Back to Journals » OncoTargets and Therapy » Volume 13
Primary Resistance to Combination Therapy with First- and Third-Generation EGFR Tyrosine Kinase Inhibitors of Lung Adenocarcinoma Harboring EGFR 19Del/T790M/in Trans-C797S Mutations with Co-Occurring CTNNB1 Alteration
Received 13 May 2020
Accepted for publication 22 June 2020
Published 9 July 2020 Volume 2020:13 Pages 6749—6753
DOI https://doi.org/10.2147/OTT.S262594
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Federico Perche
Jianxin Chen,1 Junhui Wang,2 Xilin Wu1
1Department of Medical Oncology, Quzhou People′s Hospital, Quzhou, Zhejiang, People’s Republic of China; 2Department of Radiation Oncology, Quzhou People′s Hospital, Quzhou, Zhejiang, People’s Republic of China
Correspondence: Xilin Wu
Department of Medical Oncology, Quzhou People′s Hospital, Quzhou, Zhejiang 324000, People’s Republic of China
Email [email protected]
Background: Epidermal growth factor receptor (EGFR) in trans-C797S alteration has been considered as one of the mechanisms, which leads to the resistance to third-generation tyrosine kinase inhibitors (TKI). Several reports have suggested that combination therapy with first- and third-generation TKIs might be effective for acquired trans-C797S alteration.
Case Presentation: Herein, we report a patient diagnosed with lung adenocarcinoma harboring EGFR 19Del/T790M/in trans-C797S mutations with co-occurring CTNNB1 alteration showing a negative response to combination therapy with first- and third-generation TKIs.
Conclusion: Our report highlights the importance of deploying more informed molecular diagnosis and real-time monitoring strategies to detect co-alterations that drive disease progression and drug resistance, in order to find more effective treatment.
Keywords: EGFR, in trans-C797S, CTNNB1, resistance
Introduction
The third-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs), have been recommended as the standard choice for patients diagnosed with advanced or metastatic non-small cell lung cancer (NSCLC) harboring EGFR sensitive alterations, or progressed on first line TKI acquired T790M mutation. However, resistance to third-generation TKIs seems inevitable as well. It was reported that tumor cells were resistant to third-generation TKI if EGFR T790M and C797S alterations exist in trans,1 however, they could be re-sensitive to the combination with first- and third-generation TKIs. Here, we report a patient diagnosed with lung adenocarcinoma harboring EGFR 19Del/T790M/in trans-C797S mutations with co-occurring CTNNB1 alteration, showing negative response to combination therapy with first- and third-generation TKIs, which deserves examining in clinical practice.
Case Presentation
A 52-year-old man was admitted to our institution on October 25, 2014 with a space-occupying lesion in upper lobe of left lung in association with a solitary mass on his left chest wall detected by chest computed tomography (CT) scan during a health examination in October 2014. The patient had a smoking history of 31 years with 20 cigarettes a dayand he denied any other medical or family history. According to the biopsy findings from the solitary mass on the left chest wall, the patient was diagnosed as lung adenocarcinoma with metastasis on the left chest wall, suggesting metastatic disease (Figure 1). Immunohistochemistry outcomes were presented as: TTF-1 (positive), CK (positive), Napsin-A (positive), CK5/6 (negative), P63 (negative), CD56 (negative), CK7 (positive), and Ki-67 (10%). Because of the deletion of EGFR exon 19 detected by ARMS-PCR (amplification refractory mutation system) with tumor tissue, he started on icotinib as first line treatment in November 2014. According to the criteria of response evaluation criteria in solid tumors (RECIST) version 1.1, best response during the first line was partial response (PR). After 18 months duration time with icotinib, progressive disease (PD) of the primary lesion in lung, as well as pleural effusion was detected with chest CT in May, 2016. Subsequently, he received combined chemotherapy (pemetrexed, cisplatin, docetaxel) with bevacizumab as second-, and third-line treatment, respectively (Figure 2). The cytotoxic administration was terminated because of the emerging lesion in mesocephalon in January, 2018. Next generation sequencing (NGS) using a plasma sample suggested the emerging EGFR exon 20 T790M mutation this time. Then he received systematic therapy with osimertinib and bevacizumab, as well as stereotactic body radiation therapy (SBRT) for the single lesion in mesocephalon. Response efficacy of the systematic therapy was evaluated as PR. The duration time from that regimen was 23 months. On December 23, 2019, the patient received repeated chest CT because of the aggravating chest distress, which revealed the enlarged primary mass in the upper lobe of his left lung, as well as the significantly increased pleural infusion. Oral anlotinib, a small molecule multi-targeted tyrosine kinase inhibitor, was administered as salvage treatment. Bevacizumab was also given by intrapleural injection for the control of pleural infusion. NGS, including 550 tumor-related genes using a plasma sample was conducted to search the potential targets, the results of which suggested EGFR exon 20 mutation (p.T790M, c.2369C>T, frequency as 4.17%), EGFR exon 20 mutation (p.C797S, trans to T790M, c.2390G>C, frequency as 0.37%), and CTNNB1 exon 3 alteration (p.S37F, c.110 C>T, frequency as 2.03%, Figure 3). With the results of NGS, the patient started to receive gefitinib combined with osimertinib as further-line treatment in February, 2020. However, the sudden emerging and rapid enlarged mass on his right neck saw him admitted to our hospital again on March 10, 2020. Cervical magnetic resonance imaging (MRI) showed the emerging lesion, which was finally confirmed with pathologic findings by biopsy (Figure 2). NGS (panel of 1021 cancer related genes) using tissue from the cervical mass was conducted again in April 2020, the results of which re-conformed CTNNB1 exon 3 alteration with higher frequency (p.S37F, c.110 C>T, frequency as 17.6%), as well as the other former alterations including EGFR exon 19del (p.E746-A750del), and EGFR exon 20 mutation (p.T790M, c.2369C>T). However, the C797S alteration (whether trans or cis) was missing this time. Because of the regional pain, especially at the time of feed, he received oral oxycodone 10 mg Q12h, and SBRT again for the cervical lesion. The emerging mass, as well as the pain was controlled well now. The patient is undergoing a regimen of camrelizumab and albumin-bound paclitaxel as salvage treatment. In addition, he also receives oral celecoxib 200 mg twice a day for CTNNB1 exon 3 mutation. Chest CT scans and brain MRI findings during the whole treatment are listed in the Supplementary Appendix.
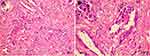 |
Figure 1 H&E staining of tumor sample by biopsy. Notes: (A) Magnified 40 times; and (B) Magnified 200 times. |
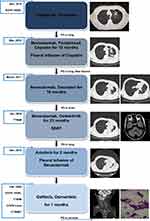 |
Figure 2 Timeline of events since the diagnosis and summary of administered treatments. |
Discussion
The present case study reported a patient diagnosed as lung adenocarcinoma harboring EGFR 19Del/T790M/in trans-C797S and CTNNB1 mutations, with a negative response to combination therapy with first- and third-generation EGFR tyrosine kinase inhibitors, which has been suggested potentially effective in such patients.2–4 This case revealed that combination therapy with first- and third-generation EGFR tyrosine kinase inhibitors might not be effective among all patients with EGFR T790M/in trans-C797S mutations. More effective agents or regimens are still urgently needed.
EGFR C797S alteration was reported as one of the main reasons responsible for the resistance of EGFR TKIs. It is located in cis with T790M most of the time (98%), and rarely in trans with that (2%). Several case reports have suggested that the combination therapy of first- and third- line TKIs might be effective in such patients, though with the potential mechanism uncovered.2,3 However, positive outcomes were not observed in the present patient. After the comparison conducted between the published literature and the present case, we found the noteworthy difference might be the co-occurring genetic alterations of CTNNB1 exon 3 alteration. CTNNB1 gene encodes β-catenin, which is involved in regulation and coordination of cell adhesion and gene transcription. Alterations of β-catenin are associated with abnormal activation of WNT/β-catenin pathway, and lead to the occurrence and progression in many cancer types including colorectal cancer, breast cancer, hepatocellular carcinoma, and endometrial cancer.5 Results from the published researches, with the genomic analysis and whole exome analysis tumor samples from EGFR mutant lung cancer patients, revealed that co-occurring alterations in CTNNB1 might be one of the main reasons giving rise to the resistance of TKI.6 They also hypothesized that the co-occurring alteration of CTNNB1 might function non-redundantly to drive tumor metastasis or limit targeted therapy response, which was partially proven in the present case.6 Limited treatment strategies including celecoxib and everolimus have been suggested potentially effective in patients with the alteration of CTNNB1, however, need further identification.7
Briefly, we present a case of lung adenocarcinoma harboring EGFR 19Del/T790M/in trans-C797S mutations co-occurring with CTNNB1 alteration, showing a negative response to combination therapy with first- and third-generation EGFR TKIs, which challenged the current view of the efficacy of the combined regimen in such patients. Hence, our report has highlighted the importance of deploying more informed molecular diagnosis and real-time monitoring strategies to detect co-alterations that drive disease progression and drug resistance, in order to find more effective treatment.
Data Sharing Statement
All data generated or analyzed during this study are included in this article.
Ethics Statements
The publication of the present case details was approved by Ethical Committee of People′s Hospital of Quzhou.
Consent for Publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Acknowledgments
The authors thank the patient for his participation and his agreement to publication of the report. They also thank Dr Lin Jun, and Dr Li Baizhou for their contribution on data collection.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Niederst MJ, Hu H, Mulvey HE, et al. The allelic context of the C797S mutation acquired upon treatment with third-generation EGFR inhibitors impacts sensitivity to subsequent treatment strategies. Clin Cancer Res. 2015;21(17):3924–3933. doi:10.1158/1078-0432.CCR-15-0560
2. Wang Z, Yang JJ, Huang J, et al. Lung adenocarcinoma harboring EGFR T790M and in trans C797S responds to combination therapy of first- and third-generation EGFR TKIs and shifts allelic configuration at resistance. J Thorac Oncol. 2017;12(11):1723–1727. doi:10.1016/j.jtho.2017.06.017
3. Zhou Z, Zhao Y, Shen S, et al. Durable clinical response of lung adenocarcinoma harboring EGFR 19Del/T790M/in trans-C797S to combination therapy of first- and third-generation EGFR tyrosine kinase inhibitors. J Thorac Oncol. 2019;14(8):2062–2070. doi:10.1016/j.jtho.2019.08.2503
4. Arulananda S, Do H, Musafer A, Mitchell P, Dobrovic A, Thomas J. Combination osimertinib and gefitinib in C797S and T790M EGFR-mutated non-small cell lung cancer. J Thorac Oncol. 2017;12(11):1728–1732. doi:10.1016/j.jtho.2017.08.006
5. Mo HY, An CH, Choi EJ, Yoo NJ. Analysis of promoter mutation in CTNNB1 gene in solid and hematologic neoplasia. Pathol Res Pract. 2019;215(11):152673. doi:10.1016/j.prp.2019.152673
6. Blakely CM, Watkins TBK, Wu W, et al. Evolution and clinical impact of co-occurring genetic alterations in advanced-stage EGFR-mutant lung cancers. Nat Genet. 2017;49(12):1693–1704. doi:10.1038/ng.3990
7. Egashira I, Takahashi-Yanaga F, Nishida R, et al. Celecoxib and 2,5-dimethylcelecoxib inhibit intestinal cancer growth by suppressing the Wnt/beta-catenin signaling pathway. Cancer Sci. 2017;108(1):108–115. doi:10.1111/cas.13106
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

