Back to Journals » Clinical Interventions in Aging » Volume 9
Relationship between cognitive impairment and apparent diffusion coefficient values from magnetic resonance-diffusion weighted imaging in elderly hypertensive patients
Authors Zhao Y, Wu G, Shi H, Xia Z, Sun T
Received 5 March 2014
Accepted for publication 8 May 2014
Published 28 July 2014 Volume 2014:9 Pages 1223—1231
DOI https://doi.org/10.2147/CIA.S63567
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Yanwei Zhao,1 Gang Wu,2 Haiming Shi,3,4 Zhijie Xia,1 Tao Sun3,4
1Emergency Department, Huashan Hospital North, Fudan University, Shanghai, People’s Republic of China; 2Emergency Department, Huashan Hospital, Fudan University, Shanghai, People’s Republic of China; 3Cardiology Division, Huashan Hospital North, 4Cardiology Division, Huashan Hospital, Fudan University, Shanghai, People’s Republic of China
Background: The purpose of this study was to determine a new method for the early diagnosis and assessment of mild cognitive impairment in elderly individuals with hypertension. Elderly hypertensive patients with cognitive impairment were assessed by the Montreal Cognitive Assessment (MoCA) and Clinical Dementia Rating Assessment (CDR). Cognitive results were compared to apparent diffusion coefficient (ADC) values from magnetic resonance-diffusion weighted imaging.
Methods: A total of 191 patients were categorized into four groups: a control group (normal cognition and no hypertension; n=20); a normal group (hypertension and normal cognition; n=33); an mild cognitive impairment group (n=80); and a vascular dementia group (n=58). The MoCA and CDR tests were used to determine cognition. ADC values in eight brain regions were calculated with magnetic resonance-diffusion weighted imaging. Other characteristics were evaluated, eg, blood pressure, MoCA, and CDR scores, and the comparisons of the four groups were made.
Results: The MoCA and CDR scores differed among the four groups (P<0.001). Systolic and diastolic blood pressure values increased as cognitive function declined (P<0.001). Cognitive function declined as ADC values increased, and they differed between elderly people with and without hypertension (P<0.001). Among elderly hypertensive participants, ADC values were significantly increased in the cortex and hippocampus.
Conclusion: The MoCA and CDR tests were sufficiently sensitive to evaluate cognition. Blood pressure was closely related to cognition, as well as to functional and structural changes in the brain. These alterations were evidenced through changes in the ADC values and were most obvious in the cortex and hippocampus. Greater cognitive decline was observed in elderly participants with hypertension compared to those without. As hypertensive stage increased, greater ADC values were observed.
Keywords: Clinical Dementia Rating Assessment, hypertension, mild cognitive impairment, Montreal Cognitive Assessment
Introduction
The global population aged 60 years or over is expected to increase from 700 million in 2009 to 2 billion by 2050, growing at a rate of 2.6% per year.1 During the last 50 years, the prevalence of hypertension in the People’s Republic of China has continuously increased, especially among the elderly. According to the structure of the Chinese population in 2010, about 200 million individuals in the People’s Republic of China (about 20% of adults) are predicted to have hypertension.2 High blood pressure (BP) and increased longevity suggest an increased incidence of age-related diseases, including cognitive impairment, the most severe of which is dementia. Increasing numbers of elderly hypertensive patients are presenting with different levels of cognitive impairment. According to surveys,3 an estimated 35.6 million people suffered from dementia worldwide in 2010. This estimate has been projected to increase to 115.4 million people by 2050.
Mild cognitive impairment (MCI) is defined as a deterioration of memory, attention, and cognitive function that exceeds what is expected for an individual’s age and education level. MCI is a common and frequently neglected condition among the elderly that may be a precursor to dementia. Petersen et al4 showed that the rate of transition from MCI to dementia was 10%–20% per year. Thus, it is important to diagnose MCI early so that modifiable risk factors during the MCI transition period can be addressed. However, the data linking dementia and MCI are sparse, especially among elderly hypertensive patients.
Therefore, this clinical study aimed to discuss the relationship between different levels of cognitive impairment and the apparent diffusion coefficient (ADC) values of magnetic resonance (MR)-diffusion weighted imaging (DWI) among elderly hypertensive people, to obtain up-to-date information regarding the early recognition and diagnosis of MCI. This information may guide intervention strategies of MCI in the future.
Materials and methods
Ethics statement
Both study sites are certified for conducting clinical trials by the Chinese State Food and Drug Administration. Prior to study initiation, approval was obtained from the ethics committees of the Renji Hospital and Huashan Hospital institutional review boards. The study was conducted in accordance with all applicable regulatory and patient privacy requirements. Written informed consent was obtained from all patients, in accordance with local regulations, before the beginning of the study.
Participants
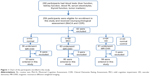
Participants were elderly hypertensive patients who were inpatients or outpatients of the Department of Neurology, Geriatrics, and Cardiology of Renji Hospital or Huashan Hospital in Shanghai, People’s Republic of China, from April 2009 to February 2011. The participants were separated into three groups (shows in Figure 1): normal group (hypertension with normal cognitive function); MCI group (hypertension with MCI); and vascular dementia (VD) group (hypertension with VD). The control group consisted of people with normal cognitive function and without hypertension.
Inclusion criteria
Participants were eligible for enrollment in this study if they satisfied the following criteria: 1) age between 60 and 79 years on their last birthday; and 2) diagnosis of hypertension according to the 2007 Guidelines for the Management of Arterial Hypertension5 as follows: for patients not taking antihypertensive drugs, a systolic blood pressure (SBP) ≥140 mmHg or diastolic blood pressure (DBP) ≥90 mmHg recorded three or more times in one day, and repeated over the course of multiple days; or, for patients taking antihypertensive drugs, a history of hypertension. All participants were assessed three times by one researcher on the same day, to ensure the accuracy of BP values. All BP results were described as mean ± standard deviation (SD) in units of mmHg.
Exclusion criteria
To control for common factors leading to cognitive impairment, except hypertension, exclusion criteria included: 1) secondary hypertension; 2) unstable angina; 3) uremia; 4) decompensated hepatic cirrhosis; 5) malignant tumors; 6) serious hematological diseases; 7) organic mental disorder or severe suicidal ideation and suicidal behavior; 8) use of antidepressant, antianxiety, or antipsychotic drugs in the previous week; 9) failure to finish the planned neuropsychiatric examinations due to diagnosed illness; 10) electrolyte imbalance; and 11) thyroid dysfunction.
Neuropsychological assessment
Criteria of mild cognitive impairment
Referring to the widely used diagnostic criteria of MCI formulated by Petersen et al in 19996 and the Montreal Cognitive Assessment (MoCA),7 which are newer, more sensitive, and more easily used than the mini-mental state examination,8 the criteria of MCI in this study were as follows: 1) a main complaint of memory loss provided by the patient, family members, or insiders; 2) Clinical Dementia Rating (CDR) assessment score of 0.5 points; 3) MoCA score of <26 points if the participant’s education history was >12 years, or <25 points otherwise; 4) memory loss that was incompatible with the participant’s age; 5) ability to maintain normal daily life; and 6) exclusion of dementia or any other physical or mental disease associated with brain dysfunction.
Criteria of VD
The diagnosis of VD was made according to the widely used criteria, which was National Institute of Neurological Disorders and Stroke and Association Internationale pour la Recherché et l’Enseignement en Neurosciences criteria [NINDS-AIREN criteria].9
Grading of hypertension
Stage 1 hypertension criteria10 included: SBP of 140–159 mmHg or DBP of 90–99 mmHg; an indication for medical intervention; recurrent or persistent (≥24 hours) symptoms; a symptomatic BP increase of >20 mmHg (diastolic) or BP >140/90 (systolic/diastolic) mmHg, if previously within normal limits; and an indication for monotherapy. Stage 2 hypertension criteria10 included: SBP ≥160 mmHg or DBP ≥100 mmHg; an indication for medical intervention; and an indication for more than one drug or more intensive therapy than previously used.
Neuroimaging
MR-DWI scan
A 3.0 T MR scanner (Philips Achieva; Koninklijke Philips N.V., Amsterdam, the Netherlands) was used for the brain MRI with a head quadrature coil. The switching rate was 150 T (m·ms−1), and a line parallel to the canthomeatal line was used to position the cross-sectional scans. First, a conventional scan sequence was used: a cross-sectional fast field echo T1 weighted image (time of repetition [TR] =180 ms, time of echo [TE] =2.3 seconds) and a fast spin echo T2 weighted image (TR =2,216 ms, TE =80 ms). If no acute lesions were found, then DWI was performed. A spin-echo echo-planar imaging pulse sequence was used for DWI with the following parameters: two diffusion gradient fields (b =0.800 s·mm−2); TR =4,521 ms; TE =136 ms; field of view =220×220 mm; acquisition matrix =512×512 mm; slice thickness =8 mm; and intervals of 2 mm. Images were collected once.
Apparent diffusion coefficient values
After importing the head MR-DWI data into the workstation, the region of interest (ROI) was selected on ADC maps manually. ROI analyses were controlled for demographic variables. The ROI size was 30±3 mm2. The Stejskal–Tanner formula:
ADC = Ln (S2/S1) (b1 − b2) | (1) |
S1, S2 represent the sensitivity of the signal intensity in diffusion-weighted imaging of different diffusion coefficients, which we called b values; b is a constant, b1, b2 represent different diffusion coefficients. This equation was used to calculate the ADC values of eight bilateral skull anatomical sites, including the centrum ovale, hippocampus, thalamus, corpus callosum, lenticular nucleus, caudate nucleus, temporal lobe cortex, and occipital lobe cortex. Each region was measured three times by one researcher on the same day to ensure that the positions of the differently sized ROIs were consistent. The results of ADC values are described as mean ± SD in units of 1×10−4 mm·s−1.
Statistical methods
Data were analyzed with SPSS version 16.0 (SPSS Inc., Chicago, IL, USA). Patient age, MoCA score, CDR score, BP values, ADC values, hypertension history, apolipoproteins A (ApoA) and B100 (ApoB100), lipoprotein (a) (LP(a)), total triglyceride (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), HDL-C/LDL-C, fasting blood glucose (FBG), postprandial blood glucose (PBG), and glycated hemoglobin (HbA1c) were described as mean ± SD. Analysis of variance was used to compare multigroup data. Data were tested for homogeneity of variance (P>0.05). A t-test was used to compare ADC values between: the control group and the normal group; the normal group and the MCI group; the MCI group and the VD group; and two stages of hypertension. Patient gender, stage of hypertension, and statin therapy status were described as numbers. Chi-squared tests were used to compare the multigroup data. In addition, multiple linear regression analysis was performed to evaluate the association between ADC value and MoCA score. Regression models were adjusted for age, gender, BP values, ADC values, hypertension history, and levels of ApoA, ApoB100, LP(a), TG, TC, HDL-C, LDL-C, FBG, PBG, and HbA1c. A small test statistic (P<0.05) indicated a statistically significant difference.
Results
Demographic characteristicsof participants
Table 1 reports the demographic characteristics of participants. Age, gender, lipid profile (ApoA, ApoB100, LP(a), TG, TC, HDL-C, LDL-C, and HDL-C/LDL-C), blood glucose profile (FBG, PBG, and HbA1c), and statin therapy status showed no significant differences between groups. According to the basic data, the influences of risk factors of cognitive impairment, including age, gender, abnormalities in lipids and blood glucose, and statin therapy status, were negligible.
Control and normal groups showed normal cognitive function, whereas the MCI and VD groups showed impaired cognitive function. The VD group showed the greatest impairment in cognition among the groups (Table 2). The MoCA and CDR scores were different between the control and normal groups. Hypertensive participants demonstrated much lower MoCA scores than those without hypertension. However, the CDR scores in all groups were zero. These results suggested that hypertension may influence cognitive function.
Participants with VD exhibited the lowest MoCA and highest CDR scores of all four groups. Participants in the MCI group exhibited much lower MoCA and higher CDR scores than control and normal group participants (MoCA F=199.850, P<0.001; CDR F=420.869, P<0.001). These findings suggest that MoCA and CDR were sufficient for assessing the cognitive condition, and that MoCA was more sensitive than CDR in this assessment. The combined use of MoCA and CDR for evaluating the cognitive condition was more accurate than either assessment alone.
Table 2 shows the BP conditions of all four groups. Participants in the normal, MCI, and VD groups were hypertensive, whereas participants in the control group exhibited normal BP. Participants with VD exhibited the highest SBP and DBP values of all groups. The BP values of the MCI participants were greater than those of normal-cognition participants with hypertension. The BP values were different among all four groups (SBP F=36.473, P<0.001; DBP F=65.234, P<0.001), as well as among the normal, MCI, and VD groups (SBP F=57.829, P=0.000; DBP F=46.245, P<0.001). Patients with VD exhibited the longest history of hypertension. The hypertensive history of participants in the MCI group was longer than that of the normal and control group participants (F=26.181, P<0.001). Significant differences were found between stage 1 and stage 2 hypertensive patients. As the stage of hypertension increased, the cognitive function impairment was more serious (P<0.001).
Cognitive impairment scores and ADC values in stage 1 and 2 hypertensive patients
As shown in Table 3, the MoCA scores of patients with stage 2 hypertension were lower than those of patients with stage 1 hypertension, while the CDR scores showed the opposite pattern (MoCA F=0.013, P=0.002; CDR F=3.401, P=0.011). The mean ADC values for all participants in the eight brain regions showed significant differences between stage 1 and 2 hypertensive patients, especially in the cortex and hippocampus. Interestingly, changes in mean ADC values were more remarkable in left hemisphere.
In multiple linear regression models adjusting for covariants, the MoCA scores were significantly associated with the ADC values (P=0.006).
Mean ADC values in brain regions in the control, normal, MCI, and VD groups
Results of region-based analyses of the mean ADC values are shown in Table 4. Comparisons among groups are shown in Table 5. Normal-cognition participants with and without hypertension showed differences in all eight brain regions (P<0.001). As these eight brain regions are related to hypertension, this finding demonstrates the important role that hypertension plays as a risk factor for cognitive impairment.
With the comparison of mean ADC values in brain regions between the normal group and the MCI group, it was found that, when the cognitive function changed from normal to MCI among the elderly hypertensive population, the mean ADC values in different brain regions also changed. Statistically significant differences were found in main brain regions, such as the centrum ovale (right F=3.463, P<0.001; left F=8.875, P<0.001), hippocampus (right F=7.751, P<0.001; left F=5.325, P<0.001), internal capsule (right F=1.222, P=0.001; left F=3.317, P<0.001), temporal lobe cortex (right F=34.122, P<0.001; left F=31.411, P<0.001), and occipital lobe cortex (right F=24.744, P<0.001; left F=34.122, P<0.001).
Discussion
The increasing age of the population brings many social problems, such as an increasing incidence of age-related diseases, including cognitive dysfunction. Studies have already confirmed that hypertension is an independent risk factor of MCI. Hypertensive patients with MCI are in transition from normal cognition to dementia, a period in which the brain undergoes dynamic pathological processes before clinical symptoms appear. Metabolic changes may manifest first among elderly hypertensive people with MCI. Therefore, the high-risk population of hypertensive patients with MCI has received much attention in recent years.
With the development of radiology, some new imaging technologies have been used in the clinic. In particular, DWI is a new MRI technology that is sensitive to early-stage ischemia and shows microdiffusion changes in ischemic areas. DWI is a relatively simple method of radiologic examination that has a higher diagnosis rate than MRI. Water molecule diffusion can be described by the diffusion tensor and diffusion coefficient D; as D increases, the diffusion speed increases. In the microcirculation, various factors influence the measurement of D, including blood flow, pulse, cerebrospinal fluid fluctuations, respiration, and tissue perfusion.11 Therefore, the value of D cannot completely describe the diffusion condition. For this reason, ADC, which integrates all of the above factors, was used in this study to replace the coefficient D.
Hypertension has been demonstrated to cause cognitive dysfunction. Studies by Rasquin et al12 suggested that hypertension leads to damage of some cognitive function domains, including speed of thought, spatial orientation abilities, and, especially, executive function. Tzourio et al13 completed a longitudinal investigation of 1,373 residents aged 59–71 years living in the western part of France. All participants had SBP ≥160 mmHg or DBP ≥90 mmHg. The authors assessed changes in the BP values and cognitive function compared to baseline after 4 years. The decline of cognitive function after the 4-year follow-up was correlated with increased BP. The relative risk of cognitive function decline was 6.0 after 2 years and 4.3 after 4 years. These results demonstrated that increased SBP or DBP is negatively correlated with decreasing cognitive scores and with multiple cognitive function domains, such as logical memory, visual reproduction, digital memories, and delayed logical memory.
However, all of the aforementioned studies used the mini-mental state examination to assess cognitive function. In the present study, we used the MoCA and CDR tests, which are more sensitive methods for the assessment of cognition.14 The MoCA and CDR scores were well-associated with cognitive function, and scores differed significantly among the four groups. A negative correlation between BP (SBP and DBP) and the degree of cognitive function was observed. The length of hypertension history was negatively correlated with cognitive function, and the differences were statistically significant between the three hypertensive groups. These observations confirmed that hypertension is one of the most important risk factors of MCI.
In summary, BP is one of the most important risk factors for cognitive function impairment. As BP and length of hypertension history increased, patients exhibited more serious cognitive impairment. Hypertension-related atherosclerosis is a predisposing factor of CVD, which could cause cerebral vascular stenosis, vasospasm, long-term chronic ischemia, and hypoxic conditions in the brain, and could lead to changes in brain function and structure, which may affect cognitive function.
Further analysis of the MoCA and CDR scores showed significant differences for stage 1 and 2 elderly hypertensive patients, indicating that increased hypertensive grade exacerbates cognitive function. Remarkable differences in ADC values were observed between these two groups in the cortex and hippocampal regions, and differences in the left hemisphere were more obvious. This result may be due to the fact that most Chinese people are right-handed, so the vascular vessels of the left side are more slender than those on the right. Thus, these vessels are more susceptible to stenosis and vascular spasm, which lead to pathological changes. Another explanation may be that the left cerebral hemisphere is the main region involved in language management and logical thinking functions. Declines in these functions are easily distinguished in daily life and in the clinic.
Reitz et al15 also found that a history of hypertension was related to a higher risk of MCI. Prevention and treatment of hypertension may have an important effect in lowering the risk of cognitive impairment. In the present study, the ADC values of the eight bilateral regions (centrum ovale, hippocampus, thalamus, corpus callosum, lenticular nucleus, caudate nucleus, temporal lobe cortex, and occipital lobe cortex) differed between people with and without hypertension. These results suggest that, as cognitive function began to decline, the function and structure of the cortex and the hippocampus noticeably changed. These changes were manifested as increased ADC values in these brain regions. When cognition declined to VD, similar results were observed.
Masdeu et al16 found that the lesion area in DWI and ADC value changes were significantly associated with cognitive function. Kantarci et al17 found that hippocampal ADC improved the ability of hippocampal volume measurements to predict future progression of MCI to AD. In the present study, in the transition from normal cognitive function to MCI and then to VD, changes in the ADC values in the cortex and hippocampus were increasingly obvious. Therefore, MR-DWI may be helpful for diagnosing MCI, and the results of this study may be useful for early therapeutic interventions of MCI.
In summary, ADC values increased with increasing impairment of cognitive function. Changes in the function and structure in the cortex and hippocampus were more obvious. With the development of cognitive impairment, the changes were more clearly observed, which suggests that these brain regions are closely related to cognitive function. The hippocampus is the region most closely associated with cognition; thus, it is expected that changes in the hippocampal area would be apparent with declining cognitive function. Cortex regions such as the centrum ovale, corpus callosum, temporal lobe cortex, and occipital lobe cortex showed a predilection for the development of hypertensive encephalopathy, as did the carotid artery and middle cerebral artery region, which are also closely related to cognition. Therefore, cognitive decline may be the main cause of ADC changes in these regions among elderly hypertensive individuals.
This study provides new ideas for identifying MCI in normal cognitive patients among the elderly hypertensive population, and may be helpful for early intervention and treatment to improve the prognosis among these patients. One limitation of the present study is that multiple comparisons increase the likelihood of type I error. To address this limitation, Bonferroni procedure was used for correction of multiple testing. Due to the small size and related experimental limitations of this study, further studies are needed to analyze in more detail the relationship between cognitive impairment and ADC values of MR-DWI among the elderly hypertensive population.
In conclusion, the BP of hypertensive patients cannot be easily controlled with increased age, leading to an increased risk of MCI. Currently, MCI is mainly diagnosed through neuropsychological assessments. However, different assessment scales exhibit different levels of reliability and validity, and an alternative is needed. This study found that cognitive impairment was related to both metabolic and structural changes in the bilateral centrum ovale, hippocampus, corpus callosum, temporal lobe cortex, and occipital lobe cortex, and that alterations in ADC values were more remarkable in the left cerebral hemisphere. In the development of declining cognition, changes in the cortex and hippocampus were more obvious. These findings suggest that clinicians should pay more attention to MR-DWI assessments in these two regions to diagnose MCI more quickly.
Acknowledgment
This work was supported by grants from the Shanghai science and technology committee to T Sun (13441900702).
Disclosure
The authors report no conflicts of interest in this work.
References
World Population Ageing [webpage on the Internet]. New York: United Nations Department of Economic and Social Affairs/Population Division; 2009. Available from: http://www.un.org/esa/population/publications/worldageing19502050/. Accessed June 30, 2014. | ||
Liu LS; Writing Group of 2010 Chinese Guidelines for the Management of Hypertension. [2010 Chinese guidelines for the management of hypertension]. Zhonghua Xin Xue Guan Bing ZaZhi. 2011;39(7):579–615. Chinese. | ||
World Alzheimer Report 2012: Overcoming the stigma of dementia [webpage on the Internet]. London: Alzheimer’s Disease international; 2012. Available from: http://www.alz.co.uk/research/world-report-2012. Accessed June 30, 2014. | ||
Petersen RC, Roberts RO, Knopman DS, et al. Mild cognitive impairment ten years later. Arch Neurol. 2009;66:1447–1455. | ||
Mansia G, De Backer G, Dominiczak A, et al. 2007 ESH-ESC Guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Blood Press. 2007;16(3):135–232. | ||
Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E. Mild cognitive impairment: clinical characterization and outcome. Arch Neurol. 1999;56:303–308. | ||
Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695–699. | ||
Sikaroodi H, Yadegari S, Miri SR. Cognitive impairments in patients with cerebrovascular risk factors: a comparison of Mini Mental Status Exam and Montreal Cognitive Assessment. Clin Neurol Neurosurg. 2013;115(8):1276–1280. | ||
Román GC, Tatemichi TK, Erkinjuntti T, et al. Vascular dementia: diagnostic criteria for research studies. Report of the NINDS-AIREN International Workshop. Neurology. 1993;43(2):250–260. | ||
Erbel R, Lehmann N, Möhlenkamp S, et al. Subclinical coronary atherosclerosis predicts cardiovascular risk in different stages of hypertension: result of the Heinz Nixdorf Recall Study. Hypertension. 2012;59:44–53. | ||
Yili Z, Xiaoyan H, Hongwen D, et al. The value of diffusion-weighted imaging in assessing the ADC changes of tissues adjacent to breast carcinoma. BMC Cancer. 2009;14(9):18. | ||
Rasquin SM, Verhey FR, Lousberg R, Lodder J. Cognitive performance after first ever stroke related to progression of vascular brain damage: a 2 year follow up CT scan study. J Neurol Neurosurg Psychiatr. 2005;76: 1075–1079. | ||
Tzourio C, Dufouil C, Ducimetière P, Alpérovitch A. Cognitive decline in individuals with high blood pressure: a longitudinal study in the elderly. EVA Study Group. Epidemiology of Vascular Aging. Neurology. 1999;53:1948–1952. | ||
Goldstein FC, Levey AI, Steenland NK. High blood pressure and cognitive decline in mild cognitive impairment. J Am Geriatr Soc. 2013; 61(1):67–73. | ||
Reitz C, Tang MX, Manly J, Mayeux R, Luchsinger JA. Hypertension and the risk of mild cognitive impairment. Arch Neurol. 2007;64(12): 1734–1740. | ||
Masdeu JC, Zubieta JL, Arbizu J. Neuroimaging as a marker of the onset and progression of Alzheimerps disease. J Neurol Sci. 2005;236: 55–64. | ||
Kantarci K, Petersen RC, Boeve BF, et al. DWI predicts future progression to Alzheimer’s Disease in amnestic mild cognitive impairment. Neurology. 2005;64(5):902–904. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.