Back to Journals » Cancer Management and Research » Volume 11
Radiation therapy-induced reactive oxygen species specifically eliminates CD19+ IgA+ B cells in nasopharyngeal carcinoma
Authors Li W, Wang L , Shen C, Xu T, Chu Y, Hu C
Received 21 January 2019
Accepted for publication 6 June 2019
Published 8 July 2019 Volume 2019:11 Pages 6299—6309
DOI https://doi.org/10.2147/CMAR.S202375
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Ahmet Emre Eşkazan
Weiwei Li,*,1,2 Luman Wang*,3, Chunying Shen1,2, Tingting Xu1,2, Yiwei Chu,3 Chaosu Hu1,2
1Department of Radiation Oncology, Fudan University Shanghai Cancer Center, Shanghai, People’s Republic of China; 2Department of Oncology, Shanghai Medical College, Fudan University, Shanghai, People’s Republic of China; 3Department of Immunology, School of Basic Medical Sciences, and Institute of Biomedical Sciences, Fudan University, Shanghai 200032, People’s Republic of China
*These authors contributed equally to this work
Purpose: Nasopharyngeal carcinoma (NPC) is one of the most common head and neck cancers and is thought to be related to the mucosal immune system. Radiation therapy (RT) is the primary treatment for NPC due to the high radiosensitivity of cancer cells. However, little is known about the impact of RT on the mucosal immune system.
Patients and methods: In this study, the expression of immune markers CD19, CD24, CD27, CD8, and IgA before and after RT, were analyzed using flow cytometry. Cytokines were assessed using the enzyme-linked immunosorbent assay. Reactive oxygen species (ROS) was assayed by flow cytometry and fluorescence staining using 2ʹ,7ʹ -dichlorofluorescein diacetate.
Results: We found that primary NPC patients had a significant increase in CD19+CD138−IgA+, B cells, which was then decreased after RT. Interestingly, the changes in CD19+CD138−IgA+, B cell frequency was accompanied by corresponding frequency changes in cytotoxic T cells (CTL), which are powerful anti-tumor lymphocytes. Mechanistically, we found that ROS release during RT specifically eliminated CD19+CD138−IgA+ B cells.
Conclusion: These findings suggest that RT may regulate the immune system and opens up new avenues for the utilization of immune-radiotherapy in NPC.
Keywords: B cells, IgA, nasopharyngeal carcinoma, radiation therapy, regulatory immune cells
Introduction
Nasopharyngeal carcinoma (NPC) is associated with the Epstein-Barr virus (EBV) and is characterized by epistaxis, nasal obstruction and discharge, and headache.1 NPC has a marked geographic distribution, where it is endemic to North Africa, Southern Asia and southern China.2 Due to its anatomic characteristics and highly sensitivity to radiation therapy (RT), NPC is generally treated with RT but rather than with surgery.3,4 The mechanism of RT for tumor treatment is complex. Studies have demonstrated that high dose irradiation directly breaks double-stranded DNA in tumor cells triggering apoptosis.5,6 On the contrary, low dose irradiation leads to the release of damage-associated molecular pattern molecules, cytokines and reactive oxygen species (ROS) from tumor cells which may interact with immune cells.7–9 However, the specific immune responses elicited after RT treatment in patient with NPC as well as its relationship with prognosis remains unknown.
Nasopharyngeal-associated lymph reticular tissue is a major mucosal inductive site, and has mucosal immune characteristics, such as high expression levels of secreted IgA (sIgA), which protects against EBV in NPC patients.10 However, the function of IgA+ B cells in the cancer microenvironment remains controversial. Several studies revealed that in addition to sIgA producing CD19−IgA+ plasma cells, there is another subset of CD19+CD138−IgA+ B cells that serve as regulatory B cells (Breg) of the mucosal immune system during cancer progression.11,12 Human Breg cells are usually identified as CD19 positive, CD24 high and CD27 positive (CD19+CD24hiCD27+).High numbers of these cells are found in the peripheral blood mononuclear cells (PBMC) of patients with autoimmune diseases, where they play a suppressive role.13,14 Thus, CD19+CD138−IgA+ Bcells, CD19+CD24hiCD27+ Breg, along with other cell subsets, including regulatory T cells (Tregs) may act as suppressive immune cells that influence the NPC prognosis. To date, the role of Bregs in NPC is poorly understood.
ROS is an electrophilic chemical species with a short half-life and is generated by incomplete oxidation. It has long been implicated as a secondary messenger involved in cell signaling activation and cell fate decisions.15,16 ROS can control B cell development, activation and apoptosis through activation of the B cell receptor or caspases signaling pathways.17–19 In the present study, we demonstrated increased percentages of CD19+CD138−IgA+ B cells in the PBMC of NPC patients where they played a suppressive function. Additionally, we showed that RT triggered the release of ROS into the tumor microenvironment, which led to the elimination of IgA+ B cells and increased cytotoxic T cell (CTL) populations, potentially augmenting the anti-tumor effect.
Materials and methods
Patients and controls
Peripheral blood was obtained from 38 primary NPC patients (mean, 47.8years; range, 15–64 years). Of those, 24 patients had blood recollected after RT. All patients had pathologically confirmed NPC and had not received prior treatment. Table 1 summarizes the characteristics of NPC patients. All patients were staged using the 7th edition of the Staging System of the American Joint Committee of Cancer. The study was approved by the medical ethics council of Fudan University Shanghai Cancer Center. Informed consent was obtained from all patients and healthy volunteers recruited in this study.
 |
Table 1 Clinical characteristics in 38 patients with NPC |
Cell isolation and flow cytometry
PBMCs were isolated using discontinuous Lymphoprep (Axis-ShieldPoCAs, Oslo, Norway) gradient centrifugation and were re-suspended in phosphate-buffered saline (PBS) containing 2% fetal bovine serum. Isolated PBMCs were collected by centrifugation, incubated with Fc-blocking antibody (purified anti-human CD16/32, BioLegend, San Diego, CA, USA) to prevent nonspecific binding and then incubated with the following antibodies (CD19: CF506236, BD Biosciences, CA, USA, 1:50; CD24: BDB563371, BD Biosciences, 1:50; CD27: BDB340424, BD Biosciences, 1:50; CD8: BDB564805, BD Biosciences, 1:50; IgA: ab193189, Abcam, CA, USA, 1:50) for 30 min on ice. After staining, cells were analyzed using CyAn-ADP (Beckman Coulter). Intracellular cytokine staining was performed by using the stimulation cocktail (eBioscience, San Diego, CA, USA) pre-stimulated for three hours as the manufacturer’s recommended protocol. Samples were analyzed by CyAn-ADP (Beckman Coulter) and data were processed using Flow Jo software (TreeStar, Ashland, OR).
Enzyme-linked immunosorbent assay (ELISA)
The serum and supernatant of radiated PBMCs were obtained, and the level of IgA, IL-10, TGF-β, TNF-α, IFN-γ and granzyme B were measured using Human Ready-SET-Go (eBioscience) according to the manufacturer’s protocol. A microplate reader (Tecan Infinite 200 PRO) was used to measure optical density at 450 nm.
In vitro ionizing radiation
Isolated PBMCs were further purified using the human pan B cell kit(StemCell Technologies, Vancouver, BC, Canada). Briefly, purified B cells were placed into 6cm diameter petri-dishes, and exposured to 4-Gy of ionizing radiation. In the diphenyleneiodonium chloride (DPI) group, cells were pretreated with 1 mM DPI30 min prior to exposure to ionizing radiation. After 18 hrs, the supernatant and cells were harvested for measurement of Cell viability using trypan blue staining.
Flow cytometry analysis of ROS
Intracellular accumulation of ROS was examined by flow cytometry after staining with the fluorescent probe, DCFH-DA (2ʹ,7ʹ-dichlorodihydro-fluorescein diacetate, 10 μM; Sigma-Aldrich). Briefly, monocytes were seeded in 24-well plates (1x106 cells/ml) and pretreated with 10 μM DCFH-DA for 30 min at 37 °C. Cells were then washed, re-suspended in PBS and prepared for further CD19 and IgA staining.
Statistical analysis
Two group comparisons were performed using Student’s t-test. Multiple group comparisons were performed using one-way analysis of variance, followed by Bonferroni correction or Mann-Whitney U testing to compare two individual groups. Statistical analysis was performed using Prism 6.0 (La Jolla, CA). The P-values of 0.05 were considered statistically significant.
Results
Enumeration and function of CD19+CD138−IgA+ B cells in healthy controls and NPC patients pre- and post-RT
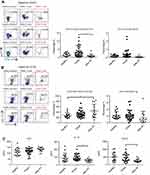
We previously demonstrated that CD19+CD138−IgA+ B cells (Plasma cells) have a suppressive role in colorectal tumors and are associated with poor prognosis.12 Due to the relation of NPC to the mucosal immune system, we assessed the CD19+IgA+ B cells in the PBMCs of patients. As shown in Figure 1A, CD19+CD138−IgA+ B cells were significantly increased in primary NPC blood cells (onset), and were subsequently decreased to a level below the control after RT. We then assessed serum cytokine expressions and found that the sIgA levels did not change with RT treatment; however, the production of TGF-β and IL-10 increased before RT and subsequently decreased to the control level after RT (Figure 1C), This trend is consistent with changes in cytokines. The percentage of CD19+IgA− cells were not affected on the other hand (Figure S1). Conventionally, Bregs (refers to CD19+CD24hiCD27+ B cells) were assessed after treatment; thus, we detected this B cell subset in our cell populations. Intriguingly, we found that CD19+CD24hiCD27+ B cells were remarkably increased after RT, in some cases resulting in an 80% CD19+CD24hiCD27+B cell population (Figure 1B).Conventional CD19+CD24hiCD27+ Breg cells do not share similar features with IgA+ B cells within the mucosal immune system and likely have inconspicuous functions in NPC. We also detected other suppressive cytokine producing cells such as Tregs. As with Bregs, the change of Tregs surprisingly increased after RT (Figure S2); however, this trend is not consistent with changes in cytokines. Collectively, we found that the PMBC CD19+CD138−IgA+B cells, rather than conventional Bregs and Tregs, may play a suppressive role in NPC onset and are specifically targeted by RT.
Changes in the percentage changes of CTLs and other immune effector cells
Next, we assessed the changes in the percentage of other immune effector cells. As shown in Figure 2A and B, the proportion of CD8+ Granzyme B (GB)+/CD8, CD8+IFN-ɣ+/CD8 and CD8+TNF-α+/CD8 were significantly increased in primary NPC (before), indicating that CD8 cells differentiate to anti-tumor CTLs after cancer onset. Importantly, the percent of CTL/CD8 cells increased to a greater extent after RT and was negatively correlated with the percentage of CD19+CD138−IgA+ B cells (Figure 2D). Secreted levels of Granzyme B, IFN-ɣ and TNF-α were detected by ELISA, and results demonstrate upregulation of IFN-ɣ and Granzyme B after RT (Figure 2C). We also detected minimal changes in the percentage of other anti-tumor effector cells, such as natural killer cells and helper T cells (Figure S3). Based on these findings, we assumed that serum cytokine changes might be caused by changes in the expression of CD19+CD138−IgA+B cell.
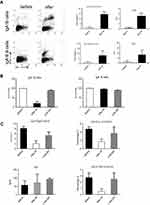
RT induced ROS eradicates CD19+CD138−IgA+ B cells in vitro
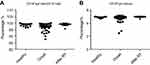
We investigated the mechanism responsible for CD19+CD138−IgA+ B cell elimination following RT. As ROS were reported to be released by the ionizing radiation (IR) used in RT, we evaluated ROS production in the peripheral blood of patients before and after RT and found increased levels following radiation therapy (Figure S4). Next, we exposed petri-dishes containing PMBC to 4Gy ionizing radiation, and detected ROS expression inside B cells and in the microenvironment. After in vitro ionizing radiation, the immune cells and their supernatants were harvested to assess ROS levels. As shown in Figure 3A, ROS was found in the supernatant and the cytoplasm of CD19+CD138−IgA+ B cells (IgA+ B cells). To confirm this result, cells were pretreated with ROS scavenging agent DPI and subsequently irradiated with 4 Gy of ionizing radiation. After 18 hrs, B cells were analyzed for apoptosis and function. As expected,CD19+CD138−IgA+ B cells were more prone to apoptosis after ionizing radiation, while most of the CD19+IgA−B cells (IgA− B cells) remained alive. Treatment with DPI reversed this phenomenon (Figure 3B). The number of CD19+CD138−IgA+ B cells decreased after exposure to IR; however, no changes were observed when cells were pretreated DPI. Intriguingly, levels of IL-10 and TGF-β produced from IgA+ cells were down-regulated after irradiation, indicating an impaired function of CD19+CD138−IgA+ B cells following exposure to ionizing radiation. However, the functions of these cells were unaffected when pretreated with DPI (Figure 3C). Levels of sIgA did not change following radiation treatment, indicating that production of sIgA is not the main function of IgA+ B cells in cancer (Figure 3C). Taken together, our data demonstrate that RT-induced ROS eliminates CD19+CD138−IgA+ B cells and results in an increased anti-tumor immune response in NPC.
Discussion
In this study, we illustrated the suppressive function of immune cells in NPC patients and determined the changes in the immune cell profile before and after RT. We found that during the onset of NPC, the percentage of CD19+CD138−IgA+ B cells and its cytokines was increased, and these numbers were subsequently decreased after RT. The percentage of other suppressive immune cells remained unchanged. Meanwhile, CTLs were slightly up-regulated during disease onset and were further increased by RT. Moreover, we observed that irradiation-induced ROS release specifically targeted CD19+CD138−IgA+ B cells.
B cells play an important suppressive role in mucosal-associated lymphoid tissues, particularly in NPC, as it is a mucosal immune-related cancer. The IgA+ plasma cells suppress inflammation by secreting anti-microbial antibodies, whereas CD19+CD138−IgA+ cells negatively regulate anti-tumor effectors by expressing PD-1 and producing TGF-β.12,20 Thus, we first detected CD19+CD138−IgA+ cells and other efficiency regulatory cells such as Tregs in patients’ PMBC. We have previously indicated that CD138− plasma cells, rather than CD138+ antibody secreting cells, play a role in mucosal immunity. Thus, we used CD138 to distinguish the subsets. The results are consistent with our previous findings, whereby CD19+CD138−IgA+ B cells increased in primary cancer microenvironment. Moreover, the anti-NPC treatment effectively eliminated this subset, indicating a possible innovative mechanism for RT used in NPC treatment. Notably, this trend was consistent with changes in cytokine expression. Thus, it can be suggested that the serum cytokine changes may be caused by changes in the expression of CD19+CD138−IgA+B cells. Furthermore, we excluded the CD19+CD24hiCD27+ Breg subset, which was involved in defining populations of B cells in this study.14
Interestingly, our current results showed that Breg cells are not involved in the RT process, despite being the only B cell subset that survived after RT. We are currently working on possible explanations of this phenomenon. Conversely, Tregs are important for suppression of anti-tumor immunity, thus were considered for analysis in this study. Results indicate that the expression of Tregs was similar to that of Bregs. In addition, we found that the expression of myeloid-derived suppressor cells remained unchanged before and after RT treatment (data not shown), indicating that they are not related to the mechanisms of radiotherapy in nasopharyngeal cancer.
CTLs are the critical effector cells in tumor immunity, and we have previously demonstrated that they can be utilized as an efficient means to treat tumors. Therefore, we sought to determine whether the decrease of CD19+CD138−IgA+ B cells after RT was accompanied by an increase of CTL cells. It was found that the proportion of CTL cells increased, along with the secretion of cytokines and particles. Moreover, the expression of CTL was inversely proportional to the expression of CD19+CD138−IgA+ Bcells, suggesting a possible correlation between the two, which will require further investigation.
RT is used to directly or indirectly eliminate cancer cells. One of the ways in which cancer can be targeted indirectly is through the release of ROS.7 Tominaga et al found that gene instability in tumor cells caused by RT could be reduced using various anti-ROS methods.21 Studies by Wang et al have shown that cancer cells can utilize changes in ROS metabolism activity to resist radiation therapy. In addition, Danhier et al outlined that cancer cells can perform a series of actions to achieve the following goals: produce sufficient energy to promote cell proliferation (biological energetics), generate debris (biosynthesis), and generate a reduction of molecular pool from oxidative stress.22–24 Most importantly, many studies have found that ROS generated by RT can lead to changes in the immune microenvironment.25 We observed that after ionizing radiation, ROS could enter the cytoplasm of all PBMCs; however, only CD19+CD138−IgA+ B cells were targeted and eliminated through decreases in TGF-β and IL-10. This is a novel and meaningful finding, as it suggests that CD19+CD138−IgA+ B cells are likely the target of RT during NPC treatment, and that these cells play a critical role in the suppression of anti-tumor immunity. Collectively, these results confirm that RT-induced ROS eliminates CD19+CD138−IgA+ B cells and results in an increased anti-tumor immune response in NPC, potentially providing a novel approach to tumor therapy.
Patients and controls
All participants, and a parent or legal guardian of participants under the age of 18 years, had written informed consent for donating their samples to Fudan University Shanghai Cancer Center.
Acknowledgments
This work was supported by the General Program of National Natural Science Founds of China (81771736) and the Young Elite Scientists Sponsorship Program by CAST (YESS20160094). The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Author contributions
L.W. and W.L. designed and performed the experiments, analyzed the data, and wrote the manuscript. H.C. and C.Y. designed the experiment and wrote the manuscript. S.C. and X.T. performed the experiments and analyzed the data. L.W. and W.L. contributed equally to this work. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accounted for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Wu L, Li C, Pan L. Nasopharyngeal carcinoma: a review of current updates. Exp Ther Med. 2018;15:3687–3692. doi:10.3892/etm.2018.5878
2. Peng H, Tang -L-L, Liu X, et al. Anti-EGFR therapy concurrently with induction chemotherapy in locoregionally advanced nasopharyngeal carcinoma. Cancer Sci. 2018;109:1609–1616. doi:10.1111/cas.13589
3. Hu J, Kong L, Gao J, Hu W, Guan X, Lu JJ. Use of radiation therapy in metastatic nasopharyngeal cancer improves survival: a SEER analysis. Sci Rep. 2017;7:721. doi:10.1038/s41598-017-00655-1
4. Toya R, Murakami R, Saito T, et al. Radiation therapy for nasopharyngeal carcinoma: the predictive value of interim survival assessment. J Radiat Res. 2016;57:541–547. doi:10.1093/jrr/rrw038
5. Baskar R, Dai J, Wenlong N, Yeo R, Yeoh KW. Biological response of cancer cells to radiation treatment. Front Mol Biosci. 2014;1:24. doi:10.3389/fmolb.2014.00024
6. Thoms J, Bristow RG. DNA repair targeting and radiotherapy: a focus on the therapeutic ratio. Semin Radiat Oncol. 2010;20:217–222. doi:10.1016/j.semradonc.2010.06.003
7. Renschler MF. The emerging role of reactive oxygen species in cancer therapy. Eur J Cancer. 2004;40:1934–1940. doi:10.1016/j.ejca.2004.02.031
8. Jeong H, Bok S, Hong B-J, Choi H-S, Ahn G-O. Radiation-induced immune responses: mechanisms and therapeutic perspectives. Blood Res. 2016;51:157–163. doi:10.5045/br.2016.51.3.157
9. Park B, Yee C, Lee KM. The effect of radiation on the immune response to cancers. Int J Mol Sci. 2014;15:927–943. doi:10.3390/ijms15010927
10. Teow SY, Liew K, Khoo AS, Peh SC. Pathogenic role of exosomes in Epstein-Barr Virus (EBV)-associated cancers. Int J Biol Sci. 2017;13:1276–1286. doi:10.7150/ijbs.19531
11. Shalapour S, Font-Burgada J, Di Caro G, et al. Immunosuppressive plasma cells impede T-cell-dependent immunogenic chemotherapy. Nature. 2015;521:94–98. doi:10.1038/nature14395
12. Liu, R, Lu Z, Gu J, et al. MicroRNAs 15A and 16–1 activate signaling pathways that mediate chemotaxis of immune regulatory B cells to colorectal tumors. Gastroenterology. 2017. doi:10.1053/j.gastro.2017.09.045
13. Iwata Y, Matsushita T, Horikawa M, et al. Characterization of a rare IL-10-competent B-cell subset in humans that parallels mouse regulatory B10 cells. Blood. 2011;117:530–541. doi:10.1182/blood-2010-07-294249blood-2010-07-294249
14. Zha B, Wang L, Liu X, et al. Decrease in proportion of CD19+ CD24(hi) CD27+ B cells and impairment of their suppressive function in Graves’ disease. PLoS One. 2012;7:e49835. doi:10.1371/journal.pone.0049835PONE-D-12-18131
15. Rhee SG. Cell signaling. H2O2, a necessary evil for cell signaling. Science. 2006;312:1882–1883. doi:10.1126/science.1130481
16. Van Acker H, Coenye T. The role of reactive oxygen species in antibiotic-mediated killing of bacteria. Trends Microbiol. 2017;25:456–466. doi:doi:10.1016/j.tim.2016.12.008
17. Park GB, Choi Y, Kim YS, Lee H-K, Kim D, Hur DY. ROS-mediated JNK/p38-MAPK activation regulates bax translocation in Sorafenib-induced apoptosis of EBV-transformed B cells. Int J Oncol. 2014;44:977–985. doi:10.3892/ijo.2014.2252
18. Polikowsky HG, Wogsland CE, Diggins KE, Huse K, Irish JM. Cutting edge: redox signaling hypersensitivity distinguishes human germinal center B cells. J Immunol. 2015;195:1364–1367. doi:10.4049/jimmunol.1500904
19. Richards SM, Clark EA. BCR-induced superoxide negatively regulates B-cell proliferation and T-cell-independent type 2 Ab responses. Eur J Immunol. 2009;39:3395–3403. doi:10.1002/eji.200939587
20. Sun R, Wang X, Li X. Correlation analysis of nasopharyngeal carcinoma TNM staging with serum EA IgA and VCA IgA in EBV and VEGF-C and -D. Med Sci Monit. 2015;21:2105–2109. doi:doi:10.12659/MSM.893415
21. Tominaga H, Kodama S, Matsuda N, Suzuki K, Watanabe M. Involvement of reactive oxygen species (ROS) in the induction of genetic instability by radiation. J Radiat Res. 2004;45:181–188. doi:10.1269/jrr.45.181
22. Sonveaux P. ROS and radiotherapy: more we care. Oncotarget. 2017;8:35482–35483. doi:doi:10.18632/oncotarget.16613
23. Danhier P, Bański P, Payen VL, et al. Cancer metabolism in space and time: beyond the Warburg effect. Biochim Biophys Acta Bioenerg. 2017;1858:556–572. doi:10.1016/j.bbabio.2017.02.001
24. Wang, H., Bouzakoura S, De Mey S, et al. Auranofin radiosensitizes tumor cells through targeting thioredoxin reductase and resulting overproduction of reactive oxygen species. Oncotarget. 2017;8:35728–35742. doi:10.18632/oncotarget.16113
25. Ozsoy HZ, Sivasubramanian N, Wieder ED, Pedersen S, Mann DL. Oxidative stress promotes ligand-independent and enhanced ligand-dependent tumor necrosis factor receptor signaling. J Biol Chem. 2008;283:23419–23428. doi:10.1074/jbc.M802967200
Supplementary materials
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.