Back to Journals » Infection and Drug Resistance » Volume 13
Pyrazinamide Resistance and Mutation Patterns Among Multidrug-Resistant Mycobacterium tuberculosis from Henan Province
Authors Shi J ![]() , Su R, Zheng D, Zhu Y, Ma X, Wang S, Li H, Sun D
, Su R, Zheng D, Zhu Y, Ma X, Wang S, Li H, Sun D
Received 7 May 2020
Accepted for publication 28 July 2020
Published 20 August 2020 Volume 2020:13 Pages 2929—2941
DOI https://doi.org/10.2147/IDR.S260161
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Suresh Antony
Jie Shi, Ruyue Su, Danwei Zheng, Yankun Zhu, Xiaoguang Ma, Shaohua Wang, Hui Li, Dingyong Sun
Henan Province Center for Disease Control and Prevention, Zhengzhou, Henan Province, People’s Republic of China
Correspondence: Jie Shi; Dingyong Sun Tel +86-15803829002
; +86-13703865596
Email [email protected]; [email protected]
Purpose: This study was designed to identify the phenotypic and genotypic characteristics of pyrazinamide (PZA) resistance among multidrug-resistant Mycobacterium tuberculosis (MDR-TB) from Henan and to evaluate the efficacy of pncA, rpsA, and panD mutations in predicting PZA resistance.
Materials and Methods: A total of 152 MDR strains were included in this study. The Bactec MGIT system was used to determine PZA susceptibility for all strains. The pncA, rpsA, and panD genes were sequenced to identify any mutations, and the sequences were then aligned with the sequence of standard strain H37Rv. Moreover, the correlations between PZA-resistant phenotypes and treatment outcomes were analysed.
Results: Of the152 strains, 105 had a PZA-resistant phenotype, and 102 harboured the pncA mutation. The PZA resistance rate was higher in the strains with resistance to all four first-line drugs and those that were pre-extensively drug-resistant (pre-XDR) and extensively drug-resistant (XDR). A total of 100 different pncA mutation patterns were identified, including 80 point mutations and 20 insertions/deletions, and 32 new pncA mutation patterns were detected. In this study, 13 strains had multiple mutations. Of the11 PZA-resistant strains without pncA mutations, two harboured the rpsA mutation, and one harboured the panD mutation. With PZA susceptibility results as the reference, single-gene pncA sequencing had sensitivity of 89.52% and specificity of 89.36%. With the combination of rpsA and panD, the sensitivity increased to 92.38%, and the specificity remained the same. No significant differences were observed in the sputum smear/culture conversion rate between PZA-resistant patients and PZA-sensitive patients. However, PZA resistance was related to the time to sputum smear/culture conversion (P = 0.018).
Conclusion: The combination of pncA, rpsA, and panD was beneficial for the timely diagnosis of PZA resistance and could provide a laboratory basis for customizing treatment regimens for MDR-TB patients.
Keywords: Mycobacterium tuberculosis, multidrug-resistant, pyrazinamide, pncA, rpsA, panD
Introduction
Multidrug-resistant Mycobacterium tuberculosis (MDR-TB, resistant to at least two of the most powerful first-line anti-TB drugs, isoniazid and rifampin) remains a critical global public health problem due to its high treatment failure rate.1–3 According to the latest 2019 report from the World Health Organization (WHO), there were approximately10millionnew cases of TB worldwide, and approximately 1.2 million HIV-negative patients died of tuberculosis. China has the second highest MDR-TB burden, after only India, with approximately 120,000 cases of MDR-TB per year. A national survey of drug-resistant tuberculosis in China showed that the incidence of TB is 5.7%, and 25.6% of retreated patients were infected with MDR-TB, significantly higher than global average level.4 The large number of patients with MDR-TB hinders the effective prevention and control of TB in China.5,6
Pyrazinamide (PZA) is an important first-line anti-TB drug, with effective bactericidal activity against both drug-sensitive and MDR strains. As a prodrug, PZA is converted by pncA-encoded pyrazinamidase (PZase) into pyrazinoic acid (POA) to exhibit bactericidal activities.7 The accumulation of POA in cells results in cytoplasmic acidification, which depletes cellular membrane potentials, inhibits various intracellular targets, and eventually leads to cell death.8 PZA is effective against persistent bacteria in macrophages, which cannot be killed by other anti-TB drugs. Due to its unique antibacterial activity, PZA reduces the treatment time from 12months to6monthswhen it is combined with isoniazid and rifampicin. Studies have shown that72-98%ofPZAresistance is due to pncA mutations,9–11 which are highly diverse and scattered in open reading frames and upstream regulatory regions.12 In addition to pncA mutations, other mechanisms can cause PZA resistance. For example, mutations in rpsA, which encodes 30S ribosomal S1 proteins, can alter the POA binding site, causing PZA resistance.13 Some studies have shown that panD is associated with PZA resistance.11 Researchers are still debating the role of rpsA and panD mutations in PZA resistance, and more experimental data are needed to clarify the contributions of rpsA and panD to PZA resistance.
PZA has antibacterial activity only at a low pH; thus, an acidic environment is needed for PZA susceptibility tests. Due to the complex procedures and the failure probability of PZA susceptibility tests, only a few laboratories perform these experiments.14 However, previous studies have shown that approximately16%of TB patients are PZA resistant. Specifically, the PZA resistance rate is 2–7.5% in non-MDR-TB patients and 36–85% in MDR-TB patients.15,16 Therefore, the reliable prediction of PZA resistance before treatment facilitates the development of more effective treatments. As the most populous region in China, Henan has the largest number of patients with TB and MDR-TB in the country, making it a hotspot for TB prevention. While PZA is widely used to treat MDR-TB patients, few studies have been conducted to investigate the prevalence of PZA resistance in MDR strains in Henan. In this study, we investigated PZA resistance in MDR-TB patients in Henan and analysed the mutation patterns of PZA resistance-related genes. Moreover, we observed the correlations between PZA resistance and treatment outcomes.
Materials and Methods
Sample Source
MDR-TB patients from 10 regions of Henan during 2018 were included in this study. The patients were followed upfor2years, with monthly sputum smears and Mycobacterium tuberculosis cultures during treatment. The study was approved by the Medical Ethics Committee of Henan Provincial Center for Disease Control and Prevention (Number: 2014-KY-012-01). All patients provided informed consent, and this study was conducted in accordance with the Declaration of Helsinki. We collected patient information from medical records. Retreated patients were defined as those receiving more than 1 month of irregular chemotherapy for tuberculosis before presenting at our hospitals.
Drug Susceptibility Testing (DST)
Drug susceptibility testing was performed with WHO-recommended proportion method. The drug concentrations in Lowenstein-Jensenmediumwere 0.2 μg/ml isoniazid (INH), 40 μg/ml rifampicin (RIF), 4μg/ml streptomycin (SM), 2 μg/ml ethambutol (EMB),30 μg/ml kanamycin (KAM), 30 μg/ml amikacin (AMK), 2 μg/ml levofloxacin (OFLX), 1 μg/ml p-aminosalicylic acid (PAS), 40 μg/ml capreomycin (CAM), and 40 μg/ml prothionamide (PTO).Resistant strains were considered when more colonies (>1%) were growing on the drug-containing medium than on the control medium. Bactec MGIT 960 was used for the PZA susceptibility test, and the concentration of PZA in the liquid medium was 100μg/mL. For strains with inconsistent results between DST and DNA sequencing, the tests were repeated, and the minimum inhibitory concentration (MIC) in liquid 7H9 medium was measured. Strains resistant to both isoniazid and rifampicin were defined as MDR strains. In addition to MDR, strains resistant to levofloxacin and at least one second-line injectable anti-tuberculosis drug (amikacin, capreomycin, or kanamycin) were considered extensively drug-resistant (XDR) strains.
DNA Extraction and PCR (Review 1), Sequencing and Characterization of Mutations in pncA, rpsA and panD (Review 2)
Fresh cultured bacteria on Lowenstein-Jensen medium were scraped into500 μL of Tris-EDTA (TE) buffer, inactivated at 85°C for30min, boiled for 5min, and then centrifuged for later use.17 Crude DNA extract was used as the template for polymerase chain reaction (PCR). The primers were as follows: pncA-5ʹ-AACAGTTCATCCCGGTTC and pncA-3ʹ-GCGTCATGGACCCTATATC;rpsA-5ʹ-CGGAGCAACCCAACAATA and rpsA-5ʹ-GTGGACAGCAACGACTTC; and panD-5ʹ-TCAACGGTTCCGGTCGGCTGCT and panD-3ʹ-TATCCGCCACTGCTGCACGACCTT. The 20μL PCR mixture was prepared as follows: 10 μl of 2 × GoldStar MasterMix (CWBIO Biotech Company, China), 5 μL of DNA template, and 0.2 μM of each primer set. The PCR conditions for amplification were 5 min at 94°C, followed by 35 cycles of 94°C for 1 min, 59°C for 1 min, 72°C for 1 min, and a final extension of 72°C for 10 min. The PCR products were sent to Shanghai Sangon Biotech Company for sequencing. All of the sequence results were aligned with the pncA, rpsA, and panDgenes from reference strain H37Rv (GenBank accession no. NC000962) using DAMAN(version6.0) software.
Genotyping
Spoligotyping analysis was performed in accordance with the previous literature.18 Briefly, the primers DR3ʹ-CCGAGAGGGGACGGAAAC and DR5ʹ-GGTTTTGGGTCTGACGAC were used for PCR of the extracted genome samples, and then the PCR products were hybridized with a membrane precoated with 43 spacer oligonucleotides to determine the results. The spoligotyping results were aligned through the SITVITWEB website (http://www.pasteur-guadeloupe.fr:8081/SITVIT_ONLINE/). The primersNTF-5ʹ-CCAGATATCGGGTGTGTCGACandNTF-3ʹ-TGCCGTTCTCGAAATCTAAACAA were designed based on the NTF (nuclear transcription factor) region of M. tuberculosis.19 The strain was defined as “modern-type” in the presence of the IS6110 insertion or “ancient type” in the absence of theIS6110 insertion according to a previous report.20
Data Analysis
SPSS software, version 19.0, was used for data analysis, and the chi-square test was performed to analyse the correlations between categorical variables.P<0.05 was considered statistically significant.
Results
PZA Susceptibility Testing and Patients Demographics
In this study, a total of 152 MDR-TB strains were included, and 105 (69.02%) were resistant to PZA. Moreover, 43 of the152 strains (28.29%) were pre-extensively drug resistant (pre-XDR), 37 (86.04%) were PZA resistant; and 25 (16.44%) were XDR, 23 (92%) were PZA resistant. In addition, 102 (71.71%) strains were resistant to all four first-line anti-tuberculosis drugs, and 82 (80.39%) were resistant to PZA. These results showed that pre-XDR strains, XDR strains, and strains resistant to all four first-line drugs were more likely to develop PZA resistance.
Of the 152 MDR strains, 119 (78.28%) were resistant to streptomycin, 85 (55.92%) were resistant to ethambutol, 35 (23.02%) were resistant to kanamycin, 63 (41.44%) were resistant to levofloxacin, 35 (23.02%) were resistant to amikacin, 25 (16.45%) were resistant to capreomycin, 23 (15.13%) were resistant to prothionamide, and 15 (9.86%) were resistant to p-aminosalicylic acid (Table 1). MDR strains that were resistant to ethambutol, streptomycin, and levofloxacin were more likely to develop PZA resistance.
 |
Table 1 PZA Resistance in Different Groups of MDR-TB Patients |
We further analysed the correlation between PZA resistance and clinical information, including sex, age, close contact history with tuberculosis patients, previous treatment, and tuberculosis genotype. The results showed that the PZA resistance rate was significantly higher among men than women (odds ratio (OR): 2.64, 95% confidence interval (CI): 1.29–5.40, P= 0.009), and the percentage of retreated MDR-TB patients in the PZA-resistant group was significantly higher than in the PZA-susceptible group (OR: 4.07,95% CI: 1.92–8.83, P<0.001). Analysis of the correlations between the genotypes of these strains and PZA resistance showed that the Beijing family had a higher frequency of PZA resistance than other genotypes and that modern-type strains were more likely to develop PZA resistance than ancient type strains.
Mutations in MDR Strains
The pncA genes of 152 strains were sequenced and analysed, and the data showed that the PZA susceptibility results were consistent with the pncA mutation for 133 strains. Eight PZA-susceptible strains harboured pncA mutations, including three synonymous mutations. Eleven strains were resistant by the test of MGIT960 but harboured no pncA mutations. As a result, with PZA susceptibility phenotype as the gold standard, pncA mutation detection showed sensitivity of 89.52% (95% CI, 81.64–94.39%) and specificity of 89.36% (95% CI, 76.10–96.01%) in predicting PZA resistance.
Of the 152 MDR strains, 50 harboured no mutations, and102 harboured pncA mutations, of which 82 strains had base mutations (including three nonsense mutations and three synonymous mutations), and 20 had frame shift mutations. Moreover, 13 strains harboured multiple mutations, including double mutations in eight strains, triple mutations in three strains, and four mutations in two strains. The remaining 89 strains harboured a single pncA mutation, either a single base mutation or a synonymous mutation (Table 2).
 |
Table 2 Correlation between pncA Mutation Patterns and PZAResistance |
Different mutation patterns were detected in these resistant strains, including 14 insertions, 6 deletions, and 80 point mutations. These mutations were scattered throughout the entire length of the pncA gene, including the promoter region. Seven strains harboured a mutation in the promoter region of pncA, including an insertion of Gat locus −7 for one strain, a T to C nucleotide substitution at locus −11 for five strains, and an A to G transition at locus −12 for one strain. For the remaining 95 strains, pncA mutations were found in the open reading frame. Three strains harboured a large segment deletion, including deletions of codons 79 to 92, codons 189 to 218, and codons 196 to 221. In this study, we detected 32 new mutations that were not found in the GMTV and TBDReaMDB databases or reported in previous studies, including 16 substitution mutations and16 frame shift mutations (Table 3).
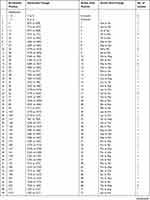 |  |  |
Table 3 Mutant Profiles of MDR-TB Isolates Within pncA |
The mutations of pncA in MDR strains from Henan were diverse. The PZA-resistant strains harboured 100 different mutant types, nine of which were found in multiple strains (Table 3). Specifically, codon 359 was associated with the highest mutation rate, and its mutation (Leuto Arg) was found in eight strains (12.3%). Locus-11 of the pncA promoter was associated with the second highest mutation rate; this mutation was found in five strains. Codon 226 mutations (Thr to Pro) were found in four strains (6.8%). In addition, six mutant types were found in two drug-resistant strains.
Eight PZA-susceptible strains harboured pncA mutations, including three synonymous mutations. It should be noted that A to T substitution at codon 535 was found in both PZA-resistant and PZA-sensitive strains. PZA susceptibility tests and pncA sequencing were repeated for these strains, and the results remained the same. The MICs of PZA were determined with7H9 liquid medium (pH 5.5) for these eight strains. The results showed that the MIC was 12.5μg/mL for three strains with a synonymous mutation, 25 μg/mL for two trains, and 50μg/mL for three strains, which included the strain with a codon535 mutation (Table 4).
 |
Table 4 MIC of PZA-Susceptible Strains Harbouring pncA Mutation(s) |
Strains without pncA mutations could have other mechanisms for PZA resistance. Therefore, other genes associated with PZA resistance were sequenced, including rpsA and panD (Table 5). No mutations were detected in the promoter regions of rpsA and panD, and most PZA-resistant strains harbour in grpsA or panD mutations were also accompanied by pncA mutations. In this study, 72.4% (76/105) of PZA-resistant and 74.5% (35/47) PZA-susceptible clinical strains had eight and one point mutations within their rpsA genes, separately. RpsA mutations were scattered, although most mutations were located in the180amino acids of the N-terminus and C-terminus. Notably, no pncA mutations were detected in PZA-resistant strains harbouring rpsA codon357 and codon532 mutations. The CGA to CGC synonymous mutation in rpsA codon 212is a non-specific mutation that occurred in both the 68PZA-resistant strains and the 35PZA-susceptible strains. Spoligotyping analysis showed that 96 (93.2%) of the 103 strains with a synonymous rpsA mutation belonged to the Beijing family and that two strains belonged to the T family. The rpsA nonsynonymous mutation frequencies among PZA-resistant XDR strains and MDR strains were 8% (2/25) and 5.5% (6/109), respectively. The XDR strains had a greater tendency to harbour rpsA mutations than the MDR strains. No panD mutations were detected in the 47PZA-susceptible strains. Two of the 105PZAdrug-resistant strains harboured panD mutations, and one of these strains harboured no pncA mutations.
 |
Table 5 Mutant Profiles of rpsA and panD among MDR-TB Isolates |
To evaluate the efficiency of DNA sequencing for PZA resistance, we compared the sequencing results and the resistance phenotype (Table 6). With the resistance phenotype as a reference, detection of mutations in pncA alone showed sensitivity of 89.52% (95% CI, 81.64–94.39%) and specificity of89.36% (95% CI, 76.10–96.01%); the kappa was 0.76 between the pncA mutation and PZA resistance. The concordance between sequencing of rpsA/panD and PZA resistance was low, indicating a low diagnostic value for PZA resistance on its own. However, the combination of rpsA and panD mutations with pncA sequencing increased the sensitivity for PZA resistance from 89.52% (95% CI, 81.64–94.39%) to 92.38% (95% CI, 85.54–96.65%) and increased the kappa to 0.80.
 |
Table 6 Efficacy Evaluation of Sequencing for PZA Resistance in MDR Strains |
Relationship Between Treatment outcome and PZA Susceptibility Phenotype
To investigate the effect of PZA resistance on treatment outcome, we followed up the patients for 24 months to obtain clinical information. Nine patients failed to provide complete treatment information, including two deaths, two refusals to followup, and five losses. The data showed that after treatment, 93 (93.93%) of the 99 PZA-resistant patients showed sputum smear/culture conversion, while 42 (95.45%) of the 44PZA-susceptible patients showed sputum smear/culture conversion (Table 7). These results indicated no significant difference in sputum conversion rate between PZA-resistant and susceptible patients. However, when the sputum conversion time was segmented and compared with PZA resistance, the results showed that at the end of 6months of treatment, the sputum smear/culture conversion rate was 1.08% in PZA-resistant patients, which was significantly lower than that in PZA-susceptible patients (7.14%) (Table 7), indicating that PZA resistance could affect the time to sputum smear/culture conversion.
 |
Table 7 Correlation Between Treatment Outcome and Drug Susceptibility for PZA |
Discussion
PZA plays an important role in the treatment of patients with MDR-TB.21,22 In this study, we investigated PZA resistance in MDR-TB patients in Henan and analysed the correlations between PZA resistance and mutations in PZA resistance-related genes. The results showed that 69% (95% CI, 57.8–71.8%) of MDR strains were PZA resistant, which was similar to that in the Chongqing area (62.4%)23 and slightly higher than those in previous reports.24–27 This study showed that retreated MDR-TB patients were more likely to develop PZA resistance (78.0% vs 46.5%), suggesting a correlation between a history of tuberculosis and PZA resistance. The higher PZA resistance rate observed in retreated MDR-TB patients could be related to their previous treatment regimens with PZA. As a result, the role of PZA should be carefully considered in the treatment of MDR-TB patients. It was necessary to perform PZA susceptibility tests to develop an optimal treatment regimen for MDR-TB patients.
Previous reports have revealed a correlation between PZA resistance and fluoroquinolone resistance.28–30 Similarly, our study showed that PZA resistance was associated with resistance to other anti-tuberculosis drugs, including EMB, SM and OFLX. When exposed to antibiotics such as rifampicin, fluoroquinolones, and aminoglycosides, bacteria will induce the generation of free oxygen radicals in the body and increase the frequency of gene mutations. Given the prolonged use of anti-tuberculosis drugs by MDR-TB patients, we hypothesized that prolonged drug exposure could induce more genetic mutations in M. tuberculosis, eventually leading to cross-resistance to PZA and other drugs.
The genotyping results showed that PZA resistance rates were significantly higher in the Beijing family of M. tuberculosis than in the non-Beijing family, and PZA resistance rates were higher in the modern subline ages than in the ancient sub line ages. In this study, approximately76.3% of Beijing family and 60% of T family strains harboured PZA resistance, the frequency was significantly higher than those among other genotypes (14.3%). However, due to the small sample size, we were unable to reach any definitive conclusions about correlations with M. tuberculosis genotypes, whereas previous studies have shown that PZA resistance was not associated with the genotype.31
In this study, the concordance rate was 91.4% between PZA-resistant phenotypes and pncA sequences, with discordant results for 13 strains. Eight strains with a pncA mutation, including three strains with a synonymous mutation, were susceptible to PZA, while eight strains with wild-type pncA were resistant to PZA. To exclude experimental errors, we repeated PZA susceptibility tests for these 13 strains and obtained the same results. The spoligotypes of 16 strains with inconsistent results between resistance and pncA gene mutations were analysed, and the results showed that 14 strains belonged to the Beijing genotype and two strains to the T family. We further determined the MICs of PZA-susceptible strains with pncA mutations. Except for three strains with a synonymous mutation, the remaining five strains showed low-level PZA resistance (12.5 μg/mL <MIC≤50μg/mL). Drug resistance is the result of the interactions of multiple macromolecules in organisms, including genes, transcripts, and proteins. In addition, some genetic mutations might not necessarily lead to a drug resistance phenotype.32,33
The pncA mutation rate in PZA-resistant strains varies among different regions, eg, 45.7% in Brazil,34 70.6% in Iran,35 75.0% in Taiwan,36 88% in Chongqing,23 and 94.1% in Sweden.37 In this study, 89.52% of the PZA-resistant strains harboured pncA mutations. The 102 mutant strains harboured 100 different mutation types (including synonymous mutations) that were scattered throughout the pncA gene. Substitution mutations and frame shifts (due to insertion or deletion) were detected in both the open reading frames and promoter regions of pncA. In this study, we detected 32 new mutations.38–42 In addition, 89 of the100mutations were found in only one strain, and the remaining 11 mutations, which contained 35 strains, were shared mutations. Mutations in the promoter region were associated with PZA resistance.43,44 Seven strains harboured mutations in the promoter region, which down-regulated the pncA transcription level, reduced PZ as activity, and ultimately led to PZA resistance. All three strains with a synonymous mutation belonged to the Beijing family. Moreover, out of 35 strains with shared mutations, 28 strains belonged to the Beijing genotype, six strains belonged to the T1 genotype, and one strain belonged to the T2 genotype. Sixteen strains with shared mutations had the same spoligotype and were genetically clustered. It was found that three of five strains with the promoter substitution mutation (T-12C) and five of seven strains with a substitution mutation at codon 359 were genetically in the Beijing family. Similar genetic classifications were found for substitution mutations at codon 226 (two of four isolates belonging to Beijing family), codon 203 (Beijing family), codon 398 (T family) and codon 416, and all of them were phenotypically resistant. Previous studies have suggested a correlation between pncA mutations and gene clustering,12,45,46 but we were unable to determine gene clustering due to the small sample size and the dominant Beijing genotype.
The11strains without pncA mutations could involve other mechanisms of PZA resistance;11,13,15 therefore, panD and rpsA mutations were analysed in this study. In 2011, Shi et al reported that rpsA could be another PZA resistance-related gene.13 However, limited data are available to explain the relationship between rpsA mutations and PZA susceptibility; thus, the contribution of the rpsA mutations conferring PZA resistance has been controversial until now. Alexander et al found no rpsA mutations in PZA-resistant isolates but one rpsA protein mutation (A364G) in 13 PZA-sensitive strains;47 Bhuju et al did not find any rpsA mutations among either PZA-resistant isolates or PZA-susceptible isolates.34 In addition, Pang et al found no genetic mutations associated with PZA resistance in the rpsA gene among MDR-TB patients.23 In this study, therpsAmutation frequency (6.7%) in MDR strains was higher than in these reports. A high synonymous mutation frequency was observed at codon212in both PZA-resistant and PZA-susceptible strains. However, no missense or deletion/insertion mutations in rpsA were detected in PZA-susceptible strains. Tan et al reported three nonsynonymous mutations in the rpsA C-terminal region;48 Gu et al also identified seven of 26 mutations occurring in the C-terminal region.24 Moreover, Khan’s research found that the majority of rpsA mutations were located in nucleotides 973–1051 of the coding region, especially nucleotides 1024–1030.49 In line with previous reports, we detected a mutation hotspot at the C-terminus of rpsA. Moreover, five of the eight rpsA mutations have never been described in previous studies, so further research is needed to investigate the effects of these new mutations on PZA resistance. In addition, the XDR strains in this study had a greater tendency to harbour rpsA mutations than the MDR strains, also demonstrating that rpsA mutations might be related to PZA resistance. While previous studies have shown that panD is a target for PZA resistance in M. tuberculosis, few studies have been conducted to investigate panD mutations in clinical strains. This study showed that two (1.9%)PZA-resistant strains harboured a panD mutation, one of which had wild-type pncA. In summary, we identified eight PZA-resistant strains without any known mutations, indicating that additional unknown mechanisms are involved in PZA resistance and requiring further research. For PZA resistance, the combination of all three genes sequenced achieved sensitivity of 92.38% and specificity of 89.36%, consistent with other reports. Considering the insufficient reliability and inconsistency between phenotypes and the drug susceptibility test, the sequencing of all three PZA-resistant genes could evaluate the resistance of PZA more effectively.
Finally, we analysed the outcomes of 124 patients under different treatment regimes. The sputum smear/culture conversion rate was slightly higher in PZA-sensitive patients than in PZA-resistant patients (95.5%vs93.9%), but the difference was not statistically significant. However, subgroup analysis showed that at the end of 6 months of treatment, the sputum smear/culture conversion rate was significantly higher in PZA-sensitive MDR-TB patients than in PZA-resistant patients (7.14%vs1.08%), suggesting that PZA resistance prolonged the time to sputum smear/culture conversion.
This study had several limitations. First, the sample size was small, and all of the strains were derived from our region. As a result, the results might not be representative and might not apply to other regions; therefore, more samples are needed in the future. Second, we detected 32 new pncA mutations and five new rpsA mutations, and further research is needed to investigate their effects on PZA resistance.
In summary, this study was the first to investigate the molecular characterization ofPZA resistance in MDR strains from Henan. The results showed that PZA resistance was substantially related to pncA mutations, which were scattered and diverse. In addition, rpsA and panD mutations were related to PZA resistance only in MDR strains without pncA mutations. With the resistance phenotype as the reference, molecular diagnosis with the combination of pncA, rpsA, and panD achieved sensitivity of98.1% and specificity of92.3%. Given the high PZA resistance rate in MDR-TB patients and that PZA resistance prolonged the time to sputum smear/culture conversion, the effects of PZA should be considered carefully for MDR-TB patients in high TB burden regions. In conclusion, the combination of pncA, rpsA, and panD enabled rapid and accurate prediction of PZA resistance, helped guide the use of PZA and optimized treatment therapy for MDR-TB patients in Henan.
Acknowledgments
This research received no specific grants from any funding agencies in the public, commercial, or not-for-profit sectors. We thank the staff of the CDC and TB dispensaries involved in this study. We are also grateful to Guolong Zhang for his constructive comments on this study.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Diacon AH, Van Baelen B, Theeuwes M. More on treatment outcomes in multidrug-resistant tuberculosis. N Engl J Med. 2016;375(26):2609–2610.
2. Johnston JC, Shahidi NC, Sadatsafavi M, Fitzgerald JM. Treatment outcomes of multidrug-resistant tuberculosis: a systematic review and meta-analysis. PLoS One. 2009;4(9):e6914. doi:10.1371/journal.pone.0006914
3. Collaborative Group for the Meta-Analysis of Individual Patient Data in MDRTBt, Ahmad N, Ahuja SD, et al. Treatment correlates of successful outcomes in pulmonary multidrug-resistant tuberculosis: an individual patient data meta-analysis. Lancet. 2018;392(10150):821–834. doi:10.1016/S0140-6736(18)31644-1.
4. Zhao Y, Xu S, Wang L, et al. National survey of drug-resistant tuberculosis in China. N Engl J Med. 2012;366(23):2161–2170. doi:10.1056/NEJMoa1108789
5. Gandhi NR, Nunn P, Dheda K, et al. Multidrug-resistant and extensively drug-resistant tuberculosis: a threat to global control of tuberculosis. Lancet. 2010;375(9728):1830–1843. doi:10.1016/S0140-6736(10)60410-2
6. Dheda K, Chang KC, Guglielmetti L, et al. Clinical management of adults and children with multidrug-resistant and extensively drug-resistant tuberculosis. Clin Microbiol Infect. 2017;23(3):131–140. doi:10.1016/j.cmi.2016.10.008
7. Scorpio A, Lindholm-Levy P, Heifets L, et al. Characterization of pncA mutations in pyrazinamide-resistant Mycobacterium tuberculosis. Antimicrob Agents Chemother. 1997;41(3):540–543. doi:10.1128/AAC.41.3.540
8. Zhang Y, Shi W, Zhang W, Mitchison D. Mechanisms of pyrazinamide action and resistance. Microbiol Spect. 2014;2(4):
9. Mestdagh M, Fonteyne PA, Realini L, et al. Relationship between pyrazinamide resistance, loss of pyrazinamidase activity, and mutations in the pncA locus in multidrug-resistant clinical isolates of Mycobacterium tuberculosis. Antimicrob Agents Chemother. 1999;43(9):2317–2319. doi:10.1128/AAC.43.9.2317
10. Stoffels K, Mathys V, Fauville-Dufaux M, Wintjens R, Bifani P. Systematic analysis of pyrazinamide-resistant spontaneous mutants and clinical isolates of Mycobacterium tuberculosis. Antimicrob Agents Chemother. 2012;56(10):5186–5193. doi:10.1128/AAC.05385-11
11. Zhang S, Chen J, Shi W, Liu W, Zhang W, Zhang Y. Mutations in panD encoding aspartate decarboxylase are associated with pyrazinamide resistance in Mycobacterium tuberculosis. Emerg Microbes Infect. 2013;2(6):e34. doi:10.1038/emi.2013.38
12. Huy NQ, Lucie C, Hoa TTT, et al. Molecular analysis of pyrazinamide resistance in Mycobacterium tuberculosis in Vietnam highlights the high rate of pyrazinamide resistance-associated mutations in clinical isolates. Emerg Microbes Infect. 2017;6(10):e86. doi:10.1038/emi.2017.73
13. Shi W, Zhang X, Jiang X, et al. Pyrazinamide inhibits trans-translation in Mycobacterium tuberculosis. Science. 2011;333(6049):1630–1632. doi:10.1126/science.1208813
14. Zhang Y, Chiu Chang K, Leung CC, et al. ‘Z(S)-MDR-TB’ versus ‘Z(R)-MDR-TB’: improving treatment of MDR-TB by identifying pyrazinamide susceptibility. Emerg Microbes Infect. 2012;1(7):e5. doi:10.1038/emi.2012.18
15. Tam KK, Leung KS, Siu GK, et al. Direct detection of pyrazinamide resistance in mycobacterium tuberculosis by use of pncA PCR sequencing. J Clin Microbiol. 2019;57:8. doi:10.1128/JCM.00145-19
16. Kurbatova EV, Cavanaugh JS, Dalton T, Click ES, Cegielski JP. Epidemiology of pyrazinamide-resistant tuberculosis in the United States, 1999-2009. Clin Infect Dis. 2013;57(8):1081–1093. doi:10.1093/cid/cit452
17. Shi J, Zheng D, Zhu Y, et al. Role of MIRU-VNTR and spoligotyping in assessing the genetic diversity of Mycobacterium tuberculosis in Henan Province, China. BMC Infect Dis. 2018;18(1):447. doi:10.1186/s12879-018-3351-y
18. Kamerbeek J, Schouls L, Kolk A, et al. Simultaneous detection and strain differentiation of Mycobacterium tuberculosis for diagnosis and epidemiology. J Clin Microbiol. 1997;35(4):907–914. doi:10.1128/JCM.35.4.907-914.1997
19. Chen YY, Chang JR, Wu CD, et al. Combining molecular typing and spatial pattern analysis to identify areas of high tuberculosis transmission in a moderate-incidence county in Taiwan. Sci Rep. 2017;7(1):5394. doi:10.1038/s41598-017-05674-6
20. Mokrousov I, Ly HM, Otten T, et al. Origin and primary dispersal of the Mycobacterium tuberculosis Beijing genotype: clues from human phylogeography. Genome Res. 2005;15(10):1357–1364. doi:10.1101/gr.3840605
21. Dookie N, Rambaran S, Padayatchi N, Mahomed S, Naidoo K. Evolution of drug resistance in Mycobacterium tuberculosis: a review on the molecular determinants of resistance and implications for personalized care. J Antimicrob Chemother. 2018;73(5):1138–1151. doi:10.1093/jac/dkx506
22. Zhang Y, Yew WW. Mechanisms of drug resistance in Mycobacterium tuberculosis: update 2015. Int J Tuberculosis Lung Dis. 2015;19(11):1276–1289. doi:10.5588/ijtld.15.0389
23. Pang Y, Zhu D, Zheng H, et al. Prevalence and molecular characterization of pyrazinamide resistance among multidrug-resistant Mycobacterium tuberculosis isolates from Southern China. BMC Infect Dis. 2017;17(1):711. doi:10.1186/s12879-017-2761-6
24. Gu Y, Yu X, Jiang G, et al. Pyrazinamide resistance among multidrug-resistant tuberculosis clinical isolates in a national referral center of China and its correlations with pncA, rpsA, and panD gene mutations. Diagn Microbiol Infect Dis. 2016;84(3):207–211. doi:10.1016/j.diagmicrobio.2015.10.017
25. Xu P, Wu J, Yang C, et al. Prevalence and transmission of pyrazinamide resistant Mycobacterium tuberculosis in China. Tuberculosis. 2016;98:56–61. doi:10.1016/j.tube.2016.02.008
26. Liu W, Chen J, Shen Y, et al. Phenotypic and genotypic characterization of pyrazinamide resistance among multidrug-resistant Mycobacterium tuberculosis clinical isolates in Hangzhou, China. Clin Microbiol Infect. 2018;24(9):1016e1011–1016 e1015. doi:10.1016/j.cmi.2017.12.012
27. Gopal P, Gruber G, Dartois V, Dick T. Pharmacological and molecular mechanisms behind the sterilizing activity of pyrazinamide. Trends Pharmacol Sci. 2019;40(12):930–940. doi:10.1016/j.tips.2019.10.005
28. Alame-Emane AK, Xu P, Pierre-Audigier C, et al. Pyrazinamide resistance in Mycobacterium tuberculosis arises after rifampicin and fluoroquinolone resistance. Int J Tuberculosis Lung Dis. 2015;19(6):679–684. doi:10.5588/ijtld.14.0768
29. Zignol M, Dean AS, Alikhanova N, et al. Population-based resistance of Mycobacterium tuberculosis isolates to pyrazinamide and fluoroquinolones: results from a multicountry surveillance project. Lancet Infect Dis. 2016;16(10):1185–1192. doi:10.1016/S1473-3099(16)30190-6
30. Zignol M, Floyd K. Resistance of Mycobacterium tuberculosis isolates to pyrazinamide and fluoroquinolones - authors’ reply. Lancet Infect Dis. 2017;17(1):25. doi:10.1016/S1473-3099(16)30538-2
31. Budzik JM, Jarlsberg LG, Higashi J, et al. Pyrazinamide resistance, Mycobacterium tuberculosis lineage and treatment outcomes in San Francisco, California. PLoS One. 2014;9(4):e95645. doi:10.1371/journal.pone.0095645
32. Suzuki S, Horinouchi T, Furusawa C. Prediction of antibiotic resistance by gene expression profiles. Nat Commun. 2014;5:5792. doi:10.1038/ncomms6792
33. Raju S, Ye CJ. How mutations express themselves in blood-cell production. Nature. 2019;571(7765):329–330. doi:10.1038/d41586-019-02028-2
34. Bhuju S, Fonseca Lde S, Marsico AG, et al. Mycobacterium tuberculosis isolates from Rio de Janeiro reveal unusually low correlation between pyrazinamide resistance and mutations in the pncA gene. Infect Genetics Evol. 2013;19:1–6. doi:10.1016/j.meegid.2013.06.008
35. Doustdar F, Khosravi AD, Farnia P. Mycobacterium tuberculosis genotypic diversity in pyrazinamide-resistant isolates of Iran. Microb Drug Resist. 2009;15(4):251–256. doi:10.1089/mdr.2009.0066
36. Jonmalung J, Prammananan T, Leechawengwongs M, Chaiprasert A. Surveillance of pyrazinamide susceptibility among multidrug-resistant Mycobacterium tuberculosis isolates from Siriraj Hospital, Thailand. BMC Microbiol. 2010;10:223. doi:10.1186/1471-2180-10-223
37. Jureen P, Werngren J, Toro JC, Hoffner S. Pyrazinamide resistance and pncA gene mutations in Mycobacterium tuberculosis. Antimicrob Agents Chemother. 2008;52(5):1852–1854. doi:10.1128/AAC.00110-08
38. Whitfield MG, Soeters HM, Warren RM, et al. A global perspective on pyrazinamide resistance: systematic review and meta-analysis. PLoS One. 2015;10(7):e0133869. doi:10.1371/journal.pone.0133869
39. Li D, Hu Y, Werngren J, et al. Multicenter study of the emergence and genetic characteristics of pyrazinamide-resistant tuberculosis in China. Antimicrob Agents Chemother. 2016;60(9):5159–5166. doi:10.1128/AAC.02687-15
40. Zheng X, Ning Z, Drobniewski F, et al. pncA mutations are associated with slower sputum conversion during standard treatment of multidrug-resistant tuberculosis. Int J Antimicrob Agents. 2017;49(2):183–188. doi:10.1016/j.ijantimicag.2016.10.012
41. Spinato J, Boivin E, Belanger-Trudelle E, Fauchon H, Tremblay C, Soualhine H. Genotypic characterization of drug resistant Mycobacterium tuberculosis in Quebec, 2002-2012. BMC Microbiol. 2016;16(1):164. doi:10.1186/s12866-016-0786-4
42. Khan MT, Malik SI, Ali S, et al. Pyrazinamide resistance and mutations in pncA among isolates of Mycobacterium tuberculosis from Khyber Pakhtunkhwa, Pakistan. BMC Infect Dis. 2019;19(1):116. doi:10.1186/s12879-019-3764-2
43. Sheen P, Lozano K, Gilman RH, et al. pncA gene expression and prediction factors on pyrazinamide resistance in Mycobacterium tuberculosis. Tuberculosis. 2013;93(5):515–522. doi:10.1016/j.tube.2013.03.005
44. Filipenko ML, Dymova MA, Cherednichenko AG, Khrapov EA, Mishukova OV, Schwartz YS. Detection of Mutations in Mycobacterium tuberculosis pncA gene by modified high-resolution melting curve analysis of PCR products. Bull Exp Biol Med. 2019;168(2):264–269. doi:10.1007/s10517-019-04688-6
45. Rodrigues Vde F, Telles MA, Ribeiro MO, Cafrune PI, Rossetti ML, Zaha A. Characterization of pncA mutations in pyrazinamide-resistant Mycobacterium tuberculosis in Brazil. Antimicrob Agents Chemother. 2005;49(1):444–446. doi:10.1128/AAC.49.1.444-446.2005
46. Rahman A, Ferdous SS, Ahmed S, et al. Pyrazinamide susceptibility and pncA mutation profiles of mycobacterium tuberculosis among multidrug-resistant tuberculosis patients in Bangladesh. Antimicrob Agents Chemother. 2017;61:9. doi:10.1128/AAC.00511-17
47. Alexander DC, Ma JH, Guthrie JL, Blair J, Chedore P, Jamieson FB. Gene sequencing for routine verification of pyrazinamide resistance in Mycobacterium tuberculosis: a role for pncA but not rpsA. J Clin Microbiol. 2012;50(11):3726–3728. doi:10.1128/JCM.00620-12
48. Tan Y, Hu Z, Zhang T, et al. Role of pncA and rpsA gene sequencing in detection of pyrazinamide resistance in Mycobacterium tuberculosis isolates from southern China. J Clin Microbiol. 2014;52(1):291–297. doi:10.1128/JCM.01903-13
49. Khan MT, Malik SI, Bhatti AI, et al. Pyrazinamide-resistant mycobacterium tuberculosis isolates from Khyber Pakhtunkhwa and rpsA mutations. J Biol Regul Homeost Agents. 2018;32(3):705–709.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
