Back to Journals » Journal of Pain Research » Volume 8
Profile of extended-release oxycodone/acetaminophen for acute pain
Authors Bekhit M
Received 18 June 2015
Accepted for publication 3 July 2015
Published 21 October 2015 Volume 2015:8 Pages 719—728
DOI https://doi.org/10.2147/JPR.S73567
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Micheal Schatman
Mary Hanna Bekhit1–5
1David Geffen School of Medicine, 2Ronald Reagan UCLA Medical Center, 3UCLA Ambulatory Surgery Center, 4UCLA Wasserman Eye Institute, 5UCLA Martin Luther King Community Hospital, University of California Los Angeles, Los Angeles, CA, USA
Abstract: This article provides a historical and pharmacological overview of a new opioid analgesic that boasts an extended-release (ER) formulation designed to provide both immediate and prolonged analgesia for up to 12 hours in patients who are experiencing acute pain. This novel medication, ER oxycodone/acetaminophen, competes with current US Food and Drug Administration (FDA)-approved opioid formulations available on the market in that it offers two benefits concurrently: a prolonged duration of action, and multimodal analgesia through a combination of an opioid (oxycodone) with a nonopioid component. Current FDA-approved combination analgesics, such as Percocet (oxycodone/acetaminophen), are available solely in immediate-release (IR) formulations.
Keywords: opioid, analgesic, xartemis, acute postsurgical pain, substance abuse, acetaminophen, extended release
Introduction
Each year, approximately 25–97 million patients in the USA experience acute pain.1 Based on estimates from the Centers for Disease Control, in the year 2010, approximately a little over 102 million surgical procedures were ordered or performed at office visits.2 In addition, another 51 million surgeries were performed in this same year on an inpatient basis.3 Further estimates by the Institute of Medicine from 2011 suggest that approximately 80% of patients undergoing surgery experienced postoperative pain, and that approximately 88% of these surgical patients described the acute postsurgical pain as ranging from moderate to severe or extreme.4
Opioid analgesics boast a well-established, long history as first-line therapy for acute surgical pain. Recently, their popularity has soared, as the sales of opioid medications have quadrupled during the past decade.5 Despite the abundance of opioids available for treatment of acute pain, as well as the large variety of different classes of analgesics in addition to the opioid family, many patients still frequently experience undertreated acute pain.1 In addition to the obvious untoward emotional, social, and psychological effects of uncontrolled pain on the patient, this level of unmanaged intense pain bears significant negative consequences on the community as a whole. It contributes significantly to decreased function, thus translating more globally into decreased productivity and a noticeable cost burden to the US businesses.6
This article provides a historical and pharmacological overview of a new opioid analgesic that boasts an extended-release (ER) formulation aimed at providing patients who are experiencing acute pain both immediate analgesia and continued, prolonged analgesia for up to 12 hours. This novel medication, ER oxycodone/acetaminophen, competes with current US Food and Drug Administration (FDA)-approved opioid formulations available on the market in that it offers two benefits concurrently: a prolonged duration of action and multimodal analgesia through combination of an opioid (oxycodone) with a nonopioid component. Current FDA-approved combination analgesics, such as Percocet (oxycodone/acetaminophen), are available solely in immediate-release (IR) formulations.
Pharmacokinetics
Oxycodone, or 4,5-alpha-epoxy-14-hydroxy-3-methoxy-17-methyl-morphinan-6-one hydrochloride, is an opioid agonist derived from the opium alkaloid thebaine.7 It is a schedule II controlled substance, with significant potential for abuse. IR formulations of oxycodone have long been available on the market for management of moderate-to-severe pain, with frequent dosing intervals – usually every 4–6 hours. ER formulations of oxycodone, though not in combination with acetaminophen, have offered the advantage of longer dosing intervals and thus the convenience of less frequent dosing.1 Opioids often are supplied as a combination pill including a nonopioid analgesic, most commonly acetaminophen. The main goal of this multimodal analgesic regimen is to achieve therapeutic benefit while using reduced doses of each individual medication in order to minimize toxic side effects.1 The combination of oxycodone and acetaminophen in IR formulations has a long-standing history of therapeutic use for moderate-to-severe pain.1 Due to the short-acting nature of the IR pill, it again requires frequent redosing every 4–6 hours, similar to IR oxycodone alone.
ER oxycodone/acetaminophen, previously the study drug MNK-795, received the FDA approval in March 2014 for the treatment of acute pain severe enough that other therapies are deemed not adequate.8 It was then marketed with the trade name Xartemis XR. One important Phase III efficacy study contributed significantly to the approval by the FDA. The study authors sought to demonstrate a statistically significant improvement in analgesia when compared with placebo in post-bunionectomy patients over 48 hours, and they were able to document that the study met this primary endpoint.6
ER oxycodone/acetaminophen comprises a combination of the opioid oxycodone hydrochloride 7.5 mg and the nonopioid analgesic acetaminophen 325 mg. It provides a much improved duration of action when compared with the previously available IR formulation of oxycodone/acetaminophen. The tablet allows dosing every 12 hours instead of every 4–6 hours, and it utilizes a dual-layer biphasic delivery mechanism that contains both IR and delayed-release layers, or components.1 When two tablets are administered as a single dose consisting of oxycodone 15 mg with acetaminophen 650 mg, the medication is designed to deliver oxycodone 3.75 mg and acetaminophen 325 mg through the IR component as well as oxycodone 11.25 mg and acetaminophen 325 mg through the ER component, which continually delivers the medication in the upper gastrointestinal (GI) tract at a steady rate.1
The oxycodone component of the medication possesses an oral bioavailability of 60%–87%.7 This is similar to that of IR medications containing oxycodone, with both single and multiple doses.7 Detectable serum levels of oxycodone occur approximately 30 minutes from ingestion of the ER tablet, and maximum plasma concentrations (Cmax) are achieved approximately 3–4 hours after administration.7 In terms of the acetaminophen component, peak plasma levels are reached in approximately 1 hour from medication administration.7 Within 24 hours, or completion of two doses of ER oxycodone/acetaminophen (dosed every 12 hours), serum concentrations of both components of the medication achieve a steady state.7
Consumption of high-fat meals will delay peak serum levels by 2 hours, and consumption of low-fat meals with administration of the medication will delay peak serum levels by 1 hour.7 The oral bioavailability of oxycodone is increased by approximately 15%, and the peak plasma level increases by 12%–25% when the medication is administered with a meal.7 Acetaminophen’s peak serum levels are reduced by approximately 23% by co-administration with a meal.7
When administered intravenously, oxycodone has a volume of distribution of 2.6 L/kg.7 At normal body temperature (37°C) and pH (7.4), it exists in plasma in a 45% protein-bound state.7 Acetaminophen’s volume of distribution is approximately 0.9 L/kg, and it exists in plasma in a 20% protein-bound state.7 It distributes through most tissues with the exception of lipids.7
Both the oxycodone and acetaminophen components undergo extensive hepatic metabolism prior to primarily renal elimination.7 The two major metabolites of oxycodone are noroxycodone and oxymorphone, as well as their glucuronides.7 Noroxycodone is an active metabolite, possessing approximately 60% the potency of its parent compound, and oxymorphone circulates in very low levels in the plasma.7 Acetaminophen undergoes three separate metabolic pathways in the liver: glucuronidation, conjugation with sulfate, or cytochrome P450-dependent oxidation.7 The metabolites of acetaminophen are inert.7
Pharmacodynamics
The oxycodone component of ER oxycodone/acetaminophen contributes to the majority of the medication’s pharmacodynamic effects on the various organ systems listed in Table 1 (summarized from http://www.rxlist.com/xartemis-xr-drug.htm). The pharmacodynamic profile is similar to that of other opioid medications.
  | Table 1 Pharmacodynamic effects of oxycodone/acetaminophen ER |
Clinical studies
One major efficacy study was published as a result of Phase III trials. This randomized, placebo-controlled, double-blind, multicenter study evaluated 303 patients undergoing unilateral first metatarsal bunionectomy. In this acute postsurgical pain model, fewer patients (85%) required ibuprofen rescue in the group that was randomized to receive two tablets of ER oxycodone/acetaminophen, when compared with the placebo group (98%). Patients who received ER oxycodone/acetaminophen also reported lower pain intensity scores at several fixed time intervals postoperatively, compared with the placebo group.7
Several pharmacokinetic studies were conducted comparing ER oxycodone/acetaminophen with commonly used IR opioids, such as Roxicodone (oxycodone hydrochloride as a 5 mg, 15 mg, or 30 mg tablet), Ultracet (tramadol hydrochloride 37.5 mg with acetaminophen 325 mg), and Percocet (IR oxycodone 7.5 mg with acetaminophen 325 mg).
Two of the studies were performed with healthy volunteers, with the first study comparing ER oxycodone/acetaminophen in both one- and two-tablet doses every 12 hours with IR oxycodone/acetaminophen in one-tablet doses every 6 hours.1 The second study evaluated ER oxycodone/acetaminophen in two-tablet doses every 12 hours compared to IR oxycodone alone, IR tramadol/acetaminophen, and IR oxycodone/acetaminophen in one-tablet doses every 6 hours.1
The authors of both studies were able to demonstrate similar plasma concentrations and steady-state pharmacokinetics for the ER formulation when compared with the various IR medication formulations.1 They were also able to ascertain that the 12-hour ER dosing delivered less variable plasma levels of oxycodone and lower serum trough levels of acetaminophen, without increased incidence of adverse events.1 In addition, the clinical effects were proportional to the doses administered with the ER formulation in one-tablet and two-tablet forms.1
Pharmacology and substance abuse
As opioid sales have dramatically increased recently over the past decade, so has the potential for substance abuse.5 Although the FDA-approved label for ER oxycodone/acetaminophen does not specifically make use of language cautioning against or deterring from narcotic abuse, Mallinckrodt continued to work closely with the FDA during the Phase I trial period to “develop more data to characterize abuse-deterrence features of Xartemis XR and other products utilizing this technology platform”.6 The pharmaceutical company continued to conduct several studies addressing the abuse potential of the medication, and was able to demonstrate that the ER formulation had no more, and in fact less, of an addictive potential, which they quantified in the study as “drug liking” and “drug high” measured by visual analog scales, than the IR formulation that had already been available on the market for many years.9
One of the Phase I trials of ER oxycodone/acetaminophen was published in August 2014. It compared the “positive subjective drug effects” of the ER formulation of oxycodone/acetaminophen with those of the IR formulation. This randomized, double-blind, placebo-controlled trial was conducted on 55 healthy volunteers, and was able to demonstrate that the ER formulation resulted in delayed peak effects, lower peak effects, and lower drug liking scores measured by visual analog scale than the IR formulation of oxycodone/acetaminophen.9 The comparison between formulations looked at two dosages, 15 mg oxycodone with 650 mg acetaminophen and 30 mg oxycodone with 1,300 mg acetaminophen, and found similar results regardless of dose administered.9
The study authors also investigated the crushed form of the ER pill and found that the crushed pill containing 30 mg oxycodone with 1,300 mg acetaminophen in ER form provided even more delayed subjective effects when compared with the crushed pill in IR form or the intact pill in ER form.9 This final investigation sought to address the attempts at chemical alteration, including crushing, of narcotic pills by substance abusers to improve the subjective effects. Therefore, the authors were able to demonstrate that the ER formulation does not present a higher addictive potential than IR formulations of oxycodone/acetaminophen. On the contrary, the ER formulation may actually prove less attractive to potential substance abusers in intact form or in chemically altered form.
The pharmacological formulation of ER oxycodone/acetaminophen, by using several inactive ingredients, acts as a deterrent to abuse, by posing additional obstacles to chemically extracting the oxycodone component.5 This requires additional effort on the part of potential substance abusers, when compared to the more straightforward extraction of the active ingredient in other opioid pills.5
The inclusion of several inactive ingredients within the large tablet of ER oxycodone/acetaminophen also poses yet another obstacle to substance abusers by making snorting or intravenous injection challenging.5 For example, the tablet transforms into a thick, gelatinous material when it dissolves into a liquid medium, thus making it difficult to aspirate into a syringe for intravenous abuse.5 It also makes snorting the drug difficult, since the humidity within the nasopharynx results in the same thick, sticky, pasty texture.5 The tablet is also difficult to dissolve, break, or crush because of one of the inactive ingredients, polyethylene oxide; the formulation also presents a significant obstacle to “cooking” the tablet on a spoon.5 As a result of all these significant impediments to the chemical alteration of ER oxycodone/acetaminophen, recreational substance abusers may view this medication as a less attractive alternative to older opioids.5
Adverse reactions
Adverse side effects of ER oxycodone/acetaminophen can be categorized in terms of frequency of occurrence. Some of the more common side effects, occurring with an incidence greater than 10%, include dizziness and nausea.10 Less frequent side effects, occurring 1%–10% of the time, include headache (10%), vomiting (9%), drowsiness (4%), constipation (4%), skin rash (2%), erythema (1%), excoriation (1%), pruritis (1%), skin blistering (1%), peripheral edema (1%), dysuria (1%), hot flashes (1%), elevated liver enzymes, diarrhea, dyspepsia, xerostomia, fatigue, insomnia, and cough.10
Significant rare side effects, some of which can be life-threatening, and occurring <1% of the time, include respiratory depression, withdrawal syndrome, palpitations, chest discomfort, cognitive dysfunction, memory impairment, excess sedation, migraine, hypersensitivity reaction, hypertension, myoclonus, and hypogonadism.11,12 Table 2 outlines more specific concerns regarding adverse side effects as well as their prevention and management.10
  | Table 2 Adverse side effect profile of extended-release oxycodone/acetaminophen |
Contraindications
Patients who should not take ER oxycodone/acetaminophen include those who have experienced a prior allergic reaction or hypersensitivity to oxycodone, acetaminophen, or other component of the pharmaceutical composition, those with severe acute asthma or severe respiratory depression predisposing them to hypercarbia, and those with ileus.10 Patients who have a known history of allergic reaction to other opioids should exercise caution, since little data exist regarding cross-reactivity for opioid hypersensitivity.10
Effect on comorbidities
Adrenal insufficiency
Chronic opioid exposure may cause secondary hypogonadism, with associated sexual dysfunction, infertility, mood disorders, and osteoporosis.11 ER oxycodone/acetaminophen should be used with caution in patients with adrenal insufficiency or Addison’s disease.
Acute abdomen
Signs/symptoms may be masked by administration of ER oxycodone/acetaminophen.10
Biliary obstruction
Patients with biliary duct disease should use ER oxycodone/acetaminophen with caution, as it may cause Sphincter of Oddi spasm and worsen biliary tract dysfunction, with possible progression to acute pancreatitis.7,10
Altered mental status
ER oxycodone/acetaminophen may potentiate CO2 retention caused by hypoventilation and as such should be used with caution in patients with central nervous system depression or coma. These patients may experience adverse intracranial effects associated with hypercarbia.10
GI pathology
Due to its chemical preparation, ER oxycodone/acetaminophen upon ingestion tends to swell and develop increased adhesiveness within the GI tract. Patients with a narrowed intestinal lumen may experience inability to pass the pill through the GI tract. An alternative analgesic regimen should be considered in patients with intestinal narrowing.10
Liver disease
Patients with alcohol-related hepatic disease, particularly those consuming more than three alcoholic beverages per day, are at increased risk of hepatotoxicity. Hepatotoxicity is associated with acetaminophen dosing greater than 4 g/day, as well as long-term exposure to ER oxycodone/acetaminophen.10
Substance abuse
As with all narcotic medications, tolerance, chemical dependency, psychological dependency, addiction, and substance abuse may develop with chronic exposure to ER oxycodone/acetaminophen.5,10 Reports exist of patients crushing, chewing, snorting, or injecting the dissolved form of the medication, which may result in the uncontrolled delivery of the oxycodone, overdose, and death.5,10 The potential for dependency or substance abuse should be assessed for each patient on an individual basis prior to administration of the medication.
Neurotrauma or elevated intracranial pressure
Similar to other opioid medications, administration of ER oxycodone/acetaminophen may potentiate hypoventilation, hypercarbia, and as a result elevated intracranial pressure (ICP) in patients with recent head trauma, intracranial lesions, or preexisting issues with ICP.10
Pulmonary disease
Respiratory depression may occur with opioid medications and can result in potentially life-threatening hypercarbia. Patients with increased susceptibility include those with preexisting obstructive pulmonary pathophysiology such as chronic obstructive pulmonary disease, cor pulmonale, and scoliosis with impaired respiratory function.10
Other
Patients with morbid obesity, renal insufficiency, seizure disorder, psychosis, G6PD deficiency, thyroid dysfunction, prostate enlargement, or urethral stricture should use caution with administration of oxycodone/acetaminophen.10
Patient-specific considerations
Neonates exposed to ER oxycodone/acetaminophen for extended periods of time in utero are at risk of developing physical dependence in utero and neonatal withdrawal syndrome after delivery, with symptoms ranging from irritability, high-pitched crying, and abnormal sleeping patterns, to nausea, vomiting, diarrhea, hyperactivity, failure to thrive, and tremors.10 If the syndrome remains undiagnosed in the neonate, these symptoms can progress to life-threatening reactions.10,13 Parturients requiring chronic opioid therapy during pregnancy must be followed by a neonatologist in the peripartum period for early recognition and treatment of the withdrawal syndrome in the neonate based on specific protocols formulated by the neonatologist.10 The neonate may also display respiratory depression or other signs of opioid toxicity at delivery. Therefore, an opioid antagonist such as naloxone, should be available for reversal of opioid-induced respiratory depression in the neonate.10
Surgical patients may develop decreased bowel motility postoperatively while taking opioid analgesics such as ER oxycodone/acetaminophen.7,10 Concomitant administration of a stool softener aids in the prevention of opioid-associated constipation.
Elderly or debilitated patients may be more susceptible to develop respiratory depression or other adverse side effects of ER oxycodone/acetaminophen.7,14 Caution should be used during administration of this medication to the frail or elderly. Renal clearance of oxycodone may also be decreased in elderly patients, necessitating appropriate dosing changes.14
In one of the Phase III trials conducted by the pharmaceutical company Mallinckrodt, 607 subjects were treated with ER oxycodone/acetaminophen. Of these, approximately 10% (63 total) were over the age of 65. Within this geriatric group, 1.6% (ten total) were older than the age of 75. There were no unfavorable or unexpected adverse reactions experienced in the elderly patients who received ER oxycodone/acetaminophen tablets.15 At the same time, however, geriatric patients may exhibit a greater sensitivity to oxycodone when compared with the younger patient population. So, special care should be taken when prescribing this medication for geriatric patients, with specific caution in ascertaining the dosing amount and frequency.16
Pregnant patients should use this opioid medication with caution, since it is transferred across the placenta to the neonate. Parturients who must take the medication expose the fetus to the risk of neonatal withdrawal syndrome at birth and should be followed by a neonatologist in the peripartum period.
ER oxycodone/acetaminophen is classified as pregnancy category C.
Unfortunately, the literature on the effects of ER oxycodone/acetaminophen on pregnant patients is sparse. No randomized controlled trials with adequate study design have been performed in this patient population to assess the pharmacokinetic effects on the parturient as well as the placental absorption to the fetus. Also lacking are data on how the pharmacokinetics of the ER formulation may compare with the pharmacokinetics and placental transfer of currently available IR formulations of oxycodone/acetaminophen.17
At this time, the literature does contain some epidemiological data on the safety profile of acetaminophen alone in the parturient population. According to data from two large population-based studies on acetaminophen use by pregnant women in their first trimesters, acetaminophen use in pregnant women seems to carry no increased risk of significant congenital deformities.15 In contrast, limited studies exist evaluating the incidence of congenital malformations related to oxycodone use in the parturient patient. Hence, as of yet, the incidence of malformations in human pregnancies has not been established for oxycodone. The baseline prevalence of major congenital deformities when looking at all pregnancies is approximately 2%–4%, without adjustments made for peripartum drug exposure.15 Also, the baseline prevalence of pregnancy loss, when including all pregnancies regardless of pharmacological history, is approximately 15%–20%.15
Currently, there are no animal studies on the reproductive or developmental effects of the combination of oxycodone with acetaminophen in the ER form.18 Animal data do exist from studies performed on the individual components separately. When rats and mice were exposed to clinically relevant doses of acetaminophen, the published literature associated those exposures to reproductive and developmental adverse events in their fetuses.15 Similarly, when pregnant rats were treated with doses of acetaminophen that are almost similar to the maximum human daily dose, this exposure was linked to increased bone variations in their fetuses as well as other fetotoxic effects.15
When a similar maximal dose was administered to pregnant rats in another study, both the mothers and the fetuses developed necrotic regions within their livers and kidneys.15 Interestingly, mice that received acetaminophen within the human clinical dosing range exhibited several adverse reproductive effects. They displayed a decreased number of litters, impaired growth, abnormal sperm in their offspring, as well as decreased weight at birth in the next generation.15
Oxycodone on the other hand has not been linked to adverse reproductive or developmental effects in animal studies. The literature on rats and rabbits has demonstrated no teratogenic or embryo–fetal toxic effects of oxycodone doses larger than human clinical doses.15
Lactating mothers should not use ER oxycodone/acetaminophen, because oxycodone is secreted in the breast milk, and may result in the accumulation of medication in the neonate.10 Potential adverse side effects from toxic serum levels in the infant may result, such as excess sedation or respiratory depression. Breast-feeding patients who do require administration of the medication should be followed closely by their physician for possible adverse effects on the neonate or infant.18
Acetaminophen is also secreted in human breast milk in small amounts. Based on results from one small study examining the effects of a 1,000 mg maternal dose of acetaminophen on breast-feeding infants, the infants were exposed to less than 1.85% of the weight-adjusted oral dose ingested by their mothers.16 Similarly, based on data from another study which examined the effects of maternal acetaminophen on breast-fed infants, looking at more than 15 nursing mothers, the calculated daily dose of acetaminophen within the infants was approximately 1%–2% of the dose the mothers had ingested.15 One well-documented case report does exist describing a rash in a breast-fed infant whose mother was taking acetaminophen while lactating. The infant’s rash was temporally related to the mother’s acetaminophen use, as it resolved when the mother stopped taking acetaminophen, and it reappeared when she began taking acetaminophen again.15 Based on the recommendations of the American Academy of Pediatrics, oral acetaminophen is considered compatible with breast-feeding.16
However, because of the potential for serious adverse reactions in nursing infants from ER oxycodone/acetaminophen specifically related to the oxycodone component, discussion between the lactating mother and her physician must weigh the risk-to-benefit ratio.15 The importance of the medication for the mother’s health needs to be taken into account and weighed against the importance of the continuation of breast-feeding. In short, the potential benefit should justify the potential risk to the newborn or infant.
Drug–drug interactions
Medications affecting cytochrome P450
The oxycodone component of ER oxycodone/acetaminophen undergoes a significant amount of hepatic metabolism via the cytochrome P450 enzyme system.15 It may therefore contribute to interactions with other medications that are also metabolized by the same enzyme group. Below is a further delineation of the specific isoenzymes within the P450 system that may contribute to drug–drug interactions, specifically isoenzyme CYP3A4 and isoenzyme CYP2D6.
Cytochrome CYP3A4 inhibitors
Medications that inhibit this enzyme’s activity could potentially impede the clearance of oxycodone, which is metabolized to a significant degree by this isoenzyme.15 This may in turn lead to increased serum oxycodone concentrations as well as possible prolonged therapeutic effects of the opioid medication.15 Simultaneous use of other medications which may have inhibitory effects on the isoenzyme CYP2D6 may further amplify these interactions, with heightened risk of respiratory depression or excess sedation.15 Patients who receive ER oxycodone/acetaminophen along with another medication that inhibits cytochrome CYP3A4 or CYP2D6 require frequent monitoring, with potential adjustments in dosing until stable medication side effects have been assessed on an individual patient basis.15 Inhibitors of CYP3A4 metabolic activity are listed in Table 3.
  | Table 3 Medications that may affect metabolism of extended-release oxycodone/acetaminophen through the cytochrome P450 system, specifically isoenzyme CYP3A4 |
Cytochrome CYP3A4 inducers
Medications that induce the activity of the isoenzyme CYP3A4 may lead to increased hepatic metabolism of oxycodone, and thus may result in increased clearance of the drug, as well as possible decreased therapeutic effect or in the worst case scenario symptoms of opioid withdrawal in patients who have been exposed to oxycodone long enough to develop chemical dependence on the medication.15 Similar to above, patients who are receiving ER oxycodone/acetaminophen along with a medication that induces the CYP3A4 isoenzyme should be monitored closely for symptoms of narcotic withdrawal or decreased efficacy, and dose adjustments should be made accordingly. Inducers of CYP3A4 are listed in Table 3.
Cytochrome CYP2D6 inhibitors
Several cardiovascular medications, including amiodorone and quinidine, as well as certain antidepressants inhibit this isoenzyme and could potentially decrease its metabolism of oxycodone to oxymorphone.15 Although the interaction has not yet led to reported clinically significant adverse outcomes, it may prove helpful to prescribers when selecting specific medications in concurrence with ER oxycodone/acetaminophen.
Cytochrome CYP2D6 inducers
Several pharmacological studies have demonstrated that this isoenzyme is not susceptible to induction by currently known medications.19 Genetics plays a significant role in the enzyme’s metabolic activity, rather than induction, with significant ethnic differences in metabolism of certain medications.19 These genetic and ethnic variations are listed in Table 4.
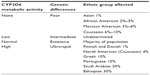  | Table 4 Genetics affecting ethnic differences in CYP2D6 metabolic activity |
Special pharmacological considerations
Other narcotic medications
Because ER oxycodone/acetaminophen has a different pharmacokinetic profile from that of IR formulations of the medication, it is not interchangeable with other oxycodone/acetaminophen drugs.16 The frequency of administration as well as the peak effect of the medication becomes challenging to calculate when the various formulations are used simultaneously.
Pediatric overdose
Accidental ingestion of ER oxycodone/acetaminophen can result in toxic, even lethal, overdose of the oxycodone component, the acetaminophen component, or both, in infants or children.14 Extra caution should be used in storage of the medication to keep it out of the reach of young children. Emergency medical attention is required in the case of pediatric overdose, for treatment of oxycodone or acetaminophen toxicity.
Opioid withdrawal
Narcotic withdrawal may be precipitated by sudden discontinuation of a mu-opioid agonist medication such as oxycodone after prolonged exposure, or by the use of an opioid agonist and antagonist at the same time. In patients who have been exposed to ER oxycodone/acetaminophen for a prolonged period of time, discontinuation of the medication should be achieved by gradual dose decreases of approximately 50% every 2–4 days.17 This gradual approach aims to prevent triggering an opioid withdrawal syndrome.
Summary
ER oxycodone/acetaminophen is a novel opioid analgesic that extends the duration of action beyond that of currently available tablets that combine an opioid with a nonopioid analgesic. Its ability to provide IR and ER components for severe, acute pain is unique, and offers an excellent therapeutic advantage within an acute care setting. Yet, it also carries significant risks of adverse side effects, similar to many opioid medications. Therefore, patients receiving this medication on an outpatient basis must be closely monitored by a physician at regular intervals for assessment of side effects and for appropriate gradual dose adjustments. Caution must be exercised when this medication is used in special patient populations, such as those at the extremes of age, debilitated patients, those with specific comorbidities that place them at heightened risk of adverse medication effects, and pregnant or lactating women due to potential effects on the fetus or neonate.
Further studies are needed to investigate the long-term outcomes, such as time to return of full function and duration of analgesic need, in postsurgical patients taking ER oxycodone/acetaminophen, and to also take a more extensive review of the efficacy of the medication for acute postoperative pain when compared with IR opioids.
Disclosure
The author has no financial interest or other conflict of interest in the publication of this article, and is in no way affiliated with the pharmaceutical company Mallinckrodt.
References
Devarakonda K, Morton T, Giuliani M, Kostenbader K, Barrett T. Steady-state pharmacokinetics of MNK-795, an extended-release oxycodone and acetaminophen combination analgesic: results from 2 active comparator studies. J Bioequiv Availab. 2014;6: 053–060. | |
CDC/NCHS, National Ambulatory Medical Care Survey. Available from: http://www.cdc.gov/nchs/data/ahcd/namcs_summary/2010_namcs_web_tables.pdf. Accessed March 30, 2015. | |
CDC, FastStats. Available from: http://www.cdc.gov/nchs/fastats/insurg.htm. Accessed March 30, 2015. | |
Committee on Advancing Pain Research, Care, and Education; Institute of Medicine. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington DC: Committee on Advancing Pain Research, Care, and Education; Institute of Medicine; 2011. | |
Nelson R. Xartemis XR Receives FDA Approval: May Reduce Opioid Abuse; 2014. Available from: http://formularyjournal. modernmedicine. | |
Available from: http://www.drugs.com/newdrugs/fda-approves-xartemis-xr-oxycodone-hydrochloride-acetaminophen-extended-release-cii-4015.html. Accessed on February 13, 2015. | |
Available from: http://www.rxlist.com/xartemis-xr-drug.htm. Accessed on February 13, 2015. | |
Available from: http://www.accessdata.fda.gov/drugsatfda_docs/label/2014/204031s000lbl.pdf. Accessed on February 13, 2015. | |
Morton T, Kostenbader K, Montgomery J, Devaradonda K, Barret T, Webster L. Comparison of subjective effects of extended-release versus immediate-release oxycodone/acetaminophen tablets in healthy nondependent recreational users of prescription opioids: a randomized trial. Postgraduate Medicine. 2014;126(4):20–32. | |
Available from: http://www.accessdata.fda.gov/drugsatfda_docs/./ 204031Orig1s000RiskR.pdf. Accessed on February 13, 2015. | |
Brennan MJ. The effect of opioid therapy on endocrine function. Am J Med. 2013;126(Suppl 1):S12–S18. | |
Debono M, Chan S, Rolfe C, Jones TH. Tramadol-induced adrenal insufficiency. Eur J Clin Pharmacol. 2011;67(8):865–867. | |
Ahlfors CE. Benzyl alcohol, kernicterus, and unbound bilirubin. J Pediatr. 2001;139(2):317–319. | |
Parker JW, Mehta R, Lehrfeld K, Manzo C. Risk Assessment and Risk Mitigation Review(s); 2014. Center for Drug Evaluation and Research. [Xartemis XR Application Number 204031Orig1s000]. | |
Available from: http://www.drugs.com/pro/xartemis-xr.html. Accessed on February 13, 2015. | |
Qiu W, Xu Y. Clinical Pharmacology and Biopharmaceutics Review(s); 2013. Center for Drug Evaluation and Research. [Xartemis XR Application Number 204031Orig1s000]. | |
Kilgore E. Medical Review (s); 2013. Center for Drug Evaluation and Research. [Xartemis XR Application Number 204031Orig1s000]. | |
Fields E. Cross Discipline Team Leader Review Addendum; 2014. Center for DrugEvaluation and Research. [Xartemis XR Application Number 20403 1Orig1s000]. | |
Horn JR and Hansten PD. Get to know an Enzyme: CYP2D6. Available from: http://www.pharmacytimes.com/publications/issue/2008/2008-07/2008-07-8624. Accessed on February 13, 2015. | |
Horn JR and Hansten PD. Get to know an Enzyme: CYP3A4. Available from: http://www.pharmacytimes.com/publications/issue/2008/2008-09/2008-09-8687#sthash.4OTK1ba4.dpuf. Accessed on February 13, 2015. | |
Accessdata.fda.gov/[homepage on the Internet]. US Food and Drug Administration. Accessed on February 13, 2015. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.