Back to Journals » OncoTargets and Therapy » Volume 12
RRAD suppresses the Warburg effect by downregulating ACTG1 in hepatocellular carcinoma
Authors Yan Y , Xu H , Zhang L , Zhou X, Qian X , Zhou J , Huang Y , Ge W, Wang W
Received 11 December 2018
Accepted for publication 17 January 2019
Published 28 February 2019 Volume 2019:12 Pages 1691—1703
DOI https://doi.org/10.2147/OTT.S197844
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Leo Jen-Liang Su
Yingcai Yan,1,2,* Hao Xu,1,2,* Linshi Zhang,1,2 Xiaohu Zhou,1,2 Xiaohui Qian,1,2 Jiarong Zhou,1,2 Yu Huang,1,2 Wenhao Ge,1,2 Weilin Wang1–4
1Division of Hepatobiliary and Pancreatic Surgery, Department of Surgery, The First Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, China; 2Key Laboratory of Precision Diagnosis and Treatment for Hepatobiliary and Pancreatic Tumor of Zhejiang Province, The First Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, China; 3Clinical Research Center of Hepatobiliary and Pancreatic Diseases of Zhejiang Province, The First Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, China; 4State Key Laboratory and Collaborative Innovation Center for Diagnosis and Treatment of Infectious Disease, Zhejiang University, Hangzhou, China
*These authors contributed equally to this work
Purpose: Hepatocellular carcinoma (HCC) is a common malignancy with poor prognosis and limited therapeutic options. Ras-related associated with diabetes (RRAD) belongs to the subfamily of Ras-related GTPases and is associated with several types of cancer, including HCC, although the mechanisms involving RRAD in HCC remains unknown.
Patients and methods: We aimed to elucidate the role of RRAD and whether it affects glucose metabolism in HCC by immunohistochemically examining tissue samples from HCC patients and assessing the effect of RRAD overexpression and knockdown on the glucose metabolism, proliferation, cell cycle, and apoptosis of HCC cell lines SK-Hep-1 and Huh7, as well as on tumor progression in vivo.
Results: We demonstrated that RRAD binds to actin gamma 1 (ACTG1). RRAD suppressed aerobic glycolysis in HCC by downregulating ACTG1. On the other hand, ACTG1 promoted HCC proliferation by regulating the cell cycle via downregulation of cyclins and cyclin-dependent kinases and inhibited apoptosis through the mitochondrial apoptosis pathway in vitro. In addition, RRAD retarded tumor growth by downregulating ACTG1 in vivo. ACTG1 was overexpressed in HCC tissues compared with adjacent normal tissues, whereas the expression of RRAD was low in tumor tissues. Low RRAD levels were significantly correlated with large tumor size and advanced tumor stage; high ACTG1 levels were significantly correlated with advanced tumor stage. Furthermore, Kaplan–Meier survival curves showed that HCC patients with high RRAD and low ACTG1 expression may have a better prognosis.
Conclusion: We have shown that RRAD exhibits a tumor-suppressing role in HCC by downregulating glucose metabolism and ACTG1 expression, thus lowering cell proliferation, arresting the cell cycle, and increasing apoptosis. These findings indicate that ACTG1 may act as a downstream effector of RRAD and open a new avenue for potential HCC treatment.
Keywords: hepatocellular carcinoma, Ras-related associated with diabetes, actin gamma 1, the Warburg effect, tumorigenicity
Introduction
Hepatocellular carcinoma (HCC) is a frequently occurring malignancy with poor prognosis, whose mortality rate ranks second among cancer-related deaths.1 With average survival rates between 6 and 20 months, a better knowledge of the pathogenic mechanisms of HCC is urgently required to develop novel strategies for treating the disease.2,3
Under normal circumstances, cells depend upon mitochondrial oxidative phosphorylation, while cancer cells prefer aerobic glycolysis, also known as the Warburg effect.4 Therefore, metabolic reprogramming must play a vital role in the biobehavioral mechanism of cancer. Increased glucose uptake levels and lactate production have been linked to tumor progression,5 and recent studies have revealed that oncogene activation and/or tumor suppressor gene mutations are related to increased glycolysis.6–8
Ras-related associated with diabetes (RRAD), which belongs to the 35–39 kDa class of the subfamily of Ras-related GTPases, was first identified in type II diabetes patients because of its abnormally high expression.9 We have previously shown that RRAD is linked to proliferation, apoptosis, and the Warburg effect in HCC cells.10 However, the exact mechanism that involves RRAD in HCC remains unknown.
In the present study, RRAD was found to play a critical role in the negative regulation of glycolysis through the induction of actin gamma 1 (ACTG1), which belongs to γ-actins, a class of proteins existing in most cell types as components of the cytoskeleton. ACTG1 is reportedly related to conditions such as hearing loss,11 while a more recent study implicated that ACTG1 overexpression could improve the proliferation and clone formation of skin cancer cells.12 However, the biological functions of ACTG1 in HCC cells have not been studied yet.
In our study, we demonstrated that RRAD binds to ACTG1. Knockdown of ACTG1 in HCC cell lines likely reduces cell proliferation and the Warburg effect and promotes apoptosis, while overexpression of ACTG1 has the opposite effect. Moreover, ACTG1 is upregulated in HCC cells, while its high expression in tumor tissues may be related to a poorer prognosis. Therefore, our results reveal a significant role of and mechanism by which RRAD may regulate the Warburg effect in HCC, thus contributing to its tumor-suppressing role.
Patients and methods
Patient data, tissue samples, and immunohistochemistry
Ninety HCC tumor samples and corresponding peri-tumor tissues were obtained from patients who underwent HCC resection in our department between 2014 and 2016. The patients had no liver-related comorbidities and did not receive any preoperative therapy. The tissues were conserved at −80°C until further analysis. Clinical data were collected and assessed by two independent physicians. The study was approved by the Human Research Ethics Committee of The First Affiliated Hospital, College of Medicine, Zhejiang University. Written informed consent was obtained from all patients. We confirmed that this was conducted in accordance with the Declaration of Helsinki.
Anti-RRAD rabbit polyclonal antibody (ab75100; Abcam, Cambridge, UK) and anti-ACTG1 mouse monoclonal antibody (ab123034; Abcam) were used as the primary antibodies for immunohistochemical staining; the staining was performed as described previously.13 All slides were examined by two pathologists, who were blinded to clinical patient data.
Cell culture
The HCC cell lines Huh7 and SK-Hep-1 were purchased from ATCC (https://www.atcc.org/) commercially and cultured in DMEM (Thermo Fisher Scientific, Waltham, MA, USA), with 10% heat-inactivated FBS (Sigma-Aldrich Co., St Louis, MO, USA). All cells were maintained in an incubator containing 95% air and 5% CO2 at 37°C.
Cell transfection
RRAD siRNAs, together with negative controls (NCs), were synthesized by GenePharma (Shanghai, China) and used to transfect the SK-Hep-1 cells at a concentration of 10 nM. The siRNA sequences were as follows: NC, 5′-UUCUCCGAACGUGUCACGUTT-3′ and antisense 5′-ACGUGACACGUUCGGAGAATT-3′; and si-RRAD, 5′-GACUCAGACGAGAGCGUUUTT-3′ and antisense 5′-AAACGCUCUCGUCUGAGUCTT-3′. The cells were treated as described previously.10 Adenoviruses overexpressing RRAD, NC/P53, or NC and replication-defective lentiviruses encoding RRAD, NC/ACTG1, or NC were constructed by Genechem. ACTG1-specific shRNA-expressing (sh-ACTG1) lentiviruses were produced by Sigma-Aldrich Co. (TRCN0000029478) using the sequence “CCGGCGCATCCTCCTCTTCTCTGGACTCGAGTCCAGAGAAGAGGAGGATGCGTTTTT”. The cells were treated as described previously.10
Western blotting
Western blotting was performed as described previously.10 GAPDH was stained (anti-GADPH [60004-1-Ig]; Proteintech, Rosemount, IL, USA) and used for normalization. Primary antibodies against RRAD, ACTG1, P53, HIS, GST, cyclin A2, cyclin D1, cyclin E1, CDK2, CDK4, caspase-3, PARP, BAX, GLUT1, LDHA, FBP1, and PKM2 (Abcam) were used.
Glucose uptake, lactate production assay, and extracellular acidification rate measurement
Glucose uptake was measured using 2-(N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)amino)-2-deoxyglucose (Thermo Fisher Scientific) as described previously.14 Lactate production was measured by Lactate Colorimetric Assay Kit II (BioVision, Milpitas, CA, USA). For extracellular acidification rate (ECAR) measurement, the Extracellular Flux Analyzer and XF Glycolysis Stress kit (Seahorse Bioscience, North Billerica, MA, USA) were used.
Co-immunoprecipitation (Co-IP) assays and LC-MS/MS analysis
Co-IP assays were performed as described previously.15 To detect the potential RRAD-binding proteins, GST-RRAD protein was pulled down by IP using anti-GST beads. Analysis was performed using liquid chromatography–tandem mass spectrometry (LC-MS/MS; Ekspert NanoLC and TripleTOF 5600+; Sciex, Framingham, MA, USA).
Cell proliferation and cell cycle analysis
Cell Counting Kit-8 (Dojindo Laboratories, Kumamoto, Japan) and EdU assay (Cell-Light™ EdU Apollo567 In Vitro Imaging Kit; Ribobio, Guangzhou, China) were used for cell proliferation analysis. Flow cytometry (Cytomics FC 500; Beckman Coulter, Miami, FL, USA) was used for cell cycle analysis, as described previously.10
Cell apoptosis assay
Forty-eight hours after transfection, the cells were harvested and double stained with 5 μL of annexin V-fluorescein isothiocyanate and 5 μL of propidium iodide in the dark for 30 minutes. Apoptosis distribution was then detected by flow cytometry.
Subcutaneous tumor growth
All experimental animal procedures were performed according to the institutional ethical requirements and were approved by the Zhejiang Medical Experimental Animal Care Commission. Cells and mice were treated as described previously,10 and corresponding data were recorded. The tumor volume was calculated using the following formula: volume = L × W2/2, where L and W are the longest and shortest diameters of the tumors, respectively.
Statistical analyses
Data were analyzed using SPSS software (version 24; IBM Corporation, Armonk, NY, USA). Figures were drawn using GraphPad Prism software (version 6.0; GraphPad, San Diego, CA, USA). The results were expressed as mean values ± SD from three independent experiments. Differences were considered statistically significant for P-values <0.05, using Student’s t-test.
Results
RRAD suppresses the Warburg effect in HCC cells
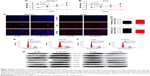
Recently, it has been shown that the viability of cancer cells is linked to glucose levels and lactate levels in the extracellular milieu.16 To evaluate whether RRAD plays a role in the regulation of glucose metabolism, we silenced RRAD in SK-Hep-1 cells using siRNA and transfected Huh7 cells with an RRAD-overexpressing lentivirus. Effective knockdown and overexpression of RRAD was confirmed (Figure 1G and H). Two hallmarks of glycolysis, glucose uptake and lactate production, were measured, and both were significantly enhanced in SK-Hep-1 cells (Figure 1A and C) and decreased in Huh7 cells (Figure 1B and D). Moreover, real-time ECAR measurement and the glycolysis stress assays were used to determine various parameters of glycolytic capacity of the cells. These showed that knockdown of RRAD in SK-Hep-1 cells with increased ECAR (Figure 1E), while overexpression of RRAD in Huh7 cells decreased ECAR (Figure 1F).
In order to determine the underlying mechanism that results in the inhibition of the Warburg effect, we detected relative protein levels. The results indicated that glucose transporter 1 (GLUT1) was upregulated when RRAD was knocked down, while GLUT1 levels were lower when RRAD was overexpressed. There was no significant difference in the levels of other proteins (Figure 1G and H).
RRAD binds to ACTG1 and is regulated by P53
To further study the mechanism by which RRAD represses the Warburg effect, STRING was used to predict the proteins that interact with RRAD.35 We found that P53 may be linked to RRAD (Figure 2A). We then transfected Huh7 cells with RRAD- and P53-overexpressing adenoviruses and found that RRAD expression was significantly upregulated when P53 was overexpressed, while there was no significant difference in P53 expression when RRAD was overexpressed (Figure 2B). Using Co-IP and LC-MS/MS assays to further explore the potential proteins that RRAD interacts with (Figure 2C) and glutathione-S-transferase (GST) pull-down assays to obtain more accurate results, we confirmed that RRAD interacts with ACTG1 (Figure 2D).
  | Figure 2 Proteins that interact with RRAD. |
ACTG1 is associated with HCC cell proliferation, cell cycle, and apoptosis
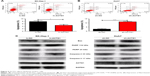
Next, we aimed to determine the effect of ACTG1 expression on HCC cells. The CCK-8 assay showed that ACTG1 overexpression in SK-Hep-1 cells and knockdown in Huh7 cells increased and decreased the viability of the cells, respectively (Figure 3A and B), which was in accordance with the results of the EdU assay (Figure 3C and D).
After silencing ACTG1, we observed an increase in the percentage of cells in G1-phase and a reduction in S-phase cells compared with the NC cells (Figure 3F). In addition, this resulted in decreased expression of cyclin A2, cyclin D1, cyclin E1, CDK2, and CDK4 (Figure 3G). As expected, ACTG1 overexpression in SK-Hep-1 cells had the opposite effect (Figure 3E and G).
In addition, overexpression of ACTG1 decreased apoptosis (Figure 4A), whereas its knockdown increased apoptosis (Figure 4B), both effects being accompanied by a corresponding alteration in the levels of the apoptosis-related proteins BAX, cleaved caspase-3, and PARP (Figure 4C).
ACTG1 promotes the Warburg effect in vitro
Glucose uptake, lactate production, and ECAR measurements showed that overexpression of ACTG1 improved glucose uptake, lactate production, and ECAR (Figure 5A, C and E) along with the upregulation of GLUT1 (Figure 5G). Contrastingly, knockdown of ACTG1 reduced these parameters (Figure 5B, D, F and H).
ACTG1 acts as a functional downstream effector of RRAD in HCC cells
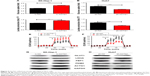
To investigate whether the tumor-suppressing role of RRAD in HCC cells is mediated by inhibiting the expression of ACTG1, SK-Hep-1 cells were co-transfected with lentiviruses encoding RRAD and ACTG1; RRAD-induced ACTG1 downregulation was rescued following the co-transfection (Figure 6F). Moreover, the inhibitory role of RRAD in glucose uptake, lactate production, ECAR, and cell proliferation was reverted when ACTG1 was overexpressed (Figure 6A–D), while the rate of apoptosis was reduced (Figure 6E). The mRNA level of ACTG1 was downregulated when RRAD was overexpressed (Figure 6F). The relative protein levels changed correspondingly (Figure 6G).
Overexpression of RRAD suppressed tumor progression through ACTG1 in vivo
To confirm the tumor-suppressing role of RRAD in vivo, a subcutaneous xenograft model was constructed. SK-Hep-1 cells transfected with RRAD− and RRAD+ ACTG1-overexpression lentiviruses, as well as NC cells, were injected into nude mice. In accordance with the in vitro results, the tumors in the mice injected with RRAD-overexpressing cells were obviously smaller, which could be rescued by ACTG1 over expression (Figure 7A–C). To further investigate the effect of RRAD on HCC in vivo, we performed an immunohistochemistry assay on the tumor removed from nude mice. The expression of GLUT1 was higher in the NC group, and the downregulation of GLUT1 induced by the overexpression of RRAD can be rescued by ACTG1 overexpression, which implied that inhibition of tumor growth by RRAD is related to suppression of the Warburg effect (Figure 7D).
Expression of RRAD is low when ACTG1 is overexpressed in HCC tumor specimens and is linked to poor prognosis
Expression of RRAD in tumor tissues was lower, while ACTG1 expression was higher, than in the corresponding peri-tumor tissues in the 90-patient cohort. In addition, low expression of RRAD was significantly correlated with increased tumor size and advanced tumor stages (Table 1), and high expression of ACTG1 was significantly correlated with advanced tumor stages (Table 2). Low RRAD expression was associated with poor prognosis according to the Kaplan–Meier survival curves, while high ACTG1 expression was correlated with poor prognosis in patients with HCC (Figure 7E and F). Taken together, these indicate that patients with high levels of RRAD and low levels of ACTG1 may have a better prognosis (Figure 7G).
Discussion
Understanding the Warburg effect is of importance because it is specific to cancer cells,4,6,17,18 wherein it is employed to meet their energy and biosynthesis demands. We have previously demonstrated that RRAD plays a role in the negative regulation of cell proliferation, cell migration, and the Warburg effect in HCC cells.10 As was one of the first members of 35-39KD class of novel Ras-related GTPases, RRAD is connected with skeletal muscle β-tropomyosin.19 It has recently been reported that it is associated with tumorigenicity, in both solid and non-solid tumors.20–22 However, the exact mechanism of how it suppresses the tumorigenicity of HCC remains unknown.
In the present study, we used STRING to predict the proteins that interact with RRAD.35 We found that P53 may be linked to RRAD and RRAD may act as a downstream factor of P53. Next, we used Co-IP and LC-MS/MS assays to screen for the potential proteins that interact with RRAD. Using the GST pull-down assay, we identified ACTG1 as an important RRAD-binding protein. ACTG1 belongs to the family of actins, which participate in a variety of cellular activities, including in cell viability and cell shape maintenance and phagocytosis.23,24 ACTG1 is essential for the stability of hair cell stereocilia,25 while its mutations may cause deafness or the Baraitser–Winter syndrome.26 A recent study revealed the oncogenic effect of ACTG1 in skin cancer, wherein its overexpression may lead to cancer cell proliferation and migration.27 However, the role of ACTG1 in HCC has not been previously reported.
Here, we showed that ACTG1 plays a crucial role in HCC. Namely, its expression was higher in HCC tissues than in the corresponding peri-tumor tissues, and this was significantly correlated to advanced tumor stages. On the other hand, the expression of RRAD was low in HCC tissues, and this was significantly correlated to large tumor size and advanced tumor stage. These findings indicate that HCC patients with high RRAD and low ACTG1 levels may have a better prognosis.
Furthermore, we demonstrated that ACTG1 has a tumor-promoting function, that is, it improved HCC cell proliferation, while its knockdown led to G1 phase arrest and downregulation of the levels of cyclins A2, D1, and E1 and CDK2 and CDK4, proteins associated with the cell cycle. Since binding of cyclin A or E to CDK2 facilitates G1/S transition28 and the cyclin D/CDK4 complex inactivates retinoblastoma proteins and leads to E2F-dependent transcription advancement,29,30 the effect of ACTG1 on HCC cell proliferation likely occurred via the upregulation of cyclins and CDKs.
In addition, as reduced apoptosis is associated with tumor development, we tested the effect of ACTG1 on the major effectors of the intrinsic and extrinsic apoptosis pathways in HCC cells.31–33 The rate of apoptosis in HCC cells, accompanied by the expression of BAX, cleaved caspase-3, and PARP, increased when ACTG1 was knocked down, while ACTG1 overexpression had the opposite effect, indicating that ACTG1 may indeed contribute to HCC development by inhibiting the intrinsic apoptosis pathway.
Finally, we have shown that RRAD is linked to glucose metabolism in HCC cells by downregulating GLUT1, which initiates glycolysis by increasing glucose import into the cytoplasm.34 The results indicated that ACTG1 may promote the Warburg effect by upregulating the expression of GLUT1 in HCC cells. However, whether the tumor-suppressing role of RRAD in HCC cells is achieved via the inhibition of ACTG1 expression remains to be revealed. Herein, we found that RRAD-induced ACTG1 downregulation was rescued following co-transfection, and the inhibitory role of RRAD in glucose uptake, lactate production, ECAR, and cell proliferation was reverted when ACTG1 was overexpressed, indicating that ACTG1 may act as a functional downstream effector of RRAD in HCC cells. These findings may open a new avenue for potential HCC treatment.
Conclusion
Our study shows for the first time that RRAD binds to ACTG1. RRAD suppressed the Warburg effect in HCC cells by downregulating ACTG1. ACTG1 promoted HCC proliferation by regulating the cell cycle and suppressing the apoptosis of HCC cells in vitro. In addition, RRAD retarded tumor growth by downregulating ACTG1 in vivo. ACTG1 was overexpressed in HCC tissues compared with corresponding peri-tumor tissues, whereas the expression of RRAD was lower in tumor tissues. The low expression of RRAD was significantly correlated to large tumor size and advanced tumor stage and high expression of ACTG1 was significantly correlated to advanced tumor stage, indicating that patients with high RRAD and low ACTG1 levels may have a better prognosis. These results identify ACTG1 as a new oncogene and enrich the recessive oncogenic role of RRAD in HCC, which may provide new methods for cancer therapy.
Acknowledgment
This work was supported by The National Natural Science Foundation of China (No 81572307, 81773096) and Major Project of Medical and Health Technology Development Program in Zhejiang Province (Nos 7211902, 2016KYB083, LGD19C040006).
Disclosure
The authors report no conflicts of interest in this work.
References
Stewart BW, Wild CP, editors. World Cancer Report. Organization World health; 2014. Available from: http://apps.who.int/bookorders/anglais/detart1.jsp?codlan=1&codcol=76&codcch=31. Accessed February 19, 2019. | ||
Byam J, Renz J, Millis JM. Liver transplantation for hepatocellular carcinoma. Hepatobiliary Surg Nutr. 2013;2(1):22–30. | ||
Bravi F, Bosetti C, Tavani A, Gallus S, La Vecchia C. Coffee reduces risk for hepatocellular carcinoma: an updated meta-analysis. Clin Gastroenterol Hepatol. 2013;11(11):1413–1421. | ||
Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science. 2009;324(5930):1029–1033. | ||
Kroemer G, Pouyssegur J. Tumor cell metabolism: cancer’s Achilles’ heel. Cancer Cell. 2008;13(6):472–482. | ||
Hitosugi T, Chen J. Post-translational modifications and the Warburg effect. Oncogene. 2014;33(34):4279–4285. | ||
DeBerardinis RJ, Lum JJ, Hatzivassiliou G, Thompson CB. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 2008;7(1):11–20. | ||
Hitosugi T, Kang S, vander Heiden MG, et al. Tyrosine phosphorylation inhibits PKM2 to promote the Warburg effect and tumor growth. Sci Signal. 2009;2(97):ra73. | ||
Reynet C, Kahn CR. Rad: a member of the Ras family overexpressed in muscle of type II diabetic humans. Science. 1993;262(5138):1441–1444. | ||
Yan Y, Xie M, Zhang L, et al. Ras-related associated with diabetes gene acts as a suppressor and inhibits Warburg effect in hepatocellular carcinoma. Onco Targets Ther. 2016;9:3925–3937. | ||
Chou CC, Davis RC, Fuller ML, et al. Gamma-actin: unusual mRNA 3′-untranslated sequence conservation and amino acid substitutions that may be cancer related. Proc Natl Acad Sci U S A. 1987;84(9):2575–2579. | ||
Dong X, Han Y, Sun Z, Xu J. Actin gamma 1, a new skin cancer pathogenic gene, identified by the biological feature-based classification. J Cell Biochem. 2018;119(2):1406–1419. | ||
Wang W, Xie Q, Zhou X, et al. Mitofusin-2 triggers mitochondria Ca2+ influx from the endoplasmic reticulum to induce apoptosis in hepatocellular carcinoma cells. Cancer Lett. 2015;358(1):47–58. | ||
Sova P, Feng Q, Geiss G. Discovery of novel methylation biomarkers in cervical carcinoma by global demethylation and microarray analysis. Cancer Epidemiol Biomark Prev. 2006;15(1):114–123. | ||
Liu J, Zhang C, Wang XL, et al. E3 ubiquitin ligase TRIM32 negatively regulates tumor suppressor p53 to promote tumorigenesis. Cell Death Differ. 2014;21(11):1792–1804. | ||
Dhup S, Dadhich RK, Porporato PE, Sonveaux P. Multiple biological activities of lactic acid in cancer: influences on tumor growth, angiogenesis and metastasis. Curr Pharm Des. 2012;18(10):1319–1330. | ||
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–674. | ||
Warburg O. On the origin of cancer cells. Science. 1956;123(3191):309–314. | ||
Goitre L, Trapani E, Trabalzini L, Retta SF. The Ras superfamily of small GTPases: the unlocked secrets. Methods Mol Biol. 2014;1120:1–18. | ||
Yeom SY, Lee SJ, Kim WS, Park C. Rad knockdown induces mitochondrial apoptosis in bortezomib resistant leukemia and lymphoma cells. Leuk Res. 2012;36(9):1172–1178. | ||
Mo Y, Midorikawa K, Zhang Z, et al. Promoter hypermethylation of Ras-related GTPase gene RRAD inactivates a tumor suppressor function in nasopharyngeal carcinoma. Cancer Lett. 2012;323(2):147–154. | ||
Suzuki M, Shigematsu H, Shames DS, et al. Methylation and gene silencing of the Ras-related GTPase gene in lung and breast cancers. Ann Surg Oncol. 2007;14(4):1397–1404. | ||
Lloyd C, Gunning P. Beta- and gamma-actin genes differ in their mechanisms of down-regulation during myogenesis. J Cell Biochem. 2002;84(2):335–342. | ||
Lloyd C, Gunning P. Noncoding regions of the gamma-actin gene influence the impact of the gene on myoblast morphology. J Cell Biol. 1993;121(1):73–82. | ||
Rendtorff ND, Zhu M, Fagerheim T, et al. A novel missense mutation in ACTG1 causes dominant deafness in a Norwegian DFNA20/26 family, but ACTG1 mutations are not frequent among families with hereditary hearing impairment. Eur J Hum Genet. 2006;14(10):1097–1105. | ||
Park G, Gim J, Kim AR, et al. Multiphasic analysis of whole exome sequencing data identifies a novel mutation of ACTG1 in a nonsyndromic hearing loss family. BMC Genomics. 2013;14:191. | ||
Dong X, Han Y, Sun Z, Xu J. Actin gamma 1, a new skin cancer pathogenic gene, identified by the biological feature-based classification. J Cell Biochem. 2018;119(2):1406–1419. | ||
Tsai LH, Harlow E, Meyerson M. Isolation of the human CDK2 gene that encodes the cyclin A- and adenovirus E1A-associated p33 kinase. Nature. 1991;353(6340):174–177. | ||
Kimura K, Hirano M, Kobayashi R, Hirano T. Phosphorylation and activation of 13S condensin by cdc2 in vitro. Science. 1998;282(5388):487–490. | ||
Massagué J, Joan M. G1 cell-cycle control and cancer. Nature. 2004;432(7015):298–306. | ||
Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007;35(4):495–516. | ||
Gu W, Li C, Yin W, et al. Shen-fu injection reduces postresuscitation myocardial dysfunction in a porcine model of cardiac arrest by modulating apoptosis. Shock. 2012;38(3):301–306. | ||
Lazebnik YA, Kaufmann SH, Desnoyers S, Poirier GG, Earnshaw WC. Cleavage of poly (ADP-ribose) polymerase by a proteinase with properties like ICE. Nature. 1994;371(6495):346–347. | ||
Jones RG, Thompson CB. Tumor suppressors and cell metabolism: a recipe for cancer growth. Genes Dev. 2009;23(5):537–548. | ||
Szklarczyk D, Franceschini A, Wyder S, et al. STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res. 2015;43(Database issue):D447–D452. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.