Back to Journals » Therapeutics and Clinical Risk Management » Volume 16
Prognostic Value of Neutrophil-to-Lymphocyte Ratio in Predicting Death Risk in Patients with Severe Hand, Foot and Mouth Disease
Authors Li Y, Wang M, Wang W, Feng D, Deng H, Zhang Y, Dang S, Zhai S
Received 23 June 2020
Accepted for publication 1 October 2020
Published 23 October 2020 Volume 2020:16 Pages 1023—1029
DOI https://doi.org/10.2147/TCRM.S268130
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Yaping Li,1 Muqi Wang,1 Wenjun Wang,1 Dandan Feng,1 Huiling Deng,1,2 Yufeng Zhang,2 Shuangsuo Dang,1 Song Zhai1
1Department of Infectious Diseases, Xi’an Jiaotong University Second Affiliated Hospital, Xi’an 710004, People’s Republic of China; 2Department of Infectious Diseases, Xi’an Children’s Hospital, Xi’an 710003, People’s Republic of China
Correspondence: Shuangsuo Dang; Song Zhai
Department of Infectious Diseases, Xi’an Jiaotong University Second Affiliated Hospital, Xi’an 710004, People’s Republic of China
Tel/Fax +86-29-87679688
; +86 18991307953
Fax +86 29-87679273
Email [email protected]; [email protected]
Introduction: Severe hand, foot, and mouth disease (HFMD) may lead to serious complications, which cause child mortality during outbreaks. The aim of this study was to determine whether neutrophil-to-lymphocyte ratio (NLR) can predict death risk in severe HFMD.
Methods: Medical records for 664 severe HFMD patients were retrospectively examined, and NLR was calculated from blood counts. Youden’s index was calculated to determine the optimal NLR cutoff. Uni- and multivariate logistic regression were used to determine death risk factors associated with severe HFMD.
Results: An NLR cutoff value of 2.01 and 2.50 respectively predicted mortality among all 664 severe HFMD and 137 critical HFMD. Among all 664 patients, the multivariate model identified the following as independently associated with death risk: high fever (OR 3.342, 95% CI 1.736– 6.432), EV71 infection (OR 3.200, 95% CI 1.529– 6.698), fasting glucose (OR 37.343, 95% CI 18.616– 74.909), and NLR (> 2.01) (OR 2.142, 95% CI 1.125– 4.079). Among 137 critical HFMD, EV71 infection (OR 3.441, 95% CI 1.132– 10.462), fasting glucose (OR 14.173, 95% CI 4.920– 40.827), and NLR (> 2.50) (OR 4.166, 95% CI 1.570– 11.051) were associated with death risk.
Conclusion: In conclusion, NLR (> 2.01) in severe HFMD and NLR (> 2.50) in critical HFMD patients may be associated with increased death risk.
Keywords: enterovirus 71, hand, foot and mouth disease, neutrophil-to-lymphocyte ratio, risk factor
Introduction
Hand, foot and mouth disease (HFMD) is a common acute infectious disease caused by a variety of human enterovirus. It usually affects infants and children and typically involves fever, painful mouth sores and a skin rash, among other symptoms.1 Most children with HFMD have mild symptoms, but some children with severe disease can suffer life-threatening complications, such as encephalitis, acute pulmonary oedema and cardiopulmonary failure. HFMD in such patients can progress rapidly with high morbidity and mortality.2–4
Most HFMD patients are given symptomatic treatments and effective therapies are lacking. From 2008 to 2019, 24 million HFMD infections in children were reported in China, approximately 45% of which were associated with enterovirus 71 (EV71). This virus accounted for nearly all 3735 fatal infections during that period, based on data from the National Health Commission (www.chinacdc.cn). HFMD outbreaks and the associated high mortality have caused an important public health problem in China.5,6 It is therefore important to search for factors that can be assessed at a patient’s first clinical visit to predict whether he or she will develop severe or life-threatening illness. This would aid clinical decision-making and disease management.
Markers of inflammation may be useful for predicting prognosis of HFMD patients, since children with severe HFMD show perturbations of cellular and humoral immunity. One candidate marker is the neutrophil-to-lymphocyte ratio (NLR), which is measured routinely in peripheral blood and serves as an index of systemic inflammation.7 The NLR is calculated by dividing the neutrophil count by the lymphocyte count.8 NLR is already used as an easily accessible inflammatory marker in adults with acute respiratory distress syndrome,9 spontaneous bacterial peritonitis,10 ischaemic heart disease, metabolic disease, cancer and other medical conditions.11 In children, NLR can differentiate between viral and bacterial pneumonia,12,13 and it can be a useful diagnostic marker of acute appendicitis and acute infection.12,14 NLR may also correlate positively with other commonly used inflammatory markers.
Here we conducted what appears to be the first investigation of whether NLR has prognostic potential in HFMD. In this large retrospective study, we analysed the relationship of NLR with severe HFMD-associated death.
Materials and Methods
Study Population
This retrospective study analysed medical records of 664 Chinese patients with severe HFMD who were admitted to Xi’an Jiaotong University Second Affiliated Hospital or to Xi’an Children’s Hospital between January 2012 and October 2016. Among the 664 patients with severe HFMD, 137 were diagnosed with critical HFMD and 97 died. This study was approved by the Medical Ethics Committees at both hospitals.
Patients were diagnosed with HFMD using the Hand, Foot and Mouth Disease Clinical Guide (2010 edition) issued by the National Health Commission of the People's Republic of China.15 These guidelines define HFMD in terms of symptoms and signs occurring during epidemics in preschool children, as well as the presence of typical exanthema on the hands, feet, mouth and/or buttocks, with or without fever. Patients were diagnosed with mild HFMD if they had rashes on their hands, feet, mouths, and/or buttocks, with or without fever. Patients were diagnosed with severe HFMD if they had neurological, respiratory or circulatory manifestations. Patients were diagnosed with critical HFMD if they showed (1) frequent convulsions, coma or cerebral hernia; (2) dyspnoea, cyanosis, bloody frothy sputum, or pulmonary rales; or (3) shock and circulatory failure. We have excluded from other underlying diseases.
Data Collection
The ethics committees of the hospitals approved the protocol for the study and all patient data accessed complied with relevant data protection and privacy regulations. Detailed demographic data including age, sex and clinical characteristics were independently extracted from medical records by three authors (LYP, ZS, DHL). Data on clinical manifestations were collected, which included peak fever temperature; vomiting; and neurological, respiratory, and circulatory manifestations. Laboratory data at the time of admission were collected on enterovirus infection, procalcitonin (PCT), white blood cell (WBC) count, fasting glucose, and NLR.
Statistical Analysis
All statistical analyses were performed using SPSS 17.0 (IBM, Chicago, IL, USA). Continuous variables were summarised as mean ± SD or median (range), and differences were assessed for significance using the Wilcoxon rank-sum test or Student’s t test. Categorical variables were summarised as numbers and percentages, and differences between cases (patients who died from HFMD) and controls (patients who survived) were assessed for significance using the χ2 test. Patients who survived served as the reference group for calculating odds ratios (ORs) and associated 95% confidence intervals (95% CIs). Univariate and multivariate logistic regression analyses were used to identify death risk factors associated with severe or critical HFMD. Receiver operating characteristic (ROC) curves were generated to define optimal NLR cutoff values. Significance for all statistical tests was defined to be P < 0.05.
Results
Patient Characteristics and Risk Factors for Severe HFMD
This study involved 664 patients diagnosed with severe HFMD (Table 1), ranging in age from 6 months to 72 months, which were divided into two groups as ≤3 years old (n=526, 79.2%) and 3–6 years old (n=138, 20.8%). Patients who survived (n=567, 85.4%) or died (n=97, 14.6%) of HFMD differed significantly in terms of age, peak temperature, central nervous symptoms, cardiopulmonary systems, fasting glucose (>8.3 mmol/L), current EV71 infection and NLR median (range) [1.92 (0.20, 19.35) vs 3.43 (0.62, 13.85), P < 0.001].
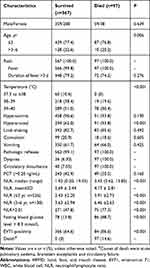 |
Table 1 Clinicodemographic Characteristics of Patients with Severe HFMD |
Based on Youden’s analysis, the ROC curve and interactive dot diagram for calculating the optimal cutoff value of NLR in predicting mortality is shown in Figure 1. An NLR cutoff value of 2.01 was used to classify patients into groups with high NLR (NLR > 2.01) or low NLR (NLR ≤ 2.01). The high-NLR group showed significantly higher death risk than the low-NLR group (P < 0.001).
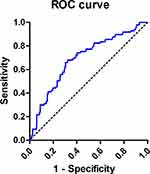 |
Figure 1 ROC curve and interactive dot diagram for calculating optimal cutoff value of NLR in in predicting mortality generated for 664 patients with critical HFMD. |
Univariate analysis of 664 patients (567 survived, 97 died) revealed significant differences (P < 0.001) in the frequency of fasting blood glucose >8.3 mmol/L, current EV71 infection, and NLR > 2.01. Multivariate analysis identified the following as independently associated with risk of death from severe HFMD (Table 2): high fever (>39.0 °C) (OR 3.342, 95% CI 1.736–6.432), EV71 infection (OR 3.200, 95% CI 1.529–6.698), fasting glucose (OR 37.343, 95% CI 18.616–74.909), and NLR > 2.01 (OR 2.142, 95% CI 1.125–4.079).
 |
Table 2 Analysis of Potential Risk Factors for Severe HFMD |
Characteristics of Patients with Severe or Critical HFMD and Risk Factors for Mortality
The study involved 664 patients diagnosed with critical HFMD, including 137 with critical disease and 97 who died (Table 3). Patients who survived or died differed significantly in age, fasting glucose >8.3 mmol/L, EV71 infection and NLR median (range) [1.87 (0.25–14.08) vs 3.43 (0.62–13.85), P < 0.001].
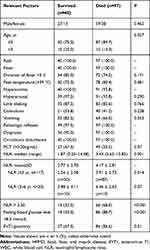 |
Table 3 Clinicodemographic Characteristics of Patients with Critical HFMD Who Died or Survived During the Study Period |
The ROC curve and interactive dot diagram for calculating the optimal cutoff value of NLR in predicting mortality generated for 137 patients with critical HFMD is shown in Figure 2. An NLR cutoff value of 2.50 was used to classify patients into those with high NLR (NLR > 2.50) or low NLR (NLR ≤ 2.50). The high-NLR group was at significantly higher death risk than the low-NLR group (P < 0.001).
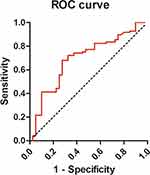 |
Figure 2 ROC curve and interactive dot diagram for calculating optimal cutoff value of NLR in predicting mortality generated for 137 patients with critical HFMD. |
Risk factors for critical HFMD are summarised in Table 4. Univariate analysis of 40 surviving and 97 dead patients revealed significant differences (P < 0.001) in the incidence of fasting blood glucose > 8.3 mmol/L, current EV71 infection, and NLR > 2.50. The multivariate model of 137 critical cases identified the following factors associated with death risk: EV71 infection (OR 3.441, 95% CI 1.132–10.462), fasting glucose (OR 14.173, 95% CI 4.920–40.827), and NLR > 2.50 (OR 4.166, 95% CI 1.570–11.051).
 |
Table 4 Analysis of Risk Factors for Mortality Associated with Severe HFMD |
Discussion
In this retrospective study, we aimed to explore the correlation between NLR and risk of mortality in patients with severe HFMD. Multivariate analysis of data from 664 patients identified EV71 infection, WBC count, fasting glucose and NLR > 2.01 as independently associated with the risk of death from severe HFMD. Uni- and multivariate analyses identified current EV71 infection, fasting glucose and NLR > 2.50 as risk factors for mortality in patients with critical HFMD. Early detection of these risk factors and timely intervention are important to control mortality due to severe HFMD.
NLR is obtained by simply dividing the number/proportion of neutrophils by that of lymphocytes, so it can be calculated from the differential white cell count test routinely performed in most clinical settings. Thus it is a convenient marker of systemic inflammatory response. Our results add to the growing list of diseases where NLR may be clinically useful as a prognostic marker. Growing evidence suggests that NLR and other haematological markers of the systemic inflammatory response can reliably predict inflammatory response and survival in patients with malignancy.9–14 Many groups have investigated the prognostic value of the NLR in a variety of inflammatory diseases at differing stages of disease.16,17
The relationship between increased NLR and poor prognosis is not fully understood in adults, and high NLR is thought to be indicative of inflammation. For example, proinflammatory cytokines in the plasma of patients with NLR >5 may establish and maintain a tumour microenvironment favouring aggressive tumour behaviour. This may explain why elevated NLR appears to be associated with recurrence of hepatocellular carcinoma.18 In fact, NLR may show greater predictive power than the classical inflammatory marker C-reactive protein or total WBC count for discriminating patients with or without Crohn’s disease.19
In children, NLR has already been associated with the activity of several infectious diseases. One study showed that the combination of NLR and C-reactive protein/mean platelet volume ratios may be useful for differential diagnosis of bacterial or viral pneumonia, as well as prediction of complications.13 NLR may be used as a diagnostic marker of febrile urinary tract infection,20 and it may be elevated in multidrug-resistant Pseudomonas aeruginosa infection in patients with hospital-acquired pneumonia.21 Our results in the present study lead us to suggest that NLR may be a useful inflammatory marker for predicting severity of HFMD and risk of associated mortality.
The early hyperdynamic phase of infection is mediated by neutrophils, macrophages and monocytes, with the release of inflammatory cytokines. Overexpression of inflammatory cytokines and chemokines can induce excessive immune responses and cause cytokine cascade reaction and cytokine storm in EV71-infected tissues.22 Severe HFMD involves the release of a large number of pro-inflammatory factors including interleukin (IL)-6, IL-12, IL-18, tumour necrosis factor-α, and interferon-γ.22 This release of proinflammatory cytokines and acute-phase factors can increase WBC count. During the systemic inflammatory response, neutrophil apoptosis is suppressed and lymphocyte apoptosis is promoted.23 Thus, NLR may be a particularly useful index because it reflects inflammatory processes (neutrophils) as well as immunoregulatory processes (lymphocytes). We speculate that the increase in NLR in HFMD is caused by cytokine-mediated recruitment of neutrophils. During HFMD progression, various cytokines and chemokines likely contribute to inflammation and may be valuable for monitoring progression and predicting prognosis. This inflammation may then help explain why increased NLR is associated with mortality risk in patients with severe HFMD.
In this large cohort of infants and young children not older than 6 years, we found that NLR >2.50 can predict mortality of severe HFMD. To the best of our knowledge, this is the first study to assess NLR as a diagnostic marker of severe HFMD in such a young population. NLR appears to correlate positively with age at least in healthy individuals,24 and neutrophil and lymphocyte counts change significantly from birth to young adulthood,12 and we verified that there were statistically significant differences in NLR values between different age groups (≤3 yr vs 3–6 yr, 2.67±2.36 vs 3.83±3.00, P < 0.001) in this study. Despite the effect of age on NLR, age-independent NLR was associated with the prognosis of HFMD patients (Tables 1 and 3). Thus, appropriate NLR cutoff values for classifying patients as low or high will depend on age. Previous work with healthy children younger than 20 years (mean age, 16 years) indicated a mean NLR value of 1.53 ± 0.56.24 In contrast, an NLR cutoff >5 has been suggested for detecting bacteraemia or sepsis in adults.25
Our study also found that EV71 infection and high fasting glucose were associated with increased risk of death from severe HFMD. Similar findings were reported in other studies.26,27 In recent years, EV71 has caused large-scale outbreaks of HFMD and substantial child mortality in the Asia-Pacific region.28,29 Now, vaccines against EV71 have become available in some Asian countries, such as in China.30–34 However, there are still a few challenges facing the worldwide use of EV71 vaccine.35 So, adequate prevention of severe cases of HFMD and determining factors that predict HFMD severity are very most important to make better clinical decisions.
A few limitations of this study are worth considering. The study was relatively small and involved patients from only two medical centres. In addition, the study was retrospective, increasing the risk of selection and analytical bias, and NLR was not observed over time. These factors make it impossible to establish causal relationships between NLR and all-cause mortality. It will be important in future work to see whether NLR increases as patient condition progressively deteriorates; in parallel, levels of various cytokines should be assayed. Our results justify further work to clarify how high NLR may contribute to severe HFMD.
Conclusions
The NLR can be easily calculated from routine differential neutrophil and lymphocyte counts, making it more straightforward and less expensive than many other inflammatory markers. NLR may be able to predict risk of HFMD-associated mortality, providing additional stratification beyond conventional risk scores.
Ethical Approval and Informed Consent
The study was approved by the Ethics Committee of the Second Affiliated Hospital of Xi’an Jiaotong University and Xi’an Children’s Hospital. Informed consent was not required for a retrospective study.
Acknowledgments
We thank the doctors of the Department of Infectious Diseases of Xi’an Children’s Hospital and Xi’an Jiaotong University Second Affiliated Hospital for their help in data collection.
Funding
This work was supported by the National Natural Science Foundation of China (grant No. 81701632).
Disclosure
The authors declare that they have no conflicts of interest.
References
1. Kushner D, Caldwell BD. Hand-foot-and-mouth disease. J Am Podiatr Med Assoc. 1996;86(6):257–259. doi:10.7547/87507315-86-6-257
2. Pathinayake PS, Hsu AC, Wark PA. Innate immunity and immune evasion by enterovirus 71. Viruses. 2015;7(12):6613–6630. doi:10.3390/v7122961
3. Huang CC, Liu CC, Chang YC, Chen CY, Wang ST, Yeh TF. Neurologic complications in children with enterovirus 71 infection. N Engl J Med. 1999;341(13):936–942. doi:10.1056/NEJM199909233411302
4. Liu SL, Pan H, Liu P, et al. Comparative epidemiology and virology of fatal and nonfatal cases of hand, foot and mouth disease in mainland China from 2008 to 2014. Rev Med Virol. 2015;5(2):115–128. doi:10.1002/rmv.1827
5. Chang LY, King CC, Hsu KH, et al. Risk factors of enterovirus 71 infection and associated hand, foot, and mouth disease/herpangina in children during an epidemic in Taiwan. Pediatrics. 2002;109(6):e88. doi:10.1542/peds.109.6.e88
6. Chang PC, Chen SC, Chen KT. The current status of the disease caused by enterovirus 71 infections: epidemiology, pathogenesis, molecular epidemiology, and vaccine development. Int J Environ Res Public Health. 2016;13(9):890. doi:10.3390/ijerph13090890
7. Guthrie GJ, Charles KA, Roxburgh CS, Horgan PG, McMillan DC, Clarke SJ. The systematic inflammation-based neutrophil-lymphocyte ratio: experience in patients with cancer. Crit Rev Oncol Hematol. 2013;88(1):218–230. doi:10.1016/j.critrevonc.2013.03.010
8. Arbel Y, Finkelstein A, Halkin A, et al. Neutrophil/lymphocyte ratio is related to the severity of coronary artery disease and clinical outcome in patients undergoing angiography. Atherosclerosis. 2012;225(2):456–460. doi:10.1016/j.atherosclerosis.2012.09.009
9. Wang Y, Ju M, Chen C, et al. Neutrophil-to-lymphocyte ratio as a prognostic marker in acute respiratory distress syndrome patients: a retrospective study. J Thorac Dis. 2018;10(1):273–282. doi:10.21037/jtd.2017.12.131
10. Mousa N, Besheer T, Abdel-Razik A, et al. Can combined blood neutrophil to lymphocyte ratio and C-reactive protein be used for diagnosis of spontaneous bacterial peritonitis? Br J Biomed Sci. 2018;75(2):71–75. doi:10.1080/09674845.2017.1396706
11. Ozmen S, Timur O, Calik I, et al. Neutrophil-lymphocyte ratio (NLR) and platelet-lymphocyte ratio (PLR) may be superior to C-reactive protein (CRP) for predicting the occurrence of differentiated thyroid cancer. Endocr Regul. 2017;51(3):131–136. doi:10.1515/enr-2017-0013
12. Hamiel U, Bahat H, Kozer E, Hamiel Y, Ziv-Baran T, Goldman M. Diagnostic markers of acute infections in infants aged 1 week to 3 months: a retrospective cohort study. BMJ Open. 2018;8(1):e018092. doi:10.1136/bmjopen-2017-018092
13. Bekdas M, Goksugur SB, Sarac EG, Erkocoglu M, Demircioglu F. Neutrophil/lymphocyte and C-reactive protein/mean platelet volume ratios in differentiating between viral and bacterial pneumonias and diagnosing early complications in children. Saudi Med J. 2014;35(5):442–447.
14. Yazici M, Ozkisacik S, Oztan MO, Gürsoy H. Neutrophil/lymphocyte ratio in the diagnosis of childhood appendicitis. Turk J Pediatr. 2010;52(4):400–403.
15. Ministry of Health of the People’s Republic of China. Hand, foot and mouth disease clinic guide (2010 edition). (in Chinese). Available from: http://www.nhc.gov.cn/xxgk/pages/viewdocument.jsp?dispatchDate=&staticUrl=/zwgkzt/wsbysj/201004/46884.shtml.
16. Imtiaz F, Shafique K, Mirza SS, Ayoob Z, Vart P, Rao S. Neutrophil lymphocyte ratio as a measure of systemic inflammation in prevalent chronic diseases in Asian population. Int Arch Med. 2012;5(1):2. doi:10.1186/1755-7682-5-2
17. Dolan RD, Lim J, McSorley ST, Horgan PG, McMillan DC. The role of the systemic inflammatory response in predicting outcomes in patients with operable cancer: systematic review and meta-analysis. Sci Rep. 2017;7(1):16717.
18. Motomura T, Shirabe K, Mano Y, et al. Neutrophil-lymphocyte ratio reflects hepatocellular carcinoma recurrence after liver transplantation via inflammatory microenvironment. J Hepatol. 2013;58(1):58–64.
19. Gao SQ, Huang LD, Dai RJ, Chen DD, Hu WJ, Shan YF. Neutrophil-lymphocyte ratio: a controversial marker in predicting Crohn’s disease severity. Int J Clin Exp Pathol. 2015;8(11):14779–14785.
20. Lee JW, Park JS, Park KB, Yoo GH, Kim SS, Lee SM. Prediction of renal cortical defect and scar using neutrophil-to-lymphocyte ratio in children with febrile urinary tract infection. Nuklearmedizin. 2017;56(3):109–114. doi:10.3413/Nukmed-0878-17-01
21. Zhou Y-Q, Feng D-Y, Li W-J, et al. Lower neutrophil-to-lymphocyte ratio predicts high risk of multidrug-resistant Pseudomonas aeruginosa infection in patients with hospital-acquired pneumonia. Ther Clin Risk Manag. 2018;14:1863–1869. doi:10.2147/TCRM.S179181
22. Shang WZ, Qian SY, Fang LJ, Han Y, Zheng CP. Association study of inflammatory cytokine and chemokine expression in hand foot and mouth disease. Oncotarget. 2017;8(45):79425–79432. doi:10.18632/oncotarget.18341
23. Wesche DE, Lomas-Neira JL, Perl M, Chung CS, Ayala A. Leukocyte apoptosis and its significance in sepsis and shock. J Leukoc Biol. 2005;78(2):325–337. doi:10.1189/jlb.0105017
24. Li J, Chen Q, Luo X, et al. Neutrophil-to-lymphocyte ratio positively correlates to age in healthy population. J Clin Lab Anal. 2015;29(6):437–443. doi:10.1002/jcla.21791
25. Gürol G, Çiftci İH, Terizi HA, Atasoy AR, Ozbek A, Köroğlu M. Are there standardized cutoff values for neutrophil-lymphocyte ratios in bacteremia or sepsis? J Microbiol Biotechnol. 2015;25(4):521–525. doi:10.4014/jmb.1408.08060
26. Long L, Gao LD, Hu SX, et al. Risk factors for death in children with severe hand, foot, and mouth disease in Hunan, China. Infect Dis (Lond). 2016;48(10):44–748.
27. Qiu J, Yan H, Cheng N, et al. The clinical and epidemiological study of children with hand, foot, and mouth disease in Hunan, China from 2013 to 2017. Sci Rep. 2019;9:11662. doi:10.1038/s41598-019-48259-1
28. Qiu J. Enterovirus 71 infection: a new threat to global public health? Lancet Neurol. 2008;7(10):868–869. doi:10.1016/S1474-4422(08)70207-2
29. Lee KY. Enterovirus 71 infection and neurological complications. Korean J Pediatr. 2016;59(10):395–401. doi:10.3345/kjp.2016.59.10.395
30. Zhu FC, Meng FY, Li JX, et al. Efficacy, safety, and immunology of an inactivated alum-adjuvant enterovirus 71 vaccine in children in China: a multicentre, randomised, double-blind, placebo-controlled, Phase 3 trial. Lancet. 2013;381(9882):2024–2032. doi:10.1016/S0140-6736(13)61049-1
31. Zhu F, Xu W, Xia J, et al. Efficacy, safety, and immunogenicity of an enterovirus 71 vaccine in China. N Engl J Med. 2014;370(9):818–828. doi:10.1056/NEJMoa1304923
32. Mao QY, Wang Y, Bian L, Xu M, Liang Z. EV71 vaccine, a new tool to control outbreaks of hand, foot and mouth disease (HFMD). Expert Rev Vaccines. 2016;15(5):599–606. doi:10.1586/14760584.2016.1138862
33. Tambyah PA, Oon J, Asli R, et al. An inactivated enterovirus 71 vaccine is safe and immunogenic in healthy adults: A Phase I, double blind, randomized, placebo-controlled, study of two dosages. Vaccine. 2019;37(31):4344–4353. doi:10.1016/j.vaccine.2019.06.023
34. Liu D, Leung K, Jit M, et al. Cost-effectiveness of bivalent versus monovalent vaccines against hand, foot and mouth disease. Clin Microbiol Infect. 2020;26(3):373–380. doi:10.1016/j.cmi.2019.06.029
35. Esposito S, Principi N. Hand, foot and mouth disease: current knowledge on clinical manifestations, epidemiology, aetiology and prevention. Eur J Clin Microbiol Infect Dis. 2018;37(3):391–398. doi:10.1007/s10096-018-3206-x
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
