Back to Journals » Drug, Healthcare and Patient Safety » Volume 8
Positive clinical outcomes derived from using Streptococcus salivarius K12 to prevent streptococcal pharyngotonsillitis in children: a pilot investigation
Authors Di Pierro F , Colombo M, Zanvit A, Rottoli AS
Received 14 July 2016
Accepted for publication 2 October 2016
Published 21 November 2016 Volume 2016:8 Pages 77—81
DOI https://doi.org/10.2147/DHPS.S117214
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Rajender R Aparasu
Francesco Di Pierro,1 Maria Colombo,2 Alberto Zanvit,3 Amilcare S Rottoli,4
1Scientific Department, Velleja Research, Milan, 2Pediatric Department, University of Parma, Parma, 3Stomatology Institute, 4Pediatric Department, Uboldo Hospital, Cernusco S/N, Milan, Italy
Background: Streptococcus salivarius K12 (BLIS K12®) is a probiotic strain producing the bacteriocins salivaricin A2 and salivaricin B, both of which strongly antagonize the growth of Streptococcus pyogenes, the most important bacterial cause of pharyngeal infections in humans. It successfully colonizes and exhibits persistence in the oral cavity and is endowed with an excellent safety profile. Previous observations of a small group of children indicated that the use of BLIS K12 could also reduce the occurrence of viral pharyngitis. The present study focused on a further evaluation of the role of BLIS K12 in the control of pediatric streptococcal disease and moreover whether its use could also help provide protection against various nonstreptococcal infections.
Methods: In total, 48 children with a recent history of recurrent pharyngeal streptococcal disease were enrolled in the treated group. The control group comprised 76 children known to have had a very low recent occurrence of oral streptococcal disease. The treated children were given BLIS K12 daily for 90 days. The number of episodes of streptococcal pharyngotonsillitis, tracheitis, viral pharyngitis, rhinitis, flu, laryngitis, acute otitis media, enteritis, and stomatitis was recorded during probiotic treatment and for a follow-up period of 9 months, and this was compared with the episodes of the control group over the corresponding period.
Results: Compared with the pretreatment time period, 2013, a 90% reduction of streptococcal pharyngeal disease was observed in 2014; compared with untreated children, a statistically significant reduction of all of the other disease conditions assessed, other than stomatitis, was detected in the probiotic-treated children.
Conclusion: In agreement with previous findings, in the present study, it was found that the daily use of BLIS K12 has been associated with a concurrent and persisting reduction in the occurrence of pharyngeal, recurrent, streptococcal disease. Moreover, the benefits to children may also extend to a reduction of nonstreptococcal diseases, including tracheitis, viral pharyngitis, rhinitis, flu, laryngitis, acute otitis media, and enteritis.
Keywords: Streptococcus salivarius K12, pediatric infections, pharyngotonsillitis, rhinitis, flu, tracheitis, laryngitis, stomatitis, enteritis, otitis
Introduction
The oral probiotic Streptococcus salivarius strain K12 (also referred to here as BLIS K12®) is known to produce the megaplasmid-encoded class I lantibiotics, salivaricin A2, and salivaricin B.1 Expression of these two salivaricins enables BLIS K12 to counteract the growth of Streptococcus pyogenes2 and, also to a lesser extent, Haemopilus influenzae, Streptococcus pneumoniae, and Moraxella catarrhalis, all of which are involved in the etiopathogenesis of acute otitis media.3 BLIS K12 colonizes the oral cavity and, to a lesser extent, the nasopharynx and adenoids,4 remaining present in the tissues for up to 1 month after the last administration.5 Characterized by excellent antibiotic sensitivity6 and safety profiles7 and consistent with the known activity spectrum of its salivaricin products, the regular use of BLIS K12 has been shown to reduce reoccurrences of streptococcal pharyngotonsillitis and acute otitis media.8–10 In addition, however, some preliminary studies have indicated that BLIS K12 may also help prevent episodes of oropharyngeal infections of viral origin.11 A potential basis for this effect has been shown, whereby the administration of BLIS K12 in adults can increase salivary γ-interferon levels without modifying the levels of either interleukin-1β (IL-1β) or tumor necrosis factor-α (TNF-α), but considerably reducing IL-8 release.3 In the present study, whether there is any associated reduction in the occurrence of episodes of tracheitis, viral pharyngitis, rhinitis, flu, laryngitis, acute otitis media, enteritis, and stomatitis in children having a history of recurrent streptococcal pharyngotonsillitis who are receiving a 3-month course of BLIS K12 was evaluated.
Materials and methods
Product
BLIS K12 was formulated in the form of slowly dissolving oral tablets by SIIT (Trezzano S/N, Milan, Italy) and notified to the Italian Ministry of Health as Bactoblis® by Omeopiacenza (Pontenure, Italy), according to the provisions of law number 169 of 2004, on July 5, 2011 (notification number: 53435). The preparation strain K12 used in the clinical trial contained >1 billion colony forming unit (CFU)/tablet of S. salivarius K12 (BLIS Technologies Ltd., Otago, New Zealand).
Clinical trial
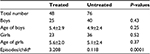
The multicenter, open, nonrandomized, controlled clinical trial was conducted on 124 pediatric individuals enrolled in the area of Milan (Italy) during 2014. The trial population consisted of 65 boys and 59 girls. The first 90 days represented the treatment period. The following 9 months was the follow-up period. The trial was conducted according to the criteria set by the Declaration of Helsinki and with the approval of the Local Ethics Committee (Milan, Italy). The parents of all the participants in the study were informed of the trial methods and signed the appropriate consent and privacy policy documents.
Inclusion criteria
All the individuals enrolled in this study were of 3–10 years of age and attended preschool or school in the Milan area. In terms of recurrent streptococcal pharyngotonsillitis, the individuals enrolled for treatment (n=48) exhibited an average of >3 episodes in the previous year (2013). The episodes were confirmed by a rapid swab positive for Group A streptococcus (Test Strep-A; Gima, Gessate, Italy). None of the individuals were clinically ill on enrolment. None of the control group subjects had experienced recurrent streptococcal pharyngotonsillitis in the previous year, and only 9 of the 76 children having single episode of streptococcal infection had been reported.
Exclusion criteria
Potential subjects were excluded from the study if they were immunocompromised, had undergone tonsillectomy or had an indication for adenotonsillectomy, had a history of rheumatic disorders, bronchospasm and/or a diagnosis of asthma and/or allergy, had diagnosed respiratory or significant systemic disorders, or were undergoing current pharmacological therapies to prevent recurrent respiratory infections. Also individuals presenting with conditions that could favor the development of acute otitis media, including severe atopy, acquired or congenital immunodeficiency, cleft palate, a chronically ruptured eardrum, craniofacial abnormalities or obstructive adenoids, sleep apnea syndrome, or placement of tympanostomy tubes, were excluded.
Study pattern
All the individuals enrolled in this study were first subjected to a general medical examination and pharyngeal swab (Test Strep-A) and then were subdivided, according to a previous diagnosis of recurrent streptococcal pharyngotonsillitis, into two groups: one group was treated with BLIS K12 in the form of strain K12 tablets, whereas the other did not receive any treatment and served as the control group. The individuals in the BLIS K12-treated group were instructed on how to use the product. The tablets were to be administered for 90 consecutive days. The children had to let one tablet dissolve slowly in the mouth immediately before going to sleep, after brushing their teeth. They were carefully instructed not to chew the tablet or to swallow it whole. They were asked not to drink or swallow anything else following the use of the product. Before administration of the first tablet, the use of a 0.2% chlorhexidine mouthwash was recommended in order to enhance the colonization process of the strain, by reducing competition from endogenous S. salivarius already inhabiting the mouth. For the trial period, it was requested that at the first sign of any oropharyngeal symptoms of infection, the subjects should be brought to the clinic for an immediate medical examination and pharyngeal rapid test. In case of a positive result, treatment was prescribed. The prescribed therapy for streptococcal infection was a combination of amoxicillin and clavulanic acid to be administered for 10 days. Following the antibiotic therapy, treatment with BLIS K12 was resumed and continued until the scheduled 90th day of the study. Viral infections accompanied by pharyngolaryngeal pain and/or a fever were treated with acetaminophen or ibuprofen. Any other pathologies present were treated according to the recommendations of the Italian Pediatric Guidelines.
Diagnosed pathologies
Diagnosis of viral infection was according to the following criteria: negative rapid swab for streptococcal disease, absence of submandibular lymphadenopathy, absence of petechiae on the palate, mild dysphagia, absence of headache, absence of abdominal pain, and absence of hyperpyrexia. From a clinical standpoint, patients with viral pharyngitis presented with modest pharyngeal hyperemia, low-grade fever, mild dysphagia, presence of rhinitis with serous secretion, and spontaneous resolution of symptoms without medication in ~48–72 hours. With regard to enteritis, according to the Italian Pediatrics Guidelines, high fever (>40°C), live blood in the stool, abdominal pain and involvement of the central nervous system may suggest the presence of pathogenic bacteria in the gut. Differently, vomiting and respiratory symptoms are more frequently associated with a viral etiology. Therefore, the diagnosis was clinical, and microbiological examination of stool was indicated only if diarrhea is prolonged for >7–8 days or is relapsing. The diagnosis of stomatitis was also clinical and according to the presence of ulcerations localized preferentially in the fornix or in gingival–labial mucosa of the lips. In all the cases, the etiology was considered to be of herpetic origin, being further and more extensively investigated only in the case of patients who were severely debilitated. With regard to rhinitis, diagnosis was done on the basis of absence of mucoid nasal secretion, absence of fever, absence adenomegaly, no signs of retropharyngeal exudate, and symptoms resolution within 72 hours. Diagnosis of tracheitis was done clinically in the presence of mild fever, mild/moderate pharyngeal redness, no submandibular lymphadenopathy, and crowing sound when inhaling. With regard to laryngitis, diagnosis was done with moderate or no fever resolving within 72 hours, pharyngeal redness with negative streptococcal rapid swab, and clear signs of dysphonia along with dysphagia. Diagnosis of acute respiratory infection (flu) was also on a clinical basis with onset with fever, headache, malaise, and myalgia followed by predominantly respiratory symptoms (eg, cough, nasal congestion, and sore throat). Virus isolation from throat swabs or sputum to identify the causative agent is only recommended in carefully selected cases, and this was not done in the present study. Acute otitis media were diagnosed by pneumatic otoscopy performed by a trained investigator.
Aims of the study
The present study aimed to evaluate the following: 1) the efficacy of the BLIS K12-containing product strain K12 in the prevention of S. pyogenes pharyngotonsillitis in young children during 3 months of treatment and a further 9-month follow-up; 2) the efficacy of strain K12 in reducing tracheitis, viral pharyngitis, rhinitis, flu, laryngitis, acute otitis media, enteritis, and stomatitis in these same subjects; and 3) the onset of side effects or toxicity while the product was being administered.
Statistical analysis
The equivalence of the two subject groups in terms of sex and age was determined by using Fisher’s exact test and the two-tailed Wilcoxon–Mann–Whitney test, respectively. The difference between the two groups in terms of numbers of streptococcal pharyngotonsillitis, tracheitis, viral pharyngitis, rhinitis, flu, laryngitis, acute otitis media, enteritis, and stomatitis episodes was determined by using the two-tailed Wilcoxon–Mann–Whitney test. Statistical software JMP Version 10 for Mac OS X was used, and the threshold for statistical significance was 95%.
Results
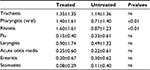
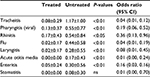
Forty-eight children having a diagnosis of recurrent streptococcal pharyngotonsillitis were enrolled as subjects to assess the preventive role associated with daily use of slowly dissolving oral tablets containing the oral probiotic S. salivarius K12 (>1 billion CFU/tablet), against S. pyogenes infection. The children were treated with 1 tablet of strain K12 each day for 90 consecutive days and then continued to be monitored for a further 9 months. The other group of 76 children, not previously diagnosed with recurrent streptococcal pharyngotonsillitis, served as controls for the same periods. Compliance assessed throughout the 90 days of strain K12 treatment was very good, and no child withdrew from the study. As shown in Table 1, the two groups did not exhibit significantly different characteristics. The only significant difference was in the diagnosis of recurrent streptococcal pharyngotonsillitis, the distinctive feature of their enrolment in the treatment group. Table 2 shows the total episodes per child of diagnosed streptococcal pharyngotonsillitis occurring during the study year 2014. When these findings are compared to those obtained in 2013, the treated children can be found to have experienced ~90% reduction in the diagnosed streptococcal pharyngitis episodes. By comparison, in the control group, a nonsignificant increase in streptococcal infections of ~30% was observed, when compared to 2013. As shown in Table 3, during 2013, the number of episodes of tracheitis, flu, acute otitis media, enteritis, and stomatitis overlapped between the two groups. A difference was present only in the episodes of viral pharyngitis and rhinitis, the incidences of which seemed to be higher and significant in the treated group versus the control group. The differences observed for laryngitis did not seem to be statistically significant. As shown in Table 4, during 2014 when strain K12 treatment was performed, the episodes per child of tracheitis, viral pharyngitis, rhinitis, flu, laryngitis, acute otitis media, and enteritis, but not of stomatitis, seemed to be significantly lower, showing an apparent protective effect associated with the use of probiotic BLIS K12. Multivariate analysis (data not shown) demonstrates the absence of any dependency between these findings and sex and/or age variables.
  | Table 2 Episodes of pharyngotonsillitis caused in 2013 and 2014 by Streptococcus pyogenes in the two study groups Note: aAverage/child. Abbreviation: ns, not significant. |
Discussion
Some intestinal probiotics have been shown to be capable of helping the consumer to counteract constipation, diarrhea, irritable bowel syndrome, and a number of other gastrointestinal disorders.12 Recently, the development of novel probiotics such as BLIS K12 from the oral cavity commensal species S. salivarius has introduced the prospect of specifically achieving oral health benefits from probiotic therapy. The BLIS K12 strain was originally isolated from the oral cavity of a young child who had no recent experience of S. pyogenes infection. The salivaricins produced by BLIS K12 have subsequently been shown to be inhibitory not only to S. pyogenes but also to oral cavity bacterial pathogens associated with acute otitis media and halitosis. Some more recent studies demonstrated that oral administration of BLIS K12, through a still not perfectly understood molecular mechanism, also reduces IL-8 plasma concentrations and increases salivary γ-interferon.3 These modulations may also rationally account for an anti-inflammatory, immunomodulating and anti-viral activity, which would augment the already-described beneficial antibacterial action of BLIS K12. The results of the current study provide further support for this proposition. BLIS K12 prophylaxis of children who appeared historically to be at an increased risk of streptococcal pharyngitis reduced streptococcal infections by ~90%, a finding consistent with those of previous studies but – and this is something new – also demonstrated an apparent reduction of tracheitis, viral pharyngitis, rhinitis, flu, laryngitis, acute otitis media, and enteritis. Only the incidence of stomatitis seemed unchanged in the treated group. This protection should not be due to a different use of antibiotics, vaccines, or immune stimulant supplements or else occurred along the year of study, being the two groups comparable in this perspective, too (data not shown). Moreover, the slight difference in terms of antibiotic administration linked to the episodes (average/child) of pharyngeal streptococcal disease between the treated and the untreated group, 0.333 and 0.157, respectively (Table 2), is not significant. Last, children resulted to be protected mostly against viral diseases, where the use of antibiotics was not effective. These results, together with the excellent tolerability and compliance found in this study, as well as the absence of side effects, show that prophylactic BLIS K12 administration could provide a safe, simple, and cost-effective preventative for a broad variety of pediatric infections and microbial dysequilibria. The authors recognize that this observational study has less validity than a double-blind, controlled, prospective, and randomized investigation and also that it may contain significant bias due to the relatively small number of treated subjects and the absence of a control, placebo, or alternative probiotic treatment. In any case, the findings of this study confirm the anti-streptococcal action of BLIS K12 and further demonstrate that its use can generate other positive outcomes. Further studies are ongoing to highlight why an oral colonizing probiotic strongly recognized as able to antagonize streptococci and also to counteract pathologies of viral etiology and/or disorders in nonoral tissues such as enteritis.
Disclosure
FDP is the main formulator of the tested product, and he is involved in the Scientific Council of the Company (Omeopiacenza®) trading the tested product. The other authors report no other conflicts of interest in this work.
References
Hyink O, Wescombe PA, Upton M, Ragland N, Burton JP, Tagg JR. Salivaricin A2 and the novel lantibiotic salivaricin B are encoded at adjacent loci on a 190-kilobase transmissible megaplasmid in the oral probiotic strain Streptococcus salivarius K12. Appl Environ Microbiol. 2007;73(4):1107–1113. | ||
Wescombe PA, Burton JP, Cadieux PA, et al. Megaplasmids encode differing combinations of lantibiotics in Streptococcus salivarius. Antonie Van Leeuwenhoek. 2006;90(3):269–280. | ||
Wescombe PA, Hale JD, Heng NC, Tagg JR. Developing oral probiotics from Streptococcus salivarius. Future Microbiol. 2012;7(12):1355–1371. | ||
Power DA, Burton JP, Chilcott CN, Dawes PJ, Tagg JR. Preliminary investigations of the colonisation of upper respiratory tract tissues of infants using a paediatric formulation of the oral probiotic Streptococcus salivarius K12. Eur J Clin Microbiol Infect Dis. 2008;27(12):1261–1263. | ||
Horz HP, Meinelt A, Houben B, Conrads G. Distribution and persistence of probiotic Streptococcus salivarius K12 in the human oral cavity as determined by real-time quantitative polymerase chain reaction. Oral Microbiol Immunol. 2007;22(2):126–130. | ||
Burton JP, Wescombe PA, Moore CJ, Chilcott CN, Tagg JR. Safety assessment of the oral cavity probiotic Streptococcus salivarius K12. Appl Environ Microbiol. 2006;72(4):3050–3053. | ||
Burton JP, Cowley S, Simon RR, McKinney J, Wescombe PA, Tagg JR. Evaluation of safety and human tolerance of the oral probiotic Streptococcus salivarius K12: a randomized, placebo-controlled, double-blind study. Food Chem Toxicol. 2011;49(9):2356–2364. | ||
Di Pierro F, Adami T, Rapacioli G, Giardini N, Streitberger C. Clinical evaluation of the oral probiotic Streptococcus salivarius K12 in the prevention of recurrent pharyngitis and/or tonsillitis caused by Streptococcus pyogenes in adults. Expert Opin Biol Ther. 2013;13(3):339–343. | ||
Di Pierro F, Donato G, Fomia F, Adami T, Careddu D, Cassandro C, Albera R. Preliminary pediatric clinical evaluation of the oral probiotic Streptococcus salivarius K12 in preventing recurrent pharyngitis and/or tonsillitis caused by Streptococcus pyogenes and recurrent acute otitis media. Int J Gen Med. 2012;5:991–997. | ||
Di Pierro F, Di Pasquale D, Di Cicco M. Oral use of Streptococcus salivarius K12 in children with secretory otitis media: preliminary results of a pilot, uncontrolled study. Int J Gen Med. 2015;8:303–308. | ||
Di Pierro F, Colombo M, Zanvit A, Risso P, Rottoli AS. Use of Streptococcus salivarius K12 in the prevention of streptococcal and viral pharyngotonsillitis in children. Drug Healthc Patient Saf. 2014;6:15–20. | ||
Hod K, Ringel Y. Probiotics in functional bowel disorders. Best Pract Res Clin Gastroenterol. 2016;30(1):89–97. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.