Back to Journals » Journal of Pain Research » Volume 13
Properties of Thermal Analgesia in a Human Chronic Low Back Pain Model
Authors Chabal C , Dunbar PJ, Painter I, Young D , Chabal DC
Received 7 May 2020
Accepted for publication 10 July 2020
Published 13 August 2020 Volume 2020:13 Pages 2083—2092
DOI https://doi.org/10.2147/JPR.S260967
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Robert B. Raffa
Video abstract of "Properties of thermal analgesia in a human chronic low back pain model" [ID 260967].
Views: 162
Charles Chabal,1 Peter J Dunbar,1 Ian Painter,2 Douglas Young,3 Darah C Chabal4
1Soovu Labs Inc., Seattle, WA, USA; 2Department of Health Services, University of Washington, Seattle, WA, USA; 3Northern California Research, Sacramento, CA, USA; 4Biology Department, University of Washington, Seattle, WA, USA
Correspondence: Charles Chabal
Soovu Labs Inc., Seattle, WA, USA
Tel +1 206-579-4910
Email [email protected]
Purpose: For years, heat has been used for comfort and analgesia is recommended as a first-line therapy in many clinical guidelines. Yet, there are questions that remain about the actual effectiveness of heat for a condition as common as chronic low back pain, and factors such as time of onset, optimal temperature, and duration of effect.
Materials and Methods: A randomized double-blinded controlled trial was designed to compare the analgesic response to heat delivered via pulses at 45°C (experimental group, N=49) to steady heat at 37°C (control group, N=51) in subjects with longstanding low back pain. Treatment lasted 30 minutes with follow-up out to four hours. The hypothesis was that the experimental group would experience a higher degree of analgesia compared to the control group. Time of onset and duration of effect were also measured.
Results: Both groups were similar in average duration of pain (10.3 years). The primary outcome measure was pain reduction at 30 minutes after the end of treatment, using a 10-points numeric pain scale. Reduction in pain was greater for the experimental group than the control group (difference in mean reduction = 0.72, 95% CI 0.15– 1.29, p = 0.014). Statistically significant differences in pain levels were observed from the first measure at 5 minutes of treatment through 120 minutes after completion of treatment. Reduction in pain associated movement was greater in the active heat group than the placebo group (p = 0.04).
Conclusion: High-level pulsed heat (45°C) produced significantly more analgesia as compared to steady heat at 37°C at the primary end point and for an additional 2 hours after treatment. The onset of analgesia was rapid, < 5 minutes of treatment. The results of this trial provide insight into the mechanisms and properties of thermal analgesia that are not well understood in a chronic low back pain model.
Keywords: thermal analgesia, heat, chronic low back pain
Plain Language Summary
Although a common and long accepted form of pain relief and comfort, there remain significant basic questions about how heat produces pain relief. A study was done in 100 people who suffered from chronic low back pain for >10 years to address some of these questions. One group (experimental) received a heating device that produced pulsed heat at 45°C and one group (control) received a heating device that produced steady heat at 37°C. The experimental group (high heat) had significantly better relief of their chronic low back pain than the group treated with the lower heat. The start of pain relief in the high heat group was very fast and occurred by the first time measurement at 5 minutes. After the 30-minute heating session, the devices were turned off, but the high heat treatment group of people had pain relief that lasted for an additional two hours. In addition, the group treated with high heat had a greater reduction in pain associated with body movements than the low heat group. This examined some important aspects of pain relief from heat and offer insight into other questions about how heat relieves pain. The results of this study may help develop an approach that could potentially be used to help treat the flair-ups of chronic low back pain.
Introduction
For thousands of years, heat has provided comfort and pain relief. Recently, heat as a nonpharmacological approach to pain management has become a key part of treatment guidelines, including the Clinical Guidelines for the American College of Physicians1 which states as part of the first recommendation, “ … clinicians and patients should select nonpharmacologic treatment with superficial heat … ”. Yet, a Cochrane Review by French et al 20062 pointed out that in spite of the frequent use of heat and clinical guidelines, there is a paucity of good studies demonstrating the effectiveness of heat in treating a condition as common as acute low back pain and even less information on treating chronic low back pain. The issues identified limiting the understanding of heat’s pain-relieving abilities, are related to the study methodology and questions about randomization, blinding, and composition of treatment groups.2
In their review, French et al (2006)2 identified four published trials judged as a higher quality based on their methodology using heat in low back pain. Nadler et al (2002)3 compared a chemical heat wrap to oral placebos, ibuprofen, or acetaminophen in subjects with acute to subacute (3 months or less) low back pain. The heat wrap provided up to 8 hours of 40°C heat produced by a chemical oxidation reaction. The heat wrap produced superior pain relief when compared to both ibuprofen and acetaminophen. Similar studies in acute low back pain (<3 months) found superior pain relief compared to oral placebo or ibuprofen when the wraps were used overnight as well over a 5-day period.4,5 Nuhr et al (2004)6 evaluated heat provided by electrical blankets in subjects with acute low back pain to treatment with an unheated blanket. The heated electrical blanket produced significantly greater pain relief than an unheated blanket alone. Finally, Mayer et al (2005)7 found that hot chemical packs in acute back pain combined with exercise were better than either intervention alone. More recent studies also support the use of heat to relieve pain in neck and back strain,8,9 knee pain,10 dysmenorrhea,11 and delayed muscle soreness related to exercise.12
The literature cited above and human experience supports that heat produces pain relief and comfort, yet our understanding of thermal analgesia (the reduction of pain intensity associated with heat) remains relatively thin. Little is known about the onset of action or how quickly heat reduces pain. For most analgesics, the onset of action is a critical measure, yet for heat, there are no published studies. In terms of mechanism of action, it is likely that heat produces some effect from interaction with the peripheral nervous system. Animal studies indicate that hotter temperatures to a point will cause increased firing of thermal receptors as measured by C fiber stimulation.13,14 However, most human studies are limited to a steady temperature of 40°C3–10 with few if any examining higher temperature stimulation. Finally, even a very fundamental outcome such as duration of action to thermal stimulation is virtually unknown.
Against this background, the present study was designed using a chronic low back pain model. The primary hypothesis was that higher level thermal stimulation would produce better analgesia than lower temperatures in a well-designed placebo controlled randomized double-blinded study. In addition, this study was designed to carefully document two poorly understood outcomes of thermal analgesia: 1) the onset and 2) duration of analgesia after 30 minutes of thermal stimulation.
Methods and Materials
The study was designed and conducted in a randomized double-blinded placebo controlled fashion and performed at the Northern California Research Inc., Sacramento, CA with IRB approval from the Western Institutional Review Board and registration on ClinicalTrials.gov (NCT04109703). Procedures followed were in accordance with the Declaration of Helsinki and all subjects gave written informed consent. Northern California Research Institute operates according to Good Clinical Research Practices (GCRP). Broadly, GCPR are standards endorsed by both the FDA15 and World Health Organization16 to ensure ethical treatment of subjects, reporting of adverse events, proper documentation standards and valid data collection and reporting procedures. In this study, no sponsoring company member had any contact with any subject. The study was designed as a randomized double-blinded study where the research assistant who set up the subject did not assist with data collection. Subject randomization was done using an online computer-generated randomization program and the CRO-study coordinator controlled the randomization schedule but did not disclose the allocation until the subject was confirmed as eligible and enrolled. The CRO-study coordinator was independent of the research assistants who interacted with the subjects.
Subjects were recruited by the Clinical Research organization (CRO) from an existing database and through community outreach. Subjects were paid $150 for the completion of the single study session.
Inclusion Criteria of the Study Were
- Chronic nonradiating low back pain. Defined as pain present for at least 6 months on more days than not.17
- Subjects must have a pretreatment level of pain 4 or greater.
- Ages 22 through 70 inclusive. The age limits were determined to include adults and those most likely to be able to use a smart phone app. FDA guidelines define adults as 22 years of age and older.18
- Pain medications can be used prior to the trial; however, none can be used during the approximate 4-hour trial (30 minutes of active treatment and 3.5 hours of post-treatment observation). Other non-pain medications are permitted as needed.
- Medications permitted prior to the trial include medications such as tramadol, codeine, NSIADs, gabapentin and acetaminophen.
- Subjects must have a phone for clinic contact and follow-up.
Exclusion Criteria of the Study Were
- Sciatica or radicular pain without non-radiating low back pain, cancer, radicular pain greater than the non-radiating component of low back pain.
- Pregnancy, skin lesions such as open skin or sores, scar tissue, skins grafts, or old burns over the treatment area.
- Current use of schedule II opioids including oxycodone, hydromorphone, hydrocodone, fentanyl and methadone.
The author (Dr Chabal) briefed the research staff on the devices and how to operate them. The sponsor (Soovu Labs Inc. Seattle, WA) did not have access to subjects or staff during the study. The originals of the study documents are securely held by CRO with images of the data sheets sent to the consultant at the University of Washington Statistics Department for analysis. Subjects were randomly assigned to either an active device that used high level (45°C) pulsed heat (experimental arm) or a device that used low level (37°C) steady heat (control arm). This physically identical device used 37° C to ensure that subjects could feel warming but would not achieve the 40°C, that may be therapeutically active.3–10 Subjects were told that the study used identical devices, but the devices were set at different temperatures and the purpose of the study was to identify which temperature worked best to relieve pain.
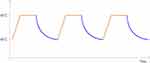
The units are advanced prototypes from Soovu Labs Inc. Experimental and control units are identical and only the programmed temperatures differed. The units (Figure 1) consisted of two heating pods with one-inch diameter metal heating plates. The heating units were attached to the subjects’ low back over the paravertebral muscles at the lumbar 4/5 vertebral level using EKG-like stickers. The units were held to the stickers by a magnetic attachment. Each subject received two heating units that were placed bilaterally, with one unit on the left paravertebral muscle at the L4/5 level and one on the right paravertebral muscle at the L4/5 level. The heating units were controlled by a phone app via Bluetooth connectivity. The app was controlled by study personnel. For this study, the app could turn the heating elements on or off and control the duration and maximum temperature of the heating device. The settings for the experimental arm included an increase of heat from baseline skin temperature to a maximum temperature of 45° C over 5 seconds, a peak temperature of 45°C held for 10 seconds and an off period that lasted for 30 seconds at 40°C. This drop in temperature was passive with no active cooling. This pattern cycle repeated for 10 minutes. At the end of 10 minutes the unit automatically shut down for 2 minutes after which the cycle started again (Figure 2). Each subject in the experimental arm underwent three 10-minute heat cycles, with two 2-minute pauses for a total time of 34 minutes. The two-minute pauses were included as a precautionary measure for safety. Subjects in the active placebo (control) arm received identical heating units. These units were programed to deliver steady heat at 37°C for three 10-minute heating periods also separated by two 2-minute pauses for a total time of 34 minutes.
Upon entering the study, subjects had a baseline pain level assessed using the 0 to 10 Numeric Pain Scale (NPS).19,20 Treatment took place for 34 minutes while sitting, after which the units were removed, and the subjects were allowed to move around for the next 4 hours. Subjects were allowed activities such as using their phones or computers, reading, eating, or watching television or movies. No subject was allowed to leave the building. Pain was assessed in both groups at baseline and after 5, 10, 15, and 30 minutes of treatment. Pain was assessed at 15, 30, 45, 60, 90, 120, 150, 180 and 210 minutes after the 30-minute treatment had ended. After 30 minutes of treatment, a pain assessment compared pain with anterior-posterior flexion and extension with the pretreatment pain level of the same movement. At the end of the 30-minute treatment session and at 4 hours, subjects completed an assessment that asked whether they felt they had received the high-temperature unit, the low-temperature unit, or were unsure.
The skin was also assessed by study personal at baseline, after the treatment session and 4-hours post treatment. The skin assessment examined any erythema or pigment or color change at the heating site. In addition, any pain or discomfort at the site was noted.
Sample Size/Power Calculation
Sample size calculations were based on the results of two pilot trials comparing an early prototype of the pulsed heat device against Thermacare™ devices (result unpublished). A standard deviation of the change in NPS of 1.75 was assumed based on the results of the pilot trials. Sample size was calculated using standard sample size calculation software for a two-sample t-test to achieve 80% power to detect an effect size of a 1.0 difference in change in NPS from baseline to end of treatment plus 30 minutes between the two groups at the alpha level of 0.05. Based on these calculations 100 total subjects were recruited.
Statistical Analysis
The software used for the analysis of data in this study was The R Project for Statistical Computing (https://www.r-project.org/). Demographic and clinical characteristics were tabulated by randomization group. The primary outcome was changed in pain score from baseline to 30 minutes after treatment ended. Linear regression was used to compare differences in primary outcome between experimental and control groups adjusting for initial pain level. Unadjusted comparisons are also presented. Change in pain scores at other time intervals were compared to baseline scores and were similarly analyzed.
Proportion of participants who somewhat or strongly believed that they received the active treatment at 30 minutes and 240 minutes post treatment were compared using fisher’s exact test.
Results
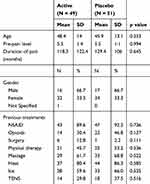
After screening, 100 subjects entered the study, and all completed the single session study. Fifty-one participants were randomized to the control group and 49 to the experimental group. There were 66 females, 33 males and one individual did not specify gender. The mean age was 47.1 (range 23–70 years). The mean duration of low back pain was 10.3 years (range 0.4–40 years). Past treatments included nonsteroidal anti-inflammatory drugs (90.1%), opioids (38.7%), surgery (7.6%), physical therapy (49.5%), massage (65.3%), heat (83.5%), ice (62.9%) and transcutaneous electrical nerve stimulation (TENS) (33.7%). Experimental versus control groups were similar in terms of age, gender distribution, duration of pain, past treatments and pre-pain scores (Table 1). Mean pain level at baseline was 5.5. Reduction in pain was negatively correlated with initial pain (correlation coefficient = −0.41, p 0.001). Reduction in pain at the primary time point (30 minutes post treatment) after adjusting for initial pain level was greater for the experimental than the control device (difference in mean reduction = 0.72, 95% CI 0.15 to 1.29, p = 0.014). Unadjusted differences were similar (mean difference = 0.72, 95% CI −0.09 to −1.35, p = 0.025). Statistically significant differences were observed from the first measure at 5 minutes of treatment through 120 minutes after completion of the treatment session for a total time of 150 minutes (Table 2 and Figure 3). When initial pain levels were examined, greater reductions in pain were observed for patients with higher initial pain levels greater than or equal to 7. Furthermore, differences in reductions in numeric pain scores between groups were much larger in patients with initial pain scores for 7 or more than in patients with initial pain scores of less than 7 (Table 3).
Early onset of pain relief in the intervention group was predictive of long-term substantial pain relief: Of those who had at least a one-point drop at time T-5, 66% had at least a 2 point drop at time post-T-90, and 50% had at least a 3 point drop; among those two showed no pain improvement at T-5, 31% had at least a 2 point drop at time post-T-90 and 7% had at least a 3 point drop.
Pain upon movement was measured at baseline and at the end of treatment (time point T-30). Reduction in pain movement score was 0.56 points greater in the experimental group than the control group (p = 0.04). Of those given a low-level heat device, 6% thought they had received a higher temperature device after 30 minutes of treatment and 2% at 4 hours post treatment.
There were no changes in skin, pigment, lasting erythema or complaints of burning pain at any assessment during or after the trial.
Discussion
The results of this study demonstrated that in a group of subjects with chronic longstanding (>10 years) low back pain, 30 minutes of pulsed heat at 45°C was statistically superior to 30 minutes of steady heat at 37°C. The differences in pain levels occurred very quickly and were evident at the first pain assessment after 5 minutes of treatment. A single 30 minutes of thermal stimulation produced pain relief that lasted for an additional 2 hours after cessation of heat. A secondary measure of pain associated with flexion and extension showed a significant reduction of pain after 30 minutes of treatment in the experimental as compared to the control group.
While one may question the utility of a single 30-minute treatment session with a four hour follow up, this study provided important fundamental information about thermal analgesia in terms of onset of action, duration of effect, and uniquely examined the analgesic responses to temperatures greater than 40°C in a human chronic pain model. To our knowledge there are no animal or human acute pain models that could provide a simpler test, thus necessitating the more complex chronic pain model used in this experiment. Clinically, this short experiment may be analogous to a common clinical occurrence, a pain exacerbation in a longtime low back pain sufferer. In this case, a short therapeutic session of heat may offer a fast effective drug-free option.
In the current study, the maximum temperature of 45°C was delivered as short pulses of 10 seconds followed by an off period where the temperature passively dropped to 40° C for 30 seconds. This was done for two reasons. Primarily, the pulsing greatly reduced the thermal energy delivered to the subjects’ skin and provided a large safety margin. No subject experienced even temporary pain at the site of heating. Although not formally tested in this design, it was also thought that the pulsing of heat could also reduce neural accommodation resulting in better pain relief.
In terms of therapeutic effect, the differential reduction in pain of 0.7 points on a 0 to 10 standard numeric pain scale was achieved by the experimental group as compared to the control group. This result is equivalent to that produced by nonsteroidal anti-inflammatory drugs (NSAIDS)21 and opioids in chronic low back pain.22,23 The effect should be further interpreted with the understanding that these study subjects started with a relatively modest level of pain on the NPS. It is more difficult to show effect in chronic longstanding pain subjects versus a commonly used analgesic model of acute pain such as a third molar extraction24,25 where initial pain levels may approach 10 on the same pain scale. A final interpretation of clinical improvement comes from the Brief Pain Inventory, a tool validated across many cultures.26 At a pain rating of 5 or 6 quality of life dimensions such as sleep, activity, mood, work and enjoyment of life are affected. As pain is reduced to a level 3 or 4, pain still affects the enjoyment of life and work but much less so for other components such as sleep, activity and mood.26 So, for many subjects who had their pain rating dropped from 5 to 4, the BPI would predict improvements in indicators of quality of life.
A relatively surprising finding was that there was a significant carryover effect in the experimental group from the 30 minutes of pulsed heat at 45°C as pain was reduced for 120 minutes after cessation of heat. Others have observed a carryover-like effect with heat, but this phenomenon has not been well studied or explained.4,27 It is unclear whether this is a result of desensitized of peripheral receptors such as transient receptor potential vanilloid 1 channel (TRPV-1) or a more central mechanism.
The temperature of 45° C was chosen for this study based on a hypothesis that some of the effectiveness of heat as an analgesic is due to an interaction initiated at the peripheral receptor level. Animal studies provide support that thermal stimulation activates receptors causing discharges in C and A delta fibers with a relationship between temperature and discharge rate.13,14 In humans, the limit temperature between the absence and presence of nociception induced by heat is approximately 45°C.28,29 The cut-off temperature between warm and burning sensations can vary greatly from one individual to another, as it is likely affected by the absolute temperature, duration of contact, thermal mass, skin pigmentation, skin thickness or blood flow.30 For safety purposes, this initial study used a maximum temperature of 45° C which was based on existing safety studies in humans30 well below temperatures and duration that could cause damage. With lower temperatures such as 40° C from chemical hot packs, the onset of analgesia is slower and may be from blood flow changes and muscle relaxation31,32 In this study, pain relief occurred very quickly, within 5 minutes, supporting a neurological effect at least initially. In addition, the temperature range of 45° C may activate TRPV1 receptors or others which are not activated by lower temperatures such as 40°C33,34 and may at least partly answer why hot showers or soaks are more comforting than lukewarm temperatures.
The design of a placebo or control arm faced challenges common to many device and drug trials. In this case, an “active” placebo was used. The control group devices heated to 37° C and maintained that temperature throughout the study. Subjects were told that the study was comparing two active devices at different temperatures. Although 37° C is a lower temperature than the chemical hot packs used successfully in previous studies,3–7 37°C was noticeably warm to subjects as the skin temperature is about 33–35° C.35 Most subjects in the low-temperature arm realized that they had received a low-temperature device but were also told that the purpose of the study was to test the effectiveness of two different temperatures and these subjects never had exposure to the hotter units. We could not rule out that the “placebo” arm may have delivered some beneficial heat benefit beyond acting solely as a placebo.
This study in subjects with long-standing low back pain demonstrated that pulsing heat to 45°C offered significantly better and clinically-meaningful pain relief as compared to low-level steady heat (37°C). In addition, the onset of action was very fast (5 minutes or less) and a single 30-minute treatment lasted for an additional 2 hours. While this study did not examine the long-term effects over many months of treatment, the results support that heat may be a fast and effective clinical treatment for exacerbations in patients who suffer from chronic low back pain.
This study offered insights about thermal analgesia in a human chronic pain model. However, many important and clinically relevant questions remain. For example, although this study used a 30-minute treatment session that produced 2 hours of pain relief, little is known about any relationship between the duration of heat and duration of action. For example, what is the duration of effect from 15, 20, or 45 minutes of thermal stimulation? In addition, multiple questions remain about the relationship between analgesic response and thermal energy transfer. Perhaps temperatures higher than 45 degrees, if pulsed briefly, could produce greater analgesia by recruitment of more receptors or even different populations of receptors without discomfort or injury. Finally, little is known about thermal stimulation and offset analgesia or condition pain modulation and whether there are different effects from direct stimulation as in this experiment. Since thermal analgesia is a drug-free effective analgesic, research exploring these important questions deserve study to potentially produce a more effective pain management option.
Conclusion
Although a common and long accepted form of pain relief and comfort there remain significant basic questions about thermal analgesia. A chronic low back pain model was used to examine some of these questions. High-level pulsed heat (45°C) in the experimental condition produced statistically significant more pain relief as compared to steady heat at 37°C in the control group, indicating that there is likely a dose-like response to the amount of thermal energy and analgesic response. The onset of thermal analgesia was rapid, <5 minutes of treatment and lasted for an additional 2 hours after termination of treatment. The high-level pulsed heat group also had a statistically significant better reduction in movement-related pain than the control group. The results of this trial provide insight into the mechanisms and properties of thermal analgesia that are not well understood in a human chronic low back pain model and could potentially be used to help treat exacerbations of chronic low back pain.
Data Sharing Statement
The datasets generated during the current study are available from the corresponding author on reasonable request for 3 years after publication.
Acknowledgment
The study was funded by Soovu Labs Inc.
Disclosure
Drs. Chabal and Dunbar are shareholders in Soovu Labs Inc. Dr Charles Chabal reports personal fees and stock from Soovu Labs Inc., during the conduct of the study. In addition, Dr Charles Chabal reports patents (see listed with Dr. Dunbar), but none from this study. Dr Peter J Dunbar reports being a founding shareholder of the predecessor company that became Soovu Labs and Chief Medical Officer of Soovu Labs, during the conduct of the study; and being a shareholder in Soovu Labs, outside the submitted work. In addition, Dr Peter J Dunbar has the following issued patents: US20200008973A1, US20190038456A1, US10603208B2, and WO2019173623A1. Dr Ian Painter reports being a paid consultant for his statistical services associated with this manuscript, from Soovu Labs, during the conduct of the study. The authors report no other potential conflicts of interest for this work.
References
1. Qaseem A, Wilt TJ, McLean RM, Forciea MA. Noninvasive treatments for acute, subacute, and chronic low back pain: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2017;166(7):514–530. doi:10.7326/M16-2367
2. French SD, Cameron M, Walker BF, Reggars JW, Esterman AJ. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;(1):Cd004750.
3. Nadler SF, Steiner DJ, Erasala GN, Hengehold DA, Abeln SB, Weingand KW. Continuous low-level heatwrap therapy for treating acute nonspecific low back pain. Arch Phys Med Rehabil. 2002;84(3):329–334. doi:10.1053/apmr.2003.50102
4. Nadler SF, Steiner DJ, Petty SR, Erasala GN, Hengehold DA, Weingand KW. Overnight use of continuous low-level heat wrap therapy for relief of low back pain. Arch Phys Med Rehabil. 2003;84(3):335–342. doi:10.1053/apmr.2003.50103
5. Nadler SF, Steiner DJ, Erasala GN, et al. Continuous low-level heat wrap therapy provides more efficacy than Ibuprofen and acetaminophen for acute low back pain. Spine. 2002;27(10):1012–1017. doi:10.1097/00007632-200205150-00003
6. Nuhr M, Hoerauf K, Bertalanffy A, et al. Active warming during emergency transport relieves acute low back pain. Spine. 2004;29(14):1499–1503. doi:10.1097/01.BRS.0000131439.87553.99
7. Mayer JM, Ralph L, Look M, et al. Treating acute low back pain with continuous low-level heat wrap therapy and/or exercise: a randomized controlled trial. Spine J. 2005;5(4):395–403. doi:10.1016/j.spinee.2005.03.009
8. Garra G, Singer AJ, Leno R, et al. Heat or cold packs for neck and back strain: a randomized controlled trial of efficacy. Acad Emerg Med. 2010;17:484–489. doi:10.1111/j.1553-2712.2010.00735.x
9. Petrofsky JS, Laymon M, Alshammari F, Khowailed IA, Lee H. Use of low level of continuous heat and Ibuprofen as an adjunct to physical therapy improves pain relief, range of motion and the compliance for home exercise in patients with nonspecific neck pain: a randomized controlled trial. J Back Musculoskelet Rehabil. 2017;30(4):
10. Petrofsky JS, Laymon MS, Alshammari FS, Lee H. Use of low level of continuous heat as an adjunct to physical therapy improves knee pain recovery and the compliance for home exercise in patients with chronic knee pain: a randomized controlled trial. J Strength Cond Res. 2016;30(11):
11. Jo J, Lee SH. Heat therapy for primary dysmenorrhea: a systematic review and meta-analysis of its effects on pain relief and quality of life. Sci Rep. 2018;8(1):16252. doi:10.1038/s41598-018-34303-z
12. Petrofsky J, Berk L, Bains G, Khowailed IA, Lee H, Laymon M. The efficacy of sustained heat treatment on delayed-onset muscle soreness. Clin J Sport Med. 2017;27(4):
13. Hensel H, Kenshalo DR. Warm receptors in the nasal region of cats. J Physiol. 1969;204(1):99–112. doi:10.1113/jphysiol.1969.sp008901
14. Hoffmann T, Sauer SK, Horch RE, Reeh PW. Sensory transduction in peripheral nerve axons elicits ectopic action potentials. J Neurosci. 2008;28(24):6281–6284. doi:10.1523/JNEUROSCI.1627-08.2008
15. FDA regulations: good clinical practice and clinical trials; 2019. Available from: https://www.fda.gov/science-research/clinical-trials-and-human-subject-protection/regulations-good-clinical-practice-and-clinical-trials.
16. Guidelines for good clinical practice (GCP) for trials on pharmaceutical products. World Health Organization WHO. 1995.
17. Treede RD, Rief W, Barke A, et al. A classification of chronic pain for ICD-11. Pain. 2015;156(6):
18. FDA. Pediatric medical devices; 2019. Available from: https://www.fda.gov/medical-devices/products-and-medical-procedures/pediatric-medical-devices.
19. Ferreira-Valente MA, Pais-Ribeiro JL, Jensen MP. Validity of four pain intensity rating scales. Pain. 2011;152(10):2399–2404. doi:10.1016/j.pain.2011.07.005
20. Hjermstad MJ, Fayers PM, Haugen DF, et al. Studies comparing numerical rating scales, verbal rating scales, and visual analogue scales for assessment of pain intensity in adults: a systematic literature review. J Pain Symptom Manage. 2011;41(6):1073–1093. doi:10.1016/j.jpainsymman.2010.08.016
21. Chou R, Deyo R, Friedly J, et al. Systemic pharmacologic therapies for low back pain: a systematic review for an American College of Physicians clinical practice guideline. Ann Intern Med. 2017;166(7):480–492. doi:10.7326/M16-2458
22. Chaparro LE, Furlan AD, Deshpande A, Mailis-Gagnon A, Atlas S, Turk DC. Opioids compared to placebo or other treatments for chronic low-back pain. Cochrane Database Syst Rev. 2013;8:CD004959.
23. Busse JW, Wang L, Kamaleldin M, et al. Opioids for chronic noncancer pain: a systematic review and meta-analysis. JAMA. 2018;320(23):2448–2460. doi:10.1001/jama.2018.18472
24. Moore RA, Derry S, McQuay HJ, Wiffen PJ. Single dose oral analgesics for acute postoperative pain in adults. Cochrane Database Syst Rev. 2011;9:Cd008659.
25. Cooper SA, Desjardins PJ. The value of the dental impaction pain model in drug development. Methods Mol Biol. 2010;617:175–190.
26. Cleeland CS, Ryan KM. Pain assessment: global use of the brief pain inventory. Ann Acad Med Singapore. 1994;23(2):
27. Michlovitz S, Hun L, Erasala GN, Hengehold DA, Weingand KW. Continuous low-level heat wrap therapy is effective for treating wrist pain. Arch Phys Med Rehabil. 2004;85(9):1409–1416. doi:10.1016/j.apmr.2003.10.016
28. Lloyd-Smith DL, Mendelssohn K. Tolerance limits to radiant heat. Br Med J. 1948;1(4559):
29. Ungar E, Stroud K, New A Approach to defining human touch temperature standards; 2010. Available from: https://ntrs.nasa.gov/search.jsp?R=20100020960 2020-06-11T22:06:35+00:00Z.
30. Ye H, De S. Thermal injury of skin and subcutaneous tissues: a review of experimental approaches and numerical models. Burns. 2017;43(5):909–932. doi:10.1016/j.burns.2016.11.014
31. O’Connor A, McCarberg W. A new look at heat treatment for pain disorders. Am Pain Soc Bull. 2004;Part 1.
32. O’Connor A, McCarberg W, O’Connor, et al. A new look at heat treatment for pain disorders. Am Pain Soc Bull. 2005;Part 2:35.
33. Hazan A, Kumar R, Matzner H, Priel A. The pain receptor TRPV1 displays agonist-dependent activation stoichiometry. Sci Rep. 2015;5:12278. doi:10.1038/srep12278
34. Hoffmann T, Kistner K, Miermeister F, et al. TRPA1 and TRPV1 are differentially involved in heat nociception of mice. Eur J Pain. 2013;17(10):1472–1482. doi:10.1002/j.1532-2149.2013.00331.x
35. Bierman W. “The Temperature of the Skin Surface”. J Am Med Assoc. 1936;106(14):1158. doi:10.1001/jama.1936.02770140020007
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.