Back to Journals » Clinical, Cosmetic and Investigational Dentistry » Volume 16
Revascularization of a Permanent Tooth with Necrotic Pulp and Apical Periodontitis
Authors Saad W ![]() , Almaslamani M
, Almaslamani M ![]() , Saleh AR
, Saleh AR ![]()
Received 21 February 2024
Accepted for publication 14 June 2024
Published 18 June 2024 Volume 2024:16 Pages 227—235
DOI https://doi.org/10.2147/CCIDE.S464214
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Christopher E. Okunseri
Wala Saad,1 Manal Almaslamani,2 Abdul Rahman Saleh2
1Department of Clinical Sciences, College of Dentistry, Ajman University, Ajman, United Arab Emirates; 2Department of Clinical Sciences, Center of Medical and Bio-Allied Health Sciences Research, College of Dentistry, Ajman University, Ajman, United Arab Emirates
Correspondence: Manal Almaslamani, Department of Clinical Sciences, Center of Medical and Bio-Allied Health Sciences Research, College of Dentistry, Ajman University, Ajman, United Arab Emirates, Email [email protected]
Abstract: This case report details a pulp revascularization treatment administered to a mature permanent tooth exhibiting pulp necrosis. A 22-years old female patient complained of the recurrence of a sinus tract labial of the maxillary right central incisor; which was tender on biting. Diagnosis of pulp necrosis and symptomatic apical periodontitis. Preoperative periapical and CBCT radiographs showed root with wide apical foramen and large apical radiolucency. Pulp revascularization procedure was performed using 1.3% sodium hypochlorite irrigation, 17% Ethylenediaminetetraacetic acid irrigation, and calcium hydroxide intracanal dressing for 2 weeks. During the last visit, intentional bleeding was induced, collagen matrix was set over the blood clot, 2 mm of mineral trioxide aggregate and glass-ionomer filling was placed. A year of follow-up, the tooth showed no signs or symptoms and responded normally to the sensibility tests. Intra-oral periapical radiograph and the CBCT showed significant reduction in the periapical lesion’s size, slight reduction in the apical foramen’s size, and hard radiopaque material deposition at the root’s middle third.
Keywords: pulp revascularization, pulp regeneration, open apex, necrotic pulp, apical periodontitis
Introduction
Regenerative Endodontic Procedures (REP) therapy utilizes stem cells, scaffolds, and growth factors within a sterile environment to facilitate the regeneration of dental tissues. Stem cells possess the unique ability to differentiate into various cell types, scaffolds provide structural support for tissue growth, and growth factors stimulate cellular activities, collectively fostering tissue regeneration during this innovative dental therapy. The aim of (REP) is to resolve any potential apical periodontitis, replacing any damaged dentin and root tissue as well as pulp-dentin complex cells.1 Consequently, post-treatment, it could yield a viable tooth capable of achieving root maturation, a crucial aspect for restoring functionality and enhancing the overall prognosis of the tooth.
REP has been described in the literature with the aim of the complete regeneration of the pulp tissue and complete root formation. Many studies reported a very high success rate of these procedures,2–4 although the exact tissue formed inside the root is yet unknown.5
In the systematic review and meta-analysis which assessed clinical, radiographic and functional retention outcomes in immature necrotic permanent teeth treated either with pulp revascularization or apexification after a minimum of three months to determine which one provides the best result, concluded that the results do not favor one treatment modality over the other.2
In more recent systematic review and meta-analysis of clinical randomized studies (a total of 3 studies were included) concluded that REPs appear as a viable treatment alternative for mature necrotic teeth with periapical lesions.4
Pulp revascularization procedure was globally recognized by Trope in 2008, who first described and used the technique on a lower right second premolar with an open apex, a fistula, and radiographic evidence of apical periodontitis. A blood clot was created at the cementum level after irrigation with 5.25% sodium hypochlorite to provide structural support for the growth of new tissue. The cervical region was then double sealed with mineral trioxide aggregate (MTA), and the restoration was completed with composite resin.3
Teeth’s apexification processes involved either the use of calcium hydroxide to create an artificial apical barrier (apical plug) or MTA to induce the formation of an apical calcified barrier (conventional apexification). With regard to the development of the apical barrier of an immature tooth, both techniques have proven to be effective. The stimulation of apical development and root maturation of developing teeth, however, is also made possible by pulp revascularization (root growth, thickening of dentinal walls, and natural apexogenesis).6,7
Trauma or infections strong enough to stop mineral deposition by obstructing blood flow may be the cause of the incomplete root development, which prevents the root from completing its formation. Several endodontic and pediatric dentistry associations advise using REP when pulp necrosis in developing teeth results from caries or developmental abnormalities, as well as dental trauma.2,4,6–8
A permanent central incisor with a history of dental trauma, apical root resorption with an open apex, and a significant periapical lesion was revascularized in this case report.
Case Report
The Preferred Reporting Items for Case Reports in Endodontics (PRICE) 2020 guidelines were followed when writing this case report. Institutional approval was obtained for publishing the case details. The patient provided consent by signing an informed consent form for the treatment and agreed to the publication of the case report.
A 22-year-old female patient with an ASA I medical status was referred to the Postgraduate Endodontic Clinic at the Department of Endodontics at Ajman University. The main complaint was that she had a sinus tract at the apex of her right central incisor tooth, which had been present on and off for the previous year and was slightly tender when she bit down on it.
The patient’s dental history revealed dental trauma 12 years ago on both upper right and left central incisors (#11, #22) resulted in uncomplicated crown fracture (enamel-dentine fracture) for both teeth.
The mucosal sinus tract was closed intra-orally on the day of the evaluation, making it impossible to trace the sinus. Pulp sensibility tests, including the cold test (Endo Frost, Roeko, Langenau, Germany) and electric pulp test (EPT) (Digitest II Parkwell – Edgewell, NY), were ineffective on tooth #11. It was completely immobile (grade 0 mobility) and sensitive to percussion.
The tooth #11 had an enamel-dentine fracture, a large root canal with thin dentinal walls, an open apical foramen, and a large periapical radiolucency were located adjacent to the nasal floor. The external walls of the root’s apical third were irregular, which was a clear sign of inflammatory apical root resorption (Figure 1A).9
Cone Beam Computed Tomography (CBCT) was indicated to measure the size of the apical foramen, the size and extent of the apical lesion, the presence and extent of the inflammatory apical root resorption, and to rule out any other root/crown fractures.10
The preoperative CBCT FOV (5 cm × 5 cm) was taken using an X-ray machine with (CS 8100 CS), the teeth were scanned with CS Imaging Software version 8.0.15.987 by Carestream Dental Technology Topco Limited. All images were saved automatically to the patient records on the dental operator CareStream-Film089.
CBCT showed in the sagittal view; tooth #11 with open apex~2mm, well-demarcated large apical radiolucency, perforating labial cortical plate that is very close to the nasal floor. The axial view showed the radiolucency with 8.9*9.5 mm, perforating the labial cortical plate (Figure 1B and C).
For tooth number 11, a diagnosis of necrotic pulp with symptomatic apical periodontitis was made.11 The treatment options were discussed before starting the pulp revascularization procedure. The patient signed the consent form after being informed of the procedure and its potential results.
First Treatment Visit: 11/10/2022
Without using a vasoconstrictor, local anesthesia was administered using 3% mepivacaine infiltration. A medium round diamond bur was used to prepare the access cavity, and no bleeding of pulp tissue was noticed. Using an electronic apex locator (Root ZX, J Morita, Tokyo, Japan) and a periapical radiograph with H-file size 60, the working length was ascertained (Figure 2).
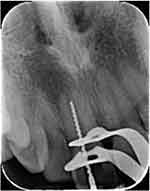 |
Figure 2 Periapical working length radiograph. |
A vented safe end needle (FANTA Dental Side Vented Needle, China) was used to apply 1.3% sodium hypochlorite for 5 minutes, 2 mm above the apical tissue, under the close monitoring through a dental operating microscope, irrigation with aseptic physiological saline (5 mL) was carried out. Finally, Irrigation with 20 mL of 17% EDTA done. As an intra-canal medication, non-setting calcium hydroxide (AH Temp, Dentsply) was applied. Teflon, hydraulic temporary restorative material (META BIOMED CO. LTD, Korea), and Glass Ionomer Cement were used to temporarily fill the tooth.
Second Treatment Visit: 25/10/2022
After 2 weeks, the patient was evaluated and, on clinical examination, the signs indicated incomplete healing of the sinus tract. The steps of pulpal canal disinfection were repeated as in the first visit.
Third Treatment Visit: 8/11/2022
After two weeks, recall revealed that all signs and symptoms had vanished completely, and the sinus tract had healed. On that day, the temporary filling was removed, irrigation with 17% EDTA was performed (20 mL, for 5 min), a side-vented needle was used and positioned 2 mm shorter than the vital apical tissue, and irrigation with sterile physiological saline was conducted (5 mL). Paper points were used to dry out the canal, and mechanical irritation of the periapical tissue caused by rotational movement of an apically pre-bent size 40 Hedstrom file caused bleeding to occur, which filled the canal with blood until 2 mm below the gingival margin.
A collagen matrix (Roeko, Gelatamp, Blister Pack, Swiss) was placed on top of the blood clot after it had formed at the coronal part of the root canal, allowing the matrix to soak up liquid to prevent the formation of a hollow space. A thin homogenous layer of about 2 mm of hydraulic silicate cement MTA (MTA- Angelus, Brazil) was applied on top of the collagen matrix beneath the cemento-enamel junction (Figure 3). Refreshment of the coronal dentine was done with round diamond bur, then a flowable light-cured dentin-bonding agent (Tetric N Flow Bulk Fill Syringes, Ivoclar Vivadent, Liechtenstein) was placed in the pulp chamber’s coronal surface to seal off the crown’s dentinal tubules followed by packable composite (3M, ESPE, Filtek z250 xt, USA) and periapical radiographic was taken.
 |
Figure 3 (A)Pre-operative intraoral photograph of the with evidence of sinus tract. (B) Post -operative after one year follow up demonstrating healing of sinus tract. |
Follow Up and Results
After three months (10/2/2023), the tooth was asymptomatic, had a normal gingival appearance, and was not sensitive to percussion. EPT revealed a positive response, but the cold test revealed no response from the tooth.
The second follow-up appointment was scheduled one year later on September 15, 2023, and the tooth was asymptomatic and responded favorably to pulp sensibility testing (cold test and EPT) at that time (Figure 4).
The size of the periapical radiolucency on the CBCT axial view (5.1*3.7 mm) was significantly reduced, the periapical foramen was significantly narrowed (1.3 mm), and hard tissue deposition (barriers) was deposited in the middle third of the root. Smaller hard tissue deposition was observed 1.7 mm from the coronal MTA, with one barrier being 4.8 mm from the MTA.
Discussion
In this presented case report, we opted for a revascularization procedure after thorough discussions with the patient, considering alternative approaches such as conventional apexification, MTA plug apexification, and pulp revascularization. Following a detailed explanation of the benefits and drawbacks associated with each option, such as the extended treatment duration for conventional apexification and the risk of crown fracture, we weighed against the potential benefits of regaining pulp sensibility and reinforcing root strength through continuous root maturation. The patient, expressing a strong desire to retain the tooth, demonstrated considerable enthusiasm for exploring this relatively new treatment modality. The pulp revascularization procedure adhered to the guidelines outlined in the position statement of the European Society of Endodontology (Table 1) for standardized and evidence-based clinical practice.6
 |
Table 1 Clinical Steps of Pulp Revascularization Procedure |
The patient was presented with a dental trauma accident, an uncomplicated crown fracture, which occurred 12 years prior and had not received any treatment. Consequently, this prolonged exposure led to the necrosis of the pulp due to exposed dentinal tubules, resulting in a wide-open apex with thin root dentin. In such cases, conventional root canal therapy may not provide optimal outcomes.7 Therefore, revascularization procedure, which can facilitate the regeneration of the pulp and strengthen the thin root dentinal walls, is considered the preferred approach for incompletely developed teeth.7
In this particular case, the observed outcome can be deemed successful based on comprehensive clinical and radiographic assessments. At the 1-year follow-up, the tooth exhibited sustained asymptomatic status, demonstrated positive responses to pulp sensibility tests, and showed evident improvements, including a notable reduction in the size of the periapical lesion and a decrease in apex width, as discernible in both periapical and CBCT radiographs. It is worth noting that an extended follow-up period, 2qa ideally spanning 12–24 months, would provide valuable insights, particularly concerning potential further increases in root length.12 However, it is essential to acknowledge that the relatively short follow-up duration represents a limitation in fully capturing the long-term dynamics of the case.
The underlying principle of revascularization lies in the capacity of apical pulp tissue to persist in unfavourable necrotic conditions while flourishing in favourable ones.13 To achieve successful pulp revascularization therapy, three essential factors must be addressed: root canal disinfection, the presence of an appropriate scaffold, and the attainment of a hermetic coronal seal.14 These factors are critical for the generation of functional tissue.15 Although blood clots have proven effective as scaffolds, they are not rich in growth factors.16,17 In this case, the induction of bleeding from the periapical tissues into the root canal is considered as a potential source of stem cells, a practice supported by other case reports.18,19
However, it is worth noting that bleeding can be unpredictable, and there may be minimal to no bleeding upon laceration of the periapical tissues. Nevertheless, the blood clot serves as a scaffold for subsequent tissue ingrowth.20–22 In comparison to other scaffolds such as platelet-rich fibrin (PRF) and platelet-rich plasma (PRP), whole blood clots exhibit superior performance in terms of root elongation, thickening, and responsiveness to vitality testing, despite PRP showing improved periapical healing.9
Given the initially thin root dentine, no instrumentation technique was deemed necessary in the pulp revascularization procedure to avoid compromising the fragile root canal walls while preserving essential stem cells.3 For root canal disinfection, we employed sodium hypochlorite (1.3% the concentration used in dental clinics and is considered safe to the tissue), the preferred endodontic irrigation solution, due to its ability to act as a solvent for necrotic tissue and its well-established antiseptic properties.23 EDTA was subsequently used as the final irrigation solution, following the recommendations of the American Association of Endodontists (AAE).12 EDTA’s chelating properties facilitate the release of growth factors trapped in the dentin, promoting stem cell proliferation.6
However, a recent study,24 conducted using an immature rat molar model, suggested that EDTA irrigation may have a detrimental effect on the regenerative process. This underscores the importance of careful consideration when employing EDTA in regenerative endodontics and highlights the need for further research to determine the optimal irrigation solution. Additionally, rinsing with saline after irrigation is advised to reduce the risk of potential precipitates and to eliminate any remaining debris and irrigation solution remnants,25 a practice that was followed in this particular case.
Numerous intracanal medicaments have been proposed in the literature for use in revascularization cases, including triple antibiotics (1:1:1 ciprofloxacin: metronidazole: minocycline), double antibiotics (without minocycline), and non-setting calcium hydroxide. In the present case, calcium hydroxide was chosen to mitigate tooth discoloration associated with triple antibiotics and due to its immediate availability in the clinic at the time of patient presentation. Moreover, several clinical studies have demonstrated favourable outcomes with the use of calcium hydroxide as a medicament in similar cases.6,8
In terms of achieving a hermetic coronal seal, hydraulic calcium silicate cements are recommended for covering the blood clot due to their ability to set in the presence of moisture.26 Careful placement of Hydraulic Silicate Cement [MTA] slightly below the Cementoenamel junction is essential to avoid undesirable outcomes such as discoloration, thereby maintaining dental health and enhancing defences against potential future infections.6
Regarding the specific type of tissue that exhibited a positive response to pulp sensibility tests, animal studies and clinical cases have indicated that newly formed tissues in revascularization are ectopic and may include soft connective tissues, cementum, or bone.27,28 In this case, positive responses to the pulp sensibility tests were observed, a reduction in the size of the periapical radiolucency, narrowing of the apical foramen, and the presence of radiopaque material deposition/barrier in the mid-root area. The precise nature of the tissue growth inside the root canal during the revascularization procedure remains a subject of ongoing debate.
A case series reported that the outcome of continued root development was not as predictable as increased thickening of the canal walls in human immature permanent teeth with infected necrotic pulp tissue and apical periodontitis/abscess after revascularization procedures.29 Another case series demonstrated a favourable outcome of the revascularization procedure in immature necrotic molars by using calcium hydroxide medication. All teeth treated (n = 6) showed radiographic evidence of complete periapical healing, progressive thickening of dentinal walls, and continued apical development in the absence of clinical symptoms.30
In a recent systematic review and meta-analysis of randomized controlled trials, it was revealed that the overall success rates for necrotic immature and mature permanent teeth were 95.6% and 95.5%, respectively. Furthermore, the asymptomatic rates for necrotic immature and mature permanent teeth were found to be 96.2% and 97.0%, respectively.31
For diagnostic purposes, treatment planning, and follow-up assessments to evaluate healing, CBCT was employed in this study, as recommended by the American Association of Endodontics.32 CBCT offers a precise examination of root canal anatomy, reducing distortion and superimposition while providing geometrically accurate images. This feature proves invaluable for diagnosing various types of resorption and root/tooth fractures following trauma and is recommended for use in the diagnosis, treatment planning, and follow-up of such cases.
The patient is very happy with the final result of the treatment.
Conclusion
The revascularization procedure described in this case report demonstrated successful indications of healing in the periapical region and a reduction in the size of the apical foramen during the follow-up period of one year. Furthermore, this technique should be considered as a well-established endodontic approach for the revitalization of a permanent tooth presenting with a necrotic pulp, apical periodontitis, and an open apex. Nevertheless, the validation of these findings could benefit from well-designed randomized controlled trials (RCTs).
Funding
There is no funding to report.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Diogenes A, Ruparel NB. Regenerative Endodontic Procedures: clinical Outcomes. Dent Clin North Am. 2017;61(1):111–125. doi:10.1016/j.cden.2016.08.004
2. Pereira AC, Oliveira ML, Cerqueira-Neto ACCL, et al. DE-Jesus-Soares A. Treatment outcomes of pulp revascularization in traumatized immature teeth using calcium hydroxide and 2% chlorhexidine gel as intracanal medication. J Appl Oral Sci. 2020;28:e20200217. doi:10.1590/1678-7757-2020-0217
3. Trope M. Regenerative potential of dental pulp. J Endod. 2008;34(7):S13–7. doi:10.1016/j.joen.2008.04.001
4. Glynis A, Foschi F, Kefalou I, Koletsi D, Tzanetakis GN. Regenerative endodontic procedures for the treatment of necrotic mature teeth with apical periodontitis: a systematic review and meta-analysis of randomized controlled trials. J Endod. 2021;47(6):873–882. doi:10.1016/j.joen.2021.03.015 PMID: 33811981.
5. Minic S, Vital S, Chaussain C, Boukpessi T, Mangione F. Tissue characteristics in endodontic regeneration: a systematic review. Int J Mol Sci. 2022;23(18):10534. doi:10.3390/ijms231810534
6. Galler KM, Krastl G, Simon S, et al. European Society of Endodontology position statement: revitalization procedures. Int Endod J. 2016;49(8):717–723. doi:10.1111/iej.12629
7. Harlamb SC. Management of incompletely developed teeth requiring root canal treatment. Aust Dent J. 2016;61(1):95–106. doi:10.1111/adj.12401
8. Murray PE. Review of guidance for the selection of regenerative endodontics, apexogenesis, apexification, pulpotomy, and other endodontic treatments for immature permanent teeth. Int Endod J. 2023;56(2):188–199. doi:10.1111/iej.13809
9. Abbott PV, Lin S. Tooth resorption-Part 2: a clinical classification. Dent Traumatol. 2022;38(4):267–285. doi:10.1111/edt.12762
10. Patel S, Brown J, Semper M, Abella F, Mannocci F. European Society of Endodontology position statement: use of cone beam computed tomography in Endodontics: European Society of Endodontology (ESE) developed by. Int Endod J. 2019;52(12):1675–1678. doi:10.1111/iej.13187
11. American Association of Endodontists. AAE Consensus Conference Recommended Diagnostic Terminology. J Endod. 2009;35(12):1634–1657. doi:10.1016/j.joen.2009.09.035
12. Wei X, Yang M, Yue L, et al. Expert consensus on regenerative endodontic procedures. Int J Oral Sci. 2022;14(1):55. doi:10.1038/s41368-022-00206-z
13. Shivashankar VY, Johns DA, Vidyanath S, Kumar MR. Platelet Rich Fibrin in the revitalization of tooth with necrotic pulp and open apex. J Conserv Dent. 2012;15(4):395–398. doi:10.4103/0972-0707.101926
14. Vijayaraghavan R, Mathian VM, Sundaram AM, Karunakaran R, Vinodh S. Triple antibiotic paste in root canal therapy. J Pharm Bioallied Sci. 2012;4(2):S230–233. doi:10.4103/0975-7406.100214
15. Galler KM, Eidt A, Schmalz G. Cell-free approaches for dental pulp tissue engineering. J Endod. 2014;40(4):S41–S45. doi:10.1016/j.joen.2014.01.014
16. Petrino JA, Boda KK, Shambarger S, Bowles WR, McClanahan SB. Challenges in regenerative endodontics: a case series. J Endod. 2010;36(3):536–541. doi:10.1016/j.joen.2009.10.006
17. Nosrat A, Seifi A, Asgary S. Regenerative endodontic treatment (revascularization) for necrotic immature permanent molars: a review and report of two cases with a new biomaterial. J Endod. 2011;37(4):562–567. doi:10.1016/j.joen.2011.01.011
18. Lin LM, Huang GT, Sigurdsson A, Kahler B. Clinical cell-based versus cell-free regenerative endodontics: clarification of concept and term. Int Endod J. 2021;54(6):887–901. doi:10.1111/iej.13471
19. Lovelace TW, Henry MA, Hargreaves KM, Diogenes A. Evaluation of the delivery of mesenchymal stem cells into the root canal space of necrotic immature teeth after clinical regenerative endodontic procedure. J Endod. 2011;37(2):133–138. doi:10.1016/j.joen.2010.10.009
20. Ritter AL, Ritter AV, Murrah V, Sigurdsson A, Trope M. Pulp revascularization of replanted immature dog teeth after treatment with minocycline and doxycycline assessed by laser Doppler flowmetry, radiography, and histology. Dent Traumatol. 2004;20(2):75–84. doi:10.1111/j.1600-4469.2004.00225.x
21. Ding RY, Cheung GS, Chen J, Yin XZ, Wang QQ, Zhang CF. Pulp revascularization of immature teeth with apical periodontitis: a clinical study. J Endod. 2009;35(5):745–749. doi:10.1016/j.joen.2009.02.009
22. Shivashankar VY, Johns DA, Maroli RK, et al. Comparison of the Effect of PRP, PRF and Induced Bleeding in the Revascularization of Teeth with Necrotic Pulp and Open Apex: a Triple Blind Randomized Clinical Trial. J Clin Diagn Res. 2017;11(6):ZC34–ZC39. doi:10.7860/JCDR/2017/22352.10056
23. Martin DE, De Almeida JF, Henry MA, et al. Concentration-dependent effect of sodium hypochlorite on stem cells of apical papilla survival and differentiation. J Endod. 2014;40(1):51. doi:10.1016/j.joen.2013.07.026
24. Reis-Prado AH D, Oliveira SC, Goto J, et al. Influence of ethylenediaminetetraacetic acid irrigation on the regenerative endodontic procedure in an immature rat molar model. Int Endod J. 2023;56(1):69–79. doi:10.1111/iej.13846
25. Trevino EG, Patwardhan AN, Henry MA, et al. Effect of irrigants on the survival of human stem cells of the apical papilla in a platelet-rich plasma scaffold in human root tips. J Endod. 2011;37(8):1109–1115. doi:10.1016/j.joen.2011.05.013
26. Lin LM, Ricucci D, Huang GT. Regeneration of the dentine-pulp complex with revitalization/revascularization therapy: challenges and hopes. Int Endod J. 2014;47(8):713–724. doi:10.1111/iej.12210
27. Wang X, Thibodeau B, Trope M, Lin LM, Huang GT. Histologic characterization of regenerated tissues in canal space after the revitalization/revascularization procedure of immature dog teeth with apical periodontitis. J Endod. 2010;36(1):56–63. doi:10.1016/j.joen.2009.09.039
28. Torabinejad M, Turman M. Revitalization of tooth with necrotic pulp and open apex by using platelet-rich plasma: a case report. J Endod. 2011;37(2):265–268. doi:10.1016/j.joen.2010.11.004
29. Cehreli Z, Sbitiren B, Sara S, Erbas G. Regenerative endodontic treatment (revascularization) of immature necrotic molars medicated with calcium hydroxide: a case series. J Endod. 2011;37(9):51–55. doi:10.1016/j.joen.2013.07.026
30. Chen M, Chen K-L, Chen C, Tayebaty F, Rosenberg P, Lin L. Responses of immature permanent teeth with infected necrotic pulp tissue and apical periodontitis/abscess to revascularization procedures. Int Endod J. 2012;45(3):294–305. doi:10.1111/j.1365-2591.2011.01978.x
31. Li J, Zheng L, Daraqel B, Liu J, Hu Y. Treatment outcome of regenerative endodontic procedures for necrotic immature and mature permanent teeth: a systematic review and meta-analysis based on randomised controlled trials. Oral Health Prev Dent. 2023;21(1):141–152. doi:10.3290/j.ohpd.b4100877 PMID: 37195330.
32. American Association of Endodontists. Clinical considerations for a regenerative procedure. 2021; 07–30.
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.