Back to Journals » Clinical Ophthalmology » Volume 15
Retrospective Study of Ellipsoid Zone Integrity Following Treatment with Intravitreal Ocriplasmin (OZONE Study)
Authors Drenser KA, Pieramici DJ, Gunn JM, Rosberger DF, Kozma P, Fineman MS, Duchateau L, Khanani AM
Received 3 November 2020
Accepted for publication 28 June 2021
Published 16 July 2021 Volume 2021:15 Pages 3109—3120
DOI https://doi.org/10.2147/OPTH.S285464
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Kimberly A Drenser,1 Dante J Pieramici,2,3 Joseph M Gunn,4 Daniel F Rosberger,5 Petra Kozma,6 Mitchell S Fineman,7,8 Luc Duchateau,9 Arshad M Khanani10
1Associated Retinal Consultants P.C., Royal Oak, MI, USA; 2California Retina Consultants, Santa Barbara, CA, USA; 3California Retina Research Foundation, Santa Barbara, CA, USA; 4Southeastern Retina Associates, Kingsport, TN, USA; 5Weill-Cornell Medical College; MaculaCare, PLLC, New York, NY, USA; 6Oxurion NV (formerly ThromboGenics NV), Leuven, Belgium; 7Mid Atlantic Retina, Philadelphia, PA, USA; 8Wills Eye Hospital, Philadelphia, PA, USA; 9Ghent University, Ghent, Belgium; 10Sierra Eye Associates, Reno, NV, USA
Correspondence: Kimberly A Drenser
Associated Retinal Consultants P.C., 3555 W 13 Mile Road# LL-20, Royal Oak, MI, 48073, USA
Tel +1 248 288 2280
Fax +1 248 288 5644
Email [email protected]
Purpose: To assess generalized (GD) and focal ellipsoid zone disruption (FD) in patients with symptomatic vitreomacular adhesion (sVMA) using spectral domain optical coherence tomography (SD-OCT) following ocriplasmin.
Patients and methods: OZONE was a Phase 4, retrospective study of patients with sVMA treated with a single intravitreal injection of ocriplasmin (0.125 mg). Data from adult patients with at least 6-month follow-up after ocriplasmin were included. SD-OCT was performed at baseline (within 30 days before ocriplasmin), before Day 21 post-injection (early observation, EO), and by last observation (LO) which was maximally 6 months post-injection. The main outcome measure was the development of new and the evolution of existing FD/GD at EO and LO.
Results: The study enrolled 134 eyes/patients from 22 sites in the USA. At baseline, 87 eyes (64.9%) had FD, 21 eyes (15.7%) had GD and 26 eyes (19.4%) had no FD/GD. Among the eyes without FD/GD at baseline, 13 (50%) and 8 (30.8%) developed FD or GD, respectively, by EO. By LO, FD/GD improvement or resolution was seen in > 80% of these eyes. Among the eyes with FD/GD at baseline, < 40% had improving/resolving EZ integrity at LO. The absence of FD/GD at baseline was associated with less persistent FD/GD at LO (P< 0.0005). The presence of FD with MH at baseline was associated with persistent FD at LO (P=0.027).
Conclusion: The fact that a large majority of eyes had FD/GD prior to ocriplasmin was unexpected and demonstrates that EZ disruptions are common in sVMA. This suggests that loss of EZ integrity may be part of the natural history of this disorder. It is hypothesized that the status of the EZ at baseline is a contributing, ocriplasmin independent modulator of subsequent EZ changes after ocriplasmin. Prospective analyses which include a sham control group would be required to test this hypothesis.
Keywords: symptomatic vitreomacular adhesion, vitreomacular traction, spectral domain optical coherence tomography, macular hole
Introduction
Spectral domain optical coherence tomography (SD-OCT) has allowed high-resolution assessment of anatomic retinal changes, including changes in the ellipsoid zone (EZ), which was previously referred to as the photoreceptor inner segment/outer segment (IS/OS) junction.1,2 Disruption or absence of EZ integrity is indicative of damage to photoreceptors.3 Studies assessing changes in the EZ have demonstrated that this may be a predictor of poor visual acuity (VA) and increased severity in a variety of retinal diseases.4–7 Disruption of the EZ may be localized to the area corresponding to an anatomic lesion (“focal disruption,” FD), or may be more generalized (“generalized disruption,” GD).
In 2012, ocriplasmin became the first commercially available pharmacologic option for the treatment of symptomatic vitreomacular adhesion (sVMA).4,8 Studies investigating the use of ocriplasmin in sVMA have frequently identified changes in the EZ following treatment, with most cases resolving in the weeks to months after treatment.9–12
The Phase 4 Ocriplasmin Ellipsoid Zone Retrospective Data Collection (OZONE) study (NCT02193945) was designed to characterize anatomic changes in the retina, including the EZ, by SD-OCT, over 6 months after a single intravitreal injection of ocriplasmin in patients with sVMA.
Patients and Methods
Inclusion/Exclusion Criteria
Investigators reviewed the medical records of patients to determine eligibility using the following criteria: aged 18 years or older with a diagnosis of sVMA and at least 6 months of follow-up following ocriplasmin treatment. Patients with epiretinal membrane (ERM) were not excluded. All patients had to have a pretreatment (baseline) visit within 30 days prior to receiving a single intravitreal injection of ocriplasmin (0.125 mg; ThromboGenics, Inc., Iselin, NJ) in the study eye. The frequency and timing of patient visits post-injection was at the discretion of the treating physician. Imaging with Spectralis® SD-OCT (Heidelberg Engineering, Franklin, MA) was required to take place at least once within 30 days before and at least twice after ocriplasmin injection. The first required post-treatment scan must have occurred from Days 1 to 21 after injection. The second required post-treatment scan must have occurred within 6 months after injection.
Patients were excluded from the study if they had been treated with ocriplasmin for medical conditions outside the product label or if they had other retinal conditions that could affect the vitreoretinal interface or lead to retinal atrophy.
Ellipsoid Zone (EZ) Assessments
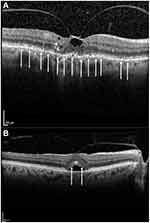
A central reading center (CRC; Digital Angiography Reading Center, New York, NY) assessed baseline and follow-up SD-OCT images obtained for each patient as part of the study. Although sVMA and vitreomacular traction (VMT) were not differentiated in the protocol, the CRC noted the presence or absence of VMT at baseline and at later time points. The CRC used a specific, standardized protocol for evaluating SD-OCT images for anatomic changes in the retina, including GD and FD of the EZ, the development of subretinal fluid (SRF), resolution of VMA, the development of macular hole (MH) including full-thickness macular hole (FTMH), and MH changes (worsening or resolution) in those patients who had MH at baseline. GD was defined as loss of continuity of the EZ line on SD-OCT outside the area of an identified anatomic lesion. FD was defined as loss of continuity of the EZ line on SD-OCT limited to the area of a corresponding anatomic lesion, such as FTMH, VMA, or sub- or intraretinal fluid. Example SD-OCT images are shown in Figure 1 and were collected at baseline from patients in OZONE. The top panel presents an image in which GD (no MH) was detected. In the bottom panel, an image in which FD (no GD or MH) was detected is presented. Arrows depict areas of EZ disruption.
Study Endpoints
The originally planned primary endpoint of the study was the proportion of eyes with new-onset GD or FD by Day 21 after injection. However, the majority of eyes already had GD or FD at baseline (before ocriplasmin treatment). Therefore, at baseline and at all other times, eyes were categorized as having no disruption, having FD, or having GD. For eyes with no EZ disruptions at baseline, both newly emergent FD and/or GD were followed across time. For eyes with FD and no GD at baseline, FD was followed across time. For eyes with GD at baseline (regardless of FD status), GD was followed across time. For each eye, a series of SD-OCT images was present, and each image was compared with the previous image. The status of pre-existing or newly emergent FD/GD was followed across time and was scored as resolved, improved, unchanged or worsened. Eyes in which FD/GD had worsened could subsequently be scored as worsened then resolved or worsened then improved. Data are reported as early (EO) and last observation (LO) as described below.
Since the timing and frequency of patient visits was at the discretion of the treating physician, there was variability among patients in the number of SD-OCT scans obtained. All scans were analyzed and compared with the scan immediately preceding it. For EO, the last scan obtained on or before Day 21 is reported. For eyes with more than one post-baseline scan prior to Day 21, it was possible to assess if newly emergent FD/GD was improved by EO as the last scan could be compared to an earlier scan. Usable data for EO were not available for some patients either because the first post-ocriplasmin scan was done after Day 21 or because the early scan(s) were uninterpretable for FD or GD. For LO, the last scan obtained before 6 months post-injection, regardless of timing, is reported. Observations post-vitrectomy were excluded from the primary analysis. In eyes that underwent vitrectomy, LO was defined as the last scan before vitrectomy (last observation carried forward).
Based on the originally planned primary endpoint, the development and evolution of GD and FD in the subgroup of eyes with neither FD nor GD (and thus without MH) at baseline was investigated. For the subgroups of eyes with either FD or GD at baseline, we investigated the evolution of FD/GD over time separately in eyes with and without MH.
Several secondary endpoints included best corrected visual acuity (BCVA) at baseline, EO and LO. Adverse drug reactions (ADRs) and the presence of ocular symptoms were also analyzed. Investigators identified ADRs that started on or after the day of ocriplasmin injection during their retrospective review. ADRs were coded using the Medical Dictionary for Regulatory Activities (MedDRA) version 18.0 and summarized by system organ class category and preferred term. In addition, the presence or absence of ocular symptoms on a solicited list of 19 terms was collected on the case report form. These terms do not necessarily correspond to MedDRA preferred terms.
Statistical Analyses
The analysis set used to perform all analyses consisted of all enrolled patients treated with ocriplasmin. Missing data were not imputed. Descriptive statistics were used to summarize continuous variables, including the number of observations (n), mean (±standard deviation [SD]), median (interquartile range), and minimum/maximum. For categorical variables, group frequencies and percentages were calculated. The Fisher's exact test was used to assess the possible relationship of MH at baseline on the persistence of FD/GD at LO. Persistent FD/GD was defined as FD/GD which was not improving or resolved at LO. This test was also used to assess the relationship of FD/GD at baseline on the persistence of FD/GD at LO. The change from baseline of BCVA at LO was compared between patients with and without disruption at baseline using a linear fixed effects model with normally distributed error and status at baseline as the independent variable. BCVA at EO and LO was compared using a linear mixed model with normally distributed error and included patient as a random effect and time (EO versus LO) as the independent variable. All computations were performed using SAS® 9.2 or higher (SAS Institute, Cary, NC) statistical software.
The study protocol was approved by Quorum Review (now Advarra) Institutional Review Board on 24 June 2014 and the study was conducted according to Good Clinical Practice and the Declaration of Helsinki. All patients provided written informed consent and the study was conducted in compliance with the trial protocol. The authors do not intend to share individual deidentified participant data or other data outside that which is presented in this manuscript.
Results
This retrospective study enrolled 134 patients/eyes from 22 retina centers across the United States (a list of sites is included at the end of this paper). Data were collected from August 22, 2014, to April 8, 2015. Baseline demographic and ocular characteristics are summarized in Table 1.
 |
Table 1 Demographics and Baseline Ocular Characteristics in the Study Eye |
At baseline, the majority of eyes had FD (± MH) without GD (87 eyes, 64.9%). Most eyes had VMT at baseline (122 eyes, 94.6%; 5 missing data). There was no clear relationship of VMT status with EZ status as VMT was absent at baseline in eyes with no FD/GD (2 eyes), FD with MH (2 eyes), GD with MH (2 eyes) and GD without MH (1 eye).
By Day 28, sVMA resolved without surgery in 52.3% (67/128) and the rate of nonsurgical resolution of FTMH was 32.4% (12/37). At LO, the nonsurgical resolution rates were 61.9% (83/134) for sVMA and 32.5% (13/40) for FTMH.
A total of 26.1% (35/134) of patients underwent a vitrectomy during the study and the median time to vitrectomy was 64 days (range Day 6-Day 147). In 12 eyes, vitrectomy was performed on or before Day 21. Reasons for vitrectomy included MH (33 patients), sVMA (6 patients), ERM (6 patients), retinal tear/detachment (5 patients) and/or a combination of these. Vitrectomy was performed in 61.8% (21/34) of eyes with FD and MH at baseline which constituted 60.0% (21/35) of all vitrectomies done. The rate of vitrectomy was no greater than 20% in any other EZ baseline group.
The mean (SD) number of SD-OCT scans prior to EO was 1.28 (0.75) and before LO was 3.51 (1.68). The mean (SD) time of LO was Day 114 (61) irrespective of vitrectomy and the range was Day 6 to Day 211. Excluding eyes which had a vitrectomy, the mean (SD) time of LO was Day 138 (50) and the range was Day 14 to Day 211. Of eyes without vitrectomy, 84 of 99 eyes (84.8%) had LO at Day 90 or later.
EZ Disruption
No Disruption at Baseline (Figure 2)
Of the 26 eyes with no disruption of the EZ at baseline, 23 eyes had data for FD status at EO and approximately half (N=12, 52.2%) of these eyes were unchanged from baseline with regard to FD status (no new FD). In 2 eyes (8.7%), FD had developed but was improving by EO. In an additional 9 eyes (39.1%), FD had developed but was not improving.
At LO, FD had never been detected (Unchanged) in 13 eyes (50.0%). In the 13 eyes in which FD had been detected at some time following ocriplasmin, FD had resolved in 5 eyes (19.2%) and improved in 6 eyes (23.1%). In 2 eyes (7.7%), FD had developed and was not improving by LO.
Of the 26 eyes with no disruption of the EZ at baseline, 24 eyes had data for GD at EO which showed that 16 eyes (66.7%) were unchanged from baseline (no new GD). In 1 eye (4.2%), GD had developed but was improving. In 7 eyes (29.2%), GD had developed but was not improving at EO.
At LO, GD had never been detected in 18 eyes (69.2%). In the 8 eyes in which GD was detected at some time following ocriplasmin, GD was resolved in 3 eyes (11.5%) and improved in 4 eyes (15.4%). In 1 eye (3.8%), GD had developed following ocriplasmin and was not improving at LO.
Of the 26 eyes with no disruption of the EZ at baseline, neither FD or GD developed in 11 eyes (42.3%) and both FD and GD developed in 6 eyes (23.1%). By LO, both FD and GD had either resolved (3 eyes) or improved (2 eyes) in all but 1 eye in which the single scan after detection of FD/GD showed no improvement and a vitrectomy was performed on Day 36.
FD at Baseline (Figure 3)
For eyes with FD at baseline (N=87), the results are described for eyes without (N=53) and with MH (N=34) separately.
Of the 53 eyes with FD without MH at baseline, EO data were available from 47 eyes. FD status did not change in almost half (N=23, 48.9%) of these eyes. FD was resolved in 2 eyes (4.3%), improved in 3 eyes (6.4%), worsened then improved in 4 eyes (8.5%), and worsened in 15 eyes (31.9%).
Of the 53 eyes with FD and without MH at baseline, LO data were available from 52 eyes. In 4 eyes (7.7%), FD had resolved and improved in 12 eyes (23.1%). FD had not changed in 14 eyes (26.9%), worsened then resolved in 7 eyes (13.5%), worsened then improved in 9 eyes (17.3%), and worsened in 6 eyes (11.5%).
Of the 34 eyes with FD and MH at baseline, EO data were available from 31 eyes. In more than half of these eyes (N=17, 54.8%) FD had worsened at EO. In 12 eyes (38.7%), FD had not changed. In 1 eye (3.2%), FD had improved and, in 1 eye (3.2%) FD had worsened then improved at EO.
At LO, FD had worsened (N=11, 32.4%) or had not changed (N=11, 32.4%) in the majority of eyes with FD and MH at baseline. However, improvement in FD status was seen in some eyes. FD was resolved in 1 eye (2.9%) and improved in 3 eyes (8.8%) (compared to baseline). In 8 eyes (23.5%), FD status had worsened at EO, but had improved by LO.
GD at Baseline (Figure 4)
For eyes with GD at baseline (N=21), results are described for eyes without (N=15) and with MH (N=6) separately. Of the 15 eyes without MH, 5 (33.3%) did not also have FD at baseline.
For the eyes without MH, GD status either did not change (N=14, 93.3%) or had worsened (N=1, 6.7%) at EO. At LO, the GD status of most eyes without MH (11/15, 73.3%) had not changed from baseline. However, 3 eyes (20.0%) had an improved GD status compared to baseline, and 1 eye (6.7%) had improved after initially worsening. The GD status of all 5 eyes with GD without FD at baseline remained unchanged at EO and LO.
For eyes with GD and MH, GD status remained the same except for 1 eye in which GD status was found to be worsened at LO but not at EO (compared to baseline).
Persistence of Disruption at LO
Of the 86 eyes with FD (± MH) at baseline, 42 eyes (48.8%) had persistent FD (unchanged without improvement/resolution or worsened without improvement/resolution) at LO. Of the 21 eyes with GD (± MH) at baseline, 17 eyes (81.0%) had persistent GD at LO. Of the 26 eyes without disruption at baseline, 2 eyes (7.7%) had persistent FD and 1 eye (3.8%) had persistent GD at LO. Fisher's exact tests indicated that the absence of FD/GD at baseline was associated with less persistent FD or GD at LO (P<0.0005).
Of the 52 eyes with FD without MH at baseline, 20 eyes (38.5%) had persistent FD at LO. Of the 34 eyes with FD and MH at baseline, 22 eyes (64.7%) had persistent FD at LO. Fisher's exact test demonstrated the persistence of FD to be higher in eyes with MH (P=0.027).
Of the 15 eyes with GD without MH at baseline, 11 eyes (73.3%) had persistent GD at LO. All (100%) of the 6 eyes with GD and MH at baseline had persistent GD at LO. The presence of MH was not associated with greater persistence of GD (P=0.281).
BCVA
As noted in Table 1, the mean (SD) BCVA was 57.4 (17.0) letters at baseline. At EO, mean BCVA was 57.9 (17.5) letters and at LO, had increased to 61.3 (19.9) letters for all eyes including those for which LO was followed by vitrectomy. These were not significantly different from baseline.
In eyes that did not undergo vitrectomy, the mean BCVA at LO was 68.1 (14.4) letters, which constituted a 7.95 (13.7) letter increase from baseline. In eyes without FD or GD at baseline which did not undergo vitrectomy, the mean BCVA at LO was 76.0 (7.2) letters, which constituted a 10.9 (13.3) letter increase from baseline. In eyes with disruption (FD and/or GD) at baseline which did not undergo vitrectomy, the mean BCVA at LO was 65.5 (15.3) letters which constituted a 7.1 (13.9) letter increase from baseline. There was a significant difference between eyes with and without disruption at baseline in the mean number of letters at LO (P=0.0017); however, the change from baseline was not significant between these two groups.
Adverse Drug Reactions and Ocular Symptoms
During the course of the study, 45 of 134 patients (33.6%) experienced at least one ADR in the study eye (Table 2). The most frequently reported adverse reactions, irrespective of vitrectomy, were vitreous floaters (12.7% of patients), reduced VA (10.4% of patients), SRF (9.0% of patients), and photopsia (flashing lights, flashing vision, 8.2% of patients). No cases of endophthalmitis or vitreoretinal dehiscence were reported.
 |
Table 3 Status of Ocular Symptoms Starting at or After Injection and at the End of Follow-Up for the Study Eye, Irrespective of Vitrectomy |
Table 3 lists the most frequently reported ocular symptoms, the reporting of which was solicited using a list of 19 terms which do not correspond to the MedDRA terms used for ADR collection. For symptoms starting at or after ocriplasmin treatment, the most common were vitreous floaters (31.3%), decreased VA (23.9%), photopsia (23.9%), and eye pain/ocular discomfort (21.6%). At the end of the follow-up period, ocular symptoms were resolved in 23 of 42 patients with floaters (54.8%) and in 14 of 32 patients with decreased VA (43.8%). By LO, photopsia resolved for 25 of 32 patients (78.1%), and eye pain/ocular discomfort resolved for 26 of 29 patients (89.7%).
Discussion
EZ disruptions have been reported in patients receiving ocriplasmin for the treatment of sVMA. Some hypothesize that pharmacologic alteration of the vitreoretinal interface may exacerbate traction and could potentially increase EZ disruption.10,11,13 The OZONE study was conducted to further characterize changes in the retina, including the EZ, over a maximum 6-month period following injection of ocriplasmin.
As noted, the originally planned primary endpoint of the study was the proportion of eyes with new-onset GD or FD by Day 21 after injection with ocriplasmin. The finding that more than 80% of eyes in the study had FD (64.9%) or GD (19.4%) at baseline was unexpected. At the time this study was designed, reports of EZ disruptions following treatment with ocriplasmin were numerous,9–13 but an association of EZ disruptions with untreated sVMA had not yet been described. The finding that the majority of eyes in this study had EZ disruptions before ocriplasmin treatment suggests that disruptions of the EZ are common in the natural history of sVMA. If EZ disruptions are common in sVMA in the absence of treatment with ocriplasmin, disease-related EZ disruptions cannot be ruled out as a contributing factor in the EZ disruptions seen following ocriplasmin. This does not imply that ocriplasmin cannot produce disruptions of the EZ, only that disease-related disruptions may modulate EZ disruptions resulting from the administration of ocriplasmin. In order to assess the role of natural history in the development and progress of EZ disruptions in sVMA, prospective studies that include sham control groups would be required, but may be ethically challenging to conduct.
Lavine et al examined quantitative changes in EZ-retinal pigment epithelium (RPE) volume in eyes with persistent VMT and in those patients who had VMT released following ocriplasmin treatment in the prospective ORBIT study.16 Both groups had decreased EZ-RPE volumes at week 1 which had recovered to near baseline levels by the final visit. Although volume losses were numerically greater at week 1 and recovery was greater by the final visit in patients with VMT release, there were no significant differences between the groups. Similar, but not significantly greater differences in total macular EZ attenuation were seen between the two groups at week 1 and at the final visit after ocriplasmin treatment. This study suggests that transient changes in the EZ occur after ocriplasmin and that the level of initial change and subsequent recovery are similar irrespective of whether VMT is released. While the findings of this study strongly suggest that ocriplasmin produces changes in the EZ, it did not address the role of disruptions in the EZ before ocriplasmin in the course of EZ disruptions after ocriplasmin.
The OZONE study took a qualitative approach and examined if disruptions of the EZ were present before ocriplasmin and how they changed over time. The finding that the presence of EZ disruptions prior to ocriplasmin treatment is predictive of persistent EZ disruptions at LO suggest that the status of the EZ at baseline is at least a contributing and an ocriplasmin independent factor to subsequent changes in the EZ after ocriplasmin. In addition, of the 26 eyes that did not have disruptions of the EZ at baseline, 8 eyes had neither a vitrectomy nor FD or GD detected at any time after ocriplasmin. Of these 8 eyes, ocriplasmin produced VMT release in 5 eyes (62.5%) suggesting that neither sVMA, nor ocriplasmin treatment, nor VMT release is always accompanied by disruptions of the EZ.
The data presented suggest that eyes with FD which also have an MH at baseline are more likely to have persistent (non-improved or resolved) FD at LO than eyes with FD and no MH. This same relationship was not found to be statistically significant for eyes with GD. However, this may be due to the small sample size of the GD population. While ~25% of eyes with GD without MH at baseline had either resolved or improved GD at LO, all eyes with GD and MH had persistent GD at LO, suggesting that MH may also be associated with a poorer prognosis of improvement of GD.
Given expectations when the study was designed, we could not select patients to power analyses sufficiently in each baseline EZ status subgroup. The small sample size of the GD groups suggests that global disruptions of the EZ are less common in sVMA than focal EZ disruptions. Although this makes intuitive sense, a larger study would be needed to confirm. The ramification in the current study is that conclusions regarding the GD at baseline groups are not as strongly supported due to small sample sizes.
While unknown, one can postulate on possible causes of widespread disruptions of the EZ in patients with sVMA with GD. VMT and sVMA are often associated with sub- or intra-retinal fluid including cystic changes.17 This may be interpreted to reflect a disturbance in the homeostasis of the metabolism in the retina, and in particular, fluids crossing the retina. Sub- and intra-retinal fluid leads to distress of the photoreceptors that is observed morphologically as EZ changes. Additionally, vitreoretinal traction outside the macular area has also been described, which could to some extent explain disruptions of the EZ outside the area of focal VMT.18
In previous studies with ocriplasmin, EZ changes were reported to be transient and often resolved between a few months and 1 year, depending on the length of the follow-up period.9,10,14–16 The data presented here for eyes without EZ disruptions at baseline are consistent with these earlier reports in that over three-quarters of these eyes had improvement/resolution at LO. It is salient to note that the mean time for LO in this study was less than 5 months after ocriplasmin treatment. Although inclusion into the study required both 6 months of follow-up and at least two SD-OCT scans following ocriplasmin, it did not dictate at what time post-ocriplasmin the last scan had to be obtained. As noted, the mean time at LO for all eyes was Day 114. For 14 eyes including 13 that underwent vitrectomy, LO was on or before Day 21. Most eyes that did not undergo vitrectomy had LO after Day 90; however, about 15% of eyes had LO earlier than this. The shorter duration of follow-up in this study may explain the higher rates of eyes with EZ disruptions at LO. Notwithstanding the fact that OZONE does not provide a clear indication of the status of the retina 6 months after treatment with ocriplasmin, the results of this study expand on earlier studies by suggesting that EZ status at baseline is a predictor whether disruptions of the EZ, principally FD, will be persistent.
In general, there was no clear relationship between EZ status and BCVA. Eyes without EZ disruptions at baseline tended to have better BCVA at LO; however, there was much variability among eyes in BCVA at LO and the timing at which LO measurements were obtained.
There were no new safety findings in this study and the types of ADRs reported were similar to those reported in previous clinical studies of ocriplasmin.8,19 All ADRs were ocular in nature, with vitreous floaters, reduced VA, SRF, and photopsia being the most commonly reported.
The ocular symptoms reported also revealed no new safety findings. The report of ocular symptoms differs from reports of ADRs in several ways. Ocular symptom terms do not necessarily correspond to MedDRA preferred terms. In this study, an ADR was recorded for reactions for which the treating physician felt there was a reasonable possibility of a causal relationship between the retrospective data and the medicinal product. Ocular symptoms were collected in a solicited fashion which may have had an impact on reporting rates as it might include capture of symptoms of sVMA present at the time of ocriplasmin administration.
The retrospective nature of the study design of OZONE is associated with several methodological limitations when comparing the results with those of prospective studies. Because the frequency and timing of patient visits were at the discretion of the treating physician, some research questions could not be answered as originally planned (eg, “time-to-event” analyses). The flexibility in the follow-up schedule and the requirement of multiple post-treatment SD-OCT scans may have resulted in the selection of patients who required more care after treatment compared with patients who responded well and did not return for extended follow-up. Also, a focus on visits in which SD-OCT scans were obtained likely prevented an accurate assessment of final BCVA outcomes following ocriplasmin treatment.
The results from this study provide new insights into the prevalence of EZ disruptions in sVMA and suggest that EZ disruptions are common in the natural history of sVMA. Further, EZ status at the time of ocriplasmin administration is likely a better predictor of what EZ status will be at LO than solely the fact that sVMA had been treated with ocriplasmin.
Acknowledgments
The authors wish to acknowledge the principal investigators and site staff who participated in the OZONE study as well as all the patients enrolled in the study. Medical writing assistance was provided by Christopher de Fiebre, PhD, from Oxurion, as well as Duprane Pedaci Young and Georgia Bakirtzi, PhD, from Fishawack Communications Inc., who were funded by ThromboGenics.
Participating Centers
Black Hills Regional Eye Institute, Rapid City, SD; California Retina Consultants, Santa Barbara, CA; Carle Foundation Hospital, Urbana, IL; Duke University Eye Center, Durham, NC; Eyesight Ophthalmic Services, Portsmouth, NH; Harvard Vanguard Medical Associates, Boston, MA; MaculaCare, New York, NY; Marietta Eye Clinic, Marietta, GA; Ophthalmic Consultants of Boston, Boston, MA; Orange County Retina Medical Group, Santa Ana, CA; Paducah Retinal Center, Paducah, KY; Retina Associates of Utah, P.C., Salt Lake City, UT; Retina Consultants of Houston, Houston, TX; Retina Specialists, Plano, TX; Retina Vitreous Associates of Florida, St. Petersburg, FL; Rush University Medical Center Department of Ophthalmology, Chicago, IL; Sierra Eye Associates, Reno, NV; Southeastern Retina Associates, P.C., Kingsport, TN; Valley Retina Institute, PA, McAllen, TX; West Virginia University Eye Institute, Morgantown, WV; William Beaumont Hospital, Royal Oak, MI; Wills Eye Institute, Philadelphia, PA
Disclosure
K. Drenser is a consultant for Allergan, Genentech, and Spark Therapeutics. She is on the DSMB for Spark Therapeutics. She is the owner of FocusROP LLC and Retinal Solutions LLC. D. Pieramici and M. Fineman previously served as consultants for ThromboGenics. L. Duchateau is a consultant for Oxurion. P. Kozma is an employee of Oxurion. A. Khanani is a consultant for Adverum, Alcon, Allegro, Allergan, EyePoint, Genentech, Inc., Kodiak, Novartis, Gemini, Graybug, Gyroscope, Opthea, Oxurion, PolyPhotonix, RecensMedical, Regenxbio. He receives research support from Adverum, Allergan, Gemini, Genentech, Inc., Gyroscope, Kodiak, Novartis, Opthea, Ophthotech, Oxurion, Regenxbio, Recens Medical. He is a speaker for Allergan and Novartis. J. Gunn and D. Rosberger report no conflicts of interest in this work.
References
1. Tao LW, Wu Z, Guymer RH, Luu CD. Ellipsoid zone on optical coherence tomography: a review. Clin Exp Ophthalmol. 2016;44(5):422–430. doi:10.1111/ceo.12685
2. Spaide RF, Curcio CA. Anatomical correlates to the bands seen in the outer retina by optical coherence tomography: literature review and model. Retina. 2011;31(8):1609–1619. doi:10.1097/IAE.0b013e3182247535
3. Zhu W, Chen H, Zhao H, et al. Automatic three-dimensional detection of photoreceptor ellipsoid zone disruption caused by trauma in the OCT. Sci Rep. 2016;6:25433. doi:10.1038/srep25433
4. García-Layana A, García-Arumí J, Ruiz-Moreno JM, Arias-Barquet L, Cabrera-López F, Figueroa MS. A review of current management of vitreomacular traction and macular hole. J Ophthalmol. 2015;2015:809640. doi:10.1155/2015/809640
5. Saxena S, Srivastav K, Cheung CM, Ng JYW, Lai TYY. Photoreceptor inner segment ellipsoid band integrity on spectral domain optical coherence tomography. Clin Ophthalmol. 2014;8:2507–2522. doi:10.2147/OPTH.S72132
6. Maheshwary AS, Oster SF, Yuson RMS, Cheng L, Mojana F, Freeman WR. The association between percent disruption of the photoreceptor inner segment-outer segment junction and visual acuity in diabetic macular edema. Am J Ophthalmol. 2010;150(1):63–67 e1. doi:10.1016/j.ajo.2010.01.039
7. Oster SF, Mojana F, Brar M, Yuson RMS, Cheng L, Freeman WR. Disruption of the photoreceptor inner segment/outer segment layer on spectral domain-optical coherence tomography is a predictor of poor visual acuity in patients with epiretinal membranes. Retina. 2010;30(5):713–718. doi:10.1097/IAE.0b013e3181c596e3
8. Stalmans P, Benz MS, Gandorfer A, et al. Enzymatic vitreolysis with ocriplasmin for vitreomacular traction and macular holes. N Engl J Med. 2012;367(7):606–615. doi:10.1056/NEJMoa1110823
9. Nudleman E, Franklin MS, Wolfe JD, Williams GA, Ruby AJ. Resolution of subretinal fluid and outer retinal changes in patients treated with ocriplasmin. Retina. 2016;36(4):738–743. doi:10.1097/IAE.0000000000000755
10. Singh RP, Li A, Bedi R, et al. Anatomical and visual outcomes following ocriplasmin treatment for symptomatic vitreomacular traction syndrome. Br J Ophthalmol. 2014;98(3):356–360. doi:10.1136/bjophthalmol-2013-304219
11. Tibbetts MD, Reichel E, Witkin AJ. Vision loss after intravitreal ocriplasmin: correlation of spectral-domain optical coherence tomography and electroretinography. JAMA Ophthalmol. 2014;132(4):487–490. doi:10.1001/jamaophthalmol.2013.8258
12. Itoh Y, Ehlers JP. Ellipsoid zone mapping and outer retinal characterization after intravitreal ocriplasmin. Retina. 2016;36(12):2290–2296. doi:10.1097/IAE.0000000000001110
13. Freund KB, Shah SA, Shah VP. Correlation of transient vision loss with outer retinal disruption following intravitreal ocriplasmin. Eye (Lond). 2013;27(6):773–774. doi:10.1038/eye.2013.94
14. Khanani AM, Duker JS, Heier JS, et al. Ocriplasmin treatment leads to symptomatic vitreomacular adhesion/vitreomacular traction resolution in the real-world setting: the Phase IV ORBIT study. Ophthalmol Retina. 2019;3(1):32–41. doi:10.1016/j.oret.2018.07.011
15. Sharma P, Juhn A, Houston SK, et al. Efficacy of intravitreal ocriplasmin on vitreomacular traction and full-thickness macular holes. Am J Ophthalmol. 2015;159(5):861–867. doi:10.1016/j.ajo.2015.01.034
16. Lavine JA, Srivastava SK, Dukkles N, Reese JL, Ehlers JP. Longitudinal ellipsoid zone and subretinal fluid mapping following ocriplasmin injection in the prospective observational ORBIT trial. Br J Ophthalmol. 2020;104(3):410–415. doi:10.1136/bjophthalmol-2019-314142
17. Duker JS, Kaiser PK, Binder S, et al. The international vitreomacular traction study group classification of vitreomacular adhesion, traction, and macular hole. Ophthalmology. 2013;120(12):2611–2619. doi:10.1016/j.ophtha.2013.07.042
18. Fukumoto M, Sato T, Oosuka S, Kida T, Oku H, Ikeda T. Clinical features of vitreomacular traction syndrome with peripheral vitreoretinal adhesion. Clin Ophthalmol. 2020;14:281–286. doi:10.2147/OPTH.S235670
19. Dugel PU, Tolentino M, Feiner L, Kozma P, Leroy A. Results of the 2-year Ocriplasmin for Treatment for Symptomatic Vitreomacular Adhesion Including Macular Hole (OASIS) randomized trial. Ophthalmology. 2016;123(10):2232–2247. doi:10.1016/j.ophtha.2016.06.043
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.