Back to Journals » Clinical Ophthalmology » Volume 13
Prefilled syringes for intravitreal drug delivery
Authors Sassalos TM, Paulus YM ![]()
Received 20 October 2018
Accepted for publication 15 February 2019
Published 23 April 2019 Volume 2019:13 Pages 701—706
DOI https://doi.org/10.2147/OPTH.S169044
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Video abstract presented by Thérèse M Sassalos.
Views: 1394
Thérèse M Sassalos,1 Yannis M Paulus1,2
1Department of Ophthalmology and Visual Sciences, W.K. Kellogg Eye Center, University of Michigan, Ann Arbor, MI, USA; 2Department of Biomedical Engineering, University of Michigan, Ann Arbor, MI, USA
Abstract: Intravitreal injections of anti-vascular endothelial growth factor (VEGF) medications play an increasingly critical role in numerous retinal vascular diseases. Initially, anti-VEGF medications came in vials that had to be drawn up by the physician into a syringe for administration. In 2018, the US Food and Drug Administration (US FDA) approved the ranibizumab 0.3 mg prefilled syringe (PFS), and in October 2016, the US FDA approved the ranibizumab 0.5 mg PFS. This article discusses the advantages of the PFS, including reduced injection time, possible reduced risk of endophthalmitis, reduction in intraocular air bubbles and silicone oil droplets, and improved precision in the volume and dose of intravitreal ranibizumab administered, along with possible disadvantages. Implications of the innovation of the PFS on intravitreal injection technique and clinical practice pattern are discussed and reviewed.
Keywords: intravitreal injection, intravitreous injection, anti-VEGF, ranibizumab, prefilled syringe, diabetic retinopathy
Introduction
The advent and ubiquity of anti-vascular endothelial growth factor (anti-VEGF) injections have led intravitreal injections to become an increasingly common and effective method of delivering many types of medications into the vitreous cavity.1 Anti-VEGF medications are designed to bind to and inhibit VEGF, which is thought to play a critical role in the formation of new blood vessels and vascular permeability of these vessels. The number of intravitreal injections being performed is increasing at epidemic proportions, with over 4 million intravitreal injections performed in the USA in 2013 and rising to an estimated 5.9 million patients receiving intravitreal injections in 2016 in the USA.2 Anti-VEGF injections have become a cornerstone in the treatment of retinal diseases.
Furthermore, randomized clinical trials have demonstrated the efficacy of anti-VEGF intravitreal injections in the treatment of retinal diseases including exudative age-related macular degeneration, diabetic retinopathy, and retinal vein occlusions.3–7 In these studies, patients demonstrated the efficacy of anti-VEGF therapy not only in avoiding visual loss but also in improving visual acuity.8–11
There are currently three anti-VEGF agents commonly used in retina practices in the USA: Avastin® (bevacizumab; Genentech, Inc., South San Francisco, CA, USA), Lucentis® (ranibizumab; Genentech, Inc.), and Eylea® (aflibercept; Regeneron, Tarrytown, NY, USA). In March 2018, the US Food and Drug Administration (US FDA) approved the ranibizumab 0.3 mg prefilled syringe (PFS) as a new method of administering intravitreal injections for the treatment of diabetic retinopathy and diabetic macular edema.12,13 The ranibizumab 0.5 mg PFS was cleared by the US FDA in 2016. This PFS (Figure 1) is packaged in a single-use, sealed sterile tray, thus allowing physicians to eliminate a number of steps in the preparation and administration of intravitreal ranibizumab. This review summarizes the literature on the administration of ranibizumab via the PFS and the implications of this innovation on intravitreal injection technique and clinical practice pattern (Table 1).
  | Figure 1 Photograph of ranibizumab PFS. |
  | Table 1 Overview of the advantages and disadvantages of the PFS for intravitreal drug delivery |
Ranibizumab PFS injection technique and ease of administration
Typical ranibizumab injection setup requires the physician/assistant to remove the vial cap, disinfect the drug vial top often with an isopropyl alcohol 70% swab, attach a filter needle to a sterile syringe, draw the medication into a sterile syringe, remove the filter needle, replace this needle with a smaller gauge sterile injection needle (often a 30 G or 32 G 1/2 inch needle), remove air bubbles, and adjust the volume of the medication prior to administration. In contrast, the ranibizumab 0.3 mg PFS is made of borosilicate glass and is packaged in a single-use, sealed sterile tray. Therefore, using the PFS, the physician simply removes the syringe cap, attaches the injection needle, and adjusts the dose prior to administration. Retina specialists and ophthalmic medical personnel have successfully performed both simulated-use and actual-use usability studies (with the PFS) with reported ease and without any critical errors.14 Reduction in these steps in the administration of intravitreal injections comes with some advantages which are detailed in the following sections.
Reduced injection time
By eliminating a number of steps in the previous injection preparation, reduced injection time is a significant advantage to using the ranibizumab PFS. Souied et al15 demonstrated a 27%–39% reduction in syringe preparation time when using the PFS rather than the standard ranibizumab vial. Likewise, a previous multicenter study comparing preparation times using traditional syringes vs the PFS revealed that the use of a PFS saved an average of 25.5 seconds.16 Although this figure may appear modest when considering a single injection, when taken in the context of a busy retina practice performing multiple injections in a day, a physician using a PFS could save substantial time using a PFS compared to conventional injection vials.
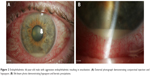
Endophthalmitis
The most visually devastating risk of intravitreal injections is infectious endophthalmitis (Figure 2). Regardless of the clinical setting or drug injected, this risk is rare, occurring on average in approximately one in 2,000 injected eyes.17–23 Although there are regional and global differences in aseptic intravitreal injection technique, the use of povidone–iodine remains a critical agent to reduce bacterial colonization and the risk of endophthalmitis.
The use of PFSs effectively eliminates a number of steps in (aseptic) injection preparation, thus decreasing the risk of iatrogenic contamination due to suboptimal drug/device handling.16,24 Although a recent report on an ongoing multicenter study has suggested that the odds of endophthalmitis with conventional intravitreal injections were higher than those using ranibizumab PFS, there are currently no comparative trials to confirm the validity of this hypothesis.25
Reduction in intraocular air bubbles and silicone oil droplets
The occurrence of intraocular air bubbles during intravitreal anti-VEGF injection has been well described.26,27 Although intravitreal air bubbles typically resorb spontaneously within 3 days, they can produce unexpected and disturbing symptoms as well as a risk for patients during air travel.28 Thought to be a result of the physician inadvertently drawing up non-sterile air from the standard drug vial (when preparing a drug for injection), air bubbles can prove difficult to eliminate once drawn into the syringe and can take significant additional physician time to remove, particularly for aflibercept.
The design features of the ranibizumab PFS include a latex-free, nonretractable plunger preventing inadvertent drawing of non-sterile air. Thus, by eliminating the need to draw up the medication and introducing a nonretractable plunger, the user is far less likely to inject intraocular air bubbles into the vitreous cavity (if proper syringe ventilation is observed).
In addition to intraocular air bubbles, recent reports have shown presumed silicone oil drops associated with intravitreal anti-VEGF injections. Specifically, a recent study demonstrated 1.73% of patients receiving bevacizumab prepared with insulin syringes to have a complication of silicone oil droplets (Figure 3).29 The culprit for these silicone oil droplets is thought to be polydimethylsiloxane, a chemical used as lubrication to reduce the friction between the syringe barrel and plunger. Much like intraocular air bubbles, silicone oil droplets are thought to result in symptoms of droplet-related floaters. Although not yet substantiated by clinical data, there is also a concern for the theoretical development of glaucoma due to retained oil droplets.30
Among the design features of the PFS is included an optimized siliconization process whereby a silicone oil-in-water emulsion is spray coated to the syringe barrel’s inner surface and then heat fixed to minimize oil migration into the ranibizumab solution. This “baked silicone” process is thought to reduce the incidence of silicone-related complications from repeated intravitreal injections.24
Accuracy and precision of intravitreal ranibizumab doses
The established therapeutic doses of anti-VEGF medications are achieved by injecting an intraocular volume of 0.05 mL. One ranibizumab PFS contains 0.165 mL of ranibizumab, ensuring that one always has adequate volume to administer a single dose of 0.05 mL. Several studies have revealed considerable variability in the accuracy and repeatability achieved with typical syringes used for intravitreal injections.31–35 Most commonly used small-volume syringes tend to over deliver their target volume.33,35 Furthermore, all syringes have unique surface markings (volume metrics) which can also lead to inaccuracies in volume dispensed. Finally, the presence of silicone oil and water deposits in the syringe can lead to deviations in volume.
The accuracy of intravitreal injection volumes directly influences their therapeutic effect on the eye. Therefore, even small deviations from intended volumes can have significant implications with regard to therapeutic response, drug toxicity, and IOP changes. Although formal comparative studies have yet to be published, the work by Loewenstein et al35 postulated that the accuracy of anti-VEGF injection volume was better achieved using a PFS compared to their conventional counterparts, although this study was not masked. They further projected that the ideal design for an intravitreal injection would consist of a low-volume, PFS with a low dead space plunger.35 The current design of the ranibizumab PFS includes a small syringe barrel (0.5 mL) which is minimally siliconized with low void volumes. This syringe design helps to offer improved dose assurance.
Disadvantages of a sterile syringe/drug assembly
Although there are several advantages to the syringe/drug complex in the PFS, one theoretical disadvantage would include the risk of drug loss due to a break in the sterile procedure. This includes the incidence of syringes which fall off the sterile injection tray or are contaminated prior to intravitreal injection. With the standard intravitreal injection procedure, the loss or contamination of a syringe (prior to drug aspiration) would simply involve replacement with a sterile syringe. A provider could incur financial loss (drug loss/replacement) from a PFS that is lost or contaminated prior to delivery to a patient.
Is the PFS a compelling leap forward?
With the advent of anti-VEGF treatment in the early 21st century, several landmark trials came which demonstrated not only the prevention of visual loss but also the significant increase in visual acuity of patients in response to treatment for retinal diseases ranging from exudative macular degeneration, diabetic macular edema, to retinal vein occlusions.36–40 Despite the marked success and widespread application of anti-VEGF treatment, there are a significant number of patients with macular edema and/or retinopathy who fail to respond to anti-VEGF treatment.41,42 Furthermore, these treatments are invasive and often require frequent administration for therapeutic effect. This has led to the evolution of novel treatments including corticosteroid-releasing implants, treatments targeting the kinin–kallikrein system, NSAIDs, vitrectomy surgery, laser therapies, immunosuppressants, and antibiotics targeting the inflammatory characteristics of early disease.43 So, while not a great leap forward, the PFS represents an incremental step that significantly reduces injection preparation time and the risk of endophthalmitis, silicone oil droplets, and air injection while improving the accuracy of drug delivery.
Conclusion
The ranibizumab PFS represents a novel method of administering intravitreal anti-VEGF therapy for the treatment of retinal diseases including exudative macular degeneration, diabetic retinopathy, and retinal vein occlusions. The PFS features a small syringe barrel which is minimally siliconized with low void volumes. In addition, the PFS totes a latex-free, nonretractable plunger and is packaged in a single-use sterile tray. This syringe design helps to offer improved dose assurance, while eliminating a number of steps in the previous injection preparation thus reducing injection times, risk of endophthalmitis, and administration of air or silicone oil droplets. In conclusion, the ranibizumab PFS improves the ease of administration to physicians for this very common intravitreal injection procedure.
Acknowledgments
This research was supported by a grant from the National Eye Institute 1K08EY027458 (YMP), unrestricted departmental support from Research to Prevent Blindness, and the Department of Ophthalmology and Visual Sciences, University of Michigan. The authors would like to thank Dr. Mark Johnson for generously allowing them to use photography from his patient for this article. Figures 1–3 are original photographs performed at the Department of Ophthalmology and Visual Sciences, Kellogg Eye Center, University of Michigan, which have not been reproduced from any other published source. Written informed consent was obtained from the patients depicted in Figures 2 and 3 to publish the content included in this manuscript.
Author contributions
TMS and YMP, contributed substantially to the conception and design of the manuscript. TMS wrote the initial draft of the manuscript, and YMP performed the critical revision of the manuscript for intellectual content. Both authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Peyman GA, Lad EM, Moshfeghi DM. Intravitreal injection of therapeutic agents. Retina. 2009;29(7):875–912. doi:10.1097/IAE.0b013e3181a94f01 | ||
Grzybowski A, Told R, Sacu S, et al. 2018 update on intravitreal injections: euretina expert consensus recommendations. Ophthalmologica. 2018;239(4):181–193. doi:10.1159/000486145 | ||
Ferrara N, Damico N, Shams M, Lowman H, Kim R. Development of ranibizumab, an anti-vascular endothelial growth factor antigen binding fragment, as therapy for neovascular age-related macular degeneration. Retina. 2006;26(8):859–870. doi:10.1097/01.iae.0000242842.14624.e7 | ||
Haritoglou C, Kook D, Neubauer A, et al. Intravitreal bevacizumab (Avastin) therapy for persistent diffuse diabetic macular edema. Retina. 2006;26(9):999–1005. doi:10.1097/01.iae.0000247165.38655.bf | ||
Iturralde D, Spaide RF, Meyerle CB, et al. Intravitreal bevacizumab (Avastin) treatment of macular edema in central retinal vein occlusion: a short-term study. Retina. 2006;26(3):279–284. | ||
Lin RC, Rosenfeld PJ. Antiangiogenic therapy in neovascular age-related macular degeneration. Int Ophthalmol Clin. 2007;47(1):117–137. doi:10.1097/IIO.0b013e31802bd873 | ||
Martin DF, Maguire MG, Ying GS, Grunwald JE, Fine SL, Jaffe GJ. Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N Engl J Med. 2011;364(20):1897–1908. doi:10.1056/NEJMoa1102673 | ||
Rosenfeld PJ, Brown DM, Heier JS, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med. 2006;355(14):1419–1431. doi:10.1056/NEJMoa054481 | ||
Mitchell P, Bandello F, Schmidt-Erfurth U, et al. The RESTORE study: ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology. 2011;118(4):615–625. doi:10.1016/j.ophtha.2011.01.031 | ||
Heier JS, Campochiaro PA, Yau L, et al. Ranibizumab for macular edema due to retinal vein occlusions: long-term follow-up in the HORIZON trial. Ophthalmology. 2012;119(4):802–809. doi:10.1016/j.ophtha.2011.12.005 | ||
Holz FG, Bandello F, Gillies M, et al. Safety of ranibizumab in routine clinical practice: 1-year retrospective pooled analysis of four European neovascular AMD registries within the LUMINOUS programme. Br J Ophthalmol. 2013;97(9):1161–1167. doi:10.1136/bjophthalmol-2013-303232 | ||
FDA approves lucentis (ranibizumab injection) prefilled syringe. [October 14, 2016]. Available from: https://www.gene.com/media/press-releases/14640/2016-10-14/fda-approves-genentechs-lucentis-ranibiz. Accessed September 14, 2018. | ||
FDA approves Genentech’s Lucentis (Ranibizumab injection) 0.3 mg prefilled syringe for diabetic macular edema and diabetic retinopathy. [March 21, 2018]. Available from: https://www.gene.com/media/press-releases/14708/2018-03-21/fda-approves-genentechs-lucentis-ranibiz. Accessed September 10, 2018. | ||
Antoszyk AN, Baker C, Calzada J, et al. Usability of the ranibizumab 0.5 mg prefilled syringe: human factors studies to evaluate critical task completion by healthcare professionals. PDA J Pharm Sci Technol. 2018;72(4):411–419. doi:10.5731/pdajpst.2017.008342 | ||
Souied E, Nghiem-Buffet S, Leteneux C, et al. Ranibizumab prefilled syringes: benefits of reduced syringe preparation times and less complex preparation procedures. Eur J Ophthalmol. 2015;25(6):529–534. doi:10.5301/ejo.5000629 | ||
Subhi Y, Kjer B, Munch IC. Prefilled syringes for intravitreal injection reduce preparation time. Dan Med J. 2016;63(4). | ||
Diago T, McCannel CA, Bakri SJ, Pulido JS, Edwards AO, Pach JM. Infectious endophthalmitis after intravitreal injection of antiangiogenic agents. Retina. 2009;29(5):601–605. doi:10.1097/IAE.0b013e31819d2591 | ||
Day S, Acquah K, Mruthyunjaya P, Grossman DS, Lee PP, Sloan FA. Ocular complications after anti-vascular endothelial growth factor therapy in medicare patients with age-related macular degeneration. Am J Ophthalmol. 2011;152(2):266–272. doi:10.1016/j.ajo.2011.01.053 | ||
McCannel CA. Meta-analysis of endophthalmitis after intravitreal injection of anti-vascular endothelial growth factor agents: causative organisms and possible prevention strategies. Retina. 2011;31(4):654–661. doi:10.1097/IAE.0b013e31820a67e4 | ||
Moshfeghi AA, Rosenfeld PJ, Flynn HW, et al. Endophthalmitis after intravitreal vascular [corrected] endothelial growth factor antagonists: a six-year experience at a university referral center. Retina. 2011;31(4):662–668. doi:10.1097/IAE.0b013e31821067c4 | ||
Shah CP, Garg SJ, Vander JF, Brown GC, Kaiser RS, Haller JA. Outcomes and risk factors associated with endophthalmitis after intravitreal injection of anti-vascular endothelial growth factor agents. Ophthalmology. 2011;118(10):2028–2034. doi:10.1016/j.ophtha.2011.02.034 | ||
Bhavsar AR, Stockdale CR, Ferris FL, Brucker AJ, Bressler NM, Glassman AR. Update on risk of endophthalmitis after intravitreal drug injections and potential impact of elimination of topical antibiotics. Arch Ophthalmol. 2012;130(6):809–810. doi:10.1001/archophthalmol.2012.227 | ||
Brown DM, Nguyen JD, Marcus DM, et al. Long-term outcomes of ranibizumab therapy for diabetic macular edema: the 36-month results from two phase III trials: RISE and RIDE. Ophthalmology. 2013;120(10):2013–2022. doi:10.1016/j.ophtha.2013.02.034 | ||
Michaud J-E, Sigg J, Boado L, et al. Ranibizumab pre-filled syringe approved in the European union: innovation to improve dose accuracy, reduce potential infection risk, and offer more efficient treatment administration. Invest Ophthalmol Vis Sci. 2014;55(13):1949. | ||
Storey P. Do prefilled syringes decrease the risk of endophthalmitis with intravitreal injection? A case-control study. Presented at: Wills Eye Conference; March 8–10, 2018; Philadelphia. | ||
Somner JE, Mansfield D. Inadvertent injection of intravitreal air during intravitreal Lucentis injection for wet age-related macular degeneration: an undescribed complication. Eye (Lond). 2009;23(8):1744. doi:10.1038/eye.2008.297 | ||
Sukgen EA, Gunay M, Kocluk Y. Occurrence of intraocular air bubbles during intravitreal injections for retinopathy of prematurity. Int Ophthalmol. 2017;37(1):215–219. doi:10.1007/s10792-016-0257-9 | ||
Mills MD, Devenyi RG, Lam WC, Berger AR, Beijer CD, Lam SR. An assessment of intraocular pressure rise in patients with gas-filled eyes during simulated air flight. Ophthalmology. 2001;108(1):40–44. | ||
Khurana RN, Chang LK, Porco TC. Incidence of presumed silicone oil droplets in the vitreous cavity after intravitreal bevacizumab injection with insulin syringes. JAMA Ophthalmol. 2017;135(7):800–803. doi:10.1001/jamaophthalmol.2017.1815 | ||
Yu JH, Gallemore E, Kim JK, Patel R, Calderon J, Gallemore RP. Silicone oil droplets following intravitreal bevacizumab injections. Am J Ophthalmol Case Rep. 2018;10:142–144. doi:10.1016/j.ajoc.2017.07.009 | ||
Gerding H, Timmermann M. Accuracy and precision of intravitreally injected ranibizumab doses: an experimental study. Klin Monbl Augenheilkd. 2010;227(4):269–272. doi:10.1055/s-0029-1245183 | ||
Meyer CH, Liu Z, Brinkmann C, Rodrigues EB, Helb HL. Accuracy, precision and repeatability in preparing the intravitreal dose with a 1.0-cc syringe. Acta Ophthalmol. 2012;90(2):e165–e166. doi:10.1111/j.1755-3768.2010.02072.x | ||
Sampat KM, Wolfe JD, Shah MK, Garg SJ. Accuracy and reproducibility of seven brands of small-volume syringes used for intraocular drug delivery. Ophthalmic Surg Lasers Imaging Retina. 2013;44(4):385–389. doi:10.3928/23258160-20130601-02 | ||
Moisseiev E, Rudell J, Tieu EV, Yiu G. Effect of syringe design on the accuracy and precision of intravitreal injections of anti-VEGF agents. Curr Eye Res. 2017;42(7):1059–1063. doi:10.1080/02713683.2016.1276195 | ||
Loewenstein I, Goldstein M, Moisseiev J, Moisseiev E. Accuracy and precision of intravitreal injection of anti-vascular endothelial growth factor agents in real life: what is actually in the syringe? Retina. Epub 2018 Apr 13. | ||
Nguyen QD, Brown DM, Marcus DM, et al. Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 2012;119(4):789–801. doi:10.1016/j.ophtha.2011.12.039 | ||
Brown DM, Kaiser PK, Michels M, et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med. 2006;355:1432–1444. doi:10.1056/NEJMoa062655 | ||
Brown DM, Michels M, Kaiser PK, Heier JS, Sy JP, Ianchulev T. Ranibizumab versus verteporfin photodynamic therapy for neovascular age-related macular degeneration: two-year results of the ANCHOR study. Ophthalmology. 2009;116:57–65.e5. doi:10.1016/j.ophtha.2008.10.018 | ||
Heier JS, Brown DM, Chong V, et al. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology. 2012;119:2537–2548. doi:10.1016/j.ophtha.2012.09.006 | ||
Martin DF, Maguire MG, Fine SL, et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology. 2012;119:1388–1398. doi:10.1016/j.ophtha.2012.03.053 | ||
Simo-Servat O, Hernandez C, Simo R. Usefulness of the vitreous fluid analysis in the translational research of diabetic retinopathy. Mediators Inflamm. 2012;2012:872978. | ||
Dabir SS, Das D, Nallathambi J, Mangalesh S, Yadav NK, Schouten JS. Differential systemic gene expression profile in patients with diabetic macular edema: responders versus nonresponders to standard treatment. Indian J Ophthalmol. 2014;62(1):66–73. doi:10.4103/0301-4738.126186 | ||
Bolinger MT, Antonetti DA. Moving past anti-VEGF: novel therapies for treating diabetic retinopathy. Int J Mol Sci. 2016;17(9):1498. doi:10.3390/ijms17091498 |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.