Back to Journals » Neuropsychiatric Disease and Treatment » Volume 13
Prefrontal cortex activation during neuropsychological tasks might predict response to pharmacotherapy in patients with obsessive–compulsive disorder
Authors Takeda T , Sumitani S , Hamatani S , Yokose Y, Shikata M, Ohmori T
Received 15 November 2016
Accepted for publication 20 January 2017
Published 23 February 2017 Volume 2017:13 Pages 577—583
DOI https://doi.org/10.2147/NDT.S127752
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Taro Kishi
Tomoya Takeda,1 Satsuki Sumitani,2 Sayo Hamatani,1 Yosuke Yokose,3 Megumi Shikata,4 Tetsuro Ohmori5
1Department of Psychiatry, Tokushima University Hospital, 2Department of Support for Students with Special Needs, Institute of Biomedical Sciences, Tokushima University Graduate School, 3Taoka Higashi Hospital, Tokushima, 4Department of Psychiatry, Ibogawa Hospital, Tatsuno, 5Department of Psychiatry, Institute of Biomedical Sciences, Tokushima University Graduate School, Tokushima, Japan
Objective: We investigated oxyhemoglobin change in the prefrontal cortex (PFC) of patients with obsessive–compulsive disorder (OCD) who showed different responses to pharmacotherapy during neuropsychological tasks with near-infrared spectroscopy.
Subjects and methods: A total of 42 patients with OCD (mean age: 35.6±9.6 years, 14 men, 28 women) and healthy control subjects (mean age: 35.4±9.7 years, 13 men, 29 women) were selected. Patients with OCD were divided into three groups (responders to selective serotonin-reuptake inhibitors (SSRIs), responders to SSRIs with antipsychotics, and nonresponders to SSRIs and SSRIs with antipsychotics) based on pharmacological response. We investigated oxyhemoglobin change in the PFC of subjects during Stroop tasks and a verbal fluency test with near-infrared spectroscopy.
Results: Responders to SSRIs showed smaller activation compared to control subjects during the Stroop incongruent task and verbal fluency test, but not during the Stroop congruent task. In contrast, responders to SSRIs with antipsychotics showed smaller activation compared to control subjects during all three tasks.
Conclusion: Our results suggest that activation of the PFC during Stroop tasks might predict responses to pharmacotherapy of patients with OCD.
Keywords: obsessive–compulsive disorder, pharmacotherapy, near-infrared spectroscopy, Stroop task, verbal fluency test
Introduction
Obsessive–compulsive disorder (OCD) is characterized by obsessive ideas and behaviors that the patient must act on to prevent anxiety and worry about persistent, recurrent thoughts. OCD occurs mostly in puberty and adolescence, and the lifetime-prevalence estimate is about 2%.1,2 Neuroimaging studies have suggested that patients with OCD have neurobiological abnormality in the orbitofrontal cortex, caudate nucleus, and thalamus. Moreover, functional magnetic resonance-imaging research has reported that patients with OCD show lower activation compared to controls in the dorsolateral prefrontal cortex and anterior cingulate cortex (ACC).3
Selective serotonin-reuptake inhibitors (SSRIs) are a well-established first-line pharmacological treatment for patients with OCD,4 suggesting that OCD is associated with abnormalities in serotonergic neurotransmission. However, 40%–60% of OCD patients treated with SSRIs do not have a satisfactory outcome.5 Atypical antipsychotics, such as risperidone, olanzapine, quetiapine, and aripiprazole, are used as an augmentation strategy for SSRIs in SSRI-resistant OCD patients.6,7 However, it is not clear how to predict response to pharmacotherapy in each OCD patient.
Our group investigated brain metabolites in OCD patients who showed different responses to pharmacotherapy using proton magnetic resonance spectroscopy.8 We found significantly lower N-acetyl-aspartate concentrations in responders to SSRIs with atypical antipsychotics, but not in responders to SSRIs, compared to control subjects in the ACC. Therefore, it was suggested that a subgroup of OCD patients who respond to SSRIs with atypical antipsychotics have distinct biological abnormalities in the ACC. However, it is known that OCD patients show cognitive impairments across several cognitive domains. OCD patients show low neurocognitive functions on the verbal memory task, Wisconsin card-sorting test, tower of Hanoi, and verbal fluency test (VFT).9–11 These neurocognitive tasks need the functions of the PFC.
The Stroop task is a cognitive task widely used to assess executive function. In the Stroop task, subjects are shown a word of a color written in colored letters and asked to name the color. When the letter color disagrees with the meaning of the word (incongruent task), cognitive interference is introduced and the subject shows more false reactions compared to when the letter color and the meaning of the word are consistent (congruent task). Previous neuroimaging studies have reported the PFC and ACC to be strongly activated during the Stroop task.12–15 It was reported the ACC plays a role in detecting conflict between competing representations and engaging the dorsolateral PFC to resolve such conflict.16 The VFT is a widely used neuropsychological task that requires subjects to generate as many words as possible beginning with a certain letter. The VFT is associated with basic neuropsychological functions, such as response initiation and attention.
Recently, several studies reported on PFC activation in OCD patients during the Stroop task and VFT using near-infrared spectroscopy (NIRS).17,18 Although these studies showed that oxyhemoglobin (oxy-Hb) change in PFCs of OCD patients during neuropsychological tests was smaller than those of control subjects, no study has investigated oxy-Hb change in PFCs of OCD patients who show different responses to pharmacotherapy. In the present study, we investigated oxy-Hb change in PFCs of OCD patients who showed different responses to pharmacotherapy during the Stroop task and VFT with NIRS. We hypothesized that OCD patients who show different responses to pharmacotherapy might have some differences in hemodynamic changes during the Stroop test and/or the VFT.
Subjects and methods
Subjects
A total of 42 OCD patients (mean age: 35.6±9.6 years, 14 men, 28 women) were selected from outpatients of the Department of Psychiatry of Tokushima University Hospital. OCD patients were diagnosed by a trained psychiatrist according to Diagnostic and Statistical Manual of Mental Disorders (DSM)-5. Three patients were drug-free, and 39 were taking psychotropic drugs, including SSRIs, atypical antipsychotics, and benzodiazepines, at the time of the experiment. The Yale–Brown Obsessive Compulsive Scale (Y-BOCS) was administered to measure the severity of OCD symptoms.19 Healthy control subjects (mean age: 35.4±9.7 years, 13 men, 29 women) were selected. These were college students, hospital employees, or their acquaintances.
Patients were divided into three groups according to pharmacological response. A total of 21 patients (mean age: 36.29±10.62 years, six men, 15 women) with >50% reduction in Y-BOCS scores after treatment with SSRI monotherapy (pretreatment Y-BOCS score: 24.14±4.02, posttreatment Y-BOCS score: 8.29±2.61, t20=19.33; P<0.01) comprised group A, while 14 SSRI treatment-refractory patients (mean age: 35.29±7.96 years, four men, ten women) with >35% reduction in Y-BOCS scores after atypical antipsychotic augmentation treatment with an SSRI (pretreatment Y-BOCS score: 26.79±4.08, posttreatment Y-BOCS score: 12.5±2.98, t13=14.92; P<0.01) comprised group B. Group A and group B patients used high-dose SSRIs (fluvoxamine [197.22±97.5 mg], paroxetine [35±14.62 mg], sertraline [96.43±47.11 mg], or escitalopram [20±0 mg]). Group B patients used antipsychotics (risperidone [1±0 mg], aripiprazole [9.5±7.02 mg], olanzapine [10±0 mg], quetiapine [100±0 mg], or perospirone [4±0 mg]) with ongoing SSRI. The rest of the patients did not show responses to either SSRI treatment or an antipsychotic augmentation treatment.
All subjects were right-handed, as assessed by the Edinburgh Handedness Inventory,20 and all subjects had their intellectual ability assessed by the Japanese Adult Reading Test.21 None of the subjects had a serious medical illness, neurodevelopment disorder, or a history of head injury or any drug or alcohol abuse. Written informed consent was obtained from all subjects prior to the experiment. Experimenters were blind to group identities during data acquisition. The ethics committee of the University of Tokushima approved this study.
Procedure
We used the Stroop task and VFT for cognitive paradigms to assess oxy-Hb change in PFCs of OCD patients who showed different responses to pharmacotherapy and control subjects. In the Stroop task, the words “red”, “green”, and “blue” were displayed on a computer screen in front of the subjects. In the congruent condition, the word’s semantic meaning was congruent with the script color (eg, the word “red” was printed in red). In the incongruent condition, the word’s semantic meaning was not congruent with the script color (eg, the word “red” was printed in green). For each task, 100 words were presented at once. Subjects were required to read the word color as quickly as possible. The investigator recorded the number of correct and incorrect responses. The task was presented in a block design that included three rest periods and two test conditions (congruent and incongruent). The two test conditions were separated by a rest period. During the rest periods, subjects were instructed to look at a circle presented on the computer screen. The duration of each test condition and rest period was 30 seconds. The task was administered in the following sequence: rest, congruent task, rest, incongruent task, and rest. NIRS measurements were performed throughout all conditions. The procedure for the Stroop task was based on that of Watanabe et al.22
In the VFT, the procedure consisted of a 30-second pretask period, 60-second VFT, and 70-second posttask period. During the pretask and posttask periods, subjects repeated the vowels (a, i, u, e, o) in Japanese. During the VFT period, subjects were requested to respond with as many words beginning with a Japanese syllable as possible. The syllables (first “a”, second “ki”, third “ha” were used) changed every 20 seconds during the VFT period. NIRS measurements were performed throughout all conditions. The procedure for the VFT was based on that of Suto et al.23 The Stroop task was conducted first, and then after 5 minutes’ rest the VFT was conducted.
NIRS measurements
NIRS measurements were performed using a 24-channel NIRS system (ETG4000; Hitachi, Tokyo, Japan) and using two wavelengths of NIR light (695 and 830 nm). The distance between the injector and the detector was 3 cm. Changes in oxy-Hb and deoxy-Hb were calculated according to the Beer–Lambert law.24 A total of 24 NIRS probes were placed bilaterally and symmetrically on the left and right hemispheres. The lowest probes were placed along the Fp1–Fp2 line, according to the international 10–20 electrode-placement system for electroencephalography.
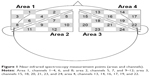
Average change in oxy-Hb during the task was analyzed by integral mode. Linear fitting was performed on the oxy-Hb before and after the task. Four areas were defined to allow investigation of the individual effects of the task parameters within different regions of NIRS measurement, in accordance with Watanabe et al: area 1, channels 1–4, 6, and 8; area 2, channels 5, 7, and 9–12; area 3, channels 15, 18, 20, 21, 23, and 24; and area 4, channels 13, 14, 16, 17, 19, and 22.22 Areas 1–4 represented the left dorsolateral PFC, left ventromedial PFC, right ventromedial PFC, and right dorsolateral PFC, respectively. The average change in oxy-Hb concentration in each area was calculated. NIRS measurements were performed as in our previous studies.25,26 The 24 probes and four areas are shown in Figure 1.
Data analysis and statistics
Comparison of demographic indices among groups was carried out using χ2 tests, unpaired t-test, and one-way analysis of variance (ANOVA). Moreover, Spearman’s rank correlation was conducted between neuropsychological measures and Y-BOCS scores in each group. Bonferroni corrections were applied to maintain an overall type I error rate of 0.05.
Next, Levene’s test for equality of variances was conducted; independent variables were drug responsiveness (group A, group B, and control group), task (Stroop task and VFT), and area (areas 1–4). As homoscedasticity was not accepted, two-way ANOVA was conducted in each task using drug responsiveness and area as independent variables. The Bonferroni test was used as a post hoc analysis.
Finally, Levene’s test for equality of variances was conducted; independent variables were medication status (SSRI, SSRI and antipsychotic, and drug-free), task (Stroop task and VFT) and area (areas 1–4). As homoscedasticity was not accepted, two-way ANOVA was conducted in each task using drug responsiveness and area as independent variables.
Results
Demographic indices and cognitive performance of groups A, B, and control are shown in Table 1. As for Stroop incongruent-task correct responses, one-way ANOVA indicated a significant main effect for groups (F2,74=8.34, P<0.01). Post hoc t-tests showed that groups A and B showed fewer correct responses than the control group. Between groups A and B, there was a significant difference in Y-BOCS score (t33=−2.62, P<0.05). There were no significant correlations between Y-BOCS score and neuropsychological measures in group A (correct responses on Stroop congruent task [r=−0.26, P=0.25], incorrect responses on Stroop congruent task [r=0.2, P=0.38], correct responses on Stroop incongruent task [r=−0.18, P=0.44], incorrect responses on Stroop incongruent task [r=−0.2, P=0.38], and total score on VFT [r=−0.31, P=0.17]) or group B (correct responses on Stroop congruent task [r=0, P=1], incorrect responses on Stroop congruent task [r=−0.24, P=0.4], correct responses on Stroop incongruent task [r=−0.04, P=0.9], incorrect responses on Stroop incongruent task [r=−0.15, P=0.6], and total score on VFT [r=0.14, P=0.63]).
As for the Stroop congruent task, the 3×4 ANOVA for change in oxy-Hb concentration indicated a significant main effect for diagnosis (F2,296=8.49, P<0.01). Post hoc t-tests showed that group B showed a smaller change in oxy-Hb than the control group. As for the Stroop incongruent task, the 3×4 ANOVA for change in oxy-Hb concentration indicated a significant main effect for the diagnosis (F2,296=10.6, P<0.01). Post hoc t-tests showed that groups A and B showed a smaller change in oxy-Hb than the control group. As for the VFT, the 3×4 ANOVA for change in oxy-Hb concentration indicated a significant main effect for diagnosis (F2,296=5.08, P<0.01) and area (F3,296=4.49, P<0.01). Post hoc t-tests showed that groups A and B showed smaller changes in oxy-Hb than the control group, and that area 1 had a greater change in oxy-Hb than area 2 or area 3. Moreover, area 4 had a greater change in oxy-Hb than area 3. The oxy-Hb change in groups during Stroop tasks and the VFT are shown in Table 2 and Figure 2.
  | Table 2 Change in oxyhemoglobin among groups |
To investigate the influence of medication, the OCD group was divided according to medication at the time of the experiment into patients using SSRI (mean age: 35.39±11.51 years, five men, 13 women), those using SSRI and an antipsychotic (mean age: 35.29±7.96 years, four men, ten women), and those not using any drug (mean age: 41.67±2.52 years, one man, two women). The 3×4 ANOVA was conducted for each task, and independent variables were medication status when NIRS were measured (patients using SSRIs, those using SSRIs and antipsychotics, and those without medication), and area (areas 1–4). There was no main effect of group. Regarding cognitive performances, correct responses on Stroop congruent task (F2,32=286.78, P=0.21), incorrect responses on Stroop congruent task (F2,32=0.2, P=0.48), correct responses on Stroop incongruent task (F2,32=8.26, P=0.92), incorrect responses on Stroop incongruent task (F2,32=0.98, P=0.39), and total VFT score (F2,32=29.39, P=0.22) showed no significant differences among medication groups.
Discussion
Numerous functional neuroimaging studies in OCD have implicated dysfunction within frontal subcortical circuits (the orbitofrontal cortex, the caudate nucleus, the globus pallidus, and the thalamus) and the limbic region, such as the ACC. Furthermore, abnormalities in the PFC in OCD are also pointed out because current studies on OCD suggest that OCD patients show a broad range of neuropsychological dysfunction across all cognitive domains (memory, attention, flexibility, inhibition, verbal fluency, planning, and decision making). Okada et al showed that oxy-Hb change in the left lateral PFC of an OCD group was significantly smaller than that of a healthy control group during the Stroop task using 24-channel NIRS.17 In addition, Hirosawa et al showed that oxy-Hb change in the right lateral PFC of an OCD group was significantly smaller than that of a healthy control group during the VFT using 42-channel NIRS.18 Our study also showed that oxy-Hb change in the PFC of the OCD group was significantly smaller than that of the healthy control group during the Stroop task and VFT by using 24-channel NIRS.
In this study, we divided OCD patients according to pharmacological response and compared the brain activation caused by Stroop tasks and the VFT. Our study showed that oxy-Hb changes in the PFC of responders to SSRIs and of responders to SSRIs with atypical antipsychotics were smaller than those of control subjects during the Stroop incongruent task and VFT. Furthermore, oxy-Hb changes in the PFC of responders to SSRIs with antipsychotics still showed smaller activation of the PFC compared to those of control subjects during the Stroop congruent task, although this task is easier than the Stroop incongruent task. The Stroop congruent task measures speed and comprehension of reading,27 and Shaywitz and Shaywitz suggested that attention function plays a role in reading.28 Therefore, the Stroop congruent task might measure attentional function underlying reading. Smaller oxy-Hb changes in the PFC during the Stroop congruent task might be related to a broad range of cognitive dysfunction in responders to SSRIs with atypical antipsychotics. However, there were no differences in oxy-Hb change in the PFC during the VFT between responders to SSRIs and responders to SSRIs with atypical antipsychotics. These results suggested that it might be possible to distinguish pharmacological responses of OCD patients by measuring levels of activation of the PFC during the two kinds of Stroop tasks. As there were no differences in task performances in the two kinds of Stroop tasks between responders to SSRIs and responders to SSRIs with atypical antipsychotics, NIRS might be more sensitive to neurological deficits than of task-performance levels. Although preliminary, our results suggest that measuring hemodynamic changes in neurocognitive tests might be a biological predictor of responses to pharmacotherapy in OCD patients.
Limitations
The current study had some limitations. First, the sample size was relatively small. Second, although there was no significant difference in the ratio of male to female patients among the three groups, the ratio was very different. Third, the subjects showed relatively mild symptoms. Therefore, there is a possibility that our results do not reflect the characteristics of all OCD patients. Fourth, it is also possible that the medication may have had an influence on our findings, although we found no significant differences in oxy-Hb changes among medication statuses when NIRS was measured.
Conclusion
In this study, we found that OCD patients with different responses to pharmacotherapy showed different oxy-Hb changes in the PFC during the two kinds of Stroop tasks. Measurement of activations in the PFC might be useful to predict the response to pharmacotherapy in OCD patients.
Disclosure
The authors report no conflicts of interest in this work.
References
Ruscio AM, Stein DJ, Chiu WT, Kessler RC. The epidemiology of obsessive-compulsive disorder in the National Comorbidity Survey Replication. Mol Psychiatry. 2010;15:53–63. | ||
Karno M, Golding JM, Sorenson SB, Burnam A. The epidemiology of obsessive-compulsive disorder in five US communities. Arch Gen Psychiatry. 1988;45:1094–1099. | ||
Nakao T, Nakagawa A, Yoshiura T, et al. A functional MRI comparison of patients with obsessive-compulsive disorder and normal controls during a Chinese character Stroop task. Psychiatry Res. 2005;139:101–114. | ||
Soomro GM, Altman D, Rajagopal S, Oakley-Browne M. Selective serotonin re-uptake inhibitors (SSRIs) versus placebo for obsessive compulsive disorder (OCD). Cochrane Database Syst Rev. 2008:CD001765. | ||
Pallanti S, Hollander E, Bienstock C, et al. Treatment non-response in OCD: methodological issue and operational definitions. Int J Neuropsychopharmacol. 2002;5:181–191. | ||
Bysrtisky A, Ackerman DL, Rosen RM, et al. Augmentation of serotonin reuptake inhibitors in refractory obsessive-compulsive disorder using adjunctive olanzapine: a placebo-controlled trial. J Clin Psychiatry. 2004;65:565–568. | ||
McDougle CJ, Epperson CN, Pelton GH, Wasylink S, Price LH. A double-blind, placebo-controlled study of risperidone addition in serotonin reuptake inhibitor-refractory obsessive-compulsive disorder. Arch Gen Psychiatry. 2000;57:794–801. | ||
Sumitani S, Harada M, Kubo H, Ohmori T. Proton magnetic resonance spectroscopy reveals an abnormality in the anterior cingulate of a subgroup of obsessive-compulsive disorder patients. Psychiatry Res. 2007;154:85–92. | ||
de Geus F, Denys DA, Sitskoorn MM, Westenberg HG. Attention and cognition in patients with obsessive-compulsive disorder. Psychiatry Clin Neurosci. 2007;61:45–53. | ||
Mataix-Cols D, Junqué C, Sànchez-Turet M, Vallejo J, Verger K, Barrios M. Neuropsychological functioning in a subclinical obsessive-compulsive sample. Biol Psychiatry. 1999;45:898–904. | ||
Head D, Bolton D, Hymas N. Deficit in cognitive shifting ability in patients with obsessive-compulsive disorder. Biol Psychiatry. 1989;25:929–937. | ||
Adleman NE, Menon V, Blasey CM, et al. A developmental fMRI study of the Stroop color-word task. Neuroimage. 2002;16:61–75. | ||
Khateb A, Michel CM, Pegna AJ, Landis T, Annoni JM. New insights into the Stroop effect: a spatiotemporal analysis of electric brain activity. Neuroreport. 2000;11:1849–1855. | ||
Prakash RS, Erickson KI, Colcombe SJ, Kim JS, Voss MW, Kramer AF. Age-related differences in the involvement of the prefrontal cortex in attentional control. Brain Cogn. 2009;71:328–335. | ||
Laird AR, McMillan KM, Lancaster JL, et al. A comparison of label-based review and ALE meta-analysis in the Stroop task. Hum Brain Mapp. 2005;25:6–21. | ||
Carter CS, van Veen V. Anterior cingulate cortex and conflict detection: an update of theory and data. Cogn Affect Behav Neurosci. 2007;7:367–379. | ||
Okada K, Ota T, Iida J, Kishimoto N, Kishimoto T. Lower prefrontal activity in adults with obsessive-compulsive disorder as measured by near-infrared spectroscopy. Prog Neuropsychopharmacol Biol Psychiatry. 2013;43:7–13. | ||
Hirosawa R, Narumoto J, Sakai Y, et al. Reduced dorsolateral prefrontal cortical hemodynamic response in adult obsessive-compulsive disorder as measured by near-infrared spectroscopy during the verbal fluency task. Neuropsychiatr Dis Treat. 2013;9:955–962. | ||
Nakajima T, Nakamura M, Taga C, et al. Reliability and validity of the Japanese version of the Yale-Brown Obsessive-Compulsive Scale. Psychiatry Clin Neurosci. 1995;49:121–126. | ||
Oldfield RC. The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia. 1971;9:97–113. | ||
Matsuoka K, Uno M, Kasai K, Koyama K, Kim Y. Estimation of premorbid IQ in individuals with Alzheimer’s disease using Japanese ideographic script (kanji) compound words: a Japanese version of NART. Psychiatry Clin Neurosci. 2006;60:332–339. | ||
Watanabe Y, Sumitani S, Hosokawa M, Ohmori T. Prefrontal activation during two Japanese Stroop tasks revealed with multi-channel near-infrared spectroscopy. J Med Invest. 2015;62:51–55. | ||
Suto T, Fukuda M, Ito M, Uehara T, Mikuni M. Multichannel near-infrared spectroscopy in depression and schizophrenia: cognitive brain activation study. Biol Psychiatry. 2004;55:501–511. | ||
Mitschele J. Beer-Lambert law. J Chem Educ. 1996;73:A260. | ||
Hosokawa M, Nakadoi Y, Watanabe Y, Sumitani S, Ohmori T. Association of autism tendency and hemodynamic changes in the prefrontal cortex during facial expression stimuli measured by multi-channel near-infrared spectroscopy. Psychiatry Clin Neurosci. 2015;69:145–152. | ||
Nakadoi Y, Sumitani S, Watanabe Y, Akiyama M, Yamashita N, Ohmori T. Multi-channel near-infrared spectroscopy shows reduced activation in the prefrontal cortex during facial expression processing in pervasive developmental disorder. Psychiatry Clin Neurosci. 2012;66:26–33. | ||
Golden CJ, Espe-Pfeifer P, Wachsler-Felder J. Neuropsychological Interpretations of Objective Psychological Tests. New York: Kluwer Academic; 2000. | ||
Shaywitz SE, Shaywitz BA. Paying attention to reading: the neurobiology of reading and dyslexia. Dev Psychopathol. 2008;20:1329–1349. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.