Back to Journals » OncoTargets and Therapy » Volume 8
Prolonged radiation time and low nadir hemoglobin during postoperative concurrent chemoradiotherapy are both poor prognostic factors with synergistic effect on locally advanced head and neck cancer patients
Authors Su N , Liu C, Leu Y, Lee J, Chen Y , Chang Y
Received 28 June 2014
Accepted for publication 8 September 2014
Published 28 January 2015 Volume 2015:8 Pages 251—258
DOI https://doi.org/10.2147/OTT.S70204
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Jianmin Xu
Nai-Wen Su,1 Chung-Ji Liu,2 Yi-Shing Leu,3 Jehn-Chuan Lee,3 Yu-Jen Chen,4 Yi-Fang Chang1,5
1Division of Medical Oncology and Hematology, Department of Internal Medicine, 2Department of Oral and Maxillofacial Surgery, 3Department of Otorhinolaryngology, 4Department of Radiation Oncology, 5Good Clinical Research Center, Department of Medical Research, Mackay Memorial Hospital, Taipei, Taiwan
Background: Anemia, a common complication of head and neck cancer treatment, is regarded as a poor prognostic factor. We evaluated the impact of low hemoglobin (Hb) levels, measured at different time points, on a consecutive cohort of patients with locally advanced squamous cell carcinoma of the head and neck (LA-SCCHN) who underwent postoperative concurrent chemoradiotherapy (CCRT).
Materials and methods: From 2002 to 2009, 140 patients were enrolled and reviewed retrospectively. Preoperative (pre-op Hb), pre-CCRT Hb, and nadir Hb during CCRT were measured and recorded. The three Hb parameters were analyzed against several well-established pathologic risk factors and radiation-associated variables. Prognostic impacts were investigated with multivariate analysis by Cox proportional hazards model.
Results: On Cox regression analysis, significantly higher risk of death was associated with pre-op Hb %13 g/dL (hazard ratio [HR] =1.8; 95% confidence interval [CI], 1.1–3.1; P=0.023), nadir Hb %11 g/dL (HR =1.9; 95% CI, 1.1–3.3; P=0.020), radiation treatment time (RTT) >7 weeks (HR =1.9; 95% CI, 1.1–3.3; P=0.022), and multiple positive lymph nodes (HR =2.1; 95% CI, 1.2–3.7; P=0.010), after adjusting for primary tumor site and pathologic lymphovascular invasion. Patients with poor prognosticators including low nadir Hb %11 g/dL and RTT >7 weeks had a higher risk of death (HR =4.0; 95% CI =1.6–10.2; P=0.004).
Conclusion: In the treatment setting of LA-SCCHN patients who underwent postoperative CCRT, coexistance of lower nadir Hb during CCRT and prolonged RTT resulted in reduced survival.
Keywords: anemia, radiation time, concurrent chemoradiotherapy, hemoglobin, head and neck cancer
Background
A multidisciplinary strategy incorporating surgery, radiotherapy (RT), and chemotherapy improves the treatment outcome in locally advanced squamous cell carcinoma of the head and neck (LA-SCCHN). Postoperative adjuvant concurrent chemoradiotherapy (CCRT) has been shown to enhance survival outcome when compared to RT alone.1–4 However, such combined treatment modalities also incur significant acute side effects and long-term morbidities. Among the acute complications, the occurrence of anemia is common5 and might be related to surgical blood loss, CCRT, or poor nutritional status.
Besides the direct physiologic consequences of anemia, anemia itself can result in an inferior treatment outcome by causing tumor hypoxia and radioresistance.6,7 Anemia and the resulting tumor hypoxia also modulate important signaling pathways that enhance expression of stem-like characters, promote metastasis, and result in chemoresistance.8–10 It has been demonstrated that patients with anemia had inferior treatment outcome in various cancers.11–13 Several clinical studies have also reported that anemia is a poor prognostic factor in head and neck cancer treatment.14–19 The optimal timing for hemoglobin (Hb) measurement to be a prognostic indicator was not established.20 The reported studies measured Hb at various time points and in different treatment settings. Moreover, only a few reports focused on patients receiving the most intense therapy, postoperative CCRT, which is associated with a higher anemia occurrence.17,21
We reported a consecutive cohort of stage III/IV SCCHN patients who received postoperative CCRT. Preoperative Hb (pre-op Hb), pre-CCRT Hb, and nadir Hb during CCRT were recorded and analyzed. Other well-established prognostic factors, such as the pathologic features of the tumor and RT-related variables, were also evaluated.
Materials and methods
Patient eligibility
This study protocol was approved by the hospital’s institutional review board. In this retrospective study, we included patients with newly diagnosed nonmetastatic squamous cell carcinoma of the oral cavity, oropharynx, hypopharynx, or larynx. Patients with a history of radiation to the head and neck region or any chemotherapy prior to the study were excluded. Patients having bone marrow dysfunction from non-cancer diseases or other hematologic diseases were not eligible for this study. All surviving patients had a minimum follow-up of 2 years. All patients received surgery with curative intent and completed the planned RT doses with concurrent chemotherapy. Each tumor was staged according to the 2002 American Joint Committee on Cancer staging classification.
After completing treatments, the follow-up schedule was once every month during the first year, and every 3 months, thereafter. Once recurrence occurred, salvage treatments were offered according to individual patient condition. The medical records of all enrolled patients were reviewed in detail and recorded.
RT
The indications for postoperative adjuvant CCRT were defined by pathologic risk features. CCRT was indicated for patients having one major risk factor, either positive surgical margin or extracapsular nodal extension, or at least two minor pathologic risk factors. The minor pathologic risk factors included T classification (T3/T4), N classification (N2b; multiple positive lymph nodes or higher), close pathologic surgical margin (1–5 mm), perineural invasion, lymphovascular invasion (LVI), and poor histological differentiation of tumor.1,5,22–24
All patients received fractionated RT consisting of five consecutive daily fractions per week with a fraction size of 1.8–2.0 Gy. Radiation was delivered via a linear accelerator using a 6 MV photon beam. Before 2007, the main RT technique was 2D conventional RT with the total dose prescribed ranging between 59.4 Gy and 64.8 Gy (1.8 Gy per fraction) at the main tumor and involved lymph nodes. Thereafter, intensity-modulated RT became the main modality with intent dose of 63 Gy (1.8 Gy per fraction) or 66 Gy (2 Gy per fraction). The RT treatment was mandated to be completed within 7 weeks. The fraction size and total dose were decided at the discretion of treating physician according to each clinical condition. RT dose was prescribed to planning target volumes.
Chemotherapy
Chemotherapy consisted primarily of cisplatin and 5-FU at two dose levels in our institution.25 One comprised cisplatin 12 mg/m2 plus 5-FU 600 mg/m2 per day administered as a 120-hour continuous infusion on week 1 and week 5 during the course of RT. The other dose level was cisplatin 15 mg/m2 plus 5-FU 750 mg/m2 with the same infusion schedule.
In this study, 15 patients received low-dose level cisplatin/5-FU and 105 patients received the high dose-level regimen. Another 20 patients received weekly 30 mg/m2 cisplatin for at least five or more doses during radiation treatment. All three regimens have been reported in literature,17,26–28 and were regularly prescribed in our institution. The Hb level was not a criterion in the selection of chemotherapy regimen, total dose, or cycles being prescribed.
Hb measurement and management
We analyzed Hb levels at three time points. The pre-op Hb and pre-CCRT Hb were measured within 1 week prior to the implemented therapy. Hb was checked weekly during CCRT and the lowest level was defined as the nadir Hb. Anemia was defined as a Hb concentration less than 13 g/dL according to World Health Organization definition. This cut-off value was used for pre-op Hb and pre-CCRT Hb. Since only a few reports contained the nadir Hb,3,15 it was analyzed as a categorical variable. The median nadir Hb (11 g/dL) of the study cohort was chosen as the cut point value for further analysis.
No predefined Hb level was maintained and all anemic situations throughout the treatment course were corrected with red blood cell transfusion at the discretion of the treating physician. No patients received erythropoietic stimulating agents (ESAs) or iron supplement before and during CCRT.
Endpoints and statistical methods
Time to events in this study was counted from the date of surgery to the event occurrence or the end of study, December 31, 2011, whichever came first. Death from any cause accounted for overall survival (OS). Disease recurrence, second primary cancer, and death from any cause were considered events for progression-free survival (PFS). Evidence of locoregional recurrence was specifically recorded as failure to locoregional control (LRC). Patients were recorded as censor if no events occurred until the last follow-up or the end of study.
Survival rates were estimated using the Kaplan–Meier statistical method. Log-rank test was applied to evaluate the possible impact on the OS, PFS, and LRC for age, primary tumor site (oral cavity or non-oral cavity primary tumor), each pathologic risk feature, RT-associated variables, and the three Hb parameters. These factors were also analyzed using the multivariate Cox proportional hazards model to estimate adjusted hazard ratio (HR) and 95% confidence interval (CI). All reported P-values were two-tailed and considered to be statistically significant if P<0.05. All statistical analyses were performed using SPSS version 12.
Results
Patient demographics and general treatment outcome
From January 2002 to December 2009, 140 patients were eligible for the study analysis. The median follow-up time was 33.5 months (interquartile range: 16–65 months). The median age of all patients in this study was 51 years (interquartile range: 44–57 years), including 135 (96.4%) male and 4 (3.6%) female patients. One hundred and 6 patients (75.7%) had their primary tumors located in the oral cavity. The pathologic stage distribution was as follows: stage III =17.9%, stage IVa =71.4%, and stage IVb =10.7%. Positive surgical margin was noted in 22.9% and nodal extracapsular extension in 14.3% of patients. The median total RT dose was 6,480 cGy (interquartile range: 6,300–6,600 cGy). Seventy-four patients (52.9%) received 2D conventional RT and the others were treated by intensity-modulated RT mode. The median time interval between operation and RT (OP–RT interval) was 6.3 weeks (interquartile range: 4.8–8.0 weeks). The median total RT treatment time (RTT) was 7.4 weeks (interquartile range: 6.7–8.3 weeks). The median treatment time between operation and the end of RT (OP–RT package time) was 13.6 weeks (interquartile range: 12.4–15.6 weeks).
The overall 5-year OS, PFS, and LRC were 52.1%, 44.9%, and 73.2%, respectively. During the study period, there were 33 and 37 events of locoregional recurrences and distant metastasis, respectively. The detailed patient demographics and tumor characters are listed in Table 1.
Hb parameters and impacts on outcome
The median pre-op Hb was 14.3 g/dL (interquartile range: 13.1–15.1 g/dL) and median pre-CCRT Hb was 11.9 g/dL (interquartile range: 11.1–12.8 g/dL). We observed that a lower pre-op Hb correlated with a higher N classification and non-oral cavity primary tumor site (Table 2). The median nadir Hb was 10.7 g/dL (interquartile range: 9.4–11.7 g/dL). Eighty-two (58.6%), 31 (22.1%), and 27 (19.3%) patients had the nadir Hb ≦11.0 g/dL, 11.1–12.0 g/dL, and >12.0 g/dL.
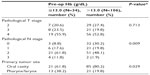
The 5-year OS, PFS, and LRC of patients with a pre-op Hb ≦13 g/dL were 29.1%, 20.7%, and 61.4%, respectively. These results were significantly worse compared to patients in the pre-op Hb >13 g/dL group (OS =59.5%, P<0.001 [Figure 1A]; PFS =51.2%, P<0.001; LRC =77.0%, P=0.019). The corresponding values, however, were not significantly different when comparing pre-CCRT Hb ≦13 g/dL versus >13 g/dL patient groups (Table 1).
Patients whose nadir Hb was ≦11.0 g/dL, between 11.1 g/dL and 12.0 g/dL, and >12.0 g/dL had a 5-year OS of 42.4%, 61.6%, and 70.6%, respectively (P=0.004; Figure 1B). The three groups of patients had a 5-year PFS of 36.6%, 49.1%, and 54.9%, respectively (P=0.012) and a 5-year LRC of 70.0%, 86.4%, and 61.7%, respectively (P=0.224). Although not statistically significant for LRC, patients with nadir Hb below 11 g/dL had more events of locoregional recurrence, distant metastasis, second primary tumors, and non-cancer death (data not shown).
Among the 82 patients with nadir Hb ≦11 g/dL, 21 (25.6%) received red blood cell transfusion during CCRT. The 5-year OS of the 82 patients with and without transfusion was 28.6% and 47.3%, respectively (P=0.095), the PFS was 22.9% and 41.6%, respectively (P=0.166), and the LRC was 64.0% and 71.7%, respectively (P=0.558; Table 1).
Multivariate analyses for treatment outcome OS
A significantly worse OS was observed in patients with non-oral cavity primary tumor (P=0.001), N classification ≧ N2b (P<0.001), and the presence of LVI (P=0.045) (Table 1). With the increasing duration of RTT, which was presumed to be ≦7 weeks, there was a trend of worse OS for patients with RTT 7–8 weeks and >8 weeks (P=0.085; Figure 1C and Table 1). We dichotomized the RTT into ≦7 weeks and >7 weeks for multivariate analysis. After adjusting for other confounders, significant HR for death was observed in patients with N classification ≧ N2b (adjusted HR =2.1; 95% CI, 1.2–3.7), RTT >7 weeks (adjusted HR =1.9, 95% CI, 1.1–3.3), pre-op Hb ≦13 g/dL (adjusted HR =1.8; 95% CI, 1.1–3.1), and nadir Hb ≦11 g/dL (adjusted HR =1.9; 95% CI, 1.1–3.3) (Table 3).
PFS
Patients with non-oral cavity primary tumor (P<0.001), N classification ≧ N2b (P=0.001), and the presence of LVI (P=0.019) had significantly reduced PFS by univariate analysis (Table 1). In multivariate analysis, non-oral cavity primary tumor, RTT >7 weeks, and pre-op Hb ≦13 g/dL were associated with worse PFS (Table 3). Nadir Hb ≦11 g/dL remained a trend toward inferior PFS (adjusted HR =1.6; 95% CI, 0.95–2.6; P=0.079).
LRC
In univariate analysis, non-oral cavity primary tumor (P=0.002) and the presence of LVI (P=0.022) were associated with inferior LRC (Table 1). A trend of worse LRC with prolonged RTT was still observed (P=0.065; Table 1). Only RTT >7 weeks strongly predicted worse LRC using multivariate analysis (Table 3). Patients with non-oral cavity primary tumor and pre-op Hb ≦13 g/dL also had a higher risk of local recurrence.
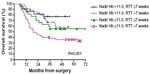
Additive effect of prolonged RTT and nadir Hb
We investigated if there was a possible interaction between prolonged RTT and low nadir Hb, both of which might attenuate CCRT efficacy. We found that patients with both nadir Hb ≦11 g/dL and RTT >7 weeks had a markedly worse 5-year OS (36.0%), PFS (27.8%), and LRC (60.3%), when compared to patients with neither of the two factors (5-year OS =77.3%, P=0.001 [Figure 2]; PFS =68.7%, P=0.002; LRC =85.9%, P=0.026). Patients with both poor prognosticators had risk of death four times higher than those with none of the two factors (HR =4.0; 95% CI, 1.6–10.2; P=0.004).
Discussion
In this study, we found that pre-op Hb ≦13 g/dL and lower nadir Hb during CCRT predicted a poorer outcome among patients with LA-SCCHN treated by postoperative CCRT. The adverse impact of anemia persisted after controlling for several well-accepted pathologic and radiation-related prognostic factors. Anemia proved to be an unfavorable factor in this patient cohort comprising predominantly of oral cancer, which was a quite different demographic compared to other reported series.14–19 However, our retrospective study design and single institutional experience were limitations of this study. Moreover, though all the chemotherapy regimens used in our study have been reported17,25–28 in the literature, this might be a limitation while interpreting and generalizing our results. Though only 8 patients had their primary tumor located at oropharynx, human papillomavirus status analysis was not investigated in this study. Our results should be verified with larger case series or clinical trials.
Only one study evaluated the influence of Hb on patients all of whom received postoperative CCRT.20 Reichel et al investigated the impact of Hb before and 48 hours after surgery, and Hb right before and after CCRT. They found that only Hb measured before surgery and Hb measured 48 hours after surgery were unfavorable factors. However, the study enrolled 12% of patients with stage I/II disease and the Hb parameters were not analyzed with other important pathologic or RT variables which might influence the true impact of anemia in this treatment setting.
Pre-op Hb, defined as pretreatment Hb in other studies, was the most common Hb parameter investigated.14–19,29–33 Our results were consistent with previous findings that low pretreatment Hb predicted a poor prognosis. In our study, we also found that non-oral cavity tumor site and higher N classification were associated with lower pre-op Hb. Only two studies had similar investigation. One revealed that lower pretreatment Hb was related to higher T classification and primary tumor site16 and the other showed an association with older age and poorer performance status.17
In this study, we would like to focus on one specific Hb parameter, nadir Hb during CCRT. It might contribute to tumor hypoxia during CCRT which is an important aspect to cause radioresistance and treatment failure. Our data showed that the lower the nadir Hb (especially for nadir Hb ≦11 g/dL), the worse the OS. Also, it might reflect a poor general condition and nutritional status of patients that non-cancer death increased with nadir Hb ≦11.0 g/dL. The occurrence of second primary malignancy as well increased in this patient group (data not shown). Only two studies investigated this Hb parameter.14,19 Both results confirmed that low nadir Hb was an unfavorable prognostic factor. However, one of the studies enrolled patients who underwent either surgery or RT as the primary treatment14 and the other study did not analyze the nadir Hb against other prognostic contributors.19 In addition, Bhide et al found that patients who received transfusions and who needed more units of blood to maintain Hb >12 g/dL during CCRT had poorer outcomes.19 Our results suggested that performing blood transfusions only when clinically necessary probably might be a rational strategy in maintaining Hb level. However, our data also showed a trend toward inferior survival in those who received blood transfusion. The optimal methods and the target level of Hb to be maintained during head and neck squamous cell carcinoma treatment were not clearly demonstrated.34,35 ESA was once considered promising but has been proven to be of no survival benefits in several clinical trials.36,37 Exogenous ESA might activate an aggressive phenotype of cancer cells through the erythropoietin receptor and its downstream pathway.38,39 On the other hand, practice with blood transfusion to correct anemia also did not improve the outcome.40,41 In contrast to the pretreatment Hb, several factors contributed to anemia during CCRT in our treatment setting, including surgical blood loss, radiation- and chemotherapy-induced anemia, and impaired nutritional status. In our study cohort, neither nasogastric nor gastroenterostomy tube was mandated which might contribute to the severity of anemia observed. We believe nadir Hb during CCRT is a factor deserving more emphasis. Best supportive care should be provided to prevent a low nadir Hb and improve the clinical outcome.
Different RT-related time factors have been investigated to affect treatment outcome in LA-SCCHN, including interval between surgery and RT,42 RT treatment time,43–45 and treatment package time.43,46 We included all three variables and observed that longer RT treatment time was the only factor being associated with inferior outcomes. It has been hypothesized that prolonged RT periods may result in cancer cell repopulation and subsequent treatment failure.47 The causes of prolonged RTT in this study were CCRT-related side effects, such as mucositis, dermatitis, fatigue, or infection. These factors should be solved with more delicate supportive care and patient education. Also, more precise radiation delivery and normal tissue protection might be helpful. Moreover, our data suggest that there may be synergistic effect of the two poor prognostic factors, prolonged RT treatment time and low nadir Hb, both of which might contribute to cancer cell repopulation and radioresistance.
In conclusion, both low nadir Hb during CCRT and prolonged RTT should be avoided in the clinical practice to maximize the efficacy of adjuvant CCRT and improve the treatment outcome in patients with locally advanced head and neck cancer.
Acknowledgment
The authors thank An-chi Lo for her dedication and great help to the statistical assistance.
Disclosure
The authors have declared no conflicts of interest.
References
Bernier J, Domenge C, Ozsahin M, et al; European Organization for Research and Treatment of Cancer Trial 22931. Postoperative irradiation with or without concomitant chemotherapy for locally advanced head and neck cancer. N Engl J Med. 2004;350(19):1945–1952. | |
Bernier J, Cooper JS, Pajak TF, et al. Defining risk levels in locally advanced head and neck cancers: a comparative analysis of concurrent postoperative radiation plus chemotherapy trials of the EORTC (#22931) and RTOG (#9501). Head Neck. 2005;27(10):843–850. | |
Fietkau R, Lautenschlager C, Sauer R, et al. Postoperative concurrent radiochemotherapy versus radiotherapy in high-risk SCCA of the head and neck: results of the German phase III trial ARO 96-3. J Clin Oncol. 2006;24:557. | |
Bachaud JM, Cohen-Jonathan E, Alzieu C, David JM, Serrano E, Daly-Schveitzer N. Combined postoperative radiotherapy and weekly cisplatin infusion for locally advanced head and neck carcinoma: final report of a randomized trial. Int J Radiat Oncol Biol Phys. 1996;36(5):999–1004. | |
Cooper JS, Pajak TF, Forastiere AA, et al. Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous cell carcinoma of the head and neck. N Engl J Med. 2004;350(19):1937–1944. | |
Becker A, Stadler P, Lavey RS, et al. Severe anemia is associated with poor tumor oxygenation in head and neck squamous cell carcinomas. Int J Radiat Oncol Biol Phys. 2000;46(2):459–466. | |
Norsmark M, Overgaard J. Tumor hypoxia is independent of hemoglobin and prognostic for loco-regional tumor control after primary radiotherapy in advanced head and neck cancer. Acta Oncol. 2004;43(4):396–403. | |
Marie-Egyptienne DT, Lohse I, Hill RP. Cancer stem cell, the epithelial to mesenchymal transition (EMT) and radioresistance: potential role of hypxia. Cancer Lett. 2013;341(1):63–72. | |
Chang J, Erler J. Hypoxia-mediated metastasis. Adv Exp Med Biol. 2014;722:55–81. | |
Ghattass K, Assah R, El-Sabban M, Gali-Muhtasib H. Targeting hypoxia for sensitization of tumors to radio- and chemotherapy. Curr Cancer Drug Targets. 2013;13(6):670–685. | |
Kim JH, Lee JM, Ryu KS, et al. The prognostic impact of duration of anemia during chemotherapy in advanced ovarian cancer. Oncologist. 2011;16(8):1154–1161. | |
Ferrandina G, Distefano M, Smaniotto D, et al. Anemia in patients with locally advanced cervical carcinoma administered preoperative radiochemotherapy: association with pathological response to treatment and clinical outcome. Gynecol Oncol. 2006;103(2):500–505. | |
Park SH, Lee J, Lee SH, et al. Anemia is the strongest prognostic factor for outcomes of 5-fluorouracil-based first-line chemotherapy in patients with advanced gastric cancer. Cancer Chemother Pharmacol. 2006;57(1):91–96. | |
Nguyen-Tan PF, Le QT, Quivey JM, et al. Treatment results and prognostic factors of advanced T3-4 laryngeal carcinoma: the university of California, San Francisco (UCSF) and Stanford university hospital (SUH) experience. Int J Radiat Oncol Biol Phys. 2001;50(5):1172–1180. | |
Denis F, Garaud P, Bardet E, et al. Final results of the 94-01 French head and neck oncology and radiotherapy group randomized trial comparing radiotherapy alone with concomitant radiochemotherapy in advanced-stage oropharynx carcinoma. J Clin Oncol. 2004;22(1):69–76. | |
Prosnitz RG, Yao B, Farrell CL, Clough R, Brizel DM. Pretreatment anemia is correlated with the reduced effectiveness of radiation and concurrent chemotherapy in advanced head and neck cancer. Int J Radiat Oncol Biol Phys. 2005;61(4):1087–1095. | |
Rades D, Stoehr M, Kazic N, et al. Locally advanced stage IV squamous cell carcinoma of the head and neck: impact of pre-radiotherapy hemoglobin level and interruption during radiotherapy. Int J Radiat Oncol Biol Phys. 2008;70(4):1108–1114. | |
Fortin A, Wang CS, Vigneault E. Effect of pretreatment anemia on treatment outcome of concurrent radiochemotherapy in patients with head and neck cancer. Int J Radiat Oncol Biol Phys. 2008;72(1):255–260. | |
Bhide SA, Ahmed M, Rengarajan V, et al. Anemia during sequential induction chemotherapy and chemoradiation for head and neck cancer: the impact of blood transfusion on treatment outcome. Int J Radiat Oncol Biol Phys. 2009;73(2):391–398. | |
Hu K, Harrison LB. Impact of anemia in patients with head and neck cancer treated with radiation therapy. Curr Treat Options Oncol. 2005;6(1):31–45. | |
Reichel O, Panzer M, Wimmer C, Duhmke E, Kastenbauer E, Suckfull M. Prognostic implications of hemoglobin levels before and after surgery as well as before and after radiochemotherapy for head and neck tumors. Eur Arch Otorhinolaryngol. 2003;260(5):248–253. | |
Chen TC, Hsu CW, Lou PJ, et al. The clinical predictive factors for subsequent distant metastasis in patients with locoregionally advanced oral squamous cell carcinoma. Oral Oncol. 2013;49(4):367–373. | |
Liao CT, Lee LY, Hsueh C, et al. Comparative outcomes in oral cavity cancer with resected pT4a and pT4b. Oral Oncol. 2013;49(4):230–236. | |
Thomas B, Stedman M, Davies L. Grade as a prognostic factor in oral squamous cell carcinoma: population-based analysis of the data. Laryngoscope. 2014;124(3):688–694. | |
Su NW, Leu YS, Lee JC, et al. Comparison of the efficacy and toxicity of two dose levels of cisplatin/5-fluorouracil as the chemoradiotherapy regimen for the treatment of locally advanced squamous cell carcinoma of the head and neck. Acta Otolaryngol. 2011;131(12):1333–1340. | |
Maguire PD, Meyerson MB, Neal CR, et al. Toxic cure: hyperfractionated radiotherapy with concurrent cisplatin and fluorouracil for stage III and IVA head-and-neck cancer in the community. Int J Radiat Oncol Biol Phys. 2004;58(3):698–704. | |
Rades D, Kronemann S, Meyners T, et al. Comparison of four cisplatin-based radiochemotherapy regimens for nonmetastatic stage III/IV squamous cell carcinoma of head and neck. Int J Radiat Oncol Biol Phys. 2011;80(4):1037–1044. | |
Morganti AG, Mignogna S, Deodato F, et al. Feasibility study of moderately accelerated intensity-modulated radiotherapy plus concurrent weekly cisplatin after induction chemotherapy in locally advanced head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2011;79(4):1073–1080. | |
van de Pol SMG, Doornaert PAH, de Bree R, Rene Leemans C, Slotman BJ, Langendijk JA. The significance of anemia in squamous cell head and neck cancer treated with surgery and postoperative radiotherapy. Oral Oncol. 2006;42(2):131–138. | |
Rutkowski T, Suwinski R, Idasiak A. The prognostic value of hemoglobin concentration in postoperative radiotherapy of 835 patients with laryngeal cancer. Int J Radiat Oncol Biol Phys. 2007;69(4):1018–1023. | |
Rades D, Fehlauer F, Wroblesky J, Albers D, Schild SE, Schimdt R. Prognostic factors in head-and-neck cancer patients treated with surgery followed by intensity-modulated radiotherapy (IMRT), 3D-conformal radiotherapy, or conventional radiotherapy. Oral Oncol. 2007;43(6):535–543. | |
Schafer U, Micke O, Muller SB, Schuller P, Willich N. Hemoglobin as an independent prognostic factor in the radiotherapy of head and neck tumors. Strahlenther Onkol. 2003;179(8):527–534. | |
McCloskey SA, Jaggernauth W, Rigual NR, et al. Radiation treatment interruptions greater than one week and low hemoglobin levels (12g/dL) are predictors of local regional failure after definite concurrent chemotherapy and intensity-modulated radiation therapy for squamous cell carcinoma of the head and neck. Am J Clin Oncol. 2009;32:587–591. | |
Hoff CM. Importance of hemoglobin concentration and its modification for the outcome of head and neck cancer patients treated with radiotherapy. Acta Oncol. 2012;51(4):419–432. | |
Kumar P. Impact of anemia in patients with head and neck cancer. Oncologist. 2000;5(Sup 2):13–18. | |
Henke M, Laszig R, Rube C, et al. Erythropoietin to treat head and neck cancer patients with anemia undergoing radiotherapy: randomized, double-blind, placebo-controlled trial. Lancet. 2003;32(9392):1255–1260. | |
Machtay M, Pajak TF, Suntharalingam M, et al; Radiation Therapy Oncology Group. Radiotherapy with or without erythropoietin for anemia patients with head and neck cancer: a randomized trial of the radiation therapy oncology group (RTOG 99-03). Int J Radiat Oncol Biol Phys. 2007;69(4):1008–1017. | |
Arcasoy MO, Amin K, Chou SC, Haroon ZA, Varia M, Raleigh JA. Erythropoietin and erythropoietin receptor expression in head and neck cancer: relationship to tumor hypoxia. Clin Cancer Res. 2005;11(1):20–27. | |
Winter SC, Shah KA, Campo L, et al. Relation of erythropoietin and erythropoietin receptor expression to hypoxia and anemia in head and neck squamous cell carcinoma. Clin Cancer Res. 2005;11(21):7614–7620. | |
Hoff CM, Hansen HS, Overgaard M, et al. The importance of hemoglobin level and effect of transfusion in HNSCC patients treated with radiotherapy – results from he randomized DAHANCA 5 study. Radiother Oncol. 2011;98(1):28–33. | |
Hoff CM, Lassen P, Eriksen JG, et al. Dose transfusion improved the outcome for HNSCC patients treated with radiotherapy? – results from the randomized DAHANCA 5 and 7 trial. Acta Oncol. 2011;50(7):1006–1014. | |
Ang KK, Trotti A, Brown BW, et al. Randomized trial addressing risk features and time factors of surgery plus radiotherapy in advanced head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2001;51(3):571–578. | |
Suwinski R, Sowa A, Rutkowski T, Wydmanski J, Tarnawski R, Maciewski B. Time factor in postoperative radiotherapy: a multivariate locoregional control analysis in 868 patients. Int J Radiat Oncol Biol Phys. 2003;56(2):399–412. | |
Sher DJ, Posner MR, Tishler RB, et al. Relationship between radiation treatment time and overall survival after induction chemotherapy for locally advanced head-and-neck carcinoma: a subset analysis of TAX 324. Int J Radiat Oncol Biol Phys. 2011;81(5):e813–e818. | |
Cannon DM, Geye HM, Hartig GK, et al. Increased local failure risk with prolonged radiation time in head and neck cancer treated with concurrent chemotherapy. Head Neck. 2014;36(8):1120–1125. | |
Rosenthal DI, Liu L, Lee JH, et al. Importance of the treatment package time in surgery and postoperative radiation therapy for squamous carcinoma of the head and neck. Head Neck. 2002;24(2):115–126. | |
Marcu LG, Bezak E. Influence of stem-cell cycle time on accelerated re-population during radiotherapy in head and neck cancer. Cell Prolif. 2012;45(5):404–412. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.