Back to Journals » Clinical Ophthalmology » Volume 8
Response of serous retinal pigment epithelial detachments to intravitreal aflibercept in polypoidal choroidal vasculopathy refractory to ranibizumab
Authors Yamashita M, Nishi T, Hasegawa T, Ogata N
Received 24 October 2013
Accepted for publication 14 November 2013
Published 3 February 2014 Volume 2014:8 Pages 343—346
DOI https://doi.org/10.2147/OPTH.S56539
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Mariko Yamashita, Tomo Nishi, Taiji Hasegawa, Nahoko Ogata
Department of Ophthalmology, Nara Medical University, Kashihara, Nara, Japan
Purpose: To report the effects of aflibercept on eyes with large retinal pigment epithelial detachment (PED) associted with polypoidal choroidal vasculopathy (PCV).
Methods: We reviewed the medical records of patients with PEDs associated with PCV that were treated with aflibercept after intravitreal ranibizumab had failed.
Results: Three eyes of patients aged 72, 79, and 80 years were studied. Reflective material was seen in the PED along the outer surface of the retinal pigment epithelium (RPE) by spectral-domain optical coherence tomography (SD-OCT). A complete resolution of the serous PEDs was found after two aflibercept injections; however, all eyes had a fibrovascular PED. In addition, one eye developed a retinal hemorrhage and a recurrent PED just after the third injection of aflibercept. The visual acuity in this eye decreased from 10/20 to 2/20.
Conclusion: The reflective material below the outer surface of the RPE in serous PED suggests the presence of neovascularization. Intravitreal aflibercept could be considered for large PEDs in eyes with PCV but should be carefully applied.
Keywords: age-related macular degeneration, retinal pigment epithelial detachment, polypoidal choroidal vasculopathy, aflibercept
Introduction
Intravitreal injections of pegaptanib or bevacizumab have been reported to be efficacious and safe treatments for retinal pigment epithelial detachments (PEDs) associated with occult choroidal neovascularization (CNV) secondary to age-related macular degeneration (AMD).1 Aflibercept, a recombinant fusion protein, has a higher binding affinity for vascular endothelial growth factor (VEGF), and it has been shown to be beneficial for patients with AMD who were refractory to multiple injections of either bevacizumab or ranibizumab.2,3 In addition, a recent study showed that intravitreal aflibercept may be an effective treatment for serous PEDs in which bevacizumab and ranibizumab were not effective.4
We report three cases of polypoidal choroidal vasculopathy (PCV) with a large PED that was refractory to ranibizumab but responded to intravitreal aflibercept. We present our spectral domain optical coherence tomographic (SD-OCT) findings, which provided some evidence on the development of the PED.
Case reports
Case 1
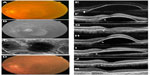
A 79-year-old man presented with a large serous PED. Fluorescein angiography (FA) displayed an occult CNV, and indocyanine green (IA) images showed a PCV. At the initial visit, his best-corrected visual acuity (BCVA) was 2/20 oculus sinister (OS). Three monthly injections of ranibizumab slightly flattened the PED and reduced the subretinal fluid (SRF), but a recurrence developed. Three additional monthly injections of ranibizumab (total of six injections) did not improve the PED, and there was an increase of the highly reflective material beneath the outer surface of the RPE in the SD-OCT images.
We switched to aflibercept and, after one intravitreal injection, the PED was slightly flattened; two additional injections of aflibercept were given, which flattened the PED over the hyper-reflective material observed by SD-OCT. The BCVA remained at 2/20 OS (Figure 1).
Case 2
A 72-year-old man presented with a large serous PED in his right eye associated with a PCV. His BCVA was 14/20 OD. He had received six ranibizumab injections over a 10-month period, but the PED worsened and the SRF recurred. Hyper-reflective materials were seen beneath the outer surface of the RPE in the SD-OCT images. The BCVA decreased to 8/20 OD.
Two monthly injections of aflibercept resulted in a complete resolution of the serous PED; however, the flattened PED contained fibrovascular material beneath the RPE layer in the SD-OCT images.
Case 3
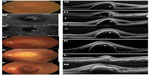
An 80-year-old man presented with a large serous PED. FA displayed an occult CNV, and IA images showed a PCV. His BCVA was 10/20 OS. Three monthly injections of ranibizumab slightly flattened the PED, but a recurrence was observed after 3 months. Three additional monthly injections of ranibizumab (total of six injections) failed to reduce the PED, and hyper-reflective materials were seen beneath the outer surface of the RPE in the SD-OCT images. We switched to aflibercept, and after the two additional monthly injections, an exudative CNV lesion was observed instead of the serous PED. OCT showed a loss of optical free space and highly reflective materials, suggesting the formation of fibrovascular PED. One day after the third monthly injection of aflibercept, a subretinal hemorrhage and recurrent PED were observed. BCVA was reduced to 2/20 (Figure 2).
Discussion
We found from three cases that intravitreal injections of aflibercept reduced or flattened large serous PEDs associated with PCV that were refractory to ranibizumab. Bird and Marshall5 and Casswell et al6 proposed that an interaction between the RPE and the Bruch membrane was central to the development of a PED, and they were not mutually exclusive.7 Alternatively, Gass8 proposed that PEDs in eyes with AMD occurred by neovascular ingrowth with subsequent exudation from the new vessels.
It is well known that PCVs frequently accompany PEDs.9 We found highly reflective materials within the serous PED beneath the outer surface of the RPE in all three cases. After aflibercept treatment, all of our cases had an apparent contracture of the accumulated material. This suggested the presence of fibrovascular tissues within the flattened PED. This is supported by the findings of Spaide,10 who examined the internal structure of PEDs in eyes with AMD and found that PEDs frequently have reflective materials on the back surface of the RPE, suggestive of CNV. These findings can be explained by the hypothesis presented by Gass8 on PED formation.
We found a sudden subretinal hemorrhage and recurrent PED in case 3. Following anti-VEGF therapy, a contracture of fibrovascular tissue has been reported in eyes with proliferative diabetic retinopathy.11 We suggest that aflibercept therapy may have enhanced fibrosis of neovascular tissues and caused a traction of the vessels, which then resulted in a sudden subretinal hemorrhage and a recurrence of the PED.
In conclusion, the reflective material below the outer surface of the RPE in serous PEDs suggests the presence of neovascularization. Intravitreal aflibercept could be considered for large PEDs in eyes with PCV but should be carefully applied.
Disclosure
The authors have no conflict of interest to declare in this work.
References
Arias L. Treatment of retinal pigment epithelial detachment with antiangiogenic therapy. Clin Ophthalmol. 2010;4:369–374. | |
Bakall B, Folk JC, Boldt HC, et al. Aflibercept therapy for exudative age-related macular degeneration resistant to bevacizumab and ranibizumab. Am J Ophthalmol. 2013;156(1):15–22. | |
Cho H, Shah CP, Weber M, Heier JS. Aflibercept for exudative AMD with persistent fluid on ranibizumab and/or bevacizumab. Br J Ophthalmol. 2013;97(8):1032–1035. | |
Patel KH, Chow CC, Rathod R, et al. Rapid response of retinal pigment epithelial detachments to intravitreal aflibercept in neovascular age-related macular degeneration refractory to bevacizumab and ranibizumab. Eye (Lond). 2013;27(5):663–667. | |
Bird AC, Marshall J. Retinal pigment epithelial detachments in the elderly. Trans Ophthalmol Soc U K. 1986;105(Pt 6):674–682. | |
Casswell AG, Kohen D, Bird AC. Retinal pigment epithelial detachments in the elderly: classification and outcome. Br J Ophthalmol. 1985;69(6):397–403. | |
Singerman LJ, Stockfish JH. Natural history of subfoveal pigment epithelial detachments associated with subfoveal or unidentifiable choroidal neovascularization complicating age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 1989;227(6):501–507. | |
Gass JD. Drusen and disciform macular detachment and degeneration. Trans Am Ophthalmol Soc. 1972:70;409–436. | |
Saito M, Iida T, Nagayama D. Cross-sectional and en face optical coherence tomographic features of polypoidal choroidal vasculopathy. Retina. 2008;28(3):459–464. | |
Spaide RF. Enhanced depth imaging optical coherence tomography of retinal pigment epithelial detachment in age-related macular degeneration. Am J Ophthalmol. 2009;147(4):644–652. | |
Arevalo JF, Maia M, Flynn HW Jr, et al. Tractional retinal detachment following intravitreal bevacizumab (Avastin) in patients with severe proliferative diabetic retinopathy. Br J Ophthalmol. 2008;92(2):213–216. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.