Back to Journals » Cancer Management and Research » Volume 13
Prognostic Value of Preoperative Prognostic Nutritional Index and Body Mass Index Combination in Patients with Unresectable Hepatocellular Carcinoma After Transarterial Chemoembolization
Authors Li S, Guo JH , Lu J, Wang C, Wang H
Received 7 November 2020
Accepted for publication 26 January 2021
Published 17 February 2021 Volume 2021:13 Pages 1637—1650
DOI https://doi.org/10.2147/CMAR.S290983
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sanjeev K. Srivastava
Shengwei Li, Jin-He Guo, Jian Lu, Chao Wang, Hao Wang
Department of Interventional Radiology, Zhongda Hospital, Southeast University, Nanjing, 210009, People’s Republic of China
Correspondence: Jin-He Guo
Department of Interventional Radiology, Zhongda Hospital, Southeast University, 87 Dingjiaqiao Road, Nanjing, 210009, People’s Republic of China
Tel +86 18551850103
Email [email protected]
Background: The aim of our study was to validate the value of combined preoperative prognostic nutritional index and body mass index in predicting overall survival (OS) and progression-free survival (PFS) in patients who underwent treatment of transarterial chemoembolization.
Methods: A single-centered retrospective study of 285 unresectable HCC patients who received treatment of transarterial chemoembolization from January 2013 to June 2015 was conducted. In our retrospective analysis, preoperative PNI and BMI data of patients were calculated and analyzed. The data of patient demographic, clinical, pathological and hematological characteristics were also systematically acquired and analyzed.
Results: Low PNI was correlated with cirrhosis, AFP ≥ 200 mg/L, Child-Pugh class B, ALT and AST ≥ 40 IU/L, TBIL≥ 21μmol/L, vascular invasion, tumor size ≥ 5 cm BCLC A/B and TNM III–IV stages (all p < 0.05). Likewise, low BMI was related to AFP ≥ 200 mg/L, Child-Pugh class B, AST ≥ 40IU/L, tumor size ≥ 5 cm, vascular invasion, BCLC C and TNM III–IV stages (all p < 0.05). In our multivariate analysis, AFP levels, tumor size, PNI and BMI were identified as independent predictive factors for OS and PFS of patients (all p < 0.05). Prominently, low PNI combined with low BMI adversely affected the overall survival and disease-free survival and the combination of PNI-BMI scores was proved to be the superior distinguished capacity compared with PNI or BMI alone because of higher area under the curve.
Conclusion: Preoperative PNI and BMI are independent predictors for prognosis in intermediate or advanced hepatocellular carcinoma patients after transarterial chemoembolization treatment. The PNI and BMI combination can promote the accuracy of prognostic prediction for patients with HCC compared with single score.
Keywords: prognostic nutritional index, body mass index, hepatocellular carcinoma, transarterial chemoembolization, prognosis
Introduction
Hepatocellular carcinoma (HCC) is one of the most common malignant tumors and the third leading cause of cancer-related death worldwide.1,2 Currently, more than half of global HCC cases and mortality occurs in China.3 Unlike other malignancies, the high incidence of HCC in China may be attributed to hepatitis B virus (HBV) infection and ethanol consumption.4 Approximately 70% of HCC patients treated with curative treatment including hepatectomy, transplantation and ablation suffer from malignant recurrence and metastasis within 5 years after therapeutic regimens.5 A majority (about 60%) of patients have progressed to an advanced phase or hepatic decompensation at first diagnosis so that curative therapies are inadaptable.6 The treatment of transarterial chemoembolization (TACE) is recommended as the standard first-line therapy in unresectable intermediate and advanced HCC.7 Some evidence showed that the treatment of TACE for prolonging overall survival period was effective for unresectable HCC.8 However, studies demonstrated that there was considerable variation in the clinical benefit patients derive from this treatment.9,10 Hence, it is crucial to determine prognostic factors for patients with HCC, leading to easier personalized therapeutic regimens and better long-term outcomes.
Recently, numerous clinical factors have been investigated and proved to possess predictive values for patient prognosis, such as tumor size, pathological differentiation, tumor invasion and several kinds of molecular histopathological biomarkers.11–14 Nevertheless, no acknowledged consensus on the standard criteria for prognostic prediction in HCC patients exists until now. Recent evidence demonstrates that immune-nutritional status is intimately correlated with prognosis in patients with many malignancies and inflammatory response which can promote tumor growth, invasion, angiogenesis and metastasis.15–17 Moreover, the presence of systematic inflammatory responses is regarded as an important independent risk factor in the occurrence of tumor-related malnutrition, leading to unsatisfactory performance status (PS) scores and higher mortality.18–20
Investigators have showed keen interest in the value of tumor-related inflammatory responses as an indicator of the long-term prognosis in patients with malignances. Apart from objective biomarkers of systemic inflammation, various combinations of immune-nutritional indices related to tumor immune-inflammatory responses have extensively researched. Prognostic nutritional index (PNI), which is based on albumin concentration and total lymphocyte count in the peripheral blood, has been proved as a simple and practical indicator of systemic inflammatory response to assess the immune-nutritional status. Recently, emerging evidence suggests that PNI maintains close ties with tumor genesis and progression in hepatocellular carcinoma.20–22 However, the practical value of PNI predicting the post-TACE prognosis in HCC remains unclear, potentially due to differences in inclusion criteria and sample sizes.21,23 On the other hand, body mass index (BMI), as an immune-nutrition-related index depending on individual height and weight, was generally accepted as a benchmark to assess body weight status and nutritional condition.24 Based on the World Health Organization (WHO) criterion, BMI<18.5kg/m2 is defined as underweight. Theoretically, low-level BMI is proved to be associated with perioperative malnutrition and metabolic disorder, which further affects systemic inflammation responses and immune-nutritional status in patients with HCC.25,26 Recent study showed that PNI combined with BMI could have potential prognostic value for patients undergoing hepatectomy in early stage.27 However, few researchers investigated the prognostic abilities of PNI and BMI to forecast the effects after treatment for unresectable HCC, especially in a prediction model based on combinative use of these two indices.
Hence, the objective of our study was to probe into the prognostic value of PNI and BMI in unresectable HCC and investigate whether preoperative PNI combined with BMI would be an effective and practical predictor for prognosis in the TACE-treated patients with HCC.
Patients and Methods
Patient Baseline Characteristics
A total of 285 patients diagnosed as intermediate or advanced hepatocellular carcinoma receiving TACE as an initial therapy at the Department of Interventional and Vascular Surgery, Zhong-Da Hospital, Southeast University (Nanjing, China) between January 2013 and June 2015 were brought into our study (Figure 1). This study was approved by the Institutional Review Board (IRB) of the Zhong-Da Hospital, Southeast University. The diagnosis of all the HCC patients included was based on the clinical diagnostic criteria recommended by the American Association for the Study of the Liver.5 Within 7 days before the first TACE treatment, a clinical routine examination was conducted, comprising routine physical, hematological and radiological examination.
 |
Figure 1 The flow chart of patient selection. |
In our study, all of the patient hematological, clinical and pathological data were collected and analyzed, including hematological parameters; albumin (ALB), total bilirubin (TBIL), alpha-fetoprotein (AFP), alanine aminotransferase (ALT), and aspartate aminotransferase (AST) levels; tumor staging information (including number of focal lesions and maximum diameter of contrast-enhancing lesions); Child–Turcotte –Pugh class and liver cirrhosis; portal vein thrombosis, Barcelona Clinic Liver Cancer (BCLC) stage and tumor-node-metastasis (TNM) stage.
Besides, preoperative PNI and BMI from all individual participants included were calculated according to the formulas. PNI was calculated using the following formula: 10 × serum albumin (g/dl) + 0.005 × total lymphocyte count (per mm3).20 Specifically, serum albumin and lymphocyte count was obtained from peripheral blood samples within 7 preoperative days. On account of the WHO definition, the score of BMI was operated as below: weight (kg) divided by height (m) squared.24 And it is acknowledged that BMI<18.5 kg/m2 is classified as low BMI. In our study, all patients included were assigned into two groups based on BMI scores: low-BMI group (<18.5 kg/m2) or high-BMI group (≥18.5 kg/m2).
The inclusion criteria were as follows: (1) receiving TACE as initial treatment; (2) Child–Pugh class A or B and BCLC stages A-C; (3) follow-up time ≥1 month and definite feedback during follow-up; (4) no other severe complications such as serious systemic infection and (5) HCC was deemed to be unresectable by HCC multidisciplinary consultation based on clinical guidelines or by patient’s refusal to surgical operation. Conversely, patients were strictly excluded if they accepted other curative therapies for HCC during the period of follow-up or they lost to follow up after TACE.
Treatment and Follow-Up
A unified treatment protocol was implemented to every patient included. After routine preoperative examination, conventional TACE was operated through the femoral artery using the Seldinger technique under local anesthesia. Then, through the arteriography of the celiac trunk and superior mesenteric artery to visualize arterial vascularization of the liver, emulsion of ethiodized oil (Lipiodol Ultra-Fluide; Andre Guerbet Laboratories, France) and doxorubicin (Adriamycin; Shenzhen Main Luck Pharmaceuticals Inc, Shenzhen, China) were injected to treat intrahepatic lesions. And the dosage of doxorubicin depended on total bilirubin level and the volume of ethiodized oil was adjusted on the size, location and haemodynamics of tumor. Subsequently, 300–500 μm Gelfoam embolization particles (Hangzhou ALICON Pharm SCI&TEC CO., LTD, Hangzhou, China) were injected selectively into the intrahepatic segmental artery at the targeting lesions.
After TACE-treatment, all participants were designedly brought into follow-up lists and periodical outpatient reexamination or interview on telephone continuously proceeded until June 2020. The follow-up evaluation included two parts: performance status, biochemistry analysis, tumor markers test per 3 months; radiological examination per 2 months, mainly enhanced CT or MRI scan.
Statistical Analysis
Statistical analysis was performed using the SPSS v 20.0 software (Chicago, IL, USA). The analysis of receiver operating characteristic (ROC) curves based on the patients’ OS status was performed to determine the cut-off values of PNI. The Youden index was calculated to select the optimal cut-off value of PNI, in accordance with the maximal sensitivity and specificity for predicting 3-year OS status in patients. Comparison of continuous normally distributed variables was evaluated by Student t-test and statistical significance of categorical variables was analyzed by chi-square test. Progression-free survival (PFS) time was defined from the date of first TACE treatment to the date of HCC recurrence, and overall survival (OS) time was from the date of first TACE treatment to the date of tumor-related death or endpoint of follow-up. The Kaplan–Meier curves analysis was conducted to compare the differences of the survival in different groups, and the statistical difference in the survival curves was assessed by Log rank test. Univariate and multivariate survival analysis was performed through the Cox proportional hazards regression model. In our statistics, p < 0.05 was identified as statistical significance.
Results
Baseline Characteristics
All eligible cases were 232 men (81.4%) and 53 women (18.6%) with a median age of 59 years (range 31–80 years). The median follow-up duration was 15.9 months (range 1.0–88.5 months). During follow-up, 259 patients (90.9%) died. In all patients included, 92 (32.3%) were defined as low BMI (<18.5kg/m2), 174 (61.1%) presented hepatitis B surface antigen (HBsAg) positive, 125 (43.9%) suffered from liver cirrhosis and 201 (70.5%) had elevated AFP levels (≥200 mg/L). Mostly, 181 (63.5%) patients still had preserved liver function (Child-Pugh class A). On the grounds of clinical and imaging evidence, 209 patients (73.3%) had tumors with the maximum diameter ≥5cm and 229 (80.4%) had multiple tumor masses. Besides, vascular invasion occurred in 63 patients (22.1%). The BCLC stages were A/B in 201 (70.5%) and C in 84 (29.5%) cases. Similarly, the TNM stages were I–II in 90 (31.6%) and III–IV in 195 (68.4%) patients. Overall, the basic data of hematological parameters, including absolute lymphocyte count, albumin (ALB), total bilirubin (TBIL), alanine aminotransferase (ALT), aspartate aminotransferase (AST), prothrombin time (PT) and prognostic nutritional index (PNI), are shown in Table 1.
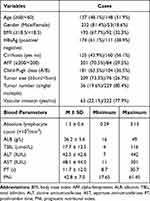 |
Table 1 Clinicopathological Characteristics of 285 Patients |
Cut-Off Value of PNI
A ROC analysis depending on survival time was carried out to select the cut-off value of PNI. With the maximum Youden index 0.503, the optimal cut-off value was 45 (44.975, accurately). Consistently, 45 represents maximal sensitivity and specificity of the PNI for predicting 3-year overall survival in ROC curve analysis (Figure 2). Subsequently, all patients were divided into two groups: PNI-high (PNI>45) group and PNI-low (PNI≤45) group.
 |
Figure 2 The optimal cut-off value of PNI on the receiver operating characteristic (ROC) curve to predict the overall survival. |
Correlation Between PNI or BMI and Clinicopathologic Characteristics of Patients
In our study, 14 clinicopathological characteristics of HCC patients were analyzed to explore the correlation between PNI or BMI and these patient characteristics. Theoretically, high PNI (>45) was more frequent in patients with Child-Pugh class A (p<0.001), ALT and AST<40 IU/L (p=0.001 and p<0.001, respectively), TBIL<21μmol/L (p=0.012), no vascular invasion (p<0.001), BCLC A/B stage (p < 0.001). On the contrary, low PNI (≤45) was more easily to be observed in patients with cirrhosis (p<0.001), AFP ≥ 200 mg/L (p<0.001), tumor size ≥5 cm (p=0.019) and TNM III–IV stages (p<0.001). Similarly, low BMI (<18.5kg/m2) was positively related to AFP ≥ 200 mg/L (p = 0.001), Child-Pugh class B (p < 0.001), AST ≥ 40IU/L (p = 0.006), tumor size ≥5 cm (p=0.031) and TNM III–IV stage (p < 0.001). Oppositely, high BMI (≥18.5kg/m2) frequently occurred in patients with BCLC A/B stages (p < 0.001) and no vascular invasion (p < 0.001) (Table 2). Besides, there was a close correlation between PNI and BMI (p < 0.001) (Table 3).
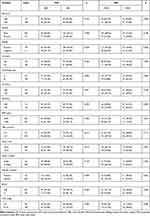 |
Table 2 Correlation Between PNI or BMI and Clinicopathologic Variables |
 |
Table 3 Correlation Between PNI and BMI in HCC Patients |
Univariate and Multivariate Analysis of Prognostic Factors
In order to identify the prognostic risk factors affecting postoperative OS and PFS, we conducted univariate and multivariate analysis on PNI, BMI and a number of possible factors in clinic. Univariate analysis revealed that AFP levels (p<0.001), Cirrhosis (p=0.023<0.05), Child-Pugh class (p<0.001), AST levels (p<0.001), TBIL (p=0.032<0.05), tumor size and number, vascular invasion, BCLC and TNM stage (all p<0.001) were significant associated with OS in HCC patients after TACE. Also, OS was found to associate with PNI and BMI (both p<0.001). Meanwhile, the analysis identified AFP levels (p<0.001), Cirrhosis (p=0.044<0.05), Child-Pugh class (p<0.001), AST levels (p=0.001), tumor size and number, vascular invasion, BCLC stage, TNM stage (all p<0.001), PNI (p=0.001) and BMI (p<0.001) as significant prognostic factors for PFS (Table 4).
 |
Table 4 Prognostic Factors for OS and PFS by Univariate Analysis |
Through the multivariate Cox proportional hazards regression model, four parameters including AFP levels, tumor size, PNI and BMI, were independently predictive factors for both OS and PFS. Specifically, multivariate analysis indicated that patient HbsAg (HR=1.353; 95% CI: 1.020–1.793; p = 0.036), AFP levels (HR=0.574; 95% CI: 0.421–0.782; p = 0.000), tumor size (HR=0.537; 95% CI: 0.330–0.873; p = 0.012), BCLC stage (HR=2.427; 95% CI: 1.429–4.123; p = 0.001), PNI (HR=0.963; 95% CI: 0.942–0.985; p=0.001) and BMI (HR=2.671; 95% CI: 1.967–3.628; p <0.001) were independent prognostic factors for OS. By contrast, AFP levels (HR=0.599; 95% CI: 0.447–0.802; p =0.001), cirrhosis (HR=1.442; 95% CI: 1.003–2.073; p =0.048), Child-Pugh class (HR=1.985; 95% CI: 1.220–3.232; p =0.036), tumor size (HR=0.560; 95% CI: 0.348–0.901; p =0.017), tumor number (HR=1.641; 95% CI: 1.112–2.421; p =0.013), vascular invasion (HR=0.354; 95% CI: 0.193–0.648; p =0.001), PNI (HR=0.962; 95% CI: 0.942–0.983; p<0.001) and BMI (HR=2.251; 95% CI: 1.676–3.024; p <0.001) were considered as prognostic factors for PFS (Table 5).
 |
Table 5 Prognostic Factors for OS and PFS by Multivariate Analysis on Cox Proportional Hazards Regression Model |
Survival Analysis Based on PNI or BMI Alone
The PNI-high group (PNI>45) in contrast to the PNI-low group (PNI ≤ 45) showed observably higher 1-year, 3-year, 5-year OS rates (86.8%, 55.3%, 24.6% vs 42.1%, 12.3%, 4.7%, respectively, p<0.001) (Figure 3A) and 1-year, 3-year, 5-year PFS rates (67.5%, 38.6%, 10.5% vs 24.0%, 7.0%, 2.3%, respectively, p<0.001) (Figure 3B). Obviously, results of survival analysis demonstrated a strong correlation between lower PNI scores and worse survival and progression-free period in HCC patients undergoing TACE treatment.
Similarly, compared with BMI-low group (BMI<18.5), the BMI-high group (BMI≥18.5) had better 1-year, 3-year, 5-year OS rates (72.0%, 40.9%, 18.7% vs 34.8%, 5.4%, 0.0%, respectively, p<0.001) (Figure 3C) and 1-year, 3-year, 5-year PFS rates (56.0%, 26.9%, 7.8% vs 10.9%, 4.3%, 1.1%, respectively, p<0.001) (Figure 3D). Therefore, it is suggested that low BMI was correlated with poor OS and PFS in patients with HCC.
Survival Analysis Based on the PNI-BMI Combination
To explore the prognostic value of PNI and BMI combination in patients undergoing TACE therapy, all patients were assigned into 4 groups on the basis of different PNI-BMI combination: Group 1, PNI-high and BMI-high; Group 2, PNI-high and BMI-low; Group 3, PNI-low and BMI-high; Group 4, PNI-low and BMI-low. Among all 285 patients included, 90 (31.6%), 24 (8.4%), 103 (36.1%) and 68 (23.9%) patients were allocated to Group 1, 2, 3 and 4, respectively. Overall, Group 1 overwhelmingly occupied the first place and Group 4 had the fourth of best OS and PFS rates (Table 6).
 |
Table 6 Comparison of OS and PFS in Different PNI-BMI Combination Groups |
The median survival time for all cases was 15.9 months (range, 1.1–89.4 months). After overall survival analysis, patients in Group 1 (compared with Group 2) had the most favorable outcomes, with higher 1-, 3-, and 5-year OS rates (91.1%, 66.7%, 31.1%, vs 70.8%, 12.5%, 0%, p<0.001) and PFS rates (78.9%, 45.6%, 12.2% vs 25.0%, 12.5%, 4.2%, p<0.001). Likewise, Group 1 (compared with Group 3) showed better 1-, 3-, and 5-year OS rates (91.1%, 66.7%, 31.1%, vs 55.3%, 18.4%, 7.8%, p< 0.001) and PFS rates (78.9%, 45.6%, 12.2% vs 35.9%, 10.7%, 3.9%, p<0.001). Also, OS and PFS rates were dramatically superior in Group 1 in contrast with Group 4 (1-, 3-, and 5-year OS: 91.1%, 66.7%, 31.1%, vs 22.1%, 2.9%, 0%; 1-, 3-, and 5-year PFS: 78.9%, 45.6%, 12.2% vs 5.9%, 1.5%, 0%, both p<0.001) (Table 6) (Figure 4).
Moreover, Group 4 took the lowest position in 1-, 3-, and 5-year OS rates (22.1%, 2.9%, 0%, respectively) and PFS rates (5.9%, 1.5%, 0%, respectively) among all groups (all p<0.001). However, OS and PFS rates in Group 2 and Group 3 had no significant difference (p>0.05). Collectively, it is suggested that the high PNI-BMI combination score is of greater possibility to have a better overall and progression-free survival (Figure 4).
Comparison of the Areas Under the ROC Curves
In order to confirm the predictive power of two indices combination, we calculated the areas under the ROC curves (AUC) of survival status at 1-year, 3-year and 5-year follow-up period. As shown in Table 7, the PNI-BMI combination had higher area under the curves of 1-year, 3-year and 5-year overall survival rates than PNI or BMI alone. Compared with PNI (0.742, 0.768,0.737) and BMI (0.670,0.687,0.685), PNI-BMI combination had higher AUC (0.790, 0.834, 0.820, respectively, all p<0.001). Undoubtedly, all AUC were greater than 0.5, which means effectiveness. Consistently, PNI-BMI visually had a splendid distinguished capacity in comparison with PNI or BMI alone (Figure 5).
 |
Table 7 Comparison of the AUC Values Between the Inflammation-Based Index and Score |
Discussion
According to the clinical practice guidelines, TACE serves as the first choice for patients diagnosed as BCLC-B stage HCC.6,7 Nevertheless, in most practical cases, some BCLC-C patients with preserved liver function also take TACE therapy to delay the advancement of tumor and remit some symptoms.7 Population suffering from unresectable HCC is composed of a large heterogeneous classification including different tumor staging systems and a large scale of immune-nutritional states and disease burdens, and undoubtedly exhibits variable outcomes as well. Hence, we convinced that novel and more precise prognostic indices are urgently essential to improve the prognostic prediction in patients suffering from unresectable HCC after TACE treatment.
Though TACE is the mainstay of palliative modalities for patients with unresectable HCC, a series of postoperative changes in metabolism and immunity are inevitable possibly leading to complications and tumor progression. In addition, malnutrition is also a prognostic factor in patients with HCC on account of concomitant hypohepatia or cirrhosis. Theoretically, immunological and malnutrition status are regarded as indicators for prognosis after TACE.25,28–31
As an immune-nutritional-related score, PNI was originally proposed to assess the perioperative status and surgical risks in patients undergoing gastro-intestinal surgery.16,32 Recently, PNI has been used to evaluate the immune-nutritional states of patients with a variety of malignancies, including HCC.16,20,32–34 Pinato et al20 who first proposed the PNI score, reported that a lower PNI score is a novel, independent and externally validated predictor of poor OS in HCC. Meanwhile, Chan et al34 demonstrated that PNI was a significant prognostic predictor of both OS and PFS in patients diagnosed as very early/early stage receiving curative therapy. However, He et al18 indicated that PNI were less valuable predicting system for TACE-treated patients. Similarly, Kinoshita et al35 suggested that there was no correlation between PNI and outcomes of patients with HCC.
Likewise, BMI is another immune-nutritional-related index, which suggests state of nutrition and immunologic function greatly affecting outcomes of patients. In other words, low BMI does not only suggest poor nutrition status but also frequently couples with decreased lymphocyte count levels on behalf of immunosuppression. Furthermore, concomitant underlying liver function lesion might reduce anti-tumor and anti-metastasis function of immune system. For example, Okamura et al26 confirmed that low BMI had a higher risk of postoperative complications and mortality in patients after hepatic resection for HCC. However, although malnutrition is proved to possess predictive power, Schutte et al25 believed that BMI was not sensitive enough to independently predict the outcomes of HCC patients. In general, the evidences collected so far cannot convincingly support BMI as an independent predictive factor of patient outcomes and tumor prognosis.
As the predictive value of these two indices are relatively weak and in need of strengthening, we explored whether a combination of these clinical scores could increase the accuracy of prognostic predictions and optimize the selection of patients who would benefit most from TACE. In addition, Ji et al27 believed that PNI combined with BMI could have potential prognostic value in early HCC patients after hepatectomy.
In our study, the cut-off value of the PNI was 45, with the peaked Youden index. In addition, both of PNI and BMI were found to be negatively correlated with AFP, Child-Pugh class, AST, tumor size, vascular invasion, TNM and BCLC stage. Further, PNI was also proved to be relevant to ALT and TBIL. Univariate analysis identified that AFP, cirrhosis, Child-Pugh class, AST, tumor size and number, vascular invasion, TNM and BCLC stage, PNI and BMI were significant prognostic factors for both OS and PFS. Nevertheless, multivariate analysis identified AFP levels, tumor size, PNI and BMI as independent prognostic factors in TACE-treated patients. After 285 patients were divided into four groups based on PNI-BMI combination, it is obvious that group with higher united PNI-BMI scores has progressively better prognosis in comparison with lower combination groups, and the differences were largely significant. Moreover, compared with PNI or BMI alone, combination of two scores could better reflect the systemic inflammatory response and immune-nutritional status for patients receiving TACE. As the AUC analysis show, the performances of PNI and BMI alone were overtaken by the effectivity of integrated PNI and BMI for predicting the OS. These data strongly support that combination of PNI and BMI can effectively enhance the accuracy of prognosis prediction.
Through our efforts, the issue that PNI or BMI alone was less accurate as a predictive factor was effectively solved by the combination of two scores. To our knowledge, our study is the first to assess the prognostic predictive capacity of combined PNI and BMI score in patients with intermediate or advanced HCC undergoing TACE. Simultaneously, it was demonstrated that low PNI-BMI combination was more frequently observed in patients with poor prognosis. On the contrary, higher PNI-BMI combination often indicates longer survival and slower tumor progression. We found that preoperative PNI and BMI had great potential to predict the outcomes of patients after TACE and combination of them can increase the predictive values. In the future, the simple and inexpensive preoperative assessment based on the PNI-BMI combination could be used to evaluate before surgery and ensure patients to receive the most appropriate therapy.
The limitation of our study should be taken into account. First, our study was retrospective and single-centered. And the sample size was relatively small which might affect the accuracy of our assessment. Second, a very tiny proportion of patients received some systemic treatment such as targeted immunotherapy, chemotherapy and traditional Chinese medicine after TACE. It was likely to produce minimal bias on results though we avoided and balanced bias as far as possible. TACE-treated patients were enrolled and the number and frequency of TACE treatment for patients were diverse, possibly causing bias. As our study is retrospective, we cannot deny the intrinsic limitations of our current study and further prospective study is needed. Third, numerous other biomarkers, including C-reactive protein, modified Glasgow Prognostic Score (mGPS), neutrophil-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), were not included in our study. Further researches are essential to determine which marker or different combination of markers could show the top performance to improve the prognostic ability.
Conclusion
Conclusively, the PNI-BMI combination can greatly improve the accuracy and efficiency of predicting prognosis in patients. Moreover, a higher PNI-BMI combination predicted a better outcome of patients while a lower PNI-BMI was more likely to indicate poor prognosis. As shown in our study, the PNI-BMI combination could serve as a novel, available and inexpensive immune-nutritional-based index to predict prognosis of unresectable HCC patients undergoing TACE in routine clinical practice.
Abbreviation
HCC, hepatocellular carcinoma; PNI, prognostic nutritional index; BMI, body mass index; TACE, transarterial chemoembolization.
Ethics Approval and Data Confidential Statement
Due to the retrospective nature of our study, patient informed consent was waived. This study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board (IRB) of the Zhong-Da Hospital, Southeast University. All patient data and personal information in our study will be confidential. When the results of our study are published, no information about patients will be disclosed. Our study complied with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Acknowledgments
All authors mentioned made a significant contribution to this study, whether that was in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was supported by the National Natural Science Foundation of China (81971716) and the Natural science foundation of jiangsu province (SBK20190350). Funding source had no involvement in the financial support for the conduct of the research and preparation of the article.
Disclosure
The authors state that there is no conflict of interest.
References
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69–90. doi:10.3322/caac.20107
2. El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132(7):2557–2576. doi:10.1053/j.gastro.2007.04.061
3. Peng W, Li C, Zhu WJ, et al. Prognostic value of the platelet to lymphocyte ratio change in liver cancer. J Surg Res. 2015;194(2):464–470. doi:10.1016/j.jss.2014.12.021
4. Schuppan D, Afdhal NH. Liver cirrhosis. Lancet. 2008;371(9615):838–851. doi:10.1016/S0140-6736(08)60383-9
5. Bruix J, Sherman M. Practice guidelines committee AAftSoLD. Management of hepatocellular carcinoma. Hepatology. 2005;42(5):1208–1236.
6. European Association For The Study Of The L, European Organisation For R, treatment of C. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2012;56(4):908–943.
7. Vogel A, Cervantes A, Chau I, et al. Correction to: “hepatocellular carcinoma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up”. Ann Oncol. 2019;30(5):871–873. doi:10.1093/annonc/mdy510
8. Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: chemoembolization improves survival. Hepatology. 2003;37(2):429–442. doi:10.1053/jhep.2003.50047
9. Bolondi L, Burroughs A, Dufour JF, et al. Heterogeneity of patients with intermediate (BCLC B) hepatocellular carcinoma: proposal for a subclassification to facilitate treatment decisions. Semin Liver Dis. 2012;32(4):348–359. doi:10.1055/s-0032-1329906
10. Piscaglia F, Ogasawara S. Patient selection for transarterial chemoembolization in hepatocellular carcinoma: importance of benefit/risk assessment. Liver Cancer. 2018;7(1):104–119. doi:10.1159/000485471
11. Kabir T, Ye M, Mohd Noor NA, Woon W, Junnarkar SP, Shelat VG. Preoperative neutrophil-to-lymphocyte ratio plus platelet-to-lymphocyte ratio predicts the outcomes after curative resection for hepatocellular carcinoma. Int J Hepatol. 2019;2019:4239463. doi:10.1155/2019/4239463
12. Ji F, Fu SJ, Shen SL, et al. The prognostic value of combined TGF-beta1 and ELF in hepatocellular carcinoma. BMC Cancer. 2015;15:116. doi:10.1186/s12885-015-1127-y
13. Fu SJ, Qi CY, Xiao WK, Li SQ, Peng BG, Liang LJ. Glypican-3 is a potential prognostic biomarker for hepatocellular carcinoma after curative resection. Surgery. 2013;154(3):536–544. doi:10.1016/j.surg.2013.02.014
14. Kim DY, Paik YH, Ahn SH, et al. PIVKA-II is a useful tumor marker for recurrent hepatocellular carcinoma after surgical resection. Oncology. 2007;72(Suppl 1):52–57. doi:10.1159/000111707
15. Du XJ, Tang LL, Mao YP, et al. Value of the prognostic nutritional index and weight loss in predicting metastasis and long-term mortality in nasopharyngeal carcinoma. J Transl Med. 2015;13:364. doi:10.1186/s12967-015-0729-0
16. Mohri Y, Inoue Y, Tanaka K, Hiro J, Uchida K, Kusunoki M. Prognostic nutritional index predicts postoperative outcome in colorectal cancer. World J Surg. 2013;37(11):2688–2692. doi:10.1007/s00268-013-2156-9
17. Lee DY, Hong SW, Chang YG, Lee WY, Lee B. Clinical significance of preoperative inflammatory parameters in gastric cancer patients. J Gastric Cancer. 2013;13(2):111–116. doi:10.5230/jgc.2013.13.2.111
18. He CB, Lin XJ. Inflammation scores predict the survival of patients with hepatocellular carcinoma who were treated with transarterial chemoembolization and recombinant human type-5 adenovirus H101. PLoS One. 2017;12(3):e0174769. doi:10.1371/journal.pone.0174769
19. Huang Z-L, Luo J, Chen M-S, Li J-Q, Shi M. Blood neutrophil-to-lymphocyte ratio predicts survival in patients with unresectable hepatocellular carcinoma undergoing transarterial chemoembolization. J Vasc Interv Radiol. 2011;22(5):702–709. doi:10.1016/j.jvir.2010.12.041
20. Pinato DJ, North BV, Sharma R. A novel, externally validated inflammation-based prognostic algorithm in hepatocellular carcinoma: the prognostic nutritional index (PNI). Br J Cancer. 2012;106(8):1439–1445. doi:10.1038/bjc.2012.92
21. Yamamura K, Sugimoto H, Kanda M, et al. Comparison of inflammation-based prognostic scores as predictors of tumor recurrence in patients with hepatocellular carcinoma after curative resection. J Hepatobiliary Pancreat Sci. 2014;21(9):682–688. doi:10.1002/jhbp.114
22. Liu C, Li L, Lu WS, et al. Neutrophil-lymphocyte ratio plus prognostic nutritional index predicts the outcomes of patients with unresectable hepatocellular carcinoma after transarterial chemoembolization. Sci Rep. 2017;7(1):13873. doi:10.1038/s41598-017-13239-w
23. Ni XC, Yi Y, Fu YP, et al. Prognostic value of the modified Glasgow prognostic score in patients undergoing radical surgery for hepatocellular carcinoma. Medicine. 2015;94(36):e1486. doi:10.1097/MD.0000000000001486
24. Consultation WHOE. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363(9403):157–163. doi:10.1016/S0140-6736(03)15268-3
25. Schutte K, Tippelt B, Schulz C, et al. Malnutrition is a prognostic factor in patients with hepatocellular carcinoma (HCC). Clin Nutr. 2015;34(6):1122–1127. doi:10.1016/j.clnu.2014.11.007
26. Okamura Y, Maeda A, Matsunaga K, Kanemoto H, Uesaka K. Negative impact of low body mass index on surgical outcomes after hepatectomy for hepatocellular carcinoma. J Hepatobiliary Pancreat Sci. 2012;19(4):449–457. doi:10.1007/s00534-011-0461-y
27. Ji F, Liang Y, Fu S, et al. Prognostic value of combined preoperative prognostic nutritional index and body mass index in HCC after hepatectomy. HPB (Oxford). 2017;19(8):695–705. doi:10.1016/j.hpb.2017.04.008
28. Souza Cunha M, Wiegert EVM, Calixto-Lima L, Oliveira LC. Relationship of nutritional status and inflammation with survival in patients with advanced cancer in palliative care. Nutrition. 2018;51–52:98–103. doi:10.1016/j.nut.2017.12.004
29. Rahman R, Hammoud GM, Almashhrawi AA, Ahmed KT, Ibdah JA. Primary hepatocellular carcinoma and metabolic syndrome: an update. World J Gastrointest Oncol. 2013;5(9):186–194. doi:10.4251/wjgo.v5.i9.186
30. McMillan DC. Systemic inflammation, nutritional status and survival in patients with cancer. Curr Opin Clin Nutr Metab Care. 2009;12(3):223–226. doi:10.1097/MCO.0b013e32832a7902
31. Colotta F, Allavena P, Sica A, Garlanda C, Mantovani A. Cancer-related inflammation, the seventh hallmark of cancer: links to genetic instability. Carcinogenesis. 2009;30(7):1073–1081. doi:10.1093/carcin/bgp127
32. Tokunaga R, Sakamoto Y, Nakagawa S, et al. Prognostic nutritional index predicts severe complications, recurrence, and poor prognosis in patients with colorectal cancer undergoing primary tumor resection. Dis Colon Rectum. 2015;58(11):1048–1057. doi:10.1097/DCR.0000000000000458
33. Hong S, Zhou T, Fang W, et al. The prognostic nutritional index (PNI) predicts overall survival of small-cell lung cancer patients. Tumour Biol. 2015;36(5):3389–3397. doi:10.1007/s13277-014-2973-y
34. Chan AW, Chan SL, Wong GL, et al. Prognostic Nutritional Index (PNI) predicts tumor recurrence of very early/early stage hepatocellular carcinoma after surgical resection. Ann Surg Oncol. 2015;22(13):4138–4148. doi:10.1245/s10434-015-4516-1
35. Kinoshita A, Onoda H, Imai N, et al. Comparison of the prognostic value of inflammation-based prognostic scores in patients with hepatocellular carcinoma. Br J Cancer. 2012;107(6):988–993. doi:10.1038/bjc.2012.354
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



