Back to Journals » International Medical Case Reports Journal » Volume 16
Sacubitril/Valsartan Shows Improvement of the Cardio-Ankle Vascular Index in a Hypertensive Patient
Authors Shimizu K , Tabata T, Iwakawa M, Sato S , Kinoshita T
Received 1 June 2023
Accepted for publication 17 August 2023
Published 22 August 2023 Volume 2023:16 Pages 461—465
DOI https://doi.org/10.2147/IMCRJ.S422196
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Kones
Kazuhiro Shimizu,1 Tsuyoshi Tabata,2 Masahiro Iwakawa,1 Shuji Sato,1 Toshio Kinoshita1,2
1Division of Cardiovascular Medicine, Department of Internal Medicine, Toho University Sakura Medical Center, Chiba, Japan; 2Department of Clinical Functional Physiology (Sakura), Toho University Sakura Medical Center, Chiba, Japan
Correspondence: Kazuhiro Shimizu, Department of Internal Medicine, Toho University Sakura Medical Center, 564-1 Shimoshizu, Sakura-shi, Chiba, 285-8741, Japan, Tel +81-43-462-8811, Fax +81-43-462-8820, Email [email protected]
Abstract: A 72-year-old man presented to our clinic with hypertension. Arterial stiffness evaluated by cardio ankle vascular index (CAVI) was markedly increased at 13.5. We treated him using 80 mg/day of valsartan for three months. CAVI was decreased from 13.5 to 13.0. However, his BP fluctuations were still high. We changed the treatment to angiotensin receptor-neprilysin inhibitor (ARNI) with increasing doses up to 400 mg. Independent of the change in blood pressure at the time of measurement, CAVI improved with ARNI dose. Hypertension treatment with an awareness of the cardio-vascular interaction might be a possibility prevents future heart failure development effectively.
Keywords: hypertension, angiotensin receptor-neprilysin inhibitor, arterial stiffness, cardio ankle vascular index
Introduction
Angiotensin receptor-neprilysin inhibitor (ARNI) is a dual-acting angiotensin II-receptor and neprilysin inhibitor in a single drug. Ruilope et al previously demonstrated significant reductions in blood pressure (BP) with ARNI compared with the angiotensin II receptor blocker valsartan.1 Moreover, Kario et al reported the effectiveness of ARNI in the treatment of Asian hypertensive patients in a randomized, double-blind, placebo-controlled study.2 ARNI is currently used worldwide for the treatment of chronic heart failure due to the evidence based on not only reduced ejection fraction3 but also preserved ejection fraction.4 To date, however, few countries have approved ARNI for the treatment of hypertension. The usual adult dosage for hypertension is 200 mg of ARNI once daily. And the maximum dosage is 400 mg. 200 mg tablet of ARNI contains 97.2 mg of sacubitril and 102.8 mg of valsartan. Left ventricular mass index (LVMI) and relative wall thickness (RWT) are closely related to the degree and history of hypertension, which becomes a potential risk for heart failure in cardiac remodeling. ARNI has shown to improve LVMI compared with Olmesartan.5 As for the mechanism and contribution factors for left ventricular geometry, it is mentioned that the degree and the duration of hypertension are very important factors, and the arterial stiffness is also supposed to be important as an afterload of the left ventricle.
Recently, the cardio-ankle vascular index (CAVI) was developed as an indicator of arterial stiffness from the origin of the aorta to the ankle.6 CAVI is a numerical index of arterial stiffness derived from the stiffness parameter beta theory and the application of the Bramwell–Hill equation for pulse wave velocity (PWV). The feature of CAVI is its independency from blood pressure at the time of measurement. As a marker of arterial diastolic-to-systolic stiffening, CAVI may be a suitable marker as arterial stiffness in the studies on the relationship between left ventricular geometrical remodeling and arterial stiffness as an afterload. This is the first case report on ARNI that explicitly states arterial stiffness without linking it to blood pressure at the time of measurement.
Case Presentation
A 72-year-old man presented to our outpatient clinic with hypertension. He was not a current smoker, but his past smoking history was 20 cigarettes per day for about 50 years. Hyperuricemia and pulmonary emphysema were confirmed by blood tests and CT scan in our hospital. He was 173-cm tall and weighed 60 kg with a body mass index of 20.0 kg/m2. Using an automated BP monitor outside the examination room, his BP was 181/117 mmHg. An electrocardiogram showed no initial left ventricular hypertrophy. The following day, he underwent an echocardiogram and measurement of CAVI in our department of clinical physiology. After resting for 10 minutes in supine position on a bed, CAVI was measured using a VaSera VS-3000 (Fukuda Denshi Co., Ltd, Tokyo, Japan). Simultaneously, BP and heart rate were measured using the VaSera system.
His BP was 141/91 in left arm and CAVI was high at 13.5. Echocardiography showed that his left ventricular function was normal (Table 1). According to the classification of the American Society of Echocardiography,7,8 the geometry classification of his heart showed concentric remodeling. Left ventricular global longitudinal strain (LVGLS) was quantitated offline, using the EchoPAC PC system, version 113 (General Electric Healthcare, Chicago, IL, USA). Echocardiography was performed by trained sonographers at the same time of the day. LVGLS was calculated as 3 cycles were recorded at a frame rate between 40 and 80 frames per second and the average negative peak of longitudinal strain in the 4-chamber, 2-chamber, and apical long-axis in 17 segments in accordance with current guidelines. His LVMI was 86.4 g/m2 and within normal range, but the RWT was increased at 0.61. We treated him using 80 mg/day of valsartan for three months. He had no other concomitant medications. On subsequent evaluation, CAVI was decreased from 13.5 to 13.0. Urine protein was disappeared after administration of valsartan (Table 1). However, his BP fluctuations evaluated by automatic home sphygmomanometer were still high. BP was measured in his left arm before breakfast and dinner. BP was measured twice at each measurement, and the average was recorded in a notebook. Intensified BP treatment was thought to be necessary. We changed the treatment from 80mg of valsartan to ARNI with increasing doses up to 400 mg. About three months later, his CAVI was markedly improved from 13.0 to 10.8. Moreover, LVMI and RWT were also improved (Figure 1). ARNI was reduced from 400 mg to 200 mg based on the good BP control. However, CAVI increased from 10.8 to 12.1 despite controlled BP (Figure 1).
 |
Table 1 Parameters Analyzed in the Follow-Up Period |
Discussion
Hypertension is a major cause of heart failure through cardiac remodeling. BP fluctuations have traditionally been a major problem due to the difficulty of assessment. To overcome this obstacle, multiple BP measurements are taken in the examination room, and home BP measurements are often used in Japan. Arterial stiffness has developed as an indicator that reflects the pathophysiology of atherosclerosis. As for a method of quantitative assessment of arterial stiffness, PWV has been used for several decades; it had been thought to be a kind of surrogate marker of arteriosclerosis. However, PWV depends inherently on BP changes at the time of measurement. CAVI, which was newly developed as an arterial stiffness index, was derived from the stiffness parameter beta theory with the application of Bramwell Hill’s equation.6,9 CAVI is measured automatically and with high measurement reproducibility. The coefficient of variation was reported to be 3.8%.6 CAVI increases with stress and smoking.9 Fortunately, this case was not in such a situation at measurement time.
In the present case, initially, the patient’s systolic BP fluctuated widely from about 140 mmHg to 180 mmHg. Especially in these cases, it is difficult to accurately assess the pathophysiology with indices that are affected by the blood pressure at the time of measurement.
Recently, the concept of cardiovascular interaction was proposed using CAVI.10–12 CAVI reflects smooth muscle cell contraction and relaxation. As for the effects on BP and CAVI, in the present case, when the sacubitril/valsartan dose was tapered from 400 mg to 200 mg, CAVI increased without any changes in cardiac function, such as global longitudinal strain (Table 1). Notably, when the ARNI dose was subsequently reduced from 400 mg to 200 mg, blood pressure at the time of measurement decreased, but CAVI increased (Figure 1). Based on these results, ARNI showed the possibility of a powerful effect on the improvement of arterial stiffness. As for the mechanism of the stabilization of BP, it was thought that ARNI relaxed vascular smooth muscle cells and reduced peripheral vascular resistance.
The established evidence of ARNI in large clinical trials of heart failure patients with standard medical therapy might be a result of strong vasoactive properties such as cardiovascular interaction (Figure 2). It has been reported that improvement in left ventricular ejection fraction after chronic heart failure treatment correlates with a decrease in CAVI.13 This phenomenon may lead to one of the keys to the ameliorative effect of ARNI on heart failure. Observational studies of multiple cases are needed to confirm this fact. In conclusion, hypertension treatment with an awareness of the cardiovascular interaction might be a possibility to prevent future heart failure development effectively.
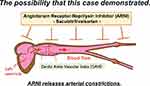 |
Figure 2 The image of ARNI’s action on heart and vessels. |
Institutional Approval
This case report was approved by Toho University (2022-154).
Consent
The authors confirm that written consent for submission and publication of this case report including images and associated text has been obtained from the patient in line with CARE guidance.
Acknowledgments
The authors thank Kohji Shirai, Honorary Professor of Toho University.
Funding
The authors declare no funding information.
Disclosure
The authors state that they have no conflict of interest.
References
1. Ruilope LM, Dukat A, Böhm M, et al. Blood-pressure reduction with LCZ696, a novel dual-acting inhibitor of the angiotensin II receptor and neprilysin: a randomised, double-blind, placebo-controlled, active comparator study. Lancet. 2010;375(9722):1255–1266. doi:10.1016/S0140-6736(09)61966-8
2. Kario K, Sun N, Chiang FT, et al. Efficacy and safety of LCZ696, a first-in-class angiotensin receptor neprilysin inhibitor, in Asian patients with hypertension: a randomized, double-blind, placebo-controlled study. Hypertension. 2014;63:698–705. doi:10.1161/HYPERTENSIONAHA.113.02002
3. McMurray JJV, Packer M, Desai AS, et al; PARADIGM-HF Investigators and Committees. Angiotensin–neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371:993–1004. doi:10.1056/NEJMoa1409077
4. Solomon SD, McMurray JJV, Anand IS, et al; PARAGON-HF Investigators and Committees. Angiotensin–neprilysin inhibition in heart failure with preserved ejection fraction. N Engl J Med. 2019;381(17):1609–1620. doi:10.1056/NEJMoa1908655
5. Schmieder RE, Wagner F, Mayr M, et al. The effect of sacubitril/valsartan compared to olmesartan on cardiovascular remodelling in subjects with essential hypertension: the results of a randomized, double-blind, active-controlled study. Eur Heart J. 2017;38(44):3308–3317. doi:10.1093/eurheartj/ehx525
6. Shirai K, Utino J, Otsuka K, et al. A novel blood pressure independent arterial wall stiffness parameter: cardio-ankle vascular index (CAVI). J Atheroscler Thromb. 2006;13(2):101–107. doi:10.5551/jat.13.101
7. Lang RM, Bierig M, Devereux RB, et al; Chamber Quantification Writing Group; American Society of Echocardiography’s Guidelines and Standards Committee; European Association of Echocardiography. Recommendations for chamber quantification: a report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr. 2005;18(12):1440–1463. doi:10.1016/j.echo.2005.10.005
8. Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2015;28(1):1–39.e14. doi:10.1016/j.echo.2014.10.003
9. Hayashi K, Yamamoto T, Takahara A, et al. Clinical assessment of arterial stiffness with cardio-ankle vascular index: theory and applications. J Hypertens. 2015;33(9):1742–1757. doi:10.1097/HJH.0000000000000651
10. Shimizu K, Tabata T, Kiyokawa H, et al. A case demonstrating the cardio-vascular interaction by a new cardio-ankle vascular index during the treatment of concentric hypertrophy. Cardiol Res. 2019;10(1):54–58. doi:10.14740/cr812
11. Tabata T, Shimizu K, Morinaga Y, et al. The relationship between cardio- ankle vascular index and left atrial phasic function in hypertensive patients with preserved ejection fraction. Front Med Technol. 2021;3:724089. doi:10.3389/fmedt.2021.724089
12. Tabata T, Sato S, Ohno R, et al. Cardio-vascular interaction evaluated by speckle-tracking echocardiography and cardio-ankle vascular index in hypertensive patients. Int J Mol Sci. 2022;23(22):14469. doi:10.3390/ijms232214469
13. Zhang C, Ohira M, Iizuka T, et al. Cardio-ankle vascular index relates to left ventricular ejection fraction in patients with heart failure. A retrospective study. Int Heart J. 2013;54(4):216–221. doi:10.1536/ihj.54.216
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

