Intensity Conditioning and Allogeneic Stem Cell Transplantation for Relapsed and Refractory Hodgkin Lymphoma. Biol Blood Marrow Transplant. 2016;22(7):1333–1337. doi:10.1016/j.bbmt.2016.03.028
102. Martínez C, Gayoso J, Canals C, et al. Post-Transplantation Cyclophosphamide-Based Haploidentical Transplantation as Alternative to Matched Sibling or Unrelated Donor Transplantation for Hodgkin Lymphoma: A Registry Study of the Lymphoma Working Party of the European Society for Blood and Marrow Transplantation. J Clin Oncol. 2017;35(30):3425–3432.
103. Gauthier J, Poiré X, Gac A-C, et al. Better outcome with haploidentical over HLA-matched related donors in patients with Hodgkin’s lymphoma undergoing allogeneic haematopoietic cell transplantation—a study by the Francophone Society of Bone Marrow Transplantation and Cellular Therapy. Bone Marrow Transplantation. 2018;53(4):400–409. doi:10.1038/s41409-017-0018-z
104. Burroughs LM, O’Donnell PV, Sandmaier BM, et al. Comparison of outcomes of HLA-matched related, unrelated, or HLA-haploidentical related hematopoietic cell transplantation following nonmyeloablative conditioning for relapsed or refractory Hodgkin lymphoma. Biol Blood Marrow Transplant. 2008;14(11):1279–1287. doi:10.1016/j.bbmt.2008.08.014
105. Raiola A, Dominietto A, Varaldo R, et al. Unmanipulated haploidentical BMT following non-myeloablative conditioning and post-transplantation CY for advanced Hodgkin’s lymphoma. Bone Marrow Transplantation. 2014;49(2):190–194. doi:10.1038/bmt.2013.166
106. Gayoso J, Balsalobre P, Pascual MJ, et al. Busulfan-based reduced intensity conditioning regimens for haploidentical transplantation in relapsed/refractory Hodgkin lymphoma: spanish multicenter experience. Bone Marrow Transplantation. 2016;51(10):1307–1312. doi:10.1038/bmt.2016.115
107. Castagna L, Bramanti S, Devillier R, et al. Haploidentical transplantation with post-infusion cyclophosphamide in advanced Hodgkin lymphoma. Bone Marrow Transplantation. 2017;52(5):683–688. doi:10.1038/bmt.2016.348
108. Gauthier J, Castagna L, Garnier F, et al. Reduced-intensity and non-myeloablative allogeneic stem cell transplantation from alternative HLA-mismatched donors for Hodgkin lymphoma: a study by the French Society of Bone Marrow Transplantation and Cellular Therapy. Bone Marrow Transplantation. 2017;52(5):689–696. doi:10.1038/bmt.2016.349
109. Marani C, Raiola AM, Morbelli S, et al. Haploidentical Transplants with Post-Transplant Cyclophosphamide for Relapsed or Refractory Hodgkin Lymphoma: the Role of Comorbidity Index and Pretransplant Positron Emission Tomography. Biol Blood Marrow Transplant. 2018;24(12):2501–2508. doi:10.1016/j.bbmt.2018.07.025
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
