Back to Journals » Clinical Interventions in Aging » Volume 17
Preoperative Correction of Low Hemoglobin Levels Can Reduce 1-Year All-Cause Mortality in Osteoporotic Hip Fracture Patients: A Retrospective Observational Study
Authors Manosroi W , Atthakomol P , Isaradech N, Phinyo P , Vaseenon T
Received 20 December 2021
Accepted for publication 4 February 2022
Published 17 February 2022 Volume 2022:17 Pages 165—173
DOI https://doi.org/10.2147/CIA.S354519
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Maddalena Illario
Worapaka Manosroi,1 Pichitchai Atthakomol,2,3 Natthanaphop Isaradech,4 Phichayut Phinyo,3,5 Tanawat Vaseenon2
1Division of Endocrinology, Department of Internal Medicine, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand; 2Department of Orthopaedics, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand; 3Clinical Epidemiology and Clinical Statistic Center, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand; 4Biomedical Informatics Center, Department of Family Medicine, Chiang Mai University, Chiang Mai, Thailand; 5Department of Family Medicine, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
Correspondence: Pichitchai Atthakomol, Department of Orthopaedics, Faculty of Medicine, Chiang Mai University, Chiang Mai, Muang Chiang Mai, Thailand, Tel +66 53 936453 Email [email protected]
Purpose: Osteoporotic hip fracture surgery is associated with a risk of morbidity and mortality. Admission hemoglobin levels < 10 g/dL have been documented as a strong predictor of mortality risk. This study aimed to investigate the mortality outcome between osteoporotic hip fracture patients who had preoperative hemoglobin levels raised to ≥ 10 g/dL and those with hemoglobin levels were < 10 g/dL.
Patients and Methods: This 5-year retrospective observational study included 226 participants with osteoporotic hip fractures that required surgery and who had admission hemoglobin levels < 10 g/dL. Patients were categorized into two groups: those with corrected preoperative hemoglobin ≥ 10 g/dL and those with either corrected or uncorrected preoperative hemoglobin < 10 g/dL. Outcomes were analyzed using Cox proportional hazard regression adjusted for confounders. Results are presented as hazard ratio (HR) and 95% confidence interval (95% CI).
Results: Among 226 the patients, the overall mortality rate was 17.25% (n=39/226) of the 226 patients, 93 (41.15%) had their hemoglobin levels raised to ≥ 10 g/dL by red blood cell transfusion. Multivariable analysis after adjustment for confounders showed a 50% lower incidence of mortality among patients with preoperative hemoglobin levels ≥ 10 g/dL than among those with hemoglobin levels < 10 g/dL (HR 0.50, 95% CI (0.25– 0.99), p=0.048).
Conclusion: In osteoporotic hip fracture patients with admission hemoglobin < 10g/dL, raising preoperative hemoglobin levels to ≥ 10 g/dL can significantly reduce the risk of mortality. Testing for and correction of low preoperative hemoglobin levels is of value in hip surgery patients.
Keywords: osteoporosis, hip fracture, anemia, mortality
Introduction
Hip fracture is a major public health concern as it is associated with an increased risk of morbidity, mortality, loss of independence and a high healthcare burden.1 A wide variation in hip fracture incidence has been reported globally. An increase in the incidence of hip fracture from 1.26 million in 1990 to 4.5 million by 2050 has been predicted.2 The first-year post-hip fracture mortality rate in the elderly is high, ranging from 18% to 30%.3–5 The risk of death increases approximately 4% per year in elderly patients. The most critical period is the first year following the fracture event.6 After 2 years, the risk of death was found to decrease noticeably but the risk was still significantly greater than in the control groups with no hip fracture.7
Predictive factors found to be related to an increased mortality rate in osteoporotic hip fracture patients include male gender, advanced age, ethnicity, physical performance, institutionalization, type of fracture, operative treatment method, longer time from injury to operation, results of some biochemical investigations and underlying medical illness.8–12 One of the most frequently reported predictive factors was low hemoglobin levels. Among geriatric hip fracture patients, approximately 40% present with hemoglobin levels less than 12 g/dL.13 In addition, admission hemoglobin levels in the anemic range (<10 g/dL) have been reported to be a strong predictor of increased mortality risk following hip fracture.10,14 In addition to an increased in mortality rate, anemia in hip fracture patients has been reported to be related to increased transfusion rates, hospital re-admission and poorer functional outcomes regardless of the severity of the anemia.15,16 Tests for hemoglobin levels prior to hip fracture surgery are widely available, easy to interpret and inexpensive. Importantly, low hemoglobin levels are modifiable predictors of mortality and can be corrected by red blood cell transfusion. Currently, evidence regarding the timing of hemoglobin correction by transfusion, ie, before, during or after hip fracture surgery, still needs to be clarified. Most studies have focused on the benefits of correction during the intra- and postoperative periods.17,18 There are as yet no studies of mortality rates in osteoporotic hip fracture patients with admission hemoglobin levels <10 g/dL who had been transfused preoperatively to raise their hemoglobin to ≥10 g/dL. The present study aimed to determine the 1-year all-cause mortality outcomes in osteoporotic hip fracture patients who had preoperative hemoglobin levels <10 g/dL and who had had preoperative transfusions to raise their hemoglobin to ≥10 g/dL.
Patients and Methods
A 5-year retrospective observational study was conducted between January 2014-December 2018 in tertiary care medical Center in Thailand. The study was approved by local ethical committee of the Faculty of Medicine, Chiang Mai University. The patients’ Clinical and biochemical data were retrieved from the electronic medical records. The inclusion criteria were 1) Thai patients age above 50 years with osteoporotic hip fracture resulting from simple fall, 2) patients who had subsequently undergone any type of hip fracture operation and 3) admission hemoglobin levels <10 g/dL. The exclusion criteria were mentioned in our previous publication.10 In brief, patients with bilateral hip fracture, previous hip fracture, more than one area of fractures, pathological hip fractures or high energy mechanism fractures were excluded. Data retrieved included basic demographics, underlying medical illness, type of fracture, type of surgery, American Association of Anesthesiologist (ASA) score19 and time to operation. Biochemical investigation information included admission hemoglobin levels, post-transfusion hemoglobin levels, creatinine with estimated glomerular filtration rate (eGFR) and serum albumin levels. The 1-year all-cause mortality data was acquired from medical records for in-hospital mortality and from the Thailand Civil Registration office in cases where data was not available in hospital records. The time to death started at the date of hip injury. eGFR was calculated using the Modification of Diet in Renal Disease Study (MDRD) formula. The admission hemoglobin level was defined as the level on the first day of admission. The preoperative hemoglobin level was defined as the level, either the corrected or uncorrected, obtained prior surgery. For patients whose level was not corrected by transfusion, admission hemoglobin levels were considered to be equivalent to preoperative levels.
The decision whether to transfuse red blood cells or not was made by each individual surgical team. The acceptable threshold level for hemoglobin before proceeding to surgery was determined by the individual surgeons. For this study, patients were categorized into one of two groups: those with corrected preoperative hemoglobin ≥10 g/dL and those with corrected or uncorrected preoperative hemoglobin <10 g/dL. All patients with preoperative hemoglobin ≥10 g/dL had been transfused with red blood cells. All methods were performed in accordance with the relevant guidelines and regulations. Informed consent was not obtained due to retrospective nature of the study.
Statistical Analysis
Statistical analysis was conducted using STATA program version 15.0. Categorical data are presented as counts and percentages. Continuous variables are presented as means and standard deviations (SD). For inferential statistics, Fisher’s exact test was used for categorical variables and Student’s t-test for continuous variables. Univariable and multivariable analyses were performed using Cox proportional hazard regression between mortality and predictors and hazard ratio (HR) with a 95% confidence interval (95% CI). In the multivariable analysis, potential confounders, including age, sex, ASA score, dementia and location of fracture, were adjusted. Time from injury to time of operation ≥48 hours was adjusted by stratified random sample method. Multiple imputation analysis was employed for predictive variables with more than 5% missing. Statistical significance was set as p < 0.05. The collinearity of each potential confounder was evaluated. Potential confounders which had a variance inflation factor (VIF) value >5 were excluded from the multivariable analysis. Subgroup analysis comparing patients that had corrected hemoglobin levels ≥10 g/dL and patients with corrected hemoglobin levels < 10g/dL was planned to demonstrate. Sample size was calculated by means of an exponential test comparing two independent hazard rates. Adequate sample size was demonstrated if the backward calculation of power of analysis was more than 0.80.
Results
There were 226 patients who had osteoporotic hip fracture with admission hemoglobin levels <10 g/dL. The overall mortality rate was 17.25% (n=39/226). There were 93 patients (41.15%) that had corrected hemoglobin levels ≥10 g/dL and 133 patients (58.84%) with corrected or uncorrected hemoglobin levels <10g/dL. Leukocyte poor packed red blood cells transfusion was used in all patients who received the blood transfusion. The majority of the patients were female (74.34%, n=168/226). The mean age was 81.51±8.28 years. Mean body mass index (BMI) was 20.06±3.74 kg/m2. The most common underlying comorbidity was hypertension (65.93%, n=149/226) followed by diabetes mellitus (23.89%, n=54/226). Intertrochanteric fracture was the most common type of injury (78.32%, n=177/226). Cephalomedullary nailing was the most frequently performed operation (56.19%). Most patients had time from injury to operation ≥48 hours (98.67%, n=223/226). BMI was significantly higher in patients with preoperative hemoglobin <10 g/dL than those with ≥10 g/dL (p = 0.044). Other Clinical factors were not significantly different between the two groups. As to admission biochemical data, all laboratory tests, including admission hemoglobin levels, were similar between the groups although preoperative hemoglobin levels were significantly higher in the group which received red blood cell transfusion (p < 0.001) (Table 1). In patients who had preoperative hemoglobin <10 g/dL (n = 133), 94 patients (70.68%, n=94/133) received the preoperative transfusion.
 |
Table 1 Baseline Clinical and Biochemical Characteristics of Hip Fracture Patients (n=226) |
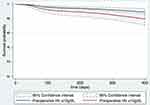
Univariable analysis revealed that the patients whose hemoglobin had been increased to ≥10 g/dL showed no significant reduction in mortality rate compared to patients whose hemoglobin remained <10 g/dL. All confounders had a variance inflation factor (VIF) value <5. We did not perform multiple imputation analysis because there was no missing data more than 5% in potential confounders. After multivariable analysis and adjusting for potential confounders including male, age at admission ≥85 years, dementia, ASA score >2, fractured neck of femur, time from injury to operation ≥48 hours, a significant reduction in mortality rate of 50% was observed (HR 0.50, 95% CI (0.25–0.99) and p = 0.048) (Table 2). The survival graph comparing survival probability between patients who had preoperative hemoglobin ≥10 g/dL and patients who had preoperative hemoglobin <10 g/dL after adjusting the confounders is shown in Figure 1. Univariable and multivariable analysis of 1-year all-cause mortality in subgroup analysis comparing 93 patients that had corrected hemoglobin levels ≥10 g/dL and 94 patients with corrected hemoglobin levels <10g/dL. Stratified by time from injury to operation ≥48 hours or <48 hours showed a strongly significant reduction in 1-year mortality of 41% in patients had corrected hemoglobin levels ≥10 g/dL compared to patients with corrected hemoglobin levels <10g/dL. After adjusting the confounders (HR 0.41, 95% CI (0.20–0.82) and p=0.013) (Table 3).
 |
Table 2 Univariable and Multivariable Analysis of 1-Year All-Cause Mortality in Hip Fracture Patients Stratified by Time from Injury to Operation ≥48 Hours or <48 Hours |
Discussion
The singular finding in this study was that an increase of preoperative hemoglobin levels to ≥10 g/dL had an association with a 50% reduction in 1-year mortality among the osteoporotic hip fracture patients. The results from subgroup analysis comparing 1-year mortality between patients had corrected hemoglobin levels ≥10 g/dL and patients with corrected hemoglobin levels <10g/dL was also confirmed our finding. This is an indication that preoperative hemoglobin level is an important and modifiable factor in osteoporotic hip fracture patients who will be undergoing surgery.
Preoperative anemia has been reported to be associated with an increased risk of morbidity and mortality in osteoporotic hip fracture patients, particularly those with hemoglobin <10 g/dL.10,14 The anemia can be the result of trauma-induced blood loss, iron deficiency, anemia from chronic comorbidities or, in the elderly, bone marrow dysfunction. However, there is currently no standard recommendation regarding who should have anemia correction by transfusion before hip fracture surgery, when such a transfusion should be administered and there is no standard cutoff values used to define the preoperative anemia.20 There has been a wide variability in studies about the timing of transfusion in hip fracture surgery, ie, whether it should be preoperative, intraoperative, or postoperative. Most studies which included a large sample explored the outcomes of postoperative transfusion.18,21 However, the benefits of preoperative optimization of hemoglobin levels have been established for other types of orthopaedic surgeries including hip arthroplasty and total knee arthroplasty. Observed benefits have included a decrease in complications, readmission, postoperative transfusion and length of hospital stay.22,23
The present study found that preoperative transfusion which raised hemoglobin to ≥10 g/dL was significantly associated with a 50% reduction in 1-year all-cause mortality. We proposed that red blood cell transfusion can facilitate an increase in the oxygen-carrying capacity of blood and that it promotes positive outcomes hip fracture surgery in multiple ways. First, a preoperatively corrected hemoglobin level can also raise the postoperative level. A study revealed that an adequate postoperative hemoglobin concentration was linked to better short-term functional recovery after hip fracture surgery24 and a reduction in symptoms of anemia, eg, fatigue, weakness and decreased physical function. Treadmill testing in a healthy population showed that reduction in VO2 max (maximum oxygen consumption) had a positive correlation with a reduction in hemoglobin levels.25 Similarly, there was an improvement in aerobic exercise capacity in patients with end-stage renal disease who had had their hemoglobin levels raised by erythropoietin injection.26 Lower functional capacity and physical fitness have been reported to be related to increased mortality risk, especially in the elderly.27 Second, a prior study demonstrated that liberal transfusion aimed at achieving hemoglobin levels of 10 g/dL (liberal strategy) showed a reduction in the rate of major cardiac events twice that with a hemoglobin target at 8 g/dL (restrictive strategy).28 In addition, patients with postoperative anemia demonstrated a higher incidence of delirium and nosocomial infection.29,30 Both prolonged delirium or cognitive impairment and nosocomial infection were associated with an increased risk of death.31 However, the present study did not include details of the causes of death.
Nevertheless, a red blood cell transfusion itself can increase morbidity in hip fracture surgery patients, with most deaths occurring during the postoperative period. An increased rate of urinary tract infection following major orthopedic surgery has also been reported.32 Other adverse events that can occur after perioperative transfusion include transfusion-associated circulatory overload, hemolytic reactions, allergic reactions, transfusion-related infection and transfusion-related lung injury.33,34 Storage of red blood cell for more than 2 weeks has been found to be related to a high risk of in-hospital mortality and postoperative complications.35 Therefore, a trade-off must be made between the risk of the negative effect of preoperative anemia and the potential adverse effects of blood transfusion.
We acknowledge some limitations in this study. As this is a retrospective study, some selection bias may have been introduced. Future studies using a randomized controlled trial are warranted to more clearly demonstrate actual benefits. Some data such as reason for transfusion or total number of red blood cell transfusion did not be recorded in all patients. The preoperative volume of crystalloid administered following red blood cell transfusion was not evaluated. An excess of crystalloid fluid could result in the dilution of the hemoglobin concentration preoperatively. There was no standard guideline of when to transfuse. The decision whether to transfuse red blood cells or not was made by each individual surgical team and the acceptable threshold of hemoglobin level was decided by the individual surgeons. Data on hemoglobin levels postoperatively and before discharge were not available in our study. In our study, the advantages of raising hemoglobin levels were postulated based on postoperative period hemoglobin levels. Thus, it was not possible to directly evaluate the association between postoperative hemoglobin levels and mortality outcomes. Also, we did not adjust for some intraoperative and postoperative factors, eg, operative time, intraoperative blood loss, postoperative infection rate, nutritional status, functional capacity after rehabilitation intervention and fall prevention strategies in the multivariable model, factors which could potentially impact mortality outcomes due to the limitation of sample size.36 As the majority of the patients with preoperative hemoglobin levels <10 g/dL were transfused, the factor which directly influenced the mortality outcome should be preoperative level of hemoglobin instead of red blood cell transfusion. Therefore, our study cannot demonstrate the association between preoperative transfusion and mortality outcome in hip fracture patients. Further study should be conducted to address this issue. This study had several strengths. First, the ASA scores, which assess overall health status, were recorded as a preoperative risk predictor. However, some comorbidities are not incorporated in that scoring system, eg, dementia and cognitive impairment. As these factors have been linked with increased mortality in the elderly, we have included both ASA scores and dementia as adjusted confounders in multivariable analysis to reduce the occurrence of bias. Second, multiple confounding factors that could potentially impact mortality outcome, eg, sex, extreme age, ASA score and dementia, have been adjusted for accordingly. Third, including the timing of preoperative transfusion in our study was novel: few studies have considered this issue. Fourth, time from injury to operation ≥48 hours was adjusted by stratified random sample in the multivariable analysis. This helps reduce the incidence of bias as there has been a report that a delayed time of surgery of ≥48 hours can increase the risk of death.37 Also, the sample size in our study is adequate for drawing conclusions. We used reverse power analysis to determine the hazard ratio of preoperative Hb >10 g/dL which demonstrated an adequate power of analysis (>0.80).
Conclusion
In osteoporotic hip fracture surgery patients, preoperative correction of hemoglobin to >10 g/dL was found to be associated with a 50% reduction in mortality. Hemoglobin level testing prior to surgery is not complicated and is available in virtually all institutions. It is also one of the easiest-to-modify among the predictive mortality factors in hip fracture surgery. We suggest that the preoperative hemoglobin levels may be considered as one of the preoperative planning in osteoporotic hip fracture surgery patients. Further research with a larger sample size and a randomized trial is warranted.
Abbreviations
ASA score, American Association of Anesthesiologist score; BMI, Body mass index; CI, Confidence interval; eGFR, Estimated glomerular filtration rate; HR, Hazard ratio; MDRD, Modification of Diet in Renal Disease Study; SD, Standard deviations; VIF, variance inflation factor.
Data Sharing Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Ethics Approval
This study was approved by local ethical committee of the Faculty of Medicine, Chiang Mai University. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. Informed consent was waived due to retrospective nature of the study. Authors confirmed that the data was anonymized or maintained with confidentiality.
Acknowledgments
The authors are grateful to Dr G. Lamar Robert, Ph.D. and Dr. Chongchit S. Robert, Ph.D., for editing the manuscript.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
The authors received no financial support in this research.
Disclosure
All authors declared no conflicts of interest for this work.
References
1. Zhang C, Feng J, Wang S, et al. Incidence of and trends in hip fracture among adults in urban China: a nationwide retrospective cohort study. PLoS Med. 2020;17(8):e1003180. doi:10.1371/journal.pmed.1003180
2. Cauley JA, Chalhoub D, Kassem AM, Fuleihan Gel H. Geographic and ethnic disparities in osteoporotic fractures. Nat Rev Endocrinol. 2014;10(6):338–351. doi:10.1038/nrendo.2014.51
3. Civinini R, Paoli T, Cianferotti L, et al. Functional outcomes and mortality in geriatric and fragility hip fractures-results of an integrated, multidisciplinary model experienced by the “Florence hip fracture unit”. Int Orthop. 2019;43(1):187–192. doi:10.1007/s00264-018-4132-3
4. Tarazona-Santabalbina FJ, Belenguer-Varea A, Rovira-Daudi E, et al. Early interdisciplinary hospital intervention for elderly patients with hip fractures: functional outcome and mortality. Clinics (Sao Paulo). 2012;67(6):547–556. doi:10.6061/clinics/2012(06)02
5. Bliuc D, Nguyen ND, Milch VE, Nguyen TV, Eisman JA, Center JR. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA. 2009;301(5):513–521. doi:10.1001/jama.2009.50
6. Schnell S, Friedman SM, Mendelson DA, Bingham KW, Kates SL. The 1-year mortality of patients treated in a hip fracture program for elders. Geriatr Orthop Surg Rehabil. 2010;1(1):6–14. doi:10.1177/2151458510378105
7. Haentjens P, Magaziner J, Colon-Emeric CS, et al. Meta-analysis: excess mortality after hip fracture among older women and men. Ann Intern Med. 2010;152(6):380–390. doi:10.7326/0003-4819-152-6-201003160-00008
8. Guzon-Illescas O, Perez Fernandez E, Crespi Villarias N, et al. Mortality after osteoporotic hip fracture: incidence, trends, and associated factors. J Orthop Surg Res. 2019;14(1):203. doi:10.1186/s13018-019-1226-6
9. Maxwell MJ, Moran CG, Moppett IK. Development and validation of a preoperative scoring system to predict 30 day mortality in patients undergoing hip fracture surgery. Br J Anaesth. 2008;101(4):511–517. doi:10.1093/bja/aen236
10. Atthakomol P, Manosroi W, Phinyo P, Pipanmekaporn T, Vaseenon T, Rojanasthien S. Prognostic factors for all-cause mortality in Thai patients with fragility fracture of hip: comorbidities and laboratory evaluations. Medicina (Kaunas, Lithuania). 2020;56(6):Jun. doi:10.3390/medicina56060311
11. Yong EL, Ganesan G, Kramer MS, et al. Risk factors and trends associated with mortality among adults with hip fracture in Singapore. JAMA Netw Open. 2020;3(2):e1919706. doi:10.1001/jamanetworkopen.2019.19706
12. Jiang L, Chou ACC, Nadkarni N, et al. Charlson comorbidity index predicts 5-year survivorship of surgically treated hip fracture patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318806442. doi:10.1177/2151459318806442
13. Halm EA, Wang JJ, Boockvar K, et al. The effect of perioperative anemia on clinical and functional outcomes in patients with hip fracture. J Orthop Trauma. 2004;18(6):369–374. doi:10.1097/00005131-200407000-00007
14. Nijmeijer WS, Folbert EC, Vermeer M, Slaets JP, Hegeman JH. Prediction of early mortality following hip fracture surgery in frail elderly: the Almelo Hip Fracture Score (AHFS). Injury. 2016;47(10):2138–2143. doi:10.1016/j.injury.2016.07.022
15. Puckeridge G, Terblanche M, Wallis M, Fung YL. Blood management in hip fractures; are we leaving it too late? A retrospective observational study. BMC Geriatr. 2019;19(1):79. doi:10.1186/s12877-019-1099-x
16. Ryan G, Nowak L, Melo L, et al. Anemia at presentation predicts acute mortality and need for readmission following geriatric hip fracture. 2020;5(3):
17. Jang SY, Cha YH, Yoo JI, et al. Blood transfusion for elderly patients with hip fracture: a nationwide cohort study. J Korean Med Sci. 2020;35(37):e313. doi:10.3346/jkms.2020.35.e313
18. Carson JL, Terrin ML, Noveck H, et al. Liberal or restrictive transfusion in high-risk patients after hip surgery. N Engl J Med. 2011;365(26):2453–2462. doi:10.1056/NEJMoa1012452
19. Abouleish AE, Leib ML, Cohen NH, Provides ASA. Examples to each ASA physical status class. ASA Newsl. 2015;79(6):38–49.
20. Mueller MM, Van Remoortel H, Meybohm P, et al. Patient blood management: recommendations from the 2018 frankfurt consensus conference. JAMA. 2019;321(10):983–997. doi:10.1001/jama.2019.0554
21. Gregersen M. Postoperative red blood cell transfusion strategy in frail anemic elderly with hip fracture. A randomized controlled trial. Dan Med J. 2016;63:4.
22. Loftus TJ, Spratling L, Stone BA, Xiao L, Jacofsky DJ, Patient Blood A. Management program in prosthetic joint arthroplasty decreases blood use and improves outcomes. J Arthroplasty. 2016;31(1):11–14. doi:10.1016/j.arth.2015.07.040
23. Kotze A, Carter LA, Scally AJ. Effect of a patient blood management programme on preoperative anaemia, transfusion rate, and outcome after primary hip or knee arthroplasty: a quality improvement cycle. Br J Anaesth. 2012;108(6):943–952. doi:10.1093/bja/aes135
24. Lawrence VA, Silverstein JH, Cornell JE, Pederson T, Noveck H, Carson JL. Higher Hb level is associated with better early functional recovery after hip fracture repair. Transfusion. 2003;43(12):1717–1722. doi:10.1046/j.0041-1132.2003.00581.x
25. Woodson RD, Wills RE, Lenfant C. Effect of acute and established anemia on O2 transport at rest, submaximal and maximal work. J Appl Physiol Respir Environ Exerc Physiol. 1978;44(1):36–43. doi:10.1152/jappl.1978.44.1.36
26. Mayer G, Thum J, Cada EM, Stummvoll HK, Graf H. Working capacity is increased following recombinant human erythropoietin treatment. Kidney Int. 1988;34(4):525–528. doi:10.1038/ki.1988.213
27. Sui X, Laditka JN, Hardin JW, Blair SN. Estimated functional capacity predicts mortality in older adults. J Am Geriatr Soc. 2007;55(12):1940–1947. doi:10.1111/j.1532-5415.2007.01455.x
28. Gregersen M, Borris LC, Damsgaard EM. Postoperative blood transfusion strategy in frail, anemic elderly patients with hip fracture: the TRIFE randomized controlled trial. Acta Orthop. 2015;86(3):363–372. doi:10.3109/17453674.2015.1006980
29. Izuel Rami M, Garcia Erce JA, Gomez-Barrera M, Cuenca Espierrez J, Abad Sazatornil R, Rabanaque Hernandez MJ. Relacion de la transfusion y la ferropenia con la infeccion nosocomial en pacientes con fractura de cadera[Relationship between allogeneic blood transfusion, iron deficiency and nosocomial infection in patients with hip fracture]. Med Clin (Barc). 2008;131(17):647–652. doi:10.1157/13128722
30. Marcantonio ER, Goldman L, Orav EJ, Cook EF, Lee TH. The association of intraoperative factors with the development of postoperative delirium. Am J Med. 1998;105(5):380–384. doi:10.1016/s0002-9343(98)00292-7
31. Holvik K, Ranhoff AH, Martinsen MI, Solheim LF. Predictors of mortality in older hip fracture inpatients admitted to an orthogeriatric unit in Oslo, Norway. J Aging Health. 2010;22(8):1114–1131. doi:10.1177/0898264310378040
32. Nguyen AQ, Foy MP, Sood A, Gonzalez MH. Preoperative risk factors for postoperative urinary tract infection after primary total hip and knee arthroplasties. J Arthroplasty. 2021;36(2):734–738. doi:10.1016/j.arth.2020.08.002
33. Meier J, Muller MM, Lauscher P, Sireis W, Seifried E, Zacharowski K. Perioperative red blood cell transfusion: harmful or beneficial to the patient? Transfus Med Hemother. 2012;39(2):98–103. doi:10.1159/000337187
34. Raghavan M, Marik PE. Anemia, allogenic blood transfusion, and immunomodulation in the critically ill. Chest. 2005;127(1):295–307. doi:10.1378/chest.127.1.295
35. Koch CG, Li L, Sessler DI, et al. Duration of red-cell storage and complications after cardiac surgery. N Engl J Med. 2008;358(12):1229–1239. doi:10.1056/NEJMoa070403
36. Peduzzi P, Concato J, Feinstein AR, Holford TR. Importance of events per independent variable in proportional hazards regression analysis. II. Accuracy and precision of regression estimates. J Clin Epidemiol. 1995;48(12):1503–1510. doi:10.1016/0895-4356(95)00048-8
37. Klestil T, Roder C, Stotter C, et al. Impact of timing of surgery in elderly hip fracture patients: a systematic review and meta-analysis. Sci Rep. 2018;8(1):13933. doi:10.1038/s41598-018-32098-7
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.