Back to Journals » Clinical Interventions in Aging » Volume 12
Relationship between tongue strength, lip strength, and nutrition-related sarcopenia in older rehabilitation inpatients: a cross-sectional study
Authors Sakai K , Nakayama E, Tohara H , Kodama K, Takehisa T, Takehisa Y, Ueda K
Received 5 May 2017
Accepted for publication 21 June 2017
Published 3 August 2017 Volume 2017:12 Pages 1207—1214
DOI https://doi.org/10.2147/CIA.S141148
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Walker
Video abstract presented by Kotomi Sakai.
Views: 3925
Kotomi Sakai,1,2 Enri Nakayama,2 Haruka Tohara,3 Keiji Kodama,4 Takahiro Takehisa,5 Yozo Takehisa,6 Koichiro Ueda2
1Department of Rehabilitation Medicine, Setagaya Memorial Hospital, 2Department of Dysphagia Rehabilitation, Nihon University School of Dentistry, 3Gerodontology and Oral Rehabilitation, Department of Gerontology and Gerodontology, Graduate School of Medical and Dental Sciences, Tokyo Medical and Dental University, 4Department of Internal Medicine, 5Department of Orthopaedic Surgery, Setagaya Memorial Hospital, Tokyo, 6Department of Internal Medicine, Hakuai Memorial Hospital, Tokushima, Japan
Objective: The objective of this study was to clarify the relationship between tongue strength, lip strength, and nutrition-related sarcopenia (NRS).
Patients and methods: A total of 201 older inpatients aged ≥65 years (70 men, median age: 84 years, interquartile range: 79–89 years) consecutively admitted for rehabilitation were included in this cross-sectional study. The main factors evaluated were the presence of NRS diagnosed by malnutrition using the Mini-Nutrition Assessment – Short Form, sarcopenia based on the criteria of the Asian Working Group for Sarcopenia, tongue strength, and lip strength. Other factors such as age, sex, comorbidity, physical function, cognitive function, and oral intake level were also assessed.
Results: In all, 78 (38.8%) patients were allocated to the NRS group, and 123 (61.2%) patients were allocated to the non-NRS group. The median tongue strength and lip strength (interquartile range) were significantly lower in the NRS group (tongue: 22.9 kPa [17.7–27.7 kPa] and lip: 7.2 N [5.6–9.8 N]) compared with the non-NRS group (tongue: 29.7 kPa [24.8–35.1 kPa] and lip: 9.9 N [8.4–12.3 N], P<0.001 for both). Multivariable logistic regression analysis showed that NRS was independently associated with tongue strength (odds ratio [OR] =0.93, 95% confidence interval [CI] 0.87–0.98, P=0.012) and lip strength (OR =0.76, 95% CI 0.66–0.88, P<0.001), even after adjusting for age, sex, comorbidity, physical function, cognitive function, and oral intake level.
Conclusion: The likelihood of occurrence of NRS decreased when tongue strength or lip strength increased. Tongue strength and lip strength may be important factors for preventing and improving NRS, regardless of the presence of low oral intake level in older rehabilitation inpatients.
Keywords: sarcopenia, rehabilitation, tongue, lip, malnutrition
Introduction
Rehabilitation hospitals have many more malnourished patients compared with other residential settings, acute hospitals, nursing homes, and in community dwelling older people.1 The prevalence of malnutrition in rehabilitation inpatients has been reported as 28%–53%.1–3 Malnutrition in older patients is important as it contributes to adverse outcomes such as decline in functional ability, prolonged hospital stay, discharge to high-care facilities, high medical care costs, and mortality.2,4 In recent years, sarcopenia, a condition of progressive and generalized loss of skeletal muscle mass and strength, has been recognized as a serious problem in older adults.5 Sarcopenia has negative effects on clinical outcomes, including longer hospital stays, readmission after discharge, and mortality.6 The prevalence of sarcopenia is reported to be approximately ≥50% in inpatients with malnutrition in rehabilitation units after acute illness.7,8 One of the causes of sarcopenia is malnutrition, which is called nutrition-related sarcopenia (NRS).5 Taken together, NRS can be a serious problem leading to adverse outcomes among older rehabilitation inpatients. Malnutrition occurs as a result of reduced dietary intake,9–11 and many rehabilitation inpatients consume less energy and protein than normal people.12 Malnutrition resulting from poor oral intake can cause sarcopenia and subsequently lead to adverse outcomes for rehabilitation patients. As oral intake involves oral function, clarification of the relationship between oral function and NRS may be important.
Oral strength plays an important role during eating and drinking. Tongue strength is related to the swallowing function and nutritional status,13–15 and many patients with difficulty swallowing (dysphagia) are malnourished.16 Decreased tongue strength has been associated with aging and sarcopenia.17–20 Therefore, decreased tongue strength may be related to NRS. A low level of lip strength is related to difficulty eating and drinking, as is a low level of tongue strength.21 In addition, lip posture alters (eg, pendency, the presence of labial fornices) with aging.22 Thus, lip strength may also be related to NRS.
The goal of the current study was to explore the relationship between tongue strength and lip strength and NRS. We hypothesized that NRS would be significantly associated with tongue strength and lip strength in older rehabilitation patients.
Patients and methods
Patients
This cross-sectional study was conducted in older inpatients (≥65 years) consecutively admitted from acute hospitals to our hospital in Tokyo, Japan, between October 2014 and March 2016. Our hospital has 200 beds and accepts patients with physical, swallowing, cognitive, or language disorders for rehabilitation after acute hospital stays. All measurements were assessed within a week of hospitalization. The study was approved by the ethical review committee of Setagaya Memorial Hospital. Written informed consent was obtained from all participants and their families or legal representatives. The exclusion criteria were as follows: 1) Mini-Mental State Examination (MMSE) score ≤20 reflecting moderate-to-severe cognitive dysfunction,23 as this could potentially cause difficulty in accurately measuring oral strength; 2) a history of cerebrovascular disease, head and neck cancer, or underlying neuromuscular disease such as Parkinson’s disease that might directly impair the nerves or muscles involved in oral strength; 3) loss of any upper or lower incisors, as a complete set of incisors is needed to use the apparatuses for measuring oral strength; 4) tooth pain or inadequate denture or mobile tooth assessed by dental hygienists, as these can affect the measurement of oral strength and diet level; and 5) patients diagnosed with depression by their attending physician, as this can potentially affect nutritional status and oral strength measurements.
Assessment of NRS
NRS was defined by the presence of both malnutrition and sarcopenia diagnosed using grip strength and calf circumference (CC). The nutritional status was evaluated by trained registered dietitians using the Mini Nutritional Assessment – Short Form (MNA-SF). The MNA-SF is a questionnaire survey recommended for use in the diagnosis of malnutrition among older patients,24 with a total score ranging from 0 to 14 points. The MNA-SF comprises six questions addressing decline in food intake and weight loss over the past 3 months, mobility, psychological stress or acute disease in the past 3 months, neuropsychological problems, and body mass index (BMI). Malnutrition is indicated by a score of ≤7, risk of malnutrition by a score of 8–11, and favorable nutritional status by a score of 12–14. In the present study, MNA-SF scores of ≤7 were used to define malnutrition with reference to a previous study.25 Sarcopenia was assessed using grip strength (<26 kg for men and <18 kg for women) and skeletal muscle mass based on the criteria of the Asian Working Group for Sarcopenia.26 Grip strength was measured in a sitting position using a digital grip strength dynamometer (TKK 5401; Takei Scientific Instruments, Tokyo, Japan). Physical therapists or occupational therapists asked participants to grip the instrument three times as hard as they could in each hand, and the maximal value was recorded. Skeletal muscle mass was assessed by measuring CC, a method that has been validated for measurement of skeletal muscle mass in the diagnosis of sarcopenia.27,28 With the participants in a seated position, the tape measure was placed around the calf and moved to obtain the maximal circumference without compressing subcutaneous tissues. In participants with fractures, measurements were taken on the healthy side. CC was measured by one trained rehabilitation therapist to eliminate interexaminer variation. The cutoff values for sarcopenia were set as CC <34 cm for men and CC <33 cm for women, as in a previous study.27 Participants were divided into the NRS group or the non-NRS group based on whether participants had NRS. Participants who did not have both malnutrition and sarcopenia were allocated to the non-NRS group.
Lip strength measurement
Lip strength was measured by trained speech-language pathologists (SLPs) with the lip closure strength indicator Lip De Cum (Cosmo Instruments Co., Ltd., Tokyo, Japan).29 The maximal strength produced to close the lips as hard as possible was measured using sensors inserted in the upper and lower plastic lip holders. This apparatus could record strength acting in the vertical direction and displayed the maximum lip strength of the upper and lower lips in combination. Measurements were taken with participants sitting in a relaxed position, three times for 5 s with a 30 s break between each measurement. The highest of three measurements was recorded as the lip strength of the participants.
Tongue strength measurement
Tongue strength was measured by trained SLPs using an apparatus with a balloon-type disposable oral probe on one end (JMS, Hiroshima, Japan). This apparatus measures the maximal isometric strength of anterior portion by pressing the balloon between the tongue and the roof of the mouth. Measurements were taken with participants sitting in a relaxed position, three times for 5 s with a 30 s break between each measurement. The highest of three measurements was recorded as the tongue strength of the participants.
Other recorded variables
Other possible factors related to NRS such as age, sex, comorbidity, primary disease, days in acute hospital, number of remaining teeth and occlusion, basic activities of daily living, inflammatory status, cognitive function, and the presence of low oral intake level were assessed. Comorbidity was evaluated using the Charlson Comorbidity Index (CCI) that lists 19 comorbid conditions, including diabetes with and without end organ damage, congestive heart failure, myocardial infarction, peripheral vascular disease, chronic pulmonary disease, dementia, cerebrovascular disease, connective tissue disease, peptic ulcer disease, mild and severe liver disease, hemiplegia, renal disease, leukemia, lymphoma, metastatic and non-metastatic tumors, and acquired immunodeficiency syndrome (AIDS), with each condition assigned a weight from 1 to 6.30 A higher CCI score indicates greater comorbidity. The CCI was assessed by the attending doctor in addition to the primary disease. The number of remaining teeth and the occlusion status, which were included as variables because of their effects on mastication, were assessed by trained dental hygienists. Occlusion status was assessed using the Eichner Index, regardless of whether the patient had natural or artificial teeth. This assessment was performed at the molar region and used to classify patients into the following groups: group A (occlusal support for molars and premolars on both sides), group B (reduced occlusal support), and group C (no occlusal support).31 We divided patients into two categories based on the results of Eichner Index: group A and group B or C. Basic activities of daily living were evaluated by physical therapists or occupational therapists using the Barthel Index (BI).32 The score ranged from 0 to 100, with higher scores representing greater independence. Inflammatory status was assessed using C-reactive protein (CRP) concentration, cognitive function using MMSE, and oral intake level, which can reflect the presence of dysphagia, using the Functional Oral Intake Scale (FOIS).33 The FOIS score ranged from level 1 (nothing by mouth) to 7 (total oral diet with no restrictions). Level 5 or less indicated the need for special meal preparation and/or nutritional supplementation such as enteral nutrition or intravenous nutrition. The relationship between FOIS and the dysphagia severity was confirmed; the former was assessed by SLPs, and the latter was diagnosed by videofluoroscopic examination of swallowing function in the previous study.33 FOIS was assessed by SLPs with >5 years of clinical experience in dysphagia management, and low oral intake level was determined by level 5 or less in the present study.
Statistical analyses
Sample size was calculated using data on malnutrition and sarcopenia from our previous study15 and the number of independent variables required for multivariable logistic regression analysis. Differences between the NRS group and the non-NRS group were analyzed using the Mann–Whitney U test for continuous variables and the chi-squared test or Fisher’s exact test for categorical variables. To assess the association between sarcopenia and malnutrition, the chi-squared test was used. To assess the correlation coefficient between tongue strength and lip strength, Spearman’s rank correlation coefficient was used. Multiple regression analyses were conducted to assess the relationship between tongue strength or lip strength and MNA-SF subitems. Univariable and multivariable logistic regression analyses using the forced entry method were conducted to assess the relationship between NRS and tongue strength and lip strength. The MNA-SF subitem “psychological stress or acute disease in the past 3 months” was excluded from all analyses because all participants had a score of 0. P<0.05 was considered as statistically significant. SPSS Statistics 23 software (IBM Corporation, Armonk, NY, USA) was used for all statistical analyses. There were no missing data, and all measurement data were available for analysis.
Results
Clinical characteristics
A total of 201 participants (70 men and 131 women) were included in the present study. Table 1 presents the patient characteristics. The median age (interquartile range) was 84 years (79–89 years), and the median MMSE score was 26 points (23–28 points) for all patients. Orthopedic conditions accounted for 56.7% (114) patients of all primary diseases, and other diseases included digestive (n=12, 6.0%), urinary tract (n=23, 11.4%), and skin (n=11, 5.5%) diseases. All patients took nutrition orally. The prevalence of malnutrition based on the MNA-SF was 44.8% (90) patients, and the prevalence of sarcopenia diagnosed by grip strength and CC was 76.1% (153) patients. According to the presence of NRS, 78 (38.8%) patients were classified as having NRS and 123 (61.2%) patients were classified as having non-NRS. There was a significant association between sarcopenia and malnutrition (P=0.002; Table 2). In the non-NRS group, 87 (70.7%) participants had either malnutrition or sarcopenia and 36 (29.3%) participants had neither malnutrition nor sarcopenia. There were significant differences between the two groups in sex (P=0.001), CCI (P<0.001), primary disease (P=0.008), MNA-SF (P<0.001), BMI (P<0.001), grip strength (P<0.001), CC (P<0.001), BI (P<0.001), MMSE (P=0.002), CRP (P=0.005), and FOIS (P<0.001). Significant differences were detected between the two groups for all MNA-SF subitems except “psychological stress or acute disease in the past 3 months” (neuropsychological problems: P=0.007, all others: P<0.001). The presence of a decline in food intake over the past 3 months (score 0 or 1) in the MNA-SF was confirmed in 65/201 (32.3%) subjects, 51/78 (65.4%) subjects in the NRS group and 14/123 (11.4%) subjects in the non-NRS group (P<0.001). The presence of low oral intake level was significantly different between the two groups (P<0.001): 47/78 (60.3%) subjects in the NRS group and 13/123 (10.6%) subjects in the non-NRS group.
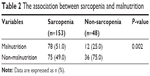  | Table 2 The association between sarcopenia and malnutrition |
Oral strength and nutritional status
The correlation coefficient between tongue strength and lip strength was 0.36 (P<0.001) in all participants. Tongue strength and lip strength were significantly lower in the NRS group compared with the non-NRS group (P<0.001 for both), although age was not significantly different. Table 3 presents the results of multiple linear regression analysis for the MNA-SF subitems with tongue strength or lip strength as a dependent variable. The decline in food intake over the past 3 months was independently associated with tongue strength and lip strength (tongue strength: P=0.009; lip strength: P=0.008).
Oral strength and the presence of NRS
Table 4 presents the results of univariable and multivariable logistic regression analyses with the presence of NRS as a dependent variable. All variables except age were significantly associated with NRS in the univariable logistic regression analysis. Tongue strength and lip strength were independently associated with the presence of NRS (tongue strength: OR =0.93, 95% CI 0.87–0.98, P=0.012; lip strength: OR =0.76, 95% CI 0.66–0.88, P<0.001) in the multivariable logistic regression analysis. In other words, tongue strength and lip strength were negatively associated with the presence of NRS. The Nagelkerke R2 was 0.51, and the omnibus test of model coefficients was significant (P<0.001) in this model.
Discussion
The present study found that tongue strength and lip strength were negatively associated with the presence of NRS. Furthermore, the greater the decline in food intake over the past 3 months, the weaker the tongue strength and lip strength. To our knowledge, this is the first study to investigate oral strength in relation to NRS. These results suggest that tongue strength and lip strength would be useful indices for the presence of NRS and that there is a causal relationship between tongue strength and lip strength and NRS.
There are some plausible mechanisms for our results. Participants in the present study did not have disease that directly impaired oral strength, such as stroke, Parkinson’s disease, or head and neck cancer. Therefore, decreased tongue strength and lip strength can occur due to sarcopenia. The association between tongue muscle and sarcopenia factors has been shown in previous studies. Tongue strength has been positively associated with grip strength in older adults,16,34 and tongue muscle thickness has been positively associated with mid-arm muscle mass in older adults.35 However, tongue strength is an important component of feeding. Decreased tongue strength is related to oral and pharyngeal residues, aspiration, lower oral intake level, longer meal times, and low food consumption.16,36–38 Thus, decreased tongue strength could occur due to sarcopenia and concurrently promote NRS. Grip strength has been positively associated with lip strength and tongue strength, independent of masticatory efficiency in older adults.39 Lips are constructed to prevent the escape of solids or liquids from the oral cavity.40 Lips also act cooperatively during chewing and are associated with chewing efficiency.41,42 Lip function is related to meal texture and malnutrition.43,44 Furthermore, the lip-closing function during meal consumption was significantly decreased in older adults compared with young adults.45 This previous finding suggests that the orbicularis oris muscle is potentially fatigued during meal consumption in adults with age-related sarcopenia. Taken together, decreased lip strength could occur due to sarcopenia and concurrently promote NRS, as could decreased tongue strength. Reduced tongue strength and lip strength were significantly associated with decline in food intake over the past 3 months. This result suggests that reduced tongue strength and lip strength occurred because of poor oral intake. Conversely, poor oral intake might have occurred because of decreased tongue strength and lip strength during acute hospital stays in the present study. The association between sarcopenia and inflammation was indicated in previous studies,46 and a significant difference in inflammation was also observed between the two groups in the present study. Although we did not include CRP in multivariable logistic regression analysis, CRP concentrations in the NRS group appeared to be low and had little effect on NRS, as demonstrated in previous studies.46,47
In the multivariable logistic regression analysis, tongue strength and lip strength were associated with NRS, independent of low oral intake level. Oral strength is typically assessed using a device that measures tongue or lip strength when patients are thought to have swallowing problems, and patients with a low oral intake level are provided with oral resistance exercises to improve tongue strength and lip strength and swallowing function.14,48 However, our results suggest that tongue strength and lip strength should also be measured for older inpatients without a low oral intake level to prevent or improve NRS. The results of the Nagelkerke R2 indicated that our findings were moderately useful for the interpretation of NRS in older rehabilitation inpatients.
Study limitations
The present study had several limitations that should be acknowledged. First, the study was cross-sectional; therefore, causal relationships could not be determined. Second, malnutrition was defined by malnutrition in MNA-SF as a screening test for nutritional status. The European Society of Clinical Nutrition and Metabolism recently developed new malnutrition criteria because of a previous lack of consensus about diagnostic criteria.24 In these criteria, patients must first fulfill the criterion for being “at risk” of malnutrition by any validated risk screening tool. Second, the diagnostic criterion of low BMI or the combined finding of weight loss with either reduced BMI or a low fat-free mass index must be met. New criteria were suggested to reduce false positives and increase specificity when compared with the MNA-SF for adverse outcomes.49 Unfortunately, these diagnostic criteria were set after our study was started. Lastly, muscle mass for diagnosis of sarcopenia was assessed using CC. Although CC was validated as an alternative index for muscle mass, it might affect the accuracy of diagnosis for sarcopenia.
Conclusion
Tongue strength and lip strength were negatively associated with the presence of NRS. Tongue strength and lip strength may be important factors in preventing and improving NRS, regardless of the presence of low oral intake level, in older rehabilitation inpatients.
Acknowledgment
This work was supported by the Japan Society for the Promotion of Science KAKENHI (grant number 16K19262).
Disclosure
The authors report no conflicts of interest in this work.
References
Kaiser MJ, Bauer JM, Ramsch C, et al. Frequency of malnutrition in older adults: a multinational perspective using the mini nutritional assessment. J Am Geriatr Soc. 2010;58(9):1734–1738. | ||
Charlton K, Nichols C, Bowden S, et al. Poor nutritional status of older subacute patients predicts clinical outcomes and mortality at 18 months of follow-up. Eur J Clin Nutr. 2012;66(11):1224–1228. | ||
Marshall S, Young A, Bauer J, Isenring E. Malnutrition in geriatric rehabilitation: prevalence, patient outcomes, and criterion validity of the scored patient-generated subjective global assessment and the mini nutritional assessment. J Acad Nutr Diet. 2016;116(5):785–794. | ||
Correia MI, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22(3):235–239. | ||
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al. Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412–423. | ||
Gariballa S, Alessa A. Sarcopenia: prevalence and prognostic significance in hospitalized patients. Clin Nutr. 2013;32(5):772–776. | ||
Sánchez-Rodríguez D, Calle A, Contra A, et al. Sarcopenia in post-acute care and rehabilitation of older adults: a review. Eur Geriatr Med. 2016;7(3):224–231. | ||
Sanchez-Rodriguez D, Marco E, Ronquillo-Moreno N, et al. Prevalence of malnutrition and sarcopenia in a post-acute care geriatric unit: applying the new ESPEN definition and EWGSOP criteria. Clin Nutr. Epub 2016 Sep 9. | ||
Kaiser MJ, Bauer JM, Ramsch C, et al. Validation of the mini nutritional assessment short-form (MNA-SF): a practical tool for identification of nutritional status. J Nutr Health Aging. 2009;13(9):782–788. | ||
Ottery FD. Definition of standardized nutritional assessment and interventional pathways in oncology. Nutrition. 1996;12(1 suppl):S15–S19. | ||
Kondrup J, Rasmussen HH, Hamberg OLE, Stanga Z; Ad Hoc ESPEN Working Group. Nutritional risk screening (NRS 2002): a new method based on an analysis of controlled clinical trials. Clin Nutr. 2003;22(3):321–336. | ||
Walton K, Williams P, Tapsell L, Batterham M. Rehabilitation inpatients are not meeting their energy and protein needs. E Spen Eur E J Clin Nutr Metab. 2007;2(6):e120–e126. | ||
Robbins J, Gangnon RE, Theis SM, Kays SA, Hewitt AL, Hind JA. The effects of lingual exercise on swallowing in older adults. J Am Geriatr Soc. 2005;53(9):1483–1489. | ||
Robbins J, Kays SA, Gangnon RE, et al. The effects of lingual exercise in stroke patients with dysphagia. Arch Phys Med Rehabil. 2007;88(2):150–158. | ||
Sakai K, Nakayama E, Tohara H, et al. Tongue strength is associated with grip strength and nutritional status in older adult inpatients of a rehabilitation hospital. Dysphagia. 2017;32(2):241–249. | ||
Carrión S, Cabré M, Monteis R, et al. Oropharyngeal dysphagia is a prevalent risk factor for malnutrition in a cohort of older patients admitted with an acute disease to a general hospital. Clin Nutr. 2015;34(3):436–442. | ||
Robbins J, Levine R, Wood J, Roecker EB, Luschei E. Age effects on lingual pressure generation as a risk factor for dysphagia. J Gerontol A Biol Sci Med Sci. 1995;50(5):M257–M262. | ||
Nicosia MA, Hind JA, Roecker EB, et al. Age effects on the temporal evolution of isometric and swallowing pressure. J Gerontol A Biol Sci Med Sci. 2000;55(11):M634–M640. | ||
Robbins J, Humpal NS, Banaszynski K, Hind J, Rogus-Pulia N. Age-related differences in pressures generated during isometric presses and swallows by healthy adults. Dysphagia. 2016;31(1):90–96. | ||
Maeda K, Akagi J. Decreased tongue pressure is associated with sarcopenia and sarcopenic dysphagia in the elderly. Dysphagia. 2015;30(1):80–87. | ||
SjÖGreen L, Lohmander A, Kiliaridis S. Exploring quantitative methods for evaluation of lip function. J Oral Rehabil. 2011;38(6):410–422. | ||
Baum BJ, Bodner L. Aging and oral motor function: evidence for altered performance among older persons. J Dent Res. 1983;62(1):2–6. | ||
Folstein MF, Folsein SE, Fanjiang G. MMSE Mini-Mental State Examination Clinical Guide. Lutz: Psychological Assessment Resources, Inc.; 2001. | ||
Cederholm T, Bosaeus I, Barazzoni R, et al. Diagnostic criteria for malnutrition – an ESPEN Consensus Statement. Clin Nutr. 2015;34(3):335–340. | ||
Koren-Hakim T, Weiss A, Hershkovitz A, et al. The relationship between nutritional status of hip fracture operated elderly patients and their functioning, comorbidity and outcome. Clin Nutr. 2012;31(6):917–921. | ||
Chen LK, Liu LK, Woo J, et al. Sarcopenia in Asia: consensus report of the Asian working group for sarcopenia. J Am Med Dir Assoc. 2014;15(2):95–101. | ||
Kawakami R, Murakami H, Sanada K, et al. Calf circumference as a surrogate marker of muscle mass for diagnosing sarcopenia in Japanese men and women. Geriatr Gerontol Int. 2015;15(8):969–976. | ||
Rolland Y, Lauwers-Cances V, Cournot M, et al. Sarcopenia, calf circumference, and physical function of elderly women: a cross-sectional study. J Am Geriatr Soc. 2003;51(8):1120–1124. | ||
Ono T, Hori K, Masuda Y, Hayashi T. Recent advances in sensing oropharyngeal swallowing function in Japan. Sensors (Basel). 2010;10(1):176–202. | ||
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383. | ||
Swoboda J, Kiyak HA, Persson RE, et al. Predictors of oral health quality of life in older adults. Spec Care Dentist. 2006;26:137–144. | ||
Mahoney FI, Barthel DW. Functional evaluation: The Barthel Index. Md State Med J. 1965;14:61–65. | ||
Crary MA, Mann GD, Groher ME. Initial psychometric assessment of a functional oral intake scale for dysphagia in stroke patients. Arch Phys Med Rehabil. 2005;86(8):1516–1520. | ||
Buehring B, Hind J, Fidler E, Krueger D, Binkley N, Robbins J. Tongue strength is associated with jumping mechanography performance and handgrip strength but not with classic functional tests in older adults. J Am Geriatr Soc. 2013;61(3):418–422. | ||
Tamura F, Kikutani T, Tohara T, Yoshida M, Yaegaki K. Tongue thickness relates to nutritional status in the elderly. Dysphagia. 2012;27(4):556–561. | ||
Ono T, Kumakura I, Arimoto M, et al. Influence of bite force and tongue pressure on oro-pharyngeal residue in the elderly. Gerodontology. 2007;24(3):143–150. | ||
Butler SG, Stuart A, Leng X, et al. The relationship of aspiration status with tongue and handgrip strength in healthy older adults. J Gerontol A Biol Sci Med Sci. 2011;66(4):452–458. | ||
Namasivayam AM, Steele CM, Keller H. The effect of tongue strength on meal consumption in long term care. Clin Nutr. 2015;35(5):1078–1083. | ||
Izuno H, Hori K, Sawada M, et al. Physical fitness and oral function in community-dwelling older people: a pilot study. Gerodontology. 2015;33(4):470–479. | ||
Kaede K, Kato T, Yamaguchi M, Nakamura N, Yamada K, Masuda Y. Effects of lip-closing training on maximum voluntary lip-closing force during lip pursing in healthy young adults. J Oral Rehabil. 2016;43(3):169–175. | ||
Takada K, Yashiro K, Sorihashi Y, Morimoto T, Sakuda M. Tongue, jaw, and lip muscle activity and jaw movement during experimental chewing efforts in man. J Dent Res. 1996;75(8):1598–1606. | ||
Schimmel M, Leemann B, Herrmann FR, Kiliaridis S, Schnider A, Müller F. Masticatory function and bite force in stroke patients. J Dent Res. 2011;90(2):230–234. | ||
Ono T, Hori K, Ikebe K, Nokubi T, Nago S, Kumakura I. Factors influencing eating ability of old in-patients in a rehabilitation hospital in Japan. Gerodontology. 2003;20(1):24–31. | ||
Van Lancker A, Verhaeghe S, Van Hecke A, Vanderwee K, Goossens J, Beeckman D. The association between malnutrition and oral health status in elderly in long-term care facilities: a systematic review. Int J Nurs Stud. 2012;49(12):1568–1581. | ||
Hiramatsu T, Kataoka H, Osaki M, Hagino H. Effect of aging on oral and swallowing function after meal consumption. Clin Interv Aging. 2015;10:229–235. | ||
Bano G, Trevisan C, Carraro S, et al. Inflammation and sarcopenia: a systematic review and meta-analysis. Maturitas. 2017;96:10–15. | ||
Rossi AP, Zanandrea V, Zoico E, et al. Inflammation and nutritional status as predictors of physical performance and strength loss during hospitalization. Eur J Clin Nutr. 2016;70(12):1439–1442. | ||
Hägg M, Anniko M. Lip muscle training in stroke patients with dysphagia. Acta Otolaryngol. 2008;128:1027–1033. | ||
Sanz-Paris A, Gomez-Candela C, Martin-Palmero A, et al. Application of the new ESPEN definition of malnutrition in geriatric diabetic patients during hospitalization: a multicentric study. Clin Nutr. 2016;35:1564–1567. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



