Back to Journals » Infection and Drug Resistance » Volume 11
Relation between flomoxef minimum inhibitory concentrations and clinical outcomes of patients treated with flomoxef for Enterobacteriaceae bacteremia
Authors Lee CH, Chen IL, Li CC, Chien CC
Received 29 August 2018
Accepted for publication 29 October 2018
Published 27 November 2018 Volume 2018:11 Pages 2471—2480
DOI https://doi.org/10.2147/IDR.S185670
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Chen-Hsiang Lee,1 I-Ling Chen,2 Chia-Chin Li,3 Chun-Chih Chien3
1Division of Infectious Diseases, Department of Internal Medicine, Kaohsiung Chang Gung Memorial Hospital, Chang Gung University College of Medicine, Kaohsiung, Taiwan; 2Department of Pharmacy, Kaohsiung Chang Gung Memorial Hospital, Kaohsiung, Taiwan; 3Department of Laboratory Medicine, Kaohsiung Chang Gung Memorial Hospital, Kaohsiung, Taiwan
Background: Flomoxef is potentially effective against β-lactamase-producing Enterobacteriaceae because limited clinical data demonstrate its effectiveness against Enterobacteriaceae bloodstream infections (BSIs) based on its minimum inhibitory concentrations (MICs). This study was conducted to determine the optimal breakpoints based on the survival of patients with Enterobacteriaceae BSIs treated with flomoxef.
Methods: The 30-day crude mortality rate was analyzed among 224 adults who initiated flomoxef monotherapy for Enterobacteriaceae BSIs at a medical center over a 3-year period, according to the flomoxef MICs of the initial isolates. The outcome was evaluated by classification and regression tree modeling and by logistic regression analysis.
Results: The 30-day crude mortality was approximately two fold greater in patients whose isolates had flomoxef MICs of ≥2 mg/L (54.9% [62/113]) than in those with isolates with MICs of ≤1 mg/L (26.1% [29/111]); the differences were significant in bivariate analysis (P<0.01) and in survival analysis (log-rank test; P<0.001). The classification and regression tree analysis revealed a split between MICs of 1 and 2 mg/L and predicted the same difference in mortality, with a P-value of <0.001. Flomoxef for Enterobacteriaceae BSIs caused by isolates with flomoxef MICs of ≥2 mg/L was an independent predictor of 30-day crude mortality (adjusted OR 3.76, 95% CI 1.94–7.29).
Conclusion: Patients with Enterobacteriaceae bacteremia who received flomoxef had a lower 30-day crude mortality when the flomoxef MICs of the isolates were ≤1 mg/L than those with MICs ≥2 mg/L.
Keywords: bloodstream infection, breakpoint, classification and regression tree modeling, mortality
Introduction
Infections caused by members of the Enterobacteriaceae family are among the major causes of hospitalization and associated morbidity and mortality.1 With the widespread emergence of multidrug-resistant strains, the clinical effectiveness of early generations of antibiotics has greatly decreased,2 leading to the introduction of broad-spectrum antibiotics such as fluoroquinolones or third-generation cephalosporins (cefotaxime, ceftriaxone, or ceftazidime). Since their first description in 1983, extended-spectrum β-lactamases (ESBLs) produced by enteric pathogens have spread worldwide.3 Carbapenems are widely regarded as the antibiotics of choice for the treatment of infections due to ESBL producers.4,5 Nevertheless, carbapenems should be cautiously used given concerns about the emergence of carbapenem-resistant organisms.6 Identification of alternative antibiotics for the treatment of infections with ESBL-producing organisms might be a feasible strategy to relieve the selective pressure.7
Cephamycins are a subclass of second-generation cephalosporins that contain an R1 medroxy group substitution. This substitution confers resistance to degradation by most ESBL enzymes with demonstration of consistent in vitro activity against ESBL-producing Enterobacteriaceae isolates. Flomoxef, one of the cephamycins, is stable in the presence of β-lactamase and may have the potential to be effective against these β-lactamase-producing strains.8 However, clinical data regarding its possible role in the treatment of ESBL-associated infections are limited to date.9–11 In our cohort study among adults with ESBL-producing Enterobacteriaceae bacteremia by propensity score-adjusted analysis, treatment with flomoxef was associated with a higher mortality rate than treatment with carbapenems, particularly in patients infected with isolates with a flomoxef minimum inhibitory concentration (MIC) of 2–8 mg/L, even though the currently suggested MIC breakpoint of flomoxef is ≤8 mg/L.8 No MIC breakpoint for flomoxef has been published by the Clinical and Laboratory Standards Institute (CLSI). While the susceptibility to flomoxef at an MIC of ≤8 mg/L for Enterobacteriaceae in the reference to the CLSI breakpoint for cefotiam is based on the similarity in pharmacokinetic data between flomoxef and cefotiam,12 no clinical studies have verified flomoxef’s effectiveness in patients with Enterobacteriaceae bloodstream infections (BSIs) according to different flomoxef MIC values. Therefore, we aimed to determine the relation between MICs and clinical outcome based on the survival of patients with Enterobacteriaceae BSIs who were treated with flomoxef.
Patients and methods
Study design and participants
This retrospective cohort study was conducted at Kaohsiung Chang Gung Memorial Hospital, a tertiary-care medical center in southern Taiwan. It was conducted in accordance with the Declaration of Helsinki. The Institutional Review Board of the hospital approved the protocol (permit no. 201801000B0) and waived the need for patient consent owing to the retrospective nature of the study. The data of patients were kept anonymous to maintain confidentiality.
During 2014–2016, blood culture records from adult patients (aged over 18 years) who had at least one positive result for Enterobacteriaceae were reviewed. In cases where a patient experienced more than one bacteremia episode, only the first episode was included. Patients who fulfilled both of the following criteria were included: 1) mono-microbial bacteremia caused by Enterobacteriaceae and 2) definitive parenteral monotherapy with flomoxef for >48 hours. The following intravenous doses or adjusted equivalents of flomoxef for renal insufficiency were prescribed: patients with creatinine clearance (CLCR) of >60 mL/(min·1.73 m2), 1 g every 8 hours; patients with CLCR of 10–60 mL/(min·1.73 m2), 1 g every 12 hours; and those with CLCR of <10 mL/(min·1.73 m2), 1 g every 24 hours. Flomoxef was normally infused within 30 minutes. In compliance with the antimicrobial stewardship system in the hospital,13 all flomoxef prescriptions had been approved by infectious-disease specialists. Patients who received an inappropriate dosage of flomoxef and those with incomplete medical records were excluded.
Definitions
The medical records of the included patients were retrospectively reviewed to collect information on demographics; duration of hospital and intensive care unit (ICU) stays; comorbidities; the presence of tubes, vascular catheters, and drainage catheters at the time of BSI onset; and timing and duration of antimicrobial drugs. Bacteremia episodes with the first positive cultures collected within 48 hours of admission were defined as community-onset infections; those obtained 48 hours after admission were categorized as hospital-onset infections. Acquisition of BSI in the ICU was considered if bacteremia occurred 48 hours after ICU admission. The onset of bacteremia was defined as the day when the blood cultures were obtained that eventually yielded a species of Enterobacteriaceae.
The severity of underlying medical conditions was classified as non-fatal, fatal, and ultimately fatal under McCabe classification.14 The severity of the infection was evaluated using the Pitt bacteremia score recorded on the day when the index blood culture was obtained.15 Patients were evaluated with the BSI mortality risk score; patients with guarded prognoses had scores ≥5 and those with good prognoses had scores <5.16 Severe sepsis was defined as the coexistence of sepsis and acute organ dysfunction or hypoperfusion. Septic shock was defined as the coexistence of severe sepsis and systolic blood pressure no higher than 90 mmHg after a crystalloid-fluid challenge or a high blood lactate concentration (>4 mmol/L).17 Inappropriate empirical antibiotic therapy was the first dose of appropriate antimicrobial agent being not administered within the first 24 hours after blood samples were drawn.18 Appropriate antibiotic therapy was considered if the route, dosage, and timing of the antimicrobial agent were in accordance with the Sanford Guide,19 and the isolated pathogens were susceptible in vitro to the prescribed agent with reference to contemporary CLSI breakpoints.20
BSI sources were determined based on the presence of an active infection site coincident with bacteremia or isolation of a microorganism from other clinical samples before or on the same date as the bacteremia onset. BSI episodes that could not be assigned to a specific site were classified as primary bacteremia. Previous use of antibiotics was defined as treatment with these drugs within 1 month before the onset of bacteremia. Adequate source control was defined as having a timely 1) surgical intervention to debride infected tissues, drain accumulated infected fluid, and control an enteric pathogen constituting an intra-abdominal source of infection that led to bacteremia; 2) removal of a central venous catheter for catheter-related bacteremia; and 3) change or removal of a urinary catheter for BSI secondary to catheter-related urinary tract infection.
The primary outcome was 30-day crude mortality, which was defined as all-cause mortality occurring within 30 days of hospitalization after the onset of BSI. No follow-up was conducted after hospital discharge unless the discharge occurred before the 30 days. In addition, early identification of patients who have been infected by isolates with suboptimal MICs of flomoxef is important. We further investigated the risk factors of acquiring isolates with suboptimal MICs of flomoxef among these patients.
Laboratory investigations
The Enterobacteriaceae isolates causing BSI were subjected to microbiological analyses. The bacteria were phenotypically identified using the Vitek 2 system (bioMerieux, Marcy l’Etoile, France). MICs of flomoxef and antimicrobial susceptibilities of other agents were determined with the use of standard broth microdilution methods and interpreted according to the breakpoints suggested by the CLSI.20
Statistical analyses
Categorical variables were expressed as percentages of all patients and were compared with the use of the χ2 or Fisher’s exact test, as appropriate. Continuous variables were expressed as mean values ± SDs and compared by the Mann–Whitney U test or Student’s t-test. Independent predictors of mortality or acquiring isolates with suboptimal MICs of flomoxef were identified via logistic regression analysis. Variables with a P-value ≤0.10 in the univariate analysis were included in the logistic regression model to identify variables with either a negative or positive impact on the 30-day crude mortality or acquiring isolates with suboptimal MICs of flomoxef. Goodness of fit was assessed by the Hosmer–Lemeshow statistic. Receiver operating characteristics (ROC) curve analysis was conducted to evaluate the predictive performance of the logistic regression model. Classification and regression tree (CART) modeling was utilized to define a split in the flomoxef MIC distribution that maximized the difference in mortality. The time to mortality was analyzed by means of the Kaplan–Meier curve and log-rank test. Data with a two-tailed P-value <0.05 were considered statistically significant. All analyses were performed using SPSS software, version 11.5 (SPSS, Chicago, IL, USA).
Results
In total, 3,658 cases of Enterobacteriaceae bacteremia were identified within this 3-year study period, including 596 (16.3%) cases of polymicrobial BSI, 2,729 (74.6%) cases of treatment with antibiotics other than flomoxef, 102 (2.8%) cases of treatment with combined antimicrobial agents, and 76 (2.1%) cases treated with flomoxef for less than 24 hours or dosage inappropriate for end-organ function. As a result, a total of 224 patients with BSIs due to Klebsiella pneumoniae (121 [54.0%]), Escherichia coli (73 [32.6%]), and other Enterobacteriaceae including 14 cases of Proteus mirabilis, eight cases of Enterobacter spp., four cases of Serratia marcescens, and four cases of Citrobacter spp. were eligible for inclusion. After initiation with flomoxef monotherapy, 46 of the 224 patients were switched to other antimicrobial agents. Among these 46 patients, 13 (28.2%) started to receive other active drugs 48–72 hours after the onset of bacteremia, 18 (39.1%) started 72–96 hours after the onset of bacteremia, and 15 (32.6%) started 96–120 hours after the onset of bacteremia. None of these alternative regimens was associated with 30-day mortality rate. Overall, the median duration of flomoxef administration was 10 days.
The 30-day crude mortality rates varied in relation to the flomoxef MICs of the Enterobacteriaceae isolates (Figure 1). The mortality rates did not differ significantly between the patients with isolates with MICs of ≤0.5 mg/L and those with MICs of 1 mg/L (25.9% vs 26.4%, P>0.99). Compared with patients with isolates with an MIC of 1 mg/L, those with isolates with an MIC of ≥2 mg/L had significantly higher 30-day mortality (54.9% vs 26.4%, P<0.01). The 30-day mortality rates of patients with isolates with an MIC of 2 mg/L did not differ significantly from those with isolates showing an MIC of ≥4 mg/L (50.0% vs 55.4%, P=0.77). However, those who were infected with isolates with an MIC of ≥2 mg/L had two fold higher 30-day mortality than those who were infected with isolates with an MIC of ≤1 mg/L (54.9% vs 26.1%, P<0.01). A division in the MICs between 1 and 2 mg/L as predictive of a difference in mortality by CART analysis was found (P<0.001) (Figure 2). Furthermore, the Kaplan–Meier survival analysis also revealed that the 30-day mortality rate was significantly higher in patients with BSIs due to Enterobacteriaceae isolates with flomoxef MICs of ≥2 mg/L than in those with isolates showing MICs of ≤1 mg/L (P<0.001 according to the log-rank test) (Figure 3).
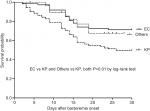
Between patients whose isolates had a flomoxef MIC of ≥2 mg/L and those with isolates with an MIC of ≤1 mg/L, no patients received flomoxef as empirical antibiotic therapy. Clinical characteristics such as age, gender, comorbidities, disease severity, BSI source, inappropriate empirical antibiotic, duration of administration of empirical and total antibiotic, and adequate source control did not differ significantly between the two groups (Table 1). The length of hospital stay (days) after bacteremia for survivors was not significantly different between the two groups, either (Table 1). However, patients infected by isolates with a flomoxef MIC of ≤1 mg/L had a better clinical outcome in terms of 14-day and 30-day crude mortality than those infected by Enterobacteriaceae with an MIC of ≥2 mg/L. We found that the acquisition of Enterobacteriaceae isolates with a flomoxef MIC of ≥2 mg/L (OR 3.76, 95% CI 1.94–7.29; P<0.001), the BSI mortality risk score ≥5 at bacteremia onset (OR 5.69, 95% CI 2.92–11.11; P<0.001), BSI acquired in the ICU (OR 2.08, 95% CI 1.05–4.12; P=0.035), the presence of ultimately or rapidly fatal comorbidity (OR 2.81, 95% CI 1.38–5.72; P=0.004), and pneumonia as the source of BSI (OR 5.27, 95% CI 1.83–15.12; P=0.002) were independent factors associated with 30-day mortality (Table 2). The survival analysis revealed a significant difference in the 30-day mortality rates between patients infected with E. coli and K. pneumoniae (P<0.01, log-rank test), and between patients infected with other Enterobacteriaceae and K. pneumoniae (P<0.01, log-rank test) (Figure S1).
According to the univariate analysis, patients who acquired Enterobacteriaceae isolates with an MIC of ≥2 mg/L were more likely to have acquired the isolates in the ICU stay, to be infected by K. pneumoniae isolates, and to have had prior use of flomoxef (Table 1). In addition, bacteremic patients who were infected by E. coli were less likely to acquire isolates with an MIC of ≥2 mg/L (Table 1). After adjustments were made, both the acquisition of an Enterobacteriaceae isolate in an ICU (OR 2.76, 95% CI 1.50–5.05; P=0.001) and bacteremia caused by K. pneumoniae (OR 5.78, 95% CI 3.12–10.48; P<0.001) were found to be independent factors associated with the acquisition of Enterobacteriaceae isolates with a flomoxef MIC of ≥2 mg/L (Table 3).
Discussion
Flomoxef has shown consistent in vitro activity against ESBL producers.9,10 Unfortunately, clinical data evaluating flomoxef for infections caused by ESBL-producing bacteria are scarce, and existing observational studies comparing flomoxef and carbapenems are limited by the inherent confounding by indication, a small sample size, and a lack of precise MIC values in most of these studies.8–11 In addition, the results of these studies are inconsistent, which may be explained at least in part by the differences in the demographics of the included study populations and in the bacterial species with a variety of MIC data. Matsumura et al found similar mortality rates among patients receiving definite therapy between 59 patients treated with flomoxef or cefmetazole and 54 treated with carbapenems, after propensity score adjustment.10 However, only five (8.6%) patients were infected by an E. coli isolate with a flomoxef MIC of ≥2 mg/L among these 58 isolates with available MIC data.10 Furthermore, our previous study found similar mortality rates between patients treated with flomoxef and those treated with carbapenems when the MIC of flomoxef was ≤1 mg/L, but not when it was 2–8 mg/L.8
In 2010, the CLSI lowered the MIC breakpoints for many β-lactam antibiotics to enhance the detection of known resistance among Enterobacteriaceae. The CLSI Antibiotic Subcommittee asserted that routine ESBL testing was no longer necessary and that treatment decisions can be based solely on MICs.20 In East Asia, flomoxef is frequently used to treat intra-abdominal infections or for prophylaxis before a surgical procedure.10,21 Thus, clinicians have sometimes used flomoxef therapy as empirical therapy or as definitive therapy for sepsis or BSIs when antimicrobial susceptibility was confirmed. Therefore, it is important to determine optimal breakpoints based on the survival of patients with Enterobacteriaceae BSIs who receive flomoxef.
Although susceptibility to flomoxef at an MIC of ≤8 mg/L has been suggested as the CLSI breakpoint for susceptibility,12 in this study, we found that the mortality rate did not differ significantly between the patients who were infected by Enterobacteriaceae isolates with flomoxef MICs 8 mg/L and those with MICs ≥16 mg/L. After controlling for confounders, acquisition of isolates with an MIC of ≥2 mg/L remained an independent factor associated with 30-day crude mortality (Table 2). These results provide clinical data to support the use of an MIC of ≥2 mg/L as a flomoxef non-susceptibility criterion. According to the current report and our previous study,8 the proportion of isolates with flomoxef MIC >1 mg/L among the infections caused by K. pneumoniae was higher than that of infections due to E. coli, regardless of the presence of ESBL genes, which may have resulted in significantly lower 30-day survival rates in patients infected with K. pneumoniae than in those infected with E. coli, according to our survival analysis (P<0.01, log-rank test) (Figure S1).
Our findings also indicate that, in the presentation of pneumonia reflected as high-inoculum infections, the 30-day crude morality that was determined in 13 patients infected by isolates with a flomoxef MIC of ≥2 mg/L and three isolates with an MIC of ≤1 mg/L was significantly higher in pneumonic BSI patients who received flomoxef therapy for isolates with an MIC of ≥2 mg/L (P=0.02, Fisher’s exact test). The clinical difference between pneumonic BSI patients who received flomoxef therapy for isolates with a flomoxef MIC of ≥2 mg/L and those with an MIC of ≤1 mg/L may be explained by the fact that flomoxef may behave differently against pathogens with different MIC data, especially in infections involving a high bacterial load. Conversely, the investigation conducted by Yang et al, which included a majority of infections originating outside the urinary tract and patients with a much higher baseline Pitt bacteremia score, found a higher rate of mortality in patients who received flomoxef therapy.11 In contrast, we found that patients with bacteremia secondary to urinary tract infections, irrespective of MIC, were at a lower risk of death.8 This discrepancy between the two studies could probably be explained by the high concentrations of flomoxef in the urine22 and a lower mortality associated with urinary tract bacteremic infections.23 Taken together, these results reinforce the importance of patient selection when considering flomoxef therapy.
Apart from the inoculum effect, an alternative explanation for poor outcomes among some patients receiving flomoxef therapy is related to the failure to meet necessary pharmacodynamic targets for isolates with high MICs. Monte Carlo simulation is a mathematical tool that may be applied to integrating multiple pharmacokinetic/pharmacodynamic (PK/PD) variables to estimate the probability of target attainment when the specific free-drug concentration (f) in the serum is above the MIC (fT>MIC) targets.24,25 When target attainment rates fall below 90%, the probability that a dosing regimen will be effective is significantly diminished, and the PK/PD cutoff becomes the highest MIC for which the target attainment exceeds 90%.24 For Enterobacteriaceae with or without ESBL producers, bactericidal effects are observed for a β-lactam when fT>MIC is 60–70% of the dosing interval.25 The probability of fT>MIC ≥70% (for isolates with flomoxef MIC90=0.5 mg/L) is seen in 67.3% of patients who receive flomoxef at a dose of 1 g every 8 hours.22 These data suggest that flomoxef may not be effectively bactericidal against Enterobacteriaceae, particularly against isolates with higher flomoxef MICs (for isolates with flomoxef MIC90 >0.5 mg/L). In the present study, all patients with Enterobacteriaceae bacteremia received the standard dose of flomoxef. Shortening the interval of flomoxef administration to 1 g every 6 hours or prolonging infusion to more than 1 hour may increase the probability of attaining pharmacodynamic targets.22 Further studies are warranted to evaluate the effect of various flomoxef dosing strategies on the clinical outcome of patients with Enterobacteriaceae BSIs.
Early identification of patients who acquired isolates with a flomoxef MIC of ≥2 mg/L is important because flomoxef therapy at standard doses is likely to fail. Our study suggests that acquisition of an Enterobacteriaceae isolate in an ICU and bacteremia caused by K. pneumoniae are independent associated factors with the isolates having flomoxef MICs of ≥2 mg/L (Table 3). Under these circumstances, clinicians should exercise caution when using flomoxef for BSIs before the flomoxef MIC data on the isolates are available.
The strength of this study is the inclusion of an adequate number of patients. This is reflected in adequate goodness of fit, and the ROC analysis indicated that the predictive performance of our logistic regression model is adequate. The limitations of this study are inherent in the retrospective design and intrinsic selection bias. Although there were some differences in terms of clinical characteristics between the patients infected by isolates with a flomoxef MIC of ≤1 mg/L and those infected by isolates with a flomoxef MIC of ≥2 mg/L, we carried out multivariate logistic regression analysis and CART analysis to minimize the bias of the uneven distribution of the clinical characteristics. All analytical results indicated the same finding, that a flomoxef MIC of ≥2 mg/L was significantly associated with mortality. The assessment of the outcomes of interest was conducted in an unblinded manner, thus raising the risk of misclassification, although the in-hospital outcome measure (30-day crude mortality) was a hard endpoint. Among these 224 patients, 134 (59.8%) acquired bacteremia during ICU admission. This may explain that high mortality rate in the current study (>25%) compared to the usual range of mortality rates with Enterobacteriaceae spp. bacteremia elsewhere (10–15%). Finally, molecular analyses of drug resistance genes were not pursued in the cefotaxime-resistant isolates, which may be an important risk factor for outcome. However, in a Japanese multicenter study investigating the in vitro activities of cephamycins toward ESBL- and plasmid-mediated AmpC β-lactamase-producing Enterobacteriaceae, in which 574 third-generation cephalosporin-resistant clinical isolates were collected,26 Matsumura et al found that flomoxef ≥4 mg/L demonstrated the best screening performance for the detection of isolates with plasmid-mediated AmpC β-lactamase genes.26 These AmpC enzymes may hydrolyze cephamycin drugs, including flomoxef, leading to treatment failure.27
Conclusion
In this study, we found that patients with Enterobacteriaceae BSIs treated with flomoxef had a more favorable outcome when the flomoxef MICs of their isolates were ≤1 mg/L than when MICs were ≥2 mg/L. Thus, proper use of flomoxef in treating bacteremia caused by Enterobacteriaceae and continuous monitoring of the outcomes among patients infected by isolates with different MICs of this antibiotic are necessary.
Acknowledgments
We would like to thank Dr Chien-Ching Hung at the Department of Internal Medicine, National Taiwan University Hospital, Taipei, for his critical review of this manuscript. This study was partially supported by grants from Chang Gung Memorial Hospital, Taiwan (CMRPG 8H0491).
Disclosure
The authors report no conflicts of interest in this work.
References
Kang CI, Kim SH, Park WB, et al. Bloodstream infections caused by Enterobacter species: predictors of 30-day mortality rate and impact of broad-spectrum cephalosporin resistance on outcome. Clin Infect Dis. 2004;39(6):812–818. | ||
Okeke IN, Laxminarayan R, Bhutta ZA, et al. Antimicrobial resistance in developing countries. Part I: recent trends and current status. Lancet Infect Dis. 2005;5(8):481–493. | ||
Rossolini GM, D’Andrea MM, Mugnaioli C. The spread of CTX-M-type extended-spectrum beta-lactamases. Clin Microbiol Infect. 2008;14 Suppl 1:33–41. | ||
Cheng WL, Hsueh PR, Lee CC, et al. Bacteremic pneumonia caused by extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae: Appropriateness of empirical treatment matters. J Microbiol Immunol Infect. 2016;49(2):208–215. | ||
Vardakas KZ, Tansarli GS, Rafailidis PI, Falagas ME. Carbapenems versus alternative antibiotics for the treatment of bacteraemia due to Enterobacteriaceae producing extended-spectrum β-lactamases: a systematic review and meta-analysis. J Antimicrob Chemother. 2012;67(12):2793–2803. | ||
Nordmann P, Naas T, Poirel L. Global spread of Carbapenemase-producing Enterobacteriaceae. Emerg Infect Dis. 2011;17(10):1791–1798. | ||
Guh AY, Limbago BM, Kallen AJ. Epidemiology and prevention of carbapenem-resistant Enterobacteriaceae in the United States. Expert Rev Anti Infect Ther. 2014;12(5):565–580. | ||
Lee CH, Su LH, Chen FJ, et al. Comparative effectiveness of flomoxef versus carbapenems in the treatment of bacteraemia due to extended-spectrum β-lactamase-producing Escherichia coli or Klebsiella pneumoniae with emphasis on minimum inhibitory concentration of flomoxef: a retrospective study. Int J Antimicrob Agents. 2015;46(6):610–615. | ||
Lee CH, Su LH, Tang YF, Liu JW. Treatment of ESBL-producing Klebsiella pneumoniae bacteraemia with carbapenems or flomoxef: a retrospective study and laboratory analysis of the isolates. J Antimicrob Chemother. 2006;58(5):1074–1077. | ||
Matsumura Y, Yamamoto M, Nagao M, et al. Multicenter retrospective study of cefmetazole and flomoxef for treatment of extended-spectrum-β-lactamase-producing Escherichia coli bacteremia. Antimicrob Agents Chemother. 2015;59(9):5107–5113. | ||
Yang CC, Li SH, Chuang FR, et al. Discrepancy between effects of carbapenems and flomoxef in treating nosocomial hemodialysis access-related bacteremia secondary to extended spectrum beta-lactamase producing Klebsiella pneumoniae in patients on maintenance hemodialysis. BMC Infect Dis. 2012;12:206. | ||
Saito A, Kato Y, Ishikawa K, et al. 6315-S (Flomoxef). Chemotherapy. 1987;35:S523–S540. Japanese. | ||
Chen IL, Lee CH, Su LH, Tang YF, Chang SJ, Liu JW. Antibiotic consumption and healthcare-associated infections caused by multidrug-resistant gram-negative bacilli at a large medical center in Taiwan from 2002 to 2009: implicating the importance of antibiotic stewardship. PLoS One. 2013;8(5):e65621. | ||
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383. | ||
Chow JW, Yu VL. Combination antibiotic therapy versus monotherapy for gram-negative bacteraemia: a commentary. Int J Antimicrob Agents. 1999;11(1):7–12. | ||
Battle SE, Bookstaver PB, Justo JA, Kohn J, Albrecht H, Al-Hasan MN. Association between inappropriate empirical antimicrobial therapy and hospital length of stay in Gram-negative bloodstream infections: stratification by prognosis. J Antimicrob Chemother. 2017;72(1):299–304. | ||
Brun-Buisson C, Doyon F, Carlet J, et al. Incidence, risk factors, and outcome of severe sepsis and septic shock in adults. A multicenter prospective study in intensive care units. French ICU Group for Severe Sepsis. JAMA. 1995;274(12):968–974. | ||
Retamar P, Portillo MM, López-Prieto MD, et al; SAEI/SAMPAC Bacteremia Group. Impact of inadequate empirical therapy on the mortality of patients with bloodstream infections: a propensity score-based analysis. Antimicrob Agents Chemother. 2012;56(1):472–478. | ||
Gilbert DN, Moellering RC, Eliopoulos GM, Chambers HF, Saag MS, editors. Selected pharmacologic features of antimicrobial agents. In: The Sanford Guide to Antimicrobial Therapy. Sperryville, VA: Antimicrobial Therapy; 2009:78–82. | ||
Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing: 20th informational supplement, CLSI document M100-S20. Wayne, PA: CLSI; 2010. | ||
Lee CH, Chen IL, Li CC, Chien CC. Clinical benefit of ertapenem compared to flomoxef for the treatment of cefotaxime-resistant Enterobacteriaceae bacteremia. Infect Drug Resist. 2018;11:257–266. | ||
Ito A, Tatsumi YM, Wajima T, Nakamura R, Tsuji M. Evaluation of antibacterial activities of flomoxef against ESBL producing Enterobacteriaceae analyzed by Monte Carlo simulation. Jpn J Antibiot. 2013;66(2):71–86. | ||
Retamar P, Portillo MM, López-Prieto MD, et al. Impact of inadequate empirical therapy on the mortality of patients with bloodstream infections: a propensity score-based analysis. Antimicrob Agents Chemother. 2012;56(1):472–478. | ||
Turnidge J, Paterson DL. Setting and revising antibacterial susceptibility breakpoints. Clin Microbiol Rev. 2007;20(3):391–408. | ||
Craig WA. Interrelationship between pharmacokinetics and pharmacodynamics in determining dosage regimens for broad-spectrum cephalosporins. Diagn Microbiol Infect Dis. 1995;22(1-2):89–96. | ||
Matsumura Y, Yamamoto M, Nagao M, Tanaka M, Takakura S, Ichiyama S. In vitro activities and detection performances of cefmetazole and flomoxef for extended-spectrum β-lactamase and plasmid-mediated AmpC β-lactamase-producing Enterobacteriaceae. Diagn Microbiol Infect Dis. 2016;84(4):322–327. | ||
Lee CH, Chu C, Liu JW, Chen YS, Chiu CJ, Su LH. Collateral damage of flomoxef therapy: in vivo development of porin deficiency and acquisition of blaDHA-1 leading to ertapenem resistance in a clinical isolate of Klebsiella pneumoniae producing CTX-M-3 and SHV-5 beta-lactamases. J Antimicrob Chemother. 2007;60(2):410–413. |
Supplementary material
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.