Back to Journals » Clinical Interventions in Aging » Volume 10
Prevalence of sarcopenia in Germany and the corresponding effect of osteoarthritis in females 70 years and older living in the community: results of the FORMoSA study
Authors Kemmler W , Teschler M, Goisser S, Bebenek M, von Stengel S, Bollheimer LC, Sieber C, Freiberger E
Received 30 May 2015
Accepted for publication 4 July 2015
Published 3 October 2015 Volume 2015:10 Pages 1565—1573
DOI https://doi.org/10.2147/CIA.S89585
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Richard Walker
Wolfgang Kemmler,1 Marc Teschler,1 Sabine Goisser,2 Michael Bebenek,1 Simon von Stengel,1 Leo Cornelius Bollheimer,2,3 Cornel C Sieber,2,3 Ellen Freiberger2
1Institute of Medical Physics, University of Erlangen-Nürnberg, Erlangen, Germany; 2Institute for Biomedicine of Aging, University Erlangen-Nürnberg, Nürnberg, Germany; 3Department of General Internal Medicine and Geriatrics, St John of God Hospital, Regensburg, Germany
Background: Although sarcopenia represents a challenging burden for health care systems around the world, its prevalence in the elderly population varies widely. The primary aim of the study was to determine the prevalence of sarcopenia in community-dwelling (CD) German women aged 70 years and older; the secondary aim was to assess the effect of osteoarthritis (OA) on sarcopenia prevalence in this cohort.
Methods: A total of 689 Caucasian females 18–35 years old and 1,325 CD females 70 years+ living in Northern Bavaria, Germany, were assessed during the initial phase of the FORMoSA research project. Anthropometry, total and regional muscle mass, were assessed by segmental multifrequency Bioelectrical Impedance Analysis. Further 10 m walking speed and handgrip strength were evaluated to apply the European Working Group on Sarcopenia in Older People definition of sarcopenia. Covariates were determined by questionnaires and interviews.
Results: Applying the algorithm of the European Working Group on Sarcopenia in Older People of two standard deviations below the mean value for appendicular skeletal muscle mass of a reference cohort of the young cohort (5.66 kg/m2), low gait speed (≤0.8 m/s), and low grip strength (<20 kg), the prevalence of sarcopenia in CD German females 70 years and older was 4.5% (70–79 years: 2.8% vs ≥80 years: 9.9%; P<0.001). Participants with OA at the hip and lower limbs (n=252) exhibited significantly higher rates of sarcopenia (OA: 9.1 vs non-OA: 3.5%). Of importance, anthropometric, demographic, health, and lifestyle parameters (except exercise participation) of our cohorts corresponded with Bavarian or German data for CD women 70 years+.
Conclusion: The prevalence of sarcopenia in CD German females 70 years+ is relatively low. However, participants with OA at the hip or lower limbs were at increased risk for sarcopenia.
Keywords: sarcopenia, prevalence, osteoarthritis, Germany
Introduction
Sarcopenia, defined as an age-related loss of muscle mass and low muscle function, may be the most prominent component of frailty, disability, and morbidity in older people.1,2 Although this relatively new geriatric syndrome3 represents a challenging burden for health care systems not only, but particularly, in developed countries,4 its prevalence in the elderly population was reported to vary widely.5 For example, for comparable European industrial nations (Belgium, Denmark, Finland, France, Holland, and Northern Italy), the prevalence of sarcopenia for female cohorts 70 years+ ranges from 0.9%6 to 52.9%.7 This finding was related to different definitions of sarcopenia with varying methods, cutoffs, and algorithms.8,9 Additionally, the specific national situation with its social background and health resources may affect the transition from independence to institution10 and thus the prevalence of sarcopenia in the older community-dwelling (CD) cohort.
In Germany, the prevalence of sarcopenia in CD older people is unknown. However, to generate tailored interventions, it would be important to identify the prevalence of this geriatric syndrome in this cohort. Thus, the primary aim of this article was to determine the prevalence of sarcopenia in Germany according to the European Working Group on Sarcopenia in Older People (EWGSOP) definition8 that may be the most accepted protocol in this area. Our second and subordinated research question addressed a more clinical issue and evaluates whether and to which degree osteoarthritis (OA) of the hip and lower limbs, which is rather common in older people, was related to higher sarcopenia prevalence rates.
Our main hypothesis was that the prevalence of sarcopenia according to the EWGSOP-proposal is relatively low (<5%) in CD German females 70 years and older. The secondary hypothesis was that the prevalence of sarcopenia is significantly higher in participants with OA at the hip and lower limbs compared with nonarthritic peers.
Methods
The FORMoSA project (Bavarian Research Association – Sarcopenia and Osteoporosis) was implemented by a Bavarian research network that addresses sarcopenia and osteoporosis under several research aspects. Our research group focuses on the prevalence and nonpharmacologic therapy of sarcopenia and sarcopenic obesity in older females. This article is based on screening data (July–November 2014) from the project. The study was initiated by the Institute of Medical Physics and the Institute of Biomedicine of Aging, University of Erlangen-Nuremberg, Germany and approved by the University Ethics committee (Ethikantrag 905, 4209, 4914 B). All study participants gave written informed consent. FORMoSA was registered under www.clinicaltrials.gov (NCT02356016).
Primary endpoint
Sarcopenia as defined by the EWGSOP.8
Participants
Using citizen registration records, the entire population of 7,908 female participants 70 years and older, living independently in the area of Erlangen, Germany, were contacted by personal letters with detailed study information including the most relevant eligibility criteria for the study. Of the 1,401 women who replied, 1,343 were included in the screening process after application of our eligibility criteria: 1) females, 70 years and older and 2) living independently (autonomous at home, in the community) in the area of Erlangen. Participants were assessed independent of their race; however, only white (Caucasian) participants were included in the analysis (n=1,325). Table 1 shows the characteristics of the eligible participants.
  | Table 1 Characteristics of CD female participants 70 years and older living (n=1,325) |
Assessment of sarcopenia
Sarcopenia was defined per the algorithm of the EWGSOP8 (Figure 1). Correspondingly, we applied a gait speed of ≤0.8 m/s and a handgrip strength of <20 kg11 to determine cutoff values for our female cohort. Following Baumgartner,12 low muscle mass was defined as a relative appendicular skeletal muscle mass (ASMM; kg/m2) of two standard deviations (SDs) below the mean value of a reference cohort of 689 Caucasian females 18–35 years old (Teschler score13). The sample was collected between February and August 2014 in seven different locations of northern Bavaria. With respect to anthropometric (eg, body mass index and body fat) and demographic (eg, family and educational status and lifestyle) parameters, this cohort can be considered as representative for young Bavarian14 and German females.15 Table 2 shows the characteristic of this cohort.
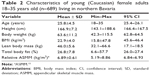  | Table 2 Characteristics of young (Caucasian) female adults 18–35 years old (n=689) living in northern Bavaria |
Assessment of anthropometry and body composition parameter
Height was assessed using a Harpenden stadiometer (Holtain Ltd, Crymych, Pembs, UK), body mass, and composition (total and regional fat and fat-free body mass) were determined using segmental multifrequency Bioelectrical Impedance Analysis (BIA; Inbody 770, Biospace Co., Ltd., Seoul, Korea). This device was used to measure impedance of the trunk, arms, and legs separately with a tetrapolar eight-point tactile electrode system that applied six frequencies (1, 5, 50, 250, 500, and 1,000 kHz).
Total ASMM was based on the sum of the lean body mass (LBM) of the extremities (upper and lower limbs). Relative ASMM was calculated by dividing the total muscle mass (kg) by the height squared (m2). Using the relative ASMM mean value of −2 SDs of the young reference sample (Table 2), the cutoff value for sarcopenia for our cohort was ≤5.66 kg/m2.
Assessment of functional sarcopenia parameters
Muscle strength
Handgrip strength was measured using a Jamar hand dynamometer (Sammons Preston Inc, Bollington, USA). The width of the dynamometers grip was individually adjusted to the hand size of the participant. Tests were performed in an upright standing position, arms down by the side. Two test trials were performed, both for the dominant and nondominant hand; the best trial was included in the analysis.
Gait speed
Gait speed was assessed according to the 10 m test protocol of Fritz and Lusardi.16 Tests were performed twice without any walking aims. Participants were asked to walk 14 m in their usual gait speed using their regular shoes. Participants started in a standing position 2 m before the first photo sensors (HL 2-31, TagHeuer, La Chaux-de-Fonds, Switzerland) and stopped 2 m after the second photo sensors, resulting in a steady-state measurement over 10 m.
Assessment of OA
Questionnaires, personal interviews, and medical letters were used to determine OA. First, all the participants were requested complete general questionnaires to report the incidence of OA and other relevant diseases (see “General characteristics, covariates, and comorbidity” section). Questionnaires were checked by research assistants in close cooperation with the participants. Subjects who listed or reported diseases after inquiry were further interviewed with respect to the corresponding disease. Initially, 246 participants suffered from OA. However, after detailed inquiry, only those subjects who said they had OA based on the diagnosis of their physician were requested to complete a questionnaire that asked for incidence, severity, and localization of OA (n=241). The research assistants then interviewed those participants to verify the data. In cases of doubt, written OA indications attested by the responsible physician were requested. OA indication of nine participants was vague and not confirmed by their physician; thus, in total, 232 subjects were included in the OA group.
General characteristics, covariates, and comorbidity
Beside general characteristics (eg, family and educational status, occupational career, and menopausal age), questionnaires and short interviews were conducted with all participants (n=1,343) to ascertain diseases, medication, and lifestyle with special emphasis on physical activity and fitness using a dedicated questionnaire.17 Participants were asked to list their medication and diseases at home, and the research assistants then checked the questionnaire for completeness and accuracy together with the participant. Specific interviews and questionnaires aimed at confirming and operationalizing specific diseases (eg, osteoporosis, coronary heart diseases, diabetes mellitus, OA) listed by the participants were finally conducted in close cooperation with the participants. In cases of doubt, written indications attested by the responsible doctor were requested.
Statistical analysis
Descriptive statistics (ie, mean values with SD (MV ± SD), and proportions) were used to describe characteristics and key parameters of the study cohort. Additionally, the range and 95% confidence interval have been given in Table 1 to characterize the young study cohort that represents the basis of our T-Score-derived relative ASMM calculation12 more clearly.
Differences between the groups categorized for either age (Tables 1 and 3) or OA (Table 4) were consistently calculated using Welch t-test for continuous variable and chi-square test or, when appropriate, Fisher’s exact test for categorical variables. In detail, the corresponding statistical tests for each group comparison can be found in the legends of Tables 1, 3, and 4. Although at least our secondary hypothesis would allow a one-tailed test procedure, for consistency and conservativeness, all the tests were analyzed with a more discrete two-sided testing procedure with a P-value of less than 0.05 considered as statistically significant. SPSS 21.0 (SPSS Inc, Chicago, IL, USA) was used for all statistical procedures.
  | Table 3 Sarcopenia parameters in 1,325 community-dwelling females 70 years and older living in northern Bavaria, Germany |
  | Table 4 Characteristics of study participants with and without osteoarthritis of the hip and lower limbs |
Results
General characteristic of the study participants were given in Table 1. With respect to anthropometric data, lifestyle, number and distribution of diseases, and medication, the results for our cohort coincide with Bavarian and German data.15,18 The same is true for family, education, and social status (not given in Table 1). Further, the age distribution among our cohort was comparable with Bavarian or German data for CD females 70 years and older.15
Prevalence of sarcopenia in German (Northern Bavaria) women 70 years and older
Table 3 shows the parameters that constituted “sarcopenia” according to the EWGSOP proposal8 for all (n=1,325), women <80 years (n=1,022) and women 80 years and older (n=303). Significant differences between the groups (<80 vs ≥80 years) were observed for all sarcopenia criteria.
Applying the algorithm of the EWGSOP proposal (Figure 1), the prevalence of sarcopenia in our cohort was 4.5% (70–79 years: 2.8% vs ≥80 years: 9.9% P<0.001) (Figure 1). In detail (Figure 1), only 55 women failed to reach the cutoff value of ≤0.8 m/s for gait speed and were thus immediately assessed for muscle mass. For the remaining 1,268 females faster than 0.8 m/s, a grip strength lower than 20 kg (n=237) led to a subsequent muscle mass assessment, while subjects with normal grip strength (and normal gait speed) were defined as nonsarcopenic. Thus, finally muscle mass was determined in the 291 remaining women. Following the EWGSOP criteria for low muscle mass (ie, ≤5.66 kg/m2 for our cohort), altogether 59 subjects were categorized as “sarcopenic” while 232 were nonsarcopenic according to the EWGSOP algorithm.8
Thus, hypothesis 1 confirmed that the prevalence of sarcopenia in CD females 70 years and older was low (<5%).
Effect of OA on sarcopenia prevalence
Table 4 shows the characteristics of participants with and without OA at the hip and lower limbs. As expected, age varies significantly between the groups; however, total difference only averaged ≈1.3 years. More importantly, the groups differ significantly (P<0.001) for parameters that may relevantly affect muscle mass and functional capacity (ie, body fat, number of diseases, long-term glucocorticoid use, physical fitness, and participation in sports and exercise).
Differences of sarcopenia parameters given in Table 4 were not consistently aligned. While functional sarcopenia parameters were significantly lower in women suffering from OA at hip and lower limbs compared with women without OA (-10% to 12%; P<0.001), a slightly higher relative ASMM (0.6%, P=0.480) was determined for the arthritic participants.
In parallel to Figure 1 and the EWGSOP algorithm described earlier, Figure 2 shows the procedure to determine sarcopenia prevalence in participants with OA and without OA (no arthritis [NA]) at the hip, knee, or ankle joint. In accordance with the descriptive sarcopenia parameters given in Table 3, the proportion of women meeting the sarcopenia criteria for gait speed (≤0.8 m/s: OA: 11.6% vs NA: 2.6) and grip strength (<20 kg: OA: 29.9% vs NA: 16.6%) differs significantly (P=0.001) between the groups. Also, the difference between OA and NA participants with low muscle mass (≤5.66 kg/m2) remained nonsignificant (OA: 23.9 vs NA: 18.7; P=0.327) during the final step (measure muscle mass) of the EWGSOP assessment. However, the low(er) sample size of remaining participants during this final step may contribute to the lack of significant results.
In summary, the prevalence of sarcopenia in women with OA compared with NA peers was significantly higher than in women without corresponding limitation.
Thus, hypothesis 2 confirmed is that the prevalence of sarcopenia was significantly higher in participants with OA compared with nonarthritic peers.
Discussion
To our knowledge, this is the first study to address the prevalence of sarcopenia in CD older females in Germany. In summary, the overall prevalence rate was relatively low (4.5%) in this cohort of Bavarian (German) females 70 years and older. This can also be applied for our subgroup of females 80 years or older (9.9%). Reviewing the literature, Cruz-Jentoft5 reported a prevalence rate of 1%–30% in women 50 years and older. Although only studies that applied the EWGSOP definition (ie, muscle mass, muscle strength, or functional performance) were included, the methods to determine these parameters vary widely. As an example, the study that reported the highest sarcopenia rate (30.1%) determined midarm circumference19 in Italian females 80–85 years old. Additionally, the inconsistency of the diagnostic muscle mass criteria (eg, LBM, SMM, and ASMM), the corresponding calculation (ie, muscle mass adjusted for height,12 weight,20 or/and fat mass21,22), and the cutoff definition (T-Score vs Z-Score based) complicate a meaningful comparison of the present studies.
Limiting the data to CD European females 70 years and older applying the EWGSOP algorithm and using dual-energy X-ray absorptiometry (DXA) or segmental multifrequency BIA devices to determine ASMM (kg/m2), sarcopenia prevalence ranges from 0.9% in 70- to 80-year-old Finnish women6 to 20.2% for a female Belgian cohort 65 years and older.23 Our result basically confirmed the lower rates of Patil et al,6 especially when taking into account that the authors used the lower ASMM cutoff value of Baumgartner et al12 (≤5.45 kg/m2). Application of this lower cutoff point resulted in a sarcopenia prevalence of 3.1% in our cohort (70–79: 1.8% vs ≥80 years: 7.6%). Revisiting the study of Beaudart et al23 in one of their eight approaches, the authors applied sarcopenia cutoff points (ASMM: ≤5.67 kg/m2,21 gait speed <0.8 m/s, grip strength <20 kg) nearly identical to the present study. However, although the Belgian cohort (n=243) was slightly younger (73.8±6.2 vs 76.4±4.9 years), its sarcopenia prevalence was more than three times as high (16%) compared with the present study. It is implausible that difference with respect to body-composition assessment (ie, DXA vs BIA) contributes to this result since both Patil et al6 and Beaudart et al23 used DXA devices. Additionally, it is rather unlikely that slight variations with respect to functional testing (eg, 4 vs 10 m walking trial, stopwatch vs automatic timers) could lead to such differences. Although Pasma et al24 reported a significantly higher gait speed for 10 m compared with the 4 m walking trial in a cohort of elderly outpatients, this result was not confirmed for healthy, CD elderly people 65 years and older.25 However, it is noticeable that gait speed among the present study (1.29±0.23 m/s) was considerably higher than described for a comparable cohort.26 In contrast, per the EWGSOP algorithm, participants with gait speed above 0.8 m/s were not excluded but assessed additionally for handgrip strength, which proved to be in the range of normative data for this cohort,27 however.
Thus, in summary, the finding that sarcopenia prevalence may considerably vary between neighboring countries despite highly comparable or even identical sarcopenia definition, criteria, method, cutoff, sex, age, social system, and origin was unexpected and should be addressed by dedicated studies.
Addressing our second and subordinated research topic, we demonstrated that sarcopenia prevalence in CD women 70+ years was significantly higher in women with OA at the hip and lower limbs.
Indeed, clinical evidence suggests that both conditions may be linked via functional and cellular pathways.28 With respect to the functional pathway, lower limb muscle weakness favors OA progression and vice versa.29,30 Indeed, habitual walking speed was significantly lower in the OA group compared with the NA group, a result confirmed by Waters et al31,32 Additionally, a higher proportion among our OA group (OA: 19% vs NA: 13%) showed OA of the hand and finger joints, which may explain33 the lower handgrip strength of this group. However, we are unable to determine a lower ASMM (or lower limb muscle mass) in the OA subgroup compared with the NA group (Table 4). This finding was not expected. In addition to the functional interaction of muscle and cartilage, chondrocytes and skeletal muscle cells share common pathological pathways;28 thus, muscle mass in participants with OA should be reduced. However, a recent review the literature on OA and sarcopenia (ie, low muscle mass)34 stated that neither a direct effect of sarcopenia on OA development nor the opposite relation can be supported on the basis of the present literature. In summary, although more dedicated studies should address this issue, the study provided some evidence that OA contributed to the development of sarcopenia in elderly females. As a clinical implication, physicians should be aware of an increased risk of sarcopenia in their patients with OA. Consequently, these patients should be encouraged to start dedicated exercise programs and protein/vitamin D supplementation to maintain or increase their muscle mass.
However, some features or limitations may reduce the evidence of the present studies, especially with respect to our secondary hypothesis: 1) It can be argued that the spatial restriction on a single German region and/or incomplete compliance of the participants contacted (16.8%) generated a sampling bias that prevents generalization of our results. We are unable to completely negate this argument, although the most important anthropometric (eg, body mass index and age distribution), demographic (eg, education and family status), and health (eg, diseases and medication) and lifestyle parameters (eg, smoking, and habitual physical activity) of our cohort corresponded with Bavarian or German data for CD women ≥70 years.15,18,35 However, participation in sports and exercise by our cohort (59.5%) was considerably higher than reported for the basic population of German females ≥65 years (50.8%). Since the study participants undertaking exercise demonstrated a significantly higher gait speed compared with their sedentary peers (1.32 vs 1.18 m/s), this may contribute to the exceptionally high gait speed in our cohort. Due to varying exercise participation in the OA vs NA group (Table 4), this factor may also affect the OA/sarcopenia issue, although the corresponding cause–effect association (ie, chicken–egg problem) remains unclear.28 2) A limitation with respect to our second research aim was the cross-sectional nature of the study with the corresponding restricted ability to determine causal effects. 3) Also our procedure for recording OA incidence via questionnaires and interviews alone was suboptimum. Retrospectively, a specific evaluation of OA using more sophisticated assessments and diagnostic tools may have allowed a more comprehensive analysis including the application of logistic regression models to determine the independent effect of OA on sarcopenia prevalence. 4) With respect to body composition assessment, we decided to use segmental multifrequency BIA for two main reasons. First, BIA is considered as a time- and cost-effective technique with portable devices and thus optimal for screening of large cohorts.36 Further, due to the lack of radiation, this technique is highly accepted and can be applied without any ethical or administrative restrictions. With respect to the accuracy of this technique, Beaudart et al37 reported a systematic overestimation of muscle mass when compared with DXA that is still the “golden standard”. However, other researchers and we do not share this opinion.38–40 Particularly, Ling et al38 reported an “excellent agreement” of BIA (InBody 720, Biospace Ltd, Seoul, Korea) and DXA (QDR 4500a; Hologic, Bedford, MA, USA). In any case, since our cutoff for “low muscle mass” is based on either a young reference cohort (<-2 SD T-Score) or the distribution (lowest quintile) within the own cohort as assessed with the same BIA device, prevalence rates should not be affected.
Acknowledgments
This work was supported by the “Bayerische Forschungsstiftung” (Munich, Germany) (grant number AZ 1044-12) and the “Universitätsbund Erlangen-Nürnberg” (grant number 21/2015).
Author contributions
All of the authors 1) made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; 2) drafted the article or revised it critically for important intellectual content; 3) finally approved the version to be published; and 4) agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Disclosure
The authors report no conflicts of interest in this work.
References
Milte R, Crotty M. Musculoskeletal health, frailty and functional decline. Best Pract Res Clin Rheumatol. 2014;28(3):395–410. | ||
Fried LP, Guralnik JM. Disability in older adults: evidence regarding significance, etiology, and risk. J Am Geriatr Soc. 1997;45(1):92–100. | ||
Cruz-Jentoft AJ, Landi F, Topinkova E, Michel JP. Understanding sarcopenia as a geriatric syndrome. Curr Opin Clin Nutr Metab Care. 2010;13(1):1–7. | ||
Janssen I, Shepard DS, Katzmarzyk PT, Roubenoff R. The healthcare costs of sarcopenia in the United States. J Am Geriatr Soc. 2004;52(1):80–85. | ||
Cruz-Jentoft AJ, Landi F, Schneider SM, et al. Prevalence of and interventions for sarcopenia in ageing adults: a systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing. 2014;43(6):748–759. | ||
Patil R, Uusi-Rasi K, Pasanen M, Kannus P, Karinkanta S, Sievanen H. Sarcopenia and osteopenia among 70-80-year-old home-dwelling Finnish women: prevalence and association with functional performance. Osteoporos Int. 2013;24(3):787–796. | ||
Zoico E, Di Francesco V, Guralnik JM, et al. Physical disability and muscular strength in relation to obesity and different body composition indexes in a sample of healthy elderly women. Int J Obes Relat Metab Disord. 2004;28(2):234–241. | ||
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412–423. | ||
Fielding RA, Vellas B, Evans WJ, et al. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International working group on sarcopenia. J Am Med Dir Assoc. 2011;12(4):249–256. | ||
Lloyd-Sherlock P. Epidemiological change and health policy for older people in developing countries: some preliminary thoughts. Ageing Horizons. 2005;2:21–24. | ||
Lauretani F, Russo CR, Bandinelli S, et al. Age-associated changes in skeletal muscles and their effect on mobility: an operational diagnosis of sarcopenia. J Appl Physiol. 2003;95(5):1851–1860. | ||
Baumgartner RN, Koehler KM, Gallagher D, et al. Epidemiology of sarcopenia among the elderly in New Mexico. Am J Epidemiol. 1998;147(8):755–763. | ||
Kemmler W, von Stengel S, Teschler M. Prävalenz von Sarkopenie und Sarcopenic Obesity bei selbstständig lebender Frauen über dem 70. Lebensjahr [Prevalence of sarcopenia and sarcopenic obesity in community dwelling females 70 years and older]. Osteologie. 2015;24(1):20. | ||
BayLfStaD. Statistisches Jahrbuch für Bayern 2014 [Statistical annual for Bavaria 2014]. München: Bayerisches Landesamt für Statistik; 2014. | ||
DESTATIS. Statistisches Jahrbuch 2014 [Statistical annual 2014]. Wiesbaden, Germany: Statistisches Bundesamt; 2014. | ||
Fritz S, Lusardi M. White paper: “walking speed: the sixth vital sign”. J Geriatr Phys Ther. 2009;32(2):46–49. | ||
Kemmler W, Weineck J, Kalender WA, Engelke K. The effect of habitual physical activity, non-athletic exercise, muscle strength, and VO2max on bone mineral density is rather low in early postmenopausal osteopenic women. J Musculoskelet Neuronal Interact. 2004;4(3):325–334. | ||
Verteilung der Bevölkerung auf Body-Mass-Index-Gruppen in Prozent. Statistisches Bundesamt; 2012. Available from: https://www.gbe-bund.de/stichworte/BMI.html. Accessed March 30, 2015. | ||
Landi F, Liperoti R, Russo A, et al. Association of anorexia with sarcopenia in a community-dwelling elderly population: results from the ilSIRENTE study. Eur J Nutr. 2013;52(3):1261–1268. | ||
Janssen I, Heymsfield SB, Ross R. Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc. 2002;50(5):889–896. | ||
Delmonico MJ, Harris TB, Lee JS, et al. Alternative definitions of sarcopenia, lower extremity performance, and functional impairment with aging in older men and women. J Am Geriatr Soc. 2007;55(5):769–774. | ||
Newman AB, Kupelian V, Visser M, et al. Sarcopenia: alternative definitions and associations with lower extremity function. J Am Geriatr Soc. 2003;51(11):1602–1609. | ||
Beaudart C, Reginster JY, Slomian J, Buckinx F, Locquet M, Bruyere O. Prevalence of sarcopenia: the impact of different diagnostic cut-off limits. J Musculoskelet Neuronal Interact. 2014;14(4):425–431. | ||
Pasma JH, Stijntjes M, Ou SS, Blauw GJ, Meskers CG, Maier AB. Walking speed in elderly outpatients depends on the assessment method. Age (Dordr). 2014;36(6):9736. | ||
Peters DM, Fritz SL, Krotish DE. Assessing the reliability and validity of a shorter walk test compared with the 10-Meter Walk Test for measurements of gait speed in healthy, older adults. J Geriatr Phys Ther. 2013;36(1):24–30. | ||
Bohannon RW, Williams Andrews A. Normal walking speed: a descriptive meta-analysis. Physiotherapy. 2011;97(3):182–189. | ||
Desrosiers J, Bravo G, Rejean H, Dutil E. Normative Data for grip strength of elderly men and women. Am J Occup Ther. 1994;49(7):637–644. | ||
De Ceuninck F, Fradin A, Pastoureau P. Bearing arms against osteoarthritis and sarcopenia: when cartilage and skeletal muscle find common interest in talking together. Drug discovery today. 2014;19(3):305–311. | ||
Conroy MB, Kwoh CK, Krishnan E, et al. Muscle strength, mass, and quality in older men and women with knee osteoarthritis. Arthritis Care Res. 2012;64(1):15–21. | ||
Scott D, Blizzard L, Fell J, Jones G. Prospective study of self-reported pain, radiographic osteoarthritis, sarcopenia progression, and falls risk in community-dwelling older adults. Arthritis Care Res. 2012;64(1):30–37. | ||
Waters RL, Perry J, Conaty P, Lunsford B, O’Meara P. The energy cost of walking with arthritis of the hip and knee. Clin Orthop Relat Res. 1987(214):278–284. | ||
Zeni JA, Higginson JS. Knee osteoarthritis affects the distribution of joint moments during gait. The Knee. 2011;18(3):156–159. | ||
Ding M, Odgaard A, Hvid I. Accuracy of cancellous bone volume fraction measured by micro-CT scanning. J Biomech. 1999;32(3):323–326. | ||
Papalia R, Zampogna B, Torre G, et al. Sarcopenia and its relationship with osteoarthritis: risk factor or direct consequence? Musculoskeletal surgery. 2014;98(1):9–14. | ||
Robert-Koch-Institut. Daten und Fakten: Ergebnisse der Studie ≫Gesundheit in Deutschland aktuell 2012≪ [Data and facts: results of the “health in germany study 2012”]. Berlin: Robert-Koch-Institut; 2014. | ||
Rubbieri G, Mossello E, Di Bari M. Techniques for the diagnosis of sarcopenia. Clinical Cases in Mineral and Bone Metabolism: the Official Journal of the Italian Society of Osteoporosis, Mineral Metabolism, and Skeletal Diseases. 2014;11(3):181–184. | ||
Beaudart C, Reginster JY, Slomian J, et al. Estimation of sarcopenia prevalence using various assessment tools. Exp Gerontol. 2015;61:31–37. | ||
Ling CH, de Craen AJ, Slagboom PE, et al. Accuracy of direct segmental multi-frequency bioimpedance analysis in the assessment of total body and segmental body composition in middle-aged adult population. Clinical Nutrition. 2011;30(5):610–615. | ||
Shafer KJ, Siders WA, Johnson LK, Lukaski HC. Validity of segmental multiple-frequency bioelectrical impedance analysis to estimate body composition of adults across a range of body mass indexes. Nutrition. 2009;25(1):25–32. | ||
von Stengel S, Kemmler W, Engelke K. Validität von BIA im Vergleich zur DXA bei der Erfassung der Körperzusammensetzung [Validity of BIA to determine body composition. A comparison with DXA]. Dtsch Z Sportmed. 2013;62(7/8):200. | ||
Schafer I, von Leitner EC, Schon G, et al. Multimorbidity patterns in the elderly: a new approach of disease clustering identifies complex interrelations between chronic conditions. PLoS One. 2010;5(12):e15941. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


