Back to Journals » Therapeutics and Clinical Risk Management » Volume 12
Safe medication management and use of narcotics in a Joint Commission International-accredited academic medical center hospital in the People’s Republic of China
Authors Fang X, Zhu L, Pan S, Xia P, Chen M, Zhou Q
Received 8 January 2016
Accepted for publication 24 February 2016
Published 6 April 2016 Volume 2016:12 Pages 535—544
DOI https://doi.org/10.2147/TCRM.S103853
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Xu Fang,1,2 Ling-ling Zhu,3 Sheng-dong Pan,4 Ping Xia,4 Meng Chen,5 Quan Zhou5
1Office of Hospital Administration, 2Office of Party and Administration Council, 3Geriatric VIP Care Ward, Division of Nursing, 4Division of Medical Administration, 5Department of Pharmacy, the Second Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang Province, People’s Republic of China
Abstract: Safe medication management and use of high-alert narcotics should arouse concern. Risk management experiences in this respect in a large-scale Joint Commission International (JCI)-accredited academic medical center hospital in the People’s Republic of China during 2011–2015, focusing on organizational, educational, motivational, and information technological measures in storage, prescribing, preparing, dispensing, administration, and monitoring of medication are summarized. The intensity of use of meperidine in hospitalized patients in 2015 was one-fourth that in 2011. A 100% implementation rate of standard storage of narcotics has been achieved in the hospital since December 2012. A “Plan, Do, Check, Act” cycle was efficient because the ratio of number of inappropriate narcotics prescriptions to total number of narcotics prescriptions for inpatients decreased from August 2014 to December 2014 (28.22% versus 2.96%, P=0.0000), and it was controlled below 6% from then on. During the journey to good pain management ward accreditation by the Ministry of Health, People’s Republic of China, (April 2012–October 2012), the medical oncology ward successfully demonstrated an increase in the pain screening rate at admission from 43.5% to 100%, cancer pain control rate from 85% to 96%, and degree of satisfaction toward pain nursing from 95.4% to 100% (all P-values <0.05). Oral morphine equivalent dosage in the good pain management ward increased from 2.3 mg/patient before June 2012 to 54.74 mg/patient in 2014. From 2011 to 2015, the oral morphine equivalent dose per discharged patient increased from 8.52 mg/person to 20.36 mg/person. A 100% implementation rate of independent double-check prior to narcotics dosing has been achieved since January 2013. From 2014 to 2015, the ratio of number of narcotics-related medication errors to number of discharged patients significantly decreased (6.95% versus 0.99%, P=0.0000). Taken together, continuous quality improvements have been achieved in safe medication management and use of narcotics by an integrated multidisciplinary collaboration during the journey to JCI accreditation and in the post-JCI accreditation era.
Keywords: medication errors, medication management, meperidine, narcotics, prescribing, quality
Introduction
Narcotics are included in the Institute for Safe Medication Practices list of high-alert medications that bear a heightened risk of causing significant patient harm when used in error.1 Literature have described that the situation in medication management and use of narcotics was not optimistic. Opioid prescribing errors were common in inpatients2 and also in emergency departments.3 Logan et al3 reported that 10.3% of enrollees who received opioid prescription by emergency departments experienced at least one indicator of potential inappropriate opioid use, such as opioid prescriptions overlapping by 1 week or more, overlapping opioid and benzodiazepine prescriptions, high daily doses, long-acting/extended-release (LA/ER) opioids for acute pain, and overlapping LA/ER opioids. Opioid analgesics represented the majority of analgesic error reports in North Carolina nursing homes, and opioid errors were more likely to be associated with wrong drug errors, wrong dose errors, and administration errors when compared with nonopioid errors.4 The Joint Commission, an independent, not-for-profit group in the USA, reports that of the opioid-related adverse events reported to the agency during 2004–2011, 47% involved wrong-dosage medication errors (MEs), 29% were due to improper patient monitoring, and 11% were caused by other factors (eg, excessive dosing, drug–drug interactions, and adverse reactions).5 Alarmed by adverse events involving opioid drugs, the Joint Commission has issued a Sentinel Alert, urging hospitals to take steps to improve safety in the prescribing of opioids.6 A prospective survey showed that 151 opioid prescribing errors were detected among 117 patients with cancer pain, indicating that opioid prescribing errors were common.7 Therefore, safe medication use of narcotics should arouse a greater concern.
However, there is limited literature on a multidisciplinary team’s integrated endeavors to continuously enhance safe medication management and use of narcotics in large-scale hospitals. The Second Affiliated Hospital of Zhejiang University (SAHZU), a comprehensive 3,200-bed academic medical center hospital with 3.51 million outpatients and emergency patients visits annually (data in 2014) in Zhejiang Province, successfully passed Joint Commission International (JCI) accreditation on February 24, 2013.8 SAHZU performed continuous quality improvements in safe medication management and use (MMU) of high-alert medications during the journey to JCI accreditation and in the post-JCI accreditation era. The aims of this paper are to summarize relevant experiences in narcotics-related risk management toward safe drug administration and to provide some reference for international counterparts.
National policy on narcotics use
The national narcotic policies give strict requirements on procurement, storage, prescribing, dispensing, administration, and monitoring of drugs.9,10 Narcotics should be prescribed by a physician who must receive special training and be authorized by the hospital where he/she serves. Besides prescribing via a computerized physician order entry (CPOE), the physician must also hand write a unified national red paper narcotic prescription. In recent years, several hospitals have attempted to implement electronic narcotic prescription and have abandoned written narcotic prescriptions.
For outpatients and emergency patients, there is a restriction on the quantity of narcotics per sheet, ie, injection should be limited to a usual dose, and sustained-release formulation and other dosage forms should not be over the total dose within 7 and 3 days, respectively. For patients with cancer pain or moderate-to-severe chronic pain, the quantity of narcotics per sheet should not be over the limit (ie, 3 days [injection], 15 days [sustained-release formulation], and 7 days [other dosage forms]). Meperidine is only allowed for use once (at a usual dose), and it must be administered within the hospital where it is prescribed. For inpatients, narcotics should be prescribed by physicians at the usual daily dose day by day.
For the purpose of analgesic treatment, physicians should follow the World Health Organization’s Three-Step Analgesic Ladder therapy. Briefly, Step 1 recommends administration of a nonopioid; Step 2, for persistent or increasing pain, administration of a weak opioid is recommended; and Step 3, for persistent or increasing pain, administration of a strong opioid is recommended. At all three steps, adjuvant medication should be administered concurrently with analgesia as needed.11
SAHZU additional organizational measures
A three-level narcotics management system in SAHZU consists of the subcommittee of narcotics and psychoactive drugs affiliated to Drug and Therapy Committee (DTC) (first level), pharmacy (second level), and departments outside of pharmacy (third level). The subcommittee of narcotics and psychoactive drugs outlines the process of how the changes, in protocols/procedures that were likely to contribute to the significant quality improvements, were developed and agreed upon (eg, retrospective data analysis, on-site inspection, and consultation with key stakeholders). The pharmacy’s drug storehouse is responsible for narcotics procurement and supply chain management. Outpatient pharmacy, emergency pharmacy, and inpatient pharmacy are responsible for appropriateness review, medication preparation, double checking, dispensing, medication management, drug utilization review, drug counseling, and monthly on-site inspection of departments outside pharmacy. Each nursing unit, Division of Anesthesia and Surgery, and other departments outside of pharmacy are responsible for their respective in-house management and use of narcotics.
The division of nursing also has a subgroup focusing on the behaviors of all nurses on medication safety. Based on the JCI accreditation standards, SAHZU established a function group named as “MMU” in 2011, which facilitated quality and patient safety associated with medications. Data on adverse drug reaction (ADRs), MEs, and sentinel events are processed and reported to the Division of Medical Administration, Division of Nursing, Pharmacy, and the Office of Quality Management, followed by potential targeted quality improvement activities. The stewardship program was approved by the Ethics Committee at SAHZU and it was in compliance with the Helsinki Declaration. The Ethics Committee at SAHZU deemed that patient consent was unnecessary, as this study is a review of previous data.
Stewardship intervention on meperidine use
Meperidine has little to no therapeutic superiority to other narcotics. It may be more susceptible to abuse and toxicity due to its neurotoxic metabolite with a long elimination half-life.12 Five deaths partially associated with meperidine were recorded at the SAHZU before August 2012, and so the SAHZU president suggested to further limit meperidine prescribing. Starting in August 28, 2012, SAHZU DTC decided to forbid meperidine use in all departments except the endoscopy center for use prior to endoscopic retrograde cholangiopancreatography, the burn ward, and the heart center (interventional cardiovascular center, cardiovascular medicine ward, and cardiac surgery ward). The Cardiovascular Medicine Department also abandoned meperidine use in August 2015. The outpatient pharmacy removed meperidine from its formulary and achieved zero appearance of this narcotic from then on. The pharmacy is required to intercept any meperidine prescriptions that do not conform to the policy. An obvious warning of the meperidine use policy will appear in the interface of computer when physicians write meperidine orders via CPOE. Shortly after the implementation of this policy, the pharmacy received a few requests from clinicians to lift the ban on meperidine. Policy explanation and alternative recommendation were given by pharmacists.
In October 2014, the Endoscopy Center experienced one death associated with meperidine. The sentinel event occurred in the department that had got permission to use meperidine. A root cause analysis was conducted, and lack of therapeutic monitoring after meperidine use was found to be the main culprit. Since then, meperidine has been included in the list of medications that must be under the moderate-to-deep sedation management, ie, patients should be monitored continuously for respiratory and heart rate, systolic and diastolic blood pressures, and oxygen saturation by registered nurses trained in advanced life support from the time of sedative drug administration. To the best of our knowledge, this special policy on meperidine is unique to the People’s Republic of China. For international processes concerning meperidine use, the Agency for Health Care Policy and Research (AHCPR) and the American Pain Society (APS) have published recommendations regarding meperidine’s place in therapy, and they recommend reserving it for patients who have a demonstrated allergy or intolerance to first-line opioids and also state that it should not be given at doses >600 mg/d or for longer than 48 hours.13–15 Our unique intervention measure seems more administrative and mandatory.
In May 2013, SAHZU initiated inpatient service at a new campus, a 20-minute bus drive away from the old campus. In April 2015, an obstetrics ward was established in the new campus. Pharmacists were counseled about the appropriateness of meperidine use in obstetrics department because meperidine is usually used during labor and delivery in the People’s Republic of China. Literature review showed the superiority of pentazocine to meperidine due to lack of side effects and similar pain relief and maternal satisfaction.16–18 Also, pentazocine is not a narcotic according to national policy. The Dean of the Obstetrics Department accepted the pharmacist’s advice. The obstetricians began to use pentazocine as an alternative to meperidine since August 2015 and have affirmed the efficacy and safety of pentazocine. In May 2015, a fatal event partly associated with meperidine use occurred at the General Surgery ward in the new campus. A root cause analysis was performed. Besides severity of disease, the additional potential causes of this event were as follows: 1) the attending physician ignored the meperidine use policy and prescribed this narcotic for the patient, 2) the pharmacy did not intercept this meperidine order, 3) the nurses did not correctly implement moderate-to-deep sedation management on meperidine, and 4) basic life support was not rapidly provided when the patient experienced loss of consciousness in the CT room of the Radiology Department after receiving meperidine injection at ward. As the Swiss cheese model of accident causation illustrated, although many layers of defense lie between hazards and accidents, there are flaws in each layer that, if aligned, can allow the accident to occur.19 Thus, suitable measures were taken, including strengthening the homogenization management of medical quality between the new campus and the old campus, promoting a sense of responsibility in clinicians, organization of basic life support skills, assessment of each staff member, and optimization of human resources and employee shift schedule. After a series of comprehensive measures, there was a dramatic decreasing trend in use intensity of meperidine in inpatients and the ratio of number of meperidine vials to number of emergency patients during 2011–2015 (Table 1). Use intensity of meperidine in 2015 was one-fourth that in 2011. The ratio of number of meperidine vials to number of emergency patients in 2015 was 5.5% the value in 2011, although annual person-time of discharged inpatients increased by 56.7% and average length of stay shortened by 17.6%. Since June 2015, zero occurrence of improper meperidine use (eg, lack of moderate-to-deep sedation monitoring and off-policy prescribing) has been achieved at SAHZU. No clinical problem has arisen from the replacement of meperidine with other opioids.
Standardized storage
Narcotics must be locked in the safe according to a rule of “double person double lock” (ie, a safe has two locks and two keys. One person keeps a key and the other person keeps the other key, only when these two persons are available at the site can narcotics be taken out of the safe). A standardized label is pasted on the corresponding narcotics site. Also, management regarding the use of look-alike and sound-alike narcotic formulations were strengthened, with these medications not kept in close proximity, to help avoid confusion. Clinical professionals should pay special attention to narcotics with different strengths, for example, Oxycontin® (oxycodone sustained release tablet, Purdue Pharma L.P. Stamford, CT, USA) has three strengths (5, 10, and 40 mg/tablet) and Durogesic® (Fentanyl transdermal patches, LZA Corporation, Vacaville, CA, USA) has two strengths (4.2 and 8.4 mg).20,21 SAHZU has achieved 100% implementation rate of standard storage of narcotics in the whole hospital since December 2012.
Quality improvements in narcotics prescribing for inpatients
A PDCA cycle (Plan, Do, Check, Act) is an effective tool for achieving continuous quality improvement in clinical practice.22,23 So, we applied it in analyzing prescribing quality of narcotics in inpatients in August 2014. The ratio (ie, number of inappropriate narcotics prescriptions identified by staff pharmacists divided by total number of narcotics prescriptions for inpatients) from March 2014 to August 2014 was calculated as preintervention data. Distribution analysis of inappropriate narcotics prescriptions during this period was performed based on total sample (n=5,353). The relative percentage of each error type was calculated, and the top four types of errors were as follows (Figure 1): incomplete patient information (46.78%), wrong drug name, dosage forms, and strengths (17.97%), incomplete description on dosage and administration (17.86%), and omission in physician’s signing (9.06%). A prevalence study of errors in opioid prescription in a large teaching hospital showed that opioid prescription error rate was 27%, and the largest error type was “unclear prescription and missing information”.24 The situation was similar with our preintervention data. We wanted to improve the issue and guarantee the achievement of a goal of below 8% inappropriateness in medical orders (plan).
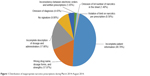  | Figure 1 Distribution of inappropriate narcotics prescriptions during March 2014–August 2014. |
Main intervention measures were as follows (do):
- Informational measures: SAHZU developed an electronic prescription and automatic print system for narcotics prescribed by anesthesiologists in the Division of Anesthesia and Surgery (where prescribing quality is the worst compared to other departments) at the end of August 2014;
- Educational measures: Physicians, pharmacists, and nurses were provided with lectures on basic knowledge concerning narcotics formulation, prescribing quality requirements, and common prescribing errors;
- Motivational measures: Staff pharmacists have been motivated with awards to document near misses related with narcotics prescribing in a special table since July 2013. However, after the initiation of this PDCA, the supervisor of inpatient pharmacy provided extra recommendation and guidance to those pharmacists who forgot documenting inappropriate prescriptions;
- Feedback measures: Sullivan et al25 reported that personalized performance feedback could reduce narcotic prescription errors in a neonatal intensive care unit, and the MEs frequency showed an 83% improvement, decreasing from every 3.94 days to every 22.63 days after the intervention. Therefore, we collected typical narcotics-associated near misses and gave feedback to the dean of clinical departments.
The number of inappropriate narcotics prescriptions was derived from table records maintained by staff pharmacists, and the ratio (ie, number of inappropriate narcotics prescriptions divided by total number of narcotics prescriptions for inpatients) was calculated and monitored monthly (check). Irrational narcotic prescriptions involving near misses significantly decreased from 28.22% (1,147/4,135) in August 2014 to 2.96% (115/3,883) in December 2014 (Pearson chi-square test, P=0.0000), and the indicator was controlled below 6% from then on (Figure 2).
  | Figure 2 Trend analysis of the proportion of inappropriate narcotics prescriptions to total narcotics prescriptions for inpatients. |
This routine monitoring was the basis for the subsequent action and for the decision of whether the maneuver needed further improvement or whether quality criteria were reached (action). We believe our preliminary intervention is successful and reaches the preestablished criteria. A further area for improvement is to achieve electronic narcotics prescriptions issued by nonanesthesiologists.
Good pain management ward and its driving effect
In July 2011, the People’s Republic of China initiated a national pilot program on constructing “Good pain management (GPM) wards”.26 SAHZU prepared to receive this challenging accreditation. In February 2012, SAHZU established a multidisciplinary team to facilitate the activities of a GPM ward. In November 2012, the medical oncology ward of SAHZU became one of the first batch of GPM wards in the People’s Republic of China. During the journey to GPM accreditation as well as JCI accreditation, management on prescribing painkillers was strengthened by SAHZU. The measures were as follows:
- DTC revised the narcotic formulary and introduced new narcotics into the hospital. The narcotics already available in SAHZU in February 2012 included injection formulations (morphine, meperidine, bucinnazine, fentanyl, sufentanil, and remifentanil), oral formulations (morphine sustained release tablet [10 and 30 mg/tablet], oxycodone sustained-release tablet [40 mg/tablet], compound platycodon tablet), and fentanyl transdermal patches (4.2 and 8.4 mg/patch). Morphine immediate-release tablet (10 mg/tablet) and oxycodone sustained-release tablet (10 mg/tablet) were introduced in March 2012 for the sake of convenience in dosage titration and breakthrough pain rescue.
- On-site inspections were regularly performed toward narcotics prescribing quality, regarding whether to rationally select narcotics, how to concomitantly use pain killers and/or prophylactic agents for avoiding side effects, how to carefully perform dynamic pain assessment, dosage titration, breakthrough pain treatment, and switch one narcotic to another therapeutic equivalent narcotic. Physicians should specify measurable goals for each patient with cancer in the electronic medical record (EMR) (eg, numerical rating scale [NRS] below 3, number of breakthrough pain and occasions of needing rescue with short-action narcotics below 3, and dosage titration time completed within 3 days).
- A full-time clinical pharmacist was appointed to work in the GPM ward, providing clinical pharmacy services including patient education; intra-ward lectures for physicians, nurses, and medical and nursing students; ward round; pain management; and regimen decision.
- Nurses should implement a standardized cancer pain nursing protocol. Discharged patients with cancer pain receiving at least World Health Organization Analgesic Ladder Step 2 therapy will be followed up via telephone regarding NRS, drug adherence, ADRs, and quality of life at 1 or 2 weeks and 1 month after discharge.
From April 2012 to October 2012, four indicators significantly improved (Pearson chi-square test, all P-values <0.05), including the percentage of pain screening at admission (43.5% versus 100%), percentage of patients with cancer pain receiving standard pain management (90% versus above 97%), percentage of cancer pain control (85% versus 96%), and the degree of satisfaction toward pain nursing from patients with cancer pain (95.4% versus 100%). The achievements continue to be maintained in the GPM ward thereafter. Meanwhile, average oral morphine equivalent dosage per discharged patient in the GPM ward increased from 2.3 mg in the first half of 2012 to 54.74 mg in 2014. Regarding inpatients with cancer pain in the GPM ward, oral morphine equivalent dosage per person in 2014 was 146.83 mg.
The construction of the GPM ward also was a driving force for improvement in other wards. We retrospectively analyzed the analgesic narcotics consumption, focusing on morphine tablet, morphine injection, meperidine injection, morphine sustained-release tablet, oxycodone sustained-release tablet, and fentanyl transdermal patch. From 2011 to 2015, the oral morphine equivalent dose in all inpatients significantly increased from 652.90 to 2,443.45 g, and oral morphine equivalent dose per discharged patient increased from 8.52 to 20.36 mg/person (Table 2).
Standardized medication preparation and dispensing
SAHZU improved the interface of inpatient pharmacy management information system for prescription auditing in January 2013. Pharmacists could see the patient’s information like age, diagnosis, allergy history, body weight, pregnancy status, clinical laboratory data (eg, hepatic and renal function), drug information (such as approved drug name, dose, route, dosing time, dosing frequency), and list of all current medications.27 Auditing pharmacists should intercept nonstandard format prescriptions (eg, omission of some information, inappropriate use of abbreviations, ambiguous writing, narcotics orders written on the prescription sheet for nonnarcotics medications, and quantities of narcotics over the limit of national policy) and inappropriate therapeutic regimen (eg, combination use of two potent opioids, combination use of an opioid and other central nervous system depressants, direct initiation of a potent opioid without dose titration, intramuscular injection of morphine rather than intravenous or subcutaneous administration, rectal administration of sustained release morphine or oxycodone tablet, and lack of detailed condition of medication administration regarding (prn orders of narcotics). Pharmacists should communicate with physicians if controversial physician orders are identified.
Considering that narcotics are fall-risk increasing medications,28 the labels of all narcotics dispensed by pharmacy must contain fall-risk increasing warnings, the symbol of high-alert medications, patient identity, barcode, and dosage and administration information. With the aid of barcode scanning, each process from prescribing to administering can be rapidly traced via an online inquiring system. An automatic voice-alarm system was created so that a stat narcotics order could be handled immediately by nurses and pharmacists. The time from physician’s prescribing to nurse’s acceptance of narcotics from inpatient pharmacy decreased from 2 hours to <30 minutes. Administration of all narcotics must receive independent double-check prior to dispensing. If outpatients and inpatients at discharge receive oral narcotics therapy, they will get special written patent education from pharmacy (eg, storage requirements, dosing time, important drug–drug interactions, awareness of increasing fall risk, and clinical presentation of ADR).
Pharmacists in hospitals and health systems can play a key role in implementing or promoting appropriate guidelines on opioid therapy, including the use of pain management agreement plans; policies to ensure adequate oversight of opioid prescribing, dispensing, and waste disposal; and educational initiatives targeting patients as well as hospital and pharmacy staff.29 Following international processes concerning narcotics use, SAHZU pharmacists are striving for excellence.
Standardized administration and monitoring
Since January 2013, SAHZU has required that two licensed health care professionals must perform a standardized independent double-check prior to barcode-assisted medication administration of narcotics, insulin infusion, chemotherapeutic drugs, and intravenous heparin.30 On-site inspection results and retrospective review of nursing record system showed a 100% implementation rate. Morriss et al31 reported that patients who were treated with an opioid in the absence of a barcode-assisted medication administration system had a 10% probability of an adverse drug event. The residue of narcotics must be appropriately disposed of under the supervision of two persons. Meanwhile, the practice of a vial of fentanyl injection being shared by multiple patients was abolished from then on.
Physicians and nurses must pay attention to pharmacotherapeutic monitoring toward narcotics use. The frequencies of pain assessment on the basis of NRS value were as follows: once (NRS, ≤3), every shift (NRS, 4–6), every hour until NRS ≤6 (NRS, 7–10), and each time following treatment with pain killers (ie, 1 hour after oral ingestion, half an hour after subcutaneous or intramuscular injection, 15 minutes after intravenous administration). The potential for opioid-induced respiratory depression should always be considered. For rescuing of narcotic poisoning, naloxone (0.4 mg/ampoule) was available at all divisions/departments in SAHZU where narcotics treatment may be excised. ADRs should be documented in the EMR following narcotics therapy.
All physicians, pharmacists, nurses, and students are encouraged to report ADRs and MEs at any process via an online no-fault reporting system within 24 hours. For sentinel events, the identifying person must report to the dean of department within 30 minutes and complete an online report within 12 hours. Root cause analysis will then be initiated following the SAHZU president’s consent. When an event is reported, a short message will be sent to the designated medication safety officer. After a preliminary review, the event will be sent to the dean of clinical department who should give feedback to the Division of Quality Management within 5 working days. The identifying person would be notified when the feedback was given by the dean of department where MEs are made. The supervisor of inpatient pharmacy and the medication safety officer collected data derived from the table records and the online reporting system. If necessary, chart review by pharmacists should be carried out. A retrospective analysis of MEs was performed every quarter.
Table 3 lists the distribution of MEs associated with narcotics. The number of MEs associated with narcotics in 2014 and 2015 was 7,272 and 1,185, respectively. The difference in ratio of number of narcotics-related MEs to number of discharged patients between 2014 and 2015 is statistically significant (Pearson chi-square test, 6.95% versus 0.99%, P=0.0000). Narcotics-related prescribing errors significantly decreased from 7,268 cases in 2014 to 1,178 cases in 2015. The difference in ratio of number of inappropriate narcotics prescriptions to number of discharged patients between 2014 and 2015 is statistically significant (Pearson chi-square test, 6.95% versus 0.98%, P=0.0000), which may be attributed to the PDCA cycle conducted by inpatient pharmacy and introduction of electronic prescribing and automatic print system in outpatient clinics and emergency department in January 20, 2015. During 2011–2015, the total number of MAEs made by nurses was 15. Among these MAEs, regarding medication categories, sustained release-tablets accounted for seven cases (oxycodone [n=6], morphine [n=1]), followed by intravenous fentanyl maintenance therapy via micropump (n=4). Regarding error types, omission accounted for seven cases, paralleled with dose errors (n=7).
There were only five narcotics-related ADRs derived from the online ADR reporting system during 2011–2015. The number of ADRs reported by SAHZU staff seemed too little despite the fact that these ADRs have been documented in patient’s EMR, indicating further opportunity of improvements in ADR surveillance.
Limitations
Although our experience may be of interest to health care professionals elsewhere, it has several limitations. First, this experience is from a single institution. Ideally, it would have been even better if we had controls (eg, non-JCI-accredited hospitals, or hospitals without GPM wards). However, it is a longitudinal follow-up study and one can appreciate the gradual improvement in outcome year by year. Second, we did not evaluate the pharmacoeconomic issue (ie, the cost/benefit ratio, the “human cost”, narcotic analgesic efficacy among inpatients except GPM ward patients, and satisfaction from medical staff). Third, we did not provide drug utilization data of other nonnarcotic analgesic agents although tramadol, pentazocine, and dezocine are not narcotics according to the People’s Republic of China national policy, and the three pain killers are type 2 psychotropic drugs that do need to undergo the same management as that for narcotics. Ideally, it is necessary to investigate the prescribing patterns of other compound formulations containing opioids, such as oxycodone and acetaminophen tablets, and tramadol and paracetamol tablets. Also, pharmacogenetic-testing-guided dosing of narcotics has not been performed at SAZHU. Studies have revealed that gene polymorphisms in UDP-glucuronosyltransferase 2B7 (UGT2B7), opioid receptor, Mu 1 (OPRM1), and ATP binding cassette subfamily B member 1 (ABCB1) contribute significantly to the variation in morphine concentrations in individual patients.32,33
Conclusion
Continuous quality improvements have been achieved in safe MMU of narcotics by an integrated multidisciplinary collaboration in an academic medical center hospital in the People’s Republic of China during the journey to JCI accreditation and in the post-JCI accreditation era.
Acknowledgment
This work was supported by Zhejiang Provincial Bureau of Education (grant N20140209).
Disclosure
The authors report no conflicts of interest in this work.
References
ISMP.org [webpage on the Internet]. ISMP high-alert medications. Available from: http://www.ismp.org/Tools/highAlertMedicationLists.asp. Accessed January 4, 2016. | ||
Denison Davies E, Schneider F, Childs S, et al. A prevalence study of errors in opioid prescribing in a large teaching hospital. Int J Clin Pract. 2011;65:923–929. | ||
Logan J, Liu Y, Paulozzi L, Zhang K, Jones C. Opioid prescribing in emergency departments: the prevalence of potentially inappropriate prescribing and misuse. Med Care. 2013;51:646–653. | ||
Desai RJ, Williams CE, Greene SB, Pierson S, Caprio AJ, Hansen RA. Analgesic medication errors in North Carolina nursing homes. J Pain Palliat Care Pharmacother. 2013;27:125–131. | ||
The Joint Commission and the FDA take steps to curb adverse events related to the use and misuse of opioid drugs. ED Manag. 2012;24:112–116. | ||
The Joint Commission. Safe use of opioids in hospitals. Sentinel Event Alert #49; 2012. Available from: http://www.jointcommission.org/assets/1/18/sea_49_opioids_8_2_12_final.pdf. Accessed February 18, 2015. | ||
Shaheen PE, Legrand SB, Walsh D, et al. Errors in opioid prescribing: a prospective survey in cancer pain. J Pain Symptom Manage. 2010;39:702–711. | ||
Jointcommissioninternational.org [webpage on the Internet]. JCI-Accredited Organizations. Available from: http://www.jointcommissioninternational.org/about-jci/jci-accredited-organizations/?c=China&a=Academic%20Medical%20Center%20Hospital%20Program. Accessed February 18, 2015. | ||
National Health and Family Planning Commission People’s Republic of China. The Health Ministry of the People’s Republic of China stipulation (No. 53) – prescription management method [updated 2006 February 14]. Available from: http://www.nhfpc.gov.cn/yzygj/s3572/200804/dd4277bbf3784ff589b9f12b6a0422ab.shtml. Accessed January 2, 2015. Chinese. | ||
National Health and Family Planning Commission People’s Republic of China. Narcotic drugs & psychotropic drugs prescription management stipulation issued by The Health Ministry of the People’s Republic of China [updated 2005 November 17]. Available from: http://www.nhfpc.gov.cn/yzygj/s3573/200804/b9df56a736e74e6d92235f10a98e2230.shtml. Accessed January 2, 2015. Chinese. | ||
Dalton JA, Youngblood R. Clinical application of the World Health Organization analgesic ladder. J Intraven Nurs. 2000;23:118–124. | ||
Friesen KJ, Falk J, Bugden S. Voluntary warnings and the limits of good prescribing behavior: the case for de-adoption of meperidine. J Pain Res. 2015;8:879–884. | ||
Stevenson JG, Pearlman M, Green CR, et al. Altering meperidine prescribing patterns in a university teaching hospital. Jt Comm J Qual Saf. 2004;30:277–281. | ||
Acute Pain Management Guideline Panel. Acute Pain Management: Operative or Medical Procedures and Trauma. Clinical Practice Guideline. AHCPR Pub. No. 92-0032. Rockville, MD: Agency for Health Care Policy and Research, Public Health Service, U.S. Department of Health and Human Services; 1992. | ||
American Pain Society (APS). Principles of Analgesic Use in the Treatment of Acute Pain and Cancer Pain. 4th ed. Glenview, IL: APS; 1999. | ||
Erskine WA, Dick A, Morrell DF, Vital M, van den Heever J. Self-administered intravenous analgesia during labour. A comparison between pentazocine and pethidine. S Afr Med J. 1985;67:764–767. | ||
Elbourne D, Wiseman RA. Types of intra-muscular opioids for maternal pain relief in labour. Cochrane Database Syst Rev. 2000;(2):CD001237. | ||
Mowat J, Garrey MM. Comparison of pentazocine and pethidine in labour. Br Med J. 1970;2:757–759. | ||
Reason J. Human error: models and management. BMJ. 2000;320:768–770. | ||
rxlist.com [webpage on the Internet]. OXYCONTIN® [updated 2015 August 28]. Available from: http://www.rxlist.com/oxycontin-drug.htm. Accessed January 2, 2015. | ||
rxlist.com [webpage on the Internet]. Duragesic® [updated 2013 October 29]. Available from: http://www.rxlist.com/duragesic-side-effects-drug-center.htm. Accessed January 2, 2015. | ||
Carter J. Small-scale study using the PDCA cycle. In: Gift R, Kinney C, Shortell S, editors. Today’s Management Methods: A Guide For The Health Care Executive. New York, NY: Wiley, John & Sons, Inc.; 1996:209–222. | ||
Zhu LL, Xu LC, Wang HQ, Jin JF, Wang HF, Zhou Q. Appropriateness of administration of nasogastric medication and preliminary intervention. Ther Clin Risk Manag. 2012;8:393–401. | ||
Davies DE, Schneider F, Childs S, et al. A prevalence study of errors in opioid prescribing in a large teaching hospital. Int J Clin Pract. 2011;65:923–929. | ||
Sullivan KM, Suh S, Monk H, Chuo J. Personalised performance feedback reduces narcotic prescription errors in a NICU. BMJ Qual Saf. 2013;22:256–262. | ||
National Health and Family Planning Commission of P.R. China. Interpretation of China’s cancer prevention and treatment three-year action plan (2015–2017) [updated 2015 September 11]. Available from: http://en.nhfpc.gov.cn/2015-09/11/content_21845393.htm. Accessed January 2, 2015. | ||
Zhu LL, Zhou Q. Intervention for improving the appropriateness of physician orders for oral medications in geriatric VIP patients during the journey to JCI accreditation. Ther Clin Risk Manag. 2013;9:273–275. | ||
Chen Y, Zhu LL, Zhou Q. Effects of drug pharmacokinetic/pharmacodynamic properties, characteristics of medication use, and relevant pharmacological interventions on fall risk in elderly patients. Ther Clin Risk Manag. 2014;10:437–448. | ||
Cobaugh DJ, Gainor C, Gaston CL, et al. The opioid abuse and misuse epidemic: implications for pharmacists in hospitals and health systems. Am J Health Syst Pharm. 2014;71:1539–1554. | ||
Wang HF, Jin JF, Feng XQ, et al. Quality improvements in decreasing medication administration errors made by nursing staff in an academic medical center hospital: a trend analysis during the journey to Joint Commission International accreditation and in the post-accreditation era. Ther Clin Risk Manag. 2015;11:393–406. | ||
Morriss FH Jr, Abramowitz PW, Nelson SP, Milavetz G, Michael SL, Gordon SN. Risk of adverse drug events in neonates treated with opioids and the effect of a bar-code-assisted medication administration system. Am J Health Syst Pharm. 2011;68:57–62. | ||
Bastami S, Gupta A, Zackrisson AL, Ahlner J, Osman A, Uppugunduri S. Influence of UGT2B7, OPRM1 and ABCB1 gene polymorphisms on postoperative morphine consumption. Basic Clin Pharmacol Toxicol. 2014;115:423–431. | ||
Hajj A, Peoc’h K, Laplanche JL, et al. Genotyping test with clinical factors: better management of acute postoperative pain? Int J Mol Sci. 2015;16:6298–6311. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



