Back to Journals » Patient Preference and Adherence » Volume 15
Preference for Easyhaler® Over Previous Dry Powder Inhalers in Asthma Patients: Results of the DPI PREFER Observational Study
Authors Alvarez-Gutiérrez FJ , Gómez-Bastero Fernández A , Medina Gallardo JF, Campo Sien C , Rytilä P, Delgado Romero J
Received 23 December 2020
Accepted for publication 30 January 2021
Published 16 February 2021 Volume 2021:15 Pages 349—358
DOI https://doi.org/10.2147/PPA.S295710
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Johnny Chen
Francisco Javier Alvarez-Gutiérrez,1 Ana Gómez-Bastero Fernández,2 Juan Francisco Medina Gallardo,1 Carlos Campo Sien,3 Paula Rytilä,4 Julio Delgado Romero5
1Unidad de Asma, Hospital Universitario Virgen del Rocio, Sevilla, Spain; 2Unidad de Asma, Hospital Universitario Virgen Macarena, Sevilla, Spain; 3Orion Pharma Iberia, Madrid, Spain; 4Orion Pharma Corporate, Espoo, Finlandia; 5Unidad de Gestión Clinica Alergología, Hospital Virgen Macarena, Sevilla, Spain
Correspondence: Francisco Javier Alvarez-Gutiérrez
Unidad de Asma, Hospital Universitario Virgen del Rocio, Av. Manuel Siurot, S/n, Sevilla, 41013, Spain
Email [email protected]
Objective: To study patient preference for and satisfaction with the Easyhaler® device and to assess ease of training and use of the inhaler in patients previously treated with a variety of dry powder inhalers (DPIs).
Methods: We designed a non-interventional, cross-sectional, single-visit observational study of adult patients with persistent asthma referred to specialized care who had previously been treated with DPI inhalers for at least 3 months. Once clinical baseline data had been checked, patients filled in questionnaires on asthma control (GINA 2019), Feeling of Satisfaction with the Inhaler (FSI-10), and adherence (TAI and Morisky-Green questionnaires). Thereafter, all patients were trained in the use of Easyhaler. We assessed ease of use and satisfaction (FSI-10) with Easyhaler, as well as inhaler device preferences.
Results: We recruited 502 patients (mean age, 50.2 ± 16.2 y; 63.1% female), of whom 485 were evaluable. In response to the main objective of the study, we compared the values of the self-completed adapted FSI-10, to measure satisfaction with the inhaler. A significantly higher score in each item of the questionnaire was recorded for Easyhaler. Overall, 38% of patients showed exclusive preference for Easyhaler (compared with 15% for the previous device) or were evenly matched in 46% of cases.
Conclusion: In the present study, Easyhaler achieved better patient ratings in terms of preference and satisfaction than previously used DPI devices. In order to improve asthma adherence strategies, patient preferences and device choice should be taken into account.
Keywords: patient satisfaction, adherence, inhaler devices, dry powder inhalers, asthma control, clinical outcomes
Plain Language Summary
Treatment of chronic diseases such as asthma has traditionally been associated with poor long-term adherence. Lack of adherence has adverse consequences on asthma control, which in turn affects quality of life. Effectiveness of treatment for chronic conditions depends widely on patient behavior and adherence. Therefore, doctors are always looking for new strategies to improve adherence and acceptance of treatments through patient education. Appropriate choice of dry powder inhaler is essential if we are to ensure control of asthma.
The DPI PREFER study was designed to compare patient preference for the Easyhaler device in comparison to other asthma devices containing a combination of the same drugs. To accomplish this, ease of training and ease of use of the Easyhaler device were assessed using rapid evaluation procedures similar to clinical practice conditions.
In our study, adult patients completed various questionnaires on ease of use, adherence, satisfaction, and preference for Easyhaler compared with other previously used inhaler devices. Easyhaler achieved better patient ratings in preference and satisfaction than other previously used dry powder inhalers. Our study also reinforces that patient preferences and device choice should be taken into account in order to improve patient adherence in asthma management.
Introduction
Long-term treatment of chronic diseases such as asthma has classically been associated with low adherence. Lack of adherence impairs control of asthma, in turn resulting in increased morbidity and mortality, poor quality of life, and increased use of healthcare resources. Although adherence is critical to clinical outcome, it is not maintained in the long term,1–5 and, depending on the assessment method used, adherence in asthma patients has been reported to range from 16% to 43%.4–6
The factors underlying lack of adherence to asthma treatment include patient education/training, ease of use of the inhaler device, patient satisfaction, age, and adverse effects of medication. Poor adherence to pharmacological treatment of asthma has adverse consequences for disease control.7–10
The effectiveness of treatment of chronic conditions depends largely on patient behavior and adherence. Guidelines recommend that when choosing a specific intervention program, physicians should take into consideration factors such as proper use of an inhaler device and patient preference.10–12
Specialists who treat affected patients on a daily basis are very aware of these issues. Therefore, expert groups and societies are always investigating new strategies to improve adherence and acceptance of treatment through patient education.13–17
Patient preference for inhaler devices has been considered a necessary part of strategies to ensure more accurate device selection. The correct choice of device can improve patient satisfaction with treatment, reduce errors in inhalation technique, and increase adherence, leading to more favorable clinical outcomes.7–10,16–20
Recent studies such as ASCONA (Asthma Satisfaction, CONtrol and Adherence) assessed the impact of patient satisfaction with an inhaler device on adherence and health outcomes. The results of this observational study reinforced the relevance of patient satisfaction with the inhaler as a critical factor influencing adherence, proper inhalation technique, and better disease control as the final outcome. Irrespective of the medication received, patient satisfaction with the inhaler device was related to improved adherence and better control of asthma.19
A post hoc analysis of a homogeneous subpopulation of the ASCONA study revealed Easyhaler® (Orion Corporation, Finland) to be the device with the highest score for patient satisfaction compared with various dry powder inhaler (DPI) devices.20
Many patients do not use inhalers properly, probably because of improper choice of device, intrinsic features of the inhaler, or insufficient training in inhaler technique. Inadequate inhaler technique correlates with both poor symptom control and more frequent asthma exacerbations. The convenience and accuracy of the Easyhaler device has been demonstrated in various conditions and clinical settings and in comparison with other DPI devices.13–20
The DPI PREFER (Dry Powder Inhaler PREFerence versus Easyhaler in Referred asthmatic patients) study was designed and carried out to compare patient preference for the Easyhaler device with that of other DPI devices containing a combination of agents from the same pharmacological family (inhaled corticosteroids and long-acting beta-2 agonists [ICS and LABA]). In order to accomplish this, patient satisfaction, adherence, ease of training, and ease of use of the Easyhaler device were assessed using rapid evaluation procedures that mimic clinical practice conditions.
Methods
Objectives
The primary objective of the DPI PREFER study was to assess preference and satisfaction with the Easyhaler in a population of patients previously treated with a variety of DPIs.
The secondary objectives were to compare the ease of training in the use of Easyhaler with that of other DPIs and to estimate patient satisfaction and ease-of-use of inhaler devices.
Study Design
DPI PREFER was a multicenter, non-interventional, non–drug-related, cross-sectional, single-visit, observational study in adult patients with persistent asthma referred to specialized care.
Inclusion and Exclusion Criteria
In order to be included in the study, patients had to be adult outpatients (≥18 years), with bronchial persistent asthma who had been referred for the first time to specialist care (allergist or pulmonologist). They also had to be have been receiving an inhaled ICS/LABA combination through a DPI device (other than Easyhaler) for at least 3 months.
We excluded patients with disabling conditions or cognitive impairment that might, in the researchers’ opinion, affect their participation in the study and patients who did not sign the informed consent document.
Assessments
All patient data were collected at a single visit. After checking clinical baseline data, patients filled in questionnaires on asthma control and satisfaction with and adherence to their inhaler. Patients were then trained in the use of Easyhaler and assessed for ease of use and device preferences. All of the instruments were validated in Spanish.
Patient satisfaction with the current inhaler was assessed using an adapted Feeling of Satisfaction with Inhaler questionnaire (FSI-10),17–19 a self-completed instrument for obtaining patient opinions regarding the portability and usability of inhaler devices, irrespective of the drug administered. Each of the questions has 5 response options on a Likert scale from poorer to greater ease of use (scored 1 to 5, respectively).16,18–20 To preserve comparability with the new device, 2 questions regarding long-term use were removed. The adapted FSI-10 comprises 7 questions, each with 5 possible responses on a 5-point Likert scale (very, fairly, somewhat, not very, hardly at all) scored from 5 to 1, respectively. The total FSI-10 Score was calculated as the parametric sum of all scores producing a whole number value in which a higher score represents greater satisfaction than a lower score.
Adherence was evaluated using the 12-item Test of Adherence to Inhalers (TAI),17 which consists of 10 items in the patient domain, plus a further 2 items in the physician domain. In these 2 items, the researcher evaluates the patient’s understanding of the dosing regimen (dose and frequency) and performance of inhalation technique without critical errors.17 Each of the 10 self-completed items is scored from 1 to 5, where 1 is the worst possible score and 5 is the best possible score. Results range from 10 to 50 points and identify the level of adherence as poor (45 or fewer points), intermediate (46–49 points), or high (50 points). In addition, the 2 items completed by the researcher can identify 3 patterns of non-adherent behavior: erratic, deliberate, and unwitting. A single patient may be classified as having more than 1 type of non-adherent behavior.17–20
Patients also completed the 4-item Morisky-Green questionnaire on adherence, which includes 4 questions with yes/no response options and is scored from 0 to 4. The tool reflects 3 levels of adherence on the basis of this score: high (0 points), intermediate (1 to 2 points), and low (3 to 4 points).21,22
Asthma control was investigated using the GINA questionnaire, which enables a simple retrospective assessment that can easily and quickly screen out patients with poor control.10,23–25
Easyhaler Training
Included patients were trained in using the new Easyhaler device. The researcher then checked the patient’s inhalation technique with both devices (Easyhaler and current DPI) and completed a training evaluation form. Finally, the patient completed a 4-item self-perception questionnaire to evaluate his/her preference with each of the devices.
Sample
A total of 500 patients were considered necessary to carry out the study. The sample was calculated based on previous FSI-10 scores attained by Easyhaler and other DPIs in the ASCONA study.18 The sample required the participation of 100 researchers to recruit 5 consecutive referred patients with bronchial asthma receiving treatment with ICS/LABA through a DPI.
Statistical Analysis
The statistical analysis was performed using SPSS 25.0 (IBM SPSS Statistics for Windows, Version 25.0, IBM Corp., Armonk, NY, USA). Statistical tests, tables, and charts were based only on the number of valid cases.
Descriptive statistics were applied for all data. Categorical variables were presented as frequencies and percentages. Quantitative variables (continuous or ordinal) were analyzed using measures of central tendency (mean, median) and dispersion (standard deviation, maximum and minimum).
Inferences were analyzed using Pearson’s chi-square test for categorical variables and through ANOVA testing for continuous variables. A multiple logistic regression model (stepwise forward method) was then applied to assess the independence of the factors detected.
A statistical analysis plan was established to achieve the objectives of the study.
Ethics
DPI PREFER was sponsored by the Spanish Society of Pulmonology and Thoracic Surgery (SEPAR) as a non-interventional study in which the exposure investigated is not a medication. Participation in DPI PREFER did not imply interference with or modification of standard therapeutic practices. Given the nature of the study, epidemiological data were recorded only during the medical visit. Before inclusion, the researcher provided the patient with detailed information on the study and obtained his/her written informed consent.
The protocol and all study materials were assessed and approved by the Clinical Research Ethics Committee (CREC) of Hospital Virgen de la Macarena, Seville, Spain. The study was implemented in accordance with the principles adopted by the 18th World Medical Assembly (Helsinki, 1964) and the amendments of the 64th General Assembly (Fortaleza, 2013) and was carried out according to Good Clinical Practice and ethics codes.
All data were obtained during a single medical visit and treated confidentially, in accordance with Spanish data protection laws.
The study was funded by a non-restrictive grant from Orion Corporate (Espoo, Finland)
Results
A total of 502 patients were recruited between November 2019 and March 2020. All 502 patients met the inclusion criteria, and of these, 485 (96.6%) completed all the questionnaires on the patient case report form. Ninety medical centers and hospitals throughout Spain participated in the study.
Patient demographic and clinical characteristics are shown in Table 1.
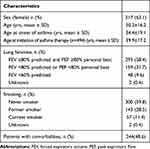 |
Table 1 Demographic and Clinical Characteristics (N=502) |
The most commonly reported comorbidities were hypertension, nasal polyposis, allergic rhinitis, and obstructive sleep apnea.
The evaluation of current treatment showed that 486 patients (96.2%) were receiving inhaled maintenance therapy, 366 (72.9%) inhaler relief therapy, and 244 (48.6%) oral treatment. The DPIs included in this comparison were Turbuhaler® (AstraZeneca, UK [173 patients]), Ellipta® (GlaxoSmithKline, UK [95 patients]), NEXThaler® (Chiesi Farmaceutici SpA, Italy [85 patients]), Accuhaler® (GlaxoSmithKline, UK [55 patients]), Spiromax® (Teva Pharmaceutical Industries, Israel [30 patients]), and other DPIs (48 patients).
According the GINA 2019 criteria, 29.9% of the total sample had mild persistent asthma, 51.6% had moderate persistent asthma, and 18.5% had severe persistent asthma. No patients had intermittent asthma; this finding was as expected because of the exclusion criteria. In addition, asthma was controlled in 184 (36.7%) patients and partially controlled or uncontrolled in 192 (38.2%) and 123 (25.1%) of patients, respectively. When the last 2 categories were considered together, asthma was partly or poorly controlled in 63.3% of patients.
According to the Morisky-Green questionnaire, 181 (36.1%) patients adhered to treatment.
The results of the Test of Adherence to Inhalers (TAI) in the 485 patients who completed the test, showed that adherence was poor in 211 patients (43.5%), intermediate in 167 (34.4%), and good in 107 (22.1%). According to the physician’s evaluation in the last 2 questions of the test, 91% of patients knew or remembered the pattern (dose and frequency) that was prescribed, and 79.3% did not make critical mistakes related to inhalation technique. The type of non-adherent behavior was identified as erratic in 25.3%, deliberate in 43.6%, and unwitting in 74.7%; it must be remembered that non-adherent patients may be classified in more than 1 of these 3 categories.
After training patients in the use of a new Easyhaler device, researchers checked the patient’s inhalation technique and ease of training with the currently used device and Easyhaler. The evaluation of training was completed by 502 patients. Training in the use of Easyhaler was considered very easy or fairly easy by 95.9% of patients; 10.8% of participants reported difficulty using Easyhaler, and critical mistakes were reported in only 4.6% (Table 2).
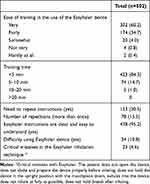 |
Table 2 Easyhaler Training Evaluation, n (%) |
The primary objective of the study was evaluated through the results of the adapted FSI-10 test in 485 valid cases (patients who completed the questionnaire). The results are shown as a qualitative evaluation (Table 3) and as a quantitative evaluation (Table 4). Satisfaction with Easyhaler was significantly higher than with the previous inhaler (31.8 ± 3.24 points vs 29.12 ± 5.15 points).
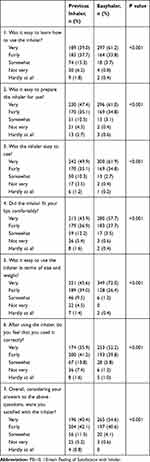 |
Table 3 FSI-10-Based Qualitative Evaluation of Easyhaler Training (n=485) |
 |
Table 4 FSI-10 Parametric Quantitative Evaluation of Satisfaction with Previous or New Inhaler (n=485) |
Age, sex, lung function, asthma severity, asthma control, inhaler adherence, and inhaler type were evaluated for their effect on the patient’s satisfaction with the inhaler. Low satisfaction with previous inhalers was significantly associated with poor asthma control and poor adherence, although mostly with inhaler type (Table 5).
 |
Table 5 Factors Influencing the Test of Satisfaction (FSI-10 Questionnaire) |
Finally, patient device preference was evaluated using a self-reported questionnaire. On the whole, 84.1% of patients preferred Easyhaler, 38.4% of patients preferred Easyhaler exclusively, while preference was similar for both devices in 45.8% of patients. Only 15.3% of patients exclusively preferred the previous device (Figure 1).
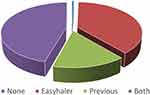 |
Figure 1 Device preference according to a self-reported questionnaire (n = 485). |
Preference for Easyhaler or the previous inhaler was analyzed according to the clinical categories used in the study. Patients reported their preference for the new device (Easyhaler) or for the previous inhaler or reported an equal preference for both devices. Table 6 shows preferences in relation to the study clinical categories.
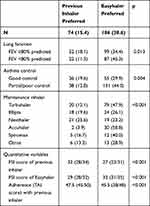 |
Table 6 Patient Preference According to the Clinical Categories Used in the Study (N=482), n (%) |
Factors that were significant in the univariate analysis of preference were included in a multiple regression analysis. The only independent predictors of device preference were the FSI-10 score and TAI score. The FSI-10 score for the previous inhaler and the TAI score significantly favored preference for the previous inhaler. Similarly, the FSI-10 score for Easyhaler strongly predicts preference for the new device, with an odds ratio of 1.79. A forest plot from the multivariate logistic regression analysis can be seen in Figure 2.
Discussion
Regardless of the inhaled medication, patient preference for a specific inhaler device is crucial in achieving long-term control of asthma. Choosing the right device increases adherence, reduces inhalation technique mistakes, and improves patient satisfaction.1–6 Giner et al16 conducted a study to evaluate preferences between 3 DPIs, (Accuhaler, Easyhaler, and Turbuhaler) in 30 adult patients with stable asthma. Patients were shown how to use each of the devices and were randomized to begin using them in different orders. Preference was assessed based on 9 features of the DPIs. Easyhaler was the first choice for 53% of patients, while Turbuhaler and Accuhaler were preferred by 27% and 20%, respectively.
Unfortunately, studies based on short-term use of various devices are not suitable in real life. Therefore, the present study used a short training course as a tool to challenge patients’ preference with respect to their previous DPI device.
Available evidence indicates that Easyhaler is an appropriate DPI for testing this hypothesis. A recent review found that Easyhaler received high patient preference ratings and may be considered one of the most convenient inhalers for daily use in patients with asthma or COPD.30 Similarly, a recent assessment of DPI features that may affect use, patient satisfaction, and clinical benefit concluded that the design features of Easyhaler make it one of the most convenient devices for daily use in patients with asthma or COPD.31
Our study showed that more participants preferred Easyhaler to other commonly used inhaler devices (Accuhaler, Turbuhaler, Spiromax, Ellipta, and NEXThaler). Preference for Easyhaler was reported by 84.1% of patients, while 61.1% preferred their previous inhaler. Preference rates were the same for both devices (previous and Easyhaler) in 45.8% of patients. Exclusive preference for Easyhaler was reported by 38.4% of patients, while only 15.3% of participants preferred exclusively the previous device.
We also evaluated patient satisfaction using the FSI-10 questionnaire, which has proven useful for assessing the degree of satisfaction with inhalation devices among patients with asthma. The instrument is comprehensive, easy to understand, and can identify differences in patient satisfaction with various inhalers.17–20 Consistent with previous findings, our results indicate that satisfaction is greater with Easyhaler than with the previous inhaler (31.8 ± 3.24 points vs 29.12 ± 5.15 points).
Valero et al20 compared patient satisfaction with 3 DPIs (Easyhaler, Turbuhaler, and Accuhaler) in a post hoc analysis of a homogeneous subpopulation (n=328) from the ASCONA study. Patients with moderate to severe asthma were paired according to age, sex, and severity. Patient satisfaction with the device was assessed using the FSI-10 questionnaire. Scores for Easyhaler were significantly better for individual FSI-10 items such as learning how to use the device, inhaler preparation, inhaler use, weight and size, and portability. Satisfaction with the inhaler was statistically significantly greater for Easyhaler (43.8 ± 7.1) than for Turbuhaler or Accuhaler (41.3 ± 7.6; p < 0.01).20
In their real-life multicenter, noncontrolled study, Gálffy et al29 assessed patients’ handling, ease of learning, and satisfaction with the Easyhaler device in 1016 asthma or COPD patients attending respiratory clinics. Patients found Easyhaler easy to learn and use, and satisfaction with the device was very high.
The level of patient satisfaction with the inhaler device has a positive influence on asthma control through its association with higher adherence.13 We found the same significant association between satisfaction with the previous inhaler and adherence, asthma control, and even pulmonary function (Table 5). The association with satisfaction was not found for Easyhaler, since the assessment was isolated and Easyhaler was not the patient’s usual inhaler. The level of adherence in our study was similar to that found in other asthma inhaler trials.13–20
Regarding device preferences, we found similar associations (Table 6), including FSI-10 scores. Remarkably, when all these factors were included in the multivariate logistic regression analysis, the predictors of device preference were the satisfaction score (FSI-10) and, albeit only marginally, the adherence score (TAI). The results of our multivariate analysis confirm the association between Easyhaler and high rates of user satisfaction. In a 12-week, real-world, multicenter, open-label study conducted in 398 and 563 adult patients with asthma and COPD, respectively, significant increases in patient satisfaction were also reported with Easyhaler compared with previously used inhalers.14
These results point to a short training session as being a suitable means for challenging patients’ preference for their current DPI inhaler. Additionally, the FSI-10 test could be the most valuable means of predicting changes in preference.
Our study is limited in that its cross-sectional design enables it to provide only a limited picture of patient satisfaction at a particular timepoint; this picture is likely to change over time because of the chronic nature of asthma. However, the patients included in the study had at least 3 months’ experience with their maintenance treatment and were therefore able to assess its suitability. Similarly, our finding of fewer critical mistakes in the Easyhaler technique could be influenced by the fact that the evaluation was performed immediately after the training session, thus likely minimizing the possibility of errors. Since inhaler errors may develop over time, the relevance of these findings for the real-life, long-term use of the devices remains unclear.
In addition, our study was observational. Since a blinded study is not feasible for assessment of device preferences, a noncontrolled evaluation of inhaler devices without random allocation always has the potential for bias. The need for homogeneous patient populations implies that randomized controlled trials would not be representative of the real-world asthma population.
Conclusion
In the present study, Easyhaler achieved better patient ratings in preference and satisfaction than the DPI device previously used by the patient. In order to improve asthma adherence strategies, patient preferences and device choice should be taken into account.
In addition, patient preference may change after some time of regular usage, therefore, follow-up studies will be needed to clarify the actual clinical benefit of the expressed preferences.
Acknowledgments
Writing and editorial assistance was provided by Content Ed Net (Madrid, Spain) with funding from Orion Pharma. The abstract of this study was presented at the 2020 ERS International Congress.
Disclosure
FJAG has received fees for consultancy, lectures or attendance to congresses and scientific meetings from ALK-Abelló, AstraZeneca, Bial, Boehringer- Ingelheim, Chiesi, GSK, Menarini, Mundipharma, Novartis, Orion Pharma, Sanofi-Genzyme and TEVA. AGBF has received fees for consultancy, lectures or attendance to congresses and scientific meetings from ALK-Abelló, AstraZeneca, Bial, Chiesi, GSK, Menarini, Mundipharma, Novartis, Orion Pharma, Sanofi-Genzyme and TEVA. JFMG has received fees for consultancy, lectures or attendance to congresses and scientific meetings from ALK-Abelló, AstraZeneca, Bial, Chiesi, GSK, Novartis, Orion Pharma y Sanofi-Genzyme. CCS and PR are employees of Orion Pharma. JDR has received fees for consultancy or lectures from GSK, Astra, Sanofi-Genzyme, TEVA, Orion Pharma, Bial, Novartis, and Leti. The authors report no other conflicts of interest in this work.
References
1. Mäkelä MJ, Backer V, Hedegaard M, Larsson K. Adherence to inhaled therapies, health outcomes and costs in patients with asthma and COPD. Respir Med. 2013;107(10):1481–1490. doi:10.1016/j.rmed.2013.04.005
2. Côté A, Godbout K, Boulet L-P. The Management of Severe Asthma in 2020. Biochem Pharmacol. 2020;179:114112. doi:10.1016/j.bcp.2020.114112
3. Bårnes CB, Ulrik CS. Asthma and adherence to inhaled corticosteroids: current status and future perspectives. Respir Care. 2015;60(3):455–468. doi:10.4187/respcare.03200
4. Boulet L-P, Vervloet D, Magar Y, Foster JM. Adherence: the goal to control asthma. Clin Chest Med. 2012;33(3):405–417. doi:10.1016/j.ccm.2012.06.002
5. Adouni Lawani M, Zongo F, Breton M-C, et al. Factors associated with adherence to asthma treatment with inhaled corticosteroids: a cross-sectional exploratory study. J Asthma. 2018;55(3):318–329. doi:10.1080/02770903.2017.1326131
6. Plaza V, López-Viña A, Entrenas LM, et al. Differences in Adherence and Non-Adherence Behaviour Patterns to Inhaler Devices Between COPD and Asthma Patients. COPD. 2016;13(5):547–554. doi:10.3109/15412555.2015.1118449
7. Zhang J, Yin C, Li H, Wei W, Gong Y, Tang F. Application of Once-Monthly Self-Reported ACT Questionnaire in Management of Adherence to Inhalers in Outpatients with Asthma. Patient Prefer Adherence. 2020;14:1027–1036. doi:10.2147/PPA.S176683
8. Capstick TG, Clifton IJ. Inhaler technique and training in people with chronic obstructive pulmonary disease and asthma. Expert Rev Respir Med. 2012;6(1):91–103. doi:10.1586/ers.11.89
9. Gregoriano C, Dieterle T, Breitenstein A-L, et al. Use and inhalation technique of inhaled medication in patients with asthma and COPD: data from a randomized controlled trial. Respir Res. 2018;19(1):237. doi:10.1186/s12931-018-0936-3
10. Global Initiative for Asthma (GINA). 2019 GINA report, global strategy for asthma management and prevention. Available from: https://ginasthma.org/wp-content/uploads/2019/06/GINA-2019-main-report-June-2019-wms.pdf.
11. Soledad AM, Cesáreo AR, Antonio G-O, et al. Spanish guideline on the management of asthma. J Invest Allergol Clin Immunol. 2016;26(Suppl Supl 1):1–92.S. doi:10.18176/jiaci.0065
12. National Asthma Education and Prevention Program, Third expert panel on the diagnosis and management of asthma. Bethesda (MD): National Heart, Lung, and Blood Institute (US); 2007 Aug. Available from https://www.ncbi.nlm.nih.gov/books/NBK7232/.
13. Small M, Anderson P, Vickers A, Kay S, Fermer S. Importance of inhaler-device satisfaction in asthma treatment: real-world observations of physician-observed compliance and clinical/patient-reported outcomes. Adv Ther. 2011;28(3):202–212. doi:10.1007/s12325-010-0108-4
14. Gálffy G, Szilasi M, Tamasi L. Effectiveness and patient satisfaction with budesonide/formoterol easyhaler® among patients with asthma or COPD switching from previous treatment: a real-world study of patient-reported outcomes. Pulm Ther. 2019;5(2):165–177. doi:10.1007/s41030-019-0097-7
15. Price D, Harrow B, Small M, Pike J, Higgins V. Establishing the relationship of inhaler satisfaction, treatment adherence, and patient outcomes: a prospective, real-world, cross-sectional survey of US adult asthma patients and physicians. World Allergy Organ J. 2015;8(1):26. doi:10.1186/s40413-015-0075-y
16. Giner J, Torrejón M, Ramos A, et al. Preferencias de los pacientes en la elección de dispositivos de inhalación en polvo. Arch Bronconeumol. 2004;40(3):106–109. doi:10.1016/S0300-2896(04)75484-8
17. Perpiñá Tordera M, Viejo JL, Sanchis J, et al. Satisfacción y preferencia del paciente asmático por los dispositivos de inhalación. Aplicación del FSI-10. Arch Bronconeumol. 2008;44(7):346–352. doi:10.1016/S0300-2896(08)70447-2
18. Plaza V, Fernández-Rodríguez C, Melero C, Cosío BG, Entrenas LM. Validation of the ‘Test of the Adherence to Inhalers’ (TAI) for Asthma and COPD Patients. J Aerosol Med Pulm Drug Deliv. 2016;29(2):142–152. doi:10.1089/jamp.2015.1212
19. Plaza V, Giner J, Calle M, et al. Impact of patient satisfaction with his or her inhaler on adherence and asthma control. Allergy Asthma Proc. 2018;39(6):437–444. doi:10.2500/aap.2018.39.4183
20. Valero A, Ribó P, Maíz L, et al. Asthma patient satisfaction with different dry powder inhalers. Expert Rev Respir Med. 2019;13(2):133–138. doi:10.1080/17476348.2019.1567339
21. Morisky DE, Green LW, Levine DM. Concurrent and predictive validity of a self-reported measure of medication adherence. Med Care. 1986;24(1):67–74. doi:10.1097/00005650-198601000-00007
22. Val Jiménez A, Amorós Ballestero G, Martínez Visa P, Fernández Ferré ML, León Sanromà M. [Descriptive study of patient compliance in pharmacologic antihypertensive treatment and validation of the Morisky and Green test]. Aten Primaria. 1992;10(5):767–770.
23. Hogan AD, Bernstein JA. GINA updated 2019: landmark changes recommended for asthma management. Ann Allergy Asthma Immunol. 2020;124(4):311–313. doi:10.1016/j.anai.2019.11.005
24. Mauer Y, Taliercio RM. Managing adult asthma: the 2019 GINA guidelines. Cleve Clin J Med. 2020;87(9):569–575. doi:10.3949/ccjm.87a.19136
25. Papi A, Blasi F, Canonica GW, Morandi L, Richeldi L, Rossi A. Treatment strategies for asthma: reshaping the concept of asthma management. Allergy Asthma Clin Immunol. 2020;16(1):75. doi:10.1186/s13223-020-00472-8
26. Levy ML, Carroll W, Izquierdo Alonso JL, Keller C, Lavorini F, Lehtimäki L. Understanding dry powder inhalers: key technical and patient preference attributes. Adv Ther. 2019;36(10):2547–2557. doi:10.1007/s12325-019-01066-6
27. Chrystyn H, Small M, Milligan G, Higgins V, Gil EG, Estruch J. Impact of patients‘ satisfaction with their inhalers on treatment compliance and health status in COPD. Respir Med. 2014;108(2):358–365. doi:10.1016/j.rmed.2013.09.021
28. Plaza V, Giner J, Curto E, et al. Determinants and differences in satisfaction with the inhaler among patients with asthma or COPD. J Allergy Clin Immunol Pract. 2020;8(2):645–653. doi:10.1016/j.jaip.2019.09.020
29. Gálffy G, Mezei G, Németh G, et al. Inhaler competence and patient satisfaction with Easyhaler®: results of two real-life multicentre studies in asthma and COPD. Drugs R D. 2013;13(3):215–222. doi:10.1007/s40268-013-0027-3
30. Lavorini F. Easyhaler®: an overview of an inhaler device for day-to-day use in patients with asthma and chronic obstructive pulmonary disease. Drugs in Context. 2019;8:212596. doi:10.7573/dic.212596
31. Chrystyn H, Lavorini F. The dry powder inhaler features of the Easyhaler that benefit the management of patients. Expert Rev Respir Med. 2020;14(4):345–351. doi:10.1080/17476348.2020.1721286
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

