Back to Journals » Clinical Ophthalmology » Volume 12
Pneumatic retinopexy: patient selection and specific factors
Received 27 October 2017
Accepted for publication 10 January 2018
Published 16 March 2018 Volume 2018:12 Pages 493—502
DOI https://doi.org/10.2147/OPTH.S137607
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Stephen Stewart, Wing Chan
Department of Ophthalmology, Royal Victoria Hospital, Belfast, UK
Abstract: Pneumatic retinopexy is a minimally invasive, non-incisional procedure for repairing retinal detachment. This procedure is associated with reduced morbidity, reduced cost and faster postoperative recovery compared to pars plana vitrectomy and scleral buckling. Superior visual acuity outcomes have also been reported. Intraoperative complications are primarily related to the intraocular pressure rise caused by gas injection, or the misdirection of the gas. There is a low rate of postoperative complications. Anatomic success rates of over 90% can be achieved when strict selection criteria are applied. However, a number of studies have revealed good outcomes in cases with expanded clinical indications. Pneumatic retinopexy is currently underutilized. With appropriate patient selection, a high success rate can be achieved, and therefore, this procedure has an important role in the armamentarium of the vitreoretinal surgeon.
Keywords: retina, vitreoretinal surgery, retinal detachment
Introduction
Pneumatic retinopexy is a minimally invasive, non-incisional procedure for repairing retinal detachment. It consists of injecting an expandable gas and applying retinal cryotherapy or laser photocoagulation to seal retinal breaks. It is an important tool in the armamentarium of the vitreoretinal surgeon, yielding good results in carefully selected patients.
In the USA, 15% of retinal detachments are repaired by pneumatic retinopexy,1 while 16% of vitreoretinal surgeons in the UK and Ireland perform this operation frequently.2 However, it is estimated that ~40% of all rhegmatogenous retinal detachments can be repaired using pneumatic retinopexy.3 The reasons for underutilization of pneumatic retinopexy include the belief that it does not relieve vitreoretinal traction, the need for skillful use of indirect ophthalmoscopy in retinal break detection and that more preoperative time is required for thorough examination of the retina to find retinal breaks and to prepare and educate patients. The techniques and instrumentation for pars plana vitrectomy and scleral buckling are reliable and familiar. Pneumatic retinopexy also has a slightly lower success rate compared to scleral buckling and pars plana vitrectomy.4,5
Background
History
The technique of repairing retinal detachments by retinopexy followed by gas endotamponade was first described by Rosengren in 1938.6 However, this method was not widely practiced until after the publication of Hilton and Grizzard’s seminal paper in 1986, in which they describe what we recognize as modern pneumatic retinopexy.3
Advantages
In appropriately selected patients, outcomes are comparable to alternative techniques of retinal detachment repair, and pneumatic retinopexy has the additional benefits of reduced morbidity, reduced costs and faster postoperative recovery.3,7 The incidence of cataract formation following pneumatic retinopexy is much lower than that associated with pars plana vitrectomy.8 The authors perform this procedure in the operating theater, but it can also be performed in an office setting, potentially avoiding delays and further reducing costs.
Outcomes
A review of 81 studies including 4,138 eyes undergoing primary pneumatic retinopexy revealed a single operation success rate of 74.4% in phakic and pseudophakic patients and a final success rate of 96.1% after further scleral buckling or vitrectomy procedures.9 In this comprehensive review (1986–2007) on “pneumatic retinopexy repair of retinal detachments”, Chan et al9 found the primary success rate in reattaching the retina with a single pneumatic retinopexy procedure in phakic patients is between 71% and 84%. In pseudophakic patients, the primary reattachment rate is between 41% and 67%. Satisfactory outcomes may still be achieved in cases with expanded indications for pneumatic retinopexy, although modification of the standard technique may be required.7,10,11
Large trials have demonstrated superior visual acuity outcomes for pneumatic retinopexy. A multicenter randomized controlled trial comparing pneumatic retinopexy and scleral buckling revealed that postoperative visual acuity of 20/50 or better at 6 months was achieved in 80% of pneumatic retinopexy cases versus 56% of scleral buckle procedures.4 Visual rehabilitation was also significantly faster in the pneumatic retinopexy group. In the PIVOT trial, patients who underwent pneumatic retinopexy had gained 4.9 more ETDRS letters at 1 year than vitrectomized eyes.12
Cost
Chang and Smiddy used stochastic modeling to analyze the costs of pneumatic retinopexy, scleral buckling and pars plana vitrectomy for primary repair of retinal detachment.13 This study estimated the cost (in USD) of pneumatic retinopexy performed in a hospital setting to be $2,343 versus $4,662 for scleral buckling and $5,061 for pars plana vitrectomy. Pneumatic retinopexy also compared favorably in an analysis of the utility of these procedures. Pneumatic retinopexy, assuming a moderate success rate of 75%, was calculated to cost $1,360 per quality-adjusted life year versus $2,243 for pars plana vitrectomy.
A retrospective study found that pneumatic retinopexy achieved comparable anatomic and functional outcomes, while being more cost-effective.7 It was estimated that pneumatic retinopexy is up to 50.9% and 59.4% less expensive than scleral buckling and pars plana vitrectomy, respectively. Tornambe calculated a cost saving of 59% for pneumatic retinopexy versus scleral buckling.14 Increased utilization of pneumatic retinopexy could, therefore, yield huge cost savings for local and national health care systems.
Technique
The specific steps of pneumatic retinopexy are dictated by the retinal pathology present and may vary due to surgeon preference, but the procedure must always comprise retinopexy, gas tamponade and posturing.
We present our preferred technique in Box 1, which could be applied to a patient with a single break or group of breaks within 1 clock hour, in the superior 8 clock hours of the retina.
  | Box 1 Authors’ recommended technique for pneumatic retinopexy |
Careful preoperative examination, including slit-lamp biomicroscopy and scleral indentation to examine the far peripheral retina, is essential in surgical planning. Any features that are likely to present intraoperative challenges or predict failure, such as the presence of media opacity, lattice degeneration or proliferative vitreoretinopathy (PVR), should be noted. For cases where it will be difficult to identify retinal breaks intraoperatively due to the presence of media opacity or a small pupil, it may be appropriate to mark the breaks with laser photocoagulation prior to injecting intraocular gas.15
The eye is anesthetized with topical proxymetacaine. Sub-Tenon anesthesia, using 2% lidocaine, is given. The periocular skin is cleaned with povidone iodine, and a drop of povidone iodine is instilled in the conjunctival sac. A sterile drape is placed over the eye and an eyelid speculum is inserted.
Transconjunctival cryopexy is applied, and is visualized with the binocular indirect ophthalmoscope to ensure the tear is entirely surrounded. Although laser is used in some cases, we prefer cryotherapy to achieve retinopexy because it facilitates a one-step procedure and, in our experience, is well tolerated by patients. Retinal cryopexy is technically easier for peripheral breaks, especially for small or hard-to-find breaks, compared to laser. Laser retinopexy must be performed 24–48 hours after gas injection, when the neurosensory retina is apposed to the retinal pigment epithelium, and can be challenging in the presence of a gas bubble.
A 27-gauge needle, attached to 1 mL syringe with the plunger removed, is used to perform a paracentesis. The needle enters the anterior chamber obliquely through the inferotemporal corneal limbus to create a self-sealing tunnel, with the tip passing over the iris to prevent lenticular touch. Removal of 0.2 mL of aqueous is usually sufficient.
We routinely use sulfur hexafluoride (SF6) for gas endotamponade. The gas bubble must be positioned correctly and should remain in the eye for long enough to close retinal breaks and facilitate resorption of subretinal fluid while chorioretinal adhesion occurs.
SF6 and perfluoropropane (C3F8) are generally the preferred agents.16 Filtered air is a non-expansile gas that is typically absorbed after 3 days and may be appropriate in some cases. The success rate for pneumatic retinopexy with filtered air has been reported as 85.7%–86.7%.17–19 Air is readily accessible, cheap and may be less toxic than SF6 and C3F8.20 It also allows a quicker recovery with a shorter duration of posturing. C2F6 has been found to be similarly effective to other tamponade agents,21 but is not widely used and is not approved by the US Food and Drug Administration for clinical use in the USA.
We recommend the injection of 0.5–0.6 mL of pure SF6, using a prefilled syringe and a 30 G needle. The needle is inserted via the pars plana, 4 mm posterior to the limbus in phakic patients and 3.5 mm posterior to the limbus in pseudophakia. Insertion is perpendicular to the sclera, penetrating the anterior hyaloid face and aiming for the center of the vitreous cavity. The needle is then withdrawn to leave 2–3 mm of the needle within the globe. Brisk injection of the gas generally avoids “fish-egg” gas bubbles, although animal studies suggest that slow and continuous injection more reliably avoids this problem.22
Iatrogenic macular detachment can be induced if there is inferior displacement of subretinal fluid toward an attached macula or inferior breaks in attached retina. Tornambe described the steamroller maneuver, which can be used to prevent this complication.23 The patient’s head position is rotated, so that the gas bubble is initially against the attached macula and then tracks along the attached retina toward the detached retina and tear.
Immediately following gas injection, the optic nerve head is viewed to assess for perfusion of the central retinal artery. If it is not perfused, another paracentesis is performed immediately to lower the intraocular pressure (IOP). The optic nerve is then rechecked, and the patient’s vision should be perception of light or better.
The prescribed head posture is dictated by the location of the retinal pathology and this is clarified with the patient after the procedure. We ask patients to posture continuously for 7 days. They are given a course of topical steroid and a cycloplegic. The patient is typically re-examined at 1 day, 1 week, 6 weeks and 6 months postoperatively. In our hands, this procedure yields a primary success rate of 80%.24
Complications
Intraoperative
Intraoperative complications are primarily related to the IOP rise caused by gas injection, or misdirection of the injected gas.
Raised IOP
A study of 22 eyes undergoing injection of SF6 during pneumatic retinopexy found that the mean IOP during gas injection was 97.2 mmHg, dropping to 46.5 mmHg immediately after.25 IOP falls toward baseline within 30–60 minutes of gas injection and typically does not rise again during the postoperative period.26 Given the pressure spike during gas injection, it is essential to visualize the optic nerve after gas injection and perform a paracentesis if the central retinal artery pulsation is absent.
Gas entrapment in pre-hyaloid space
Injected gas may track into the canal of Petit, which is the potential space between the anterior hyaloid, the lens-zonular diaphragm and the pars plana epithelium, creating the “sausage sign”.27,28 If there is a large amount of gas trapped, additional maneuvers are necessary to relocate the bubble. Facedown posturing within the first 24 hours postoperatively should allow the expanding bubble to break through the anterior hyaloid face and move toward the posterior pole. Alternatively, a 27-gauge needle attached to a 1 mL syringe filled with balanced salt solution can be inserted through the injection site, creating a drain sealed under water.29 Gas can then be reinjected at a new injection site.
Suprachoroidal gas
Suprachoroidal gas injection has been described, and the authors recommend care should be taken to ensure the needle tip is within the vitreous cavity, rather than the potential space between the sclera and pars plana, when injecting gas.30
Subconjunctival gas
Gas may enter the subconjunctival space if there is leakage through the needle track following gas injection.29 Vitreous incarceration can occur by a similar mechanism. This can be avoided by ensuring proper insertion of the needle into the vitreous cavity and by placing a dressed applicator on the needle track during withdrawal.
Corneal wound dehiscence
There is a risk of clear corneal wound dehiscence during gas injection, if pneumatic retinopexy is undertaken in the early postoperative period before corneal wounds have healed adequately.31
Postoperative
Surgical failure
Tornambe found that, for a series of 302 patients, the failure rate after a single operation was 32%.14 In this group, factors predictive of failure included pseudophakia or aphakia, the extent of retinal detachment and the number of retinal breaks. Failure of pneumatic retinopexy and need for subsequent scleral buckling or pars plana vitrectomy does not compromise the final visual acuity.32
New or missed breaks
A meta-analysis of 81 studies, which included 4,138 eyes, reported an incidence of new and missed retinal breaks of 11.7%.9 Studies have found that the most common cause of re-detachment is the formation of a new break with associated subretinal fluid.33,34 New breaks tend to occur during the first postoperative month and are likely related to an evolving posterior vitreous detachment, which may be accelerated by the presence of an expanding gas bubble.4,35 These breaks tend to be located in the superior retina, and approximately half are within 3 clock hours of the primary breaks.36 Careful examination during the early postoperative follow-up period is warranted. New retinal detachments do not necessarily require scleral buckling or pars plana vitrectomy, and may be managed successfully with repeat pneumatic retinopexy. Giant retinal tears have been reported following pneumatic retinopexy, but are rare.37,38
Extension, or re-opening, of original break
Failure of pneumatic retinopexy can also occur due to extension, or re-opening, of the original retinal breaks. This is usually due to progressive vitreoretinal traction or inadequate treatment to the retinal breaks. This can usually be avoided by ensuring satisfactory cryotherapy or laser is applied to the retinal breaks in the first instance.
Cataract progression
The development or progression of lens opacity is an uncommon complication of pneumatic retinopexy.4,29,39–42 In Tornambe’s multicenter randomized controlled trial of pneumatic retinopexy versus scleral buckling, looking at 198 eyes with at least 6 months follow-up, only 1 of the 103 eyes undergoing pneumatic retinopexy was noted to have developed cataract postoperatively.4 Two-year follow-up data for this cohort found progressive lens opacity in 19% in the pneumatic retinopexy group compared to 47% in the scleral buckle group, with 4% and 18% of eyes undergoing cataract surgery, respectively.42
A retrospective study of 193 eyes found a rate of cataract progression of 42% following small gauge pars plana vitrectomy, compared with 7% for pneumatic retinopexy.43 A smaller prospective study of 33 eyes with 24 months postoperative follow-up found no cataract progression during this period.41
Anterior chamber gas entrapment
Gas migration into the anterior chamber has been reported in phakic eyes following pneumatic retinopexy, and this can cause elevated IOP.44,45 Zonular dehiscence is thought to allow pre-hyaloidal gas to track into the anterior chamber. Paracentesis can be performed to evacuate this gas.
Subretinal gas
The presence of subretinal gas postoperatively may cause persistent retinal detachment.46 This phenomenon is thought to be related to the migration of “fish-egg” gas bubbles through the retinal breaks.14,29,46 This complication can be managed by posturing the patient, so that the bubbles are not in contact with large breaks until they coalesce, or by encouraging the bubbles to coalesce by flicking the globe intraoperatively with a dressed applicator. Subretinal gas can be eliminated with posturing, or may require vitrectomy with internal drainage.
Endophthalmitis
Post-pneumatic retinopexy endophthalmitis is rare.4,39,47,48 Infection can be managed with intravitreal antibiotics, although persistent retinal detachment in the setting of endophthalmitis will likely require further vitreoretinal surgery.48
Macular holes
The development of macular holes has been reported following pneumatic retinopexy49,50 and may occur in ~1% of cases.14 It is thought that this occurs as a result of dynamic changes in pre-existing vitreomacular traction, triggered by intraocular gas injection. These macular holes can be successfully managed by vitrectomy.51,52
Cystoid macular edema (CMO)
An observational study revealed CMO to be angiographically evident in 11% of patients undergoing pneumatic retinopexy versus 29% of scleral buckle patients.53 CMO was more common in patients with a macula-off detachment and in those who were pseudophakic. The development of CMO had a detrimental effect on the visual outcome.
Proliferative vitreoretinopathy
PVR is perhaps the most challenging complication in retinal detachment surgery. The reported incidence of PVR following pneumatic retinopexy ranges from 3% to 9.8%.4,14,29,54 Interestingly, Tornambe found no difference in the rate of PVR formation between those treated with cryopexy versus laser retinopexy.13 Re-detachment that is complicated by PVR typically warrants pars plana vitrectomy and membrane peeling.
Aniseikonia
Pneumatic retinopexy does not cause a refractive shift, but micropsic aniseikonia may be demonstrated postoperatively, particularly if the patient had a macula-off detachment. However, patients are generally asymptomatic.55
Patient selection
Pneumatic retinopexy is primarily indicated for the repair of uncomplicated retinal detachments without PVR. The ideal patients are those with the following:
- One break or a group of breaks within 1 clock hour.
- Retinal breaks involving the superior 8 clock hours of the fundus.
Phakic patients undergoing pneumatic retinopexy tend to do better than those who are pseudophakic or aphakic.42,54,56,57 Phakic patients have a single operation success rate of between 71% and 84%, whereas pseudophakic patients have a success rate of 41%–67%.9 This may be because aphakic and pseudophakic eyes are more prone to tiny retinal breaks in the far periphery, or in multiple quadrants, which may be missed preoperatively.28 However, other studies have not found lens status to affect outcomes.32,58 One-year results from the PIVOT trial showed that there was no statistically significant difference in outcomes between phakic and pseudophakic eyes, with both groups achieving a final reattachment rate of 99% after further vitrectomy or scleral buckling surgery.12
Pneumatic retinopexy is a desirable procedure in certain patients who would be unsuitable for placement of a scleral buckle. Patients with a single break under the superior rectus would be at risk of iatrogenic vertical diplopia following placement of a segmental buckle.28 Pneumatic retinopexy would obviate this risk.
Patients with comprised conjunctival or scleral integrity may also be better candidates for pneumatic retinopexy than scleral buckle. This includes patients who have previously had a glaucoma filtering procedure, thin sclera, previous strabismus surgery or pre-existing conjunctival scarring.
Unsuitable patients
Hilton and Grizzard also outlined three patient groups in which pneumatic retinopexy is contraindicated (Table 1).3
  | Table 1 Clinical features that make patients ideal, unsuitable or possible candidates for pneumatic retinopexy |
The presence of PVR grades C (marked) or D (massive) often warrants management by pars plana vitrectomy, given the increased risk of new breaks and re-detachment following pneumatic retinopexy. However, Tornambe found that a study of 40 eyes, including 7 eyes with PVR grade B or grade C in not more than two quadrants, had an overall success rate of 75%.59 There was no progression of PVR in these eyes following the procedure.
Pneumatic retinopexy is also contraindicated in patients with advanced glaucoma because of the risk of an IOP spike during gas injection. If there is end-stage thinning of the neuroretinal rim, transiently high pressure can cause snuff out.
The presence of media opacity that prevents adequate examination of the entire retina can also be considered a contraindication to pneumatic retinopexy due to the risk of missed retinal breaks or inadequate treatment to existing retinal breaks.
Post-procedure posturing is essential in achieving adequate tamponade of retinal breaks. Pneumatic retinopexy should not be undertaken in patients who are likely to have poor compliance because of physical or mental comorbidities.
Possible candidates
Patients with recurrent retinal detachment following scleral buckling may benefit from pneumatic retinopexy rather than the more invasive options of buckle revision or pars plana vitrectomy. A series of 40 such cases found a 90% success rate in this patient group.60 This is a useful rescue procedure for new or missed breaks. In this series, cases in which breaks were held open by vitreous traction were excluded, as these cases would be better suited to buckle revision or vitrectomy.
A retrospective study of seven eyes with retinal detachment that had previously undergone vitrectomy found that pneumatic retinopexy achieved reattachment in four cases.61 Of these four cases, two had undergone PPV for macular hole repair: one for retained lens fragments and one for epiretinal membrane peel. Pneumatic retinopexy was not successful in the one patient who had undergone pars plana vitrectomy for retinal detachment repair, and subsequent combined pars plana vitrectomy and scleral buckling was required. A larger study of 42 eyes demonstrated that rescue pneumatic retinopexy achieved equally good outcomes whether the primary surgery was scleral buckling or pars plana vitrectomy.62
Extensive lattice degeneration is a contraindication to pneumatic retinopexy, given the high risk of new breaks. However, lattice degeneration extending 3 clock hours or less does not affect the single operation success rate.14
Pneumatic retinopexy may also be useful in patients who are infirmed and unable to undergo a lengthy surgical procedure in a prone position.
Special considerations
Inferior breaks
Pneumatic retinopexy may have an adjunctive role for retinal detachments with inferior breaks.10 A nonconsecutive, retrospective case series of 11 patients demonstrated a single operation reattachment rate of 82%. The described technique in this study involves maintaining an inverted posture for 8 hours following the procedure. This method is, therefore, restricted to highly motivated patients without any significant mobility issues. There is also evidence that IOP increases significantly in the head-down position and, therefore, may not be appropriate for patients with glaucomatous optic neuropathy.63,64
Alternative techniques have been suggested to avoid the extreme inverted position (Table 2). A technique of posturing with a combined 10° Trendelenburg position, 10° neck hyperextension and 10° ocular supraduction, following pneumatic retinopexy for inferior retinal breaks after failed scleral buckle, was found to have a 88% success rate in 17 eyes.11 A lateral recumbent posture was found to yield a 77% success rate in 11 eyes with inferior detachments.65 The use of a temporary buckle, placed in the inferior fornix, and combined with a face-down position with 5°–10° of neck flexion achieved an 88% primary reattachment rate in 33 eyes.66
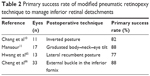  | Table 2 Primary success rate of modified pneumatic retinopexy technique to manage inferior retinal detachments |
It is important to note that very few of these cases had successful reattachment with pneumatic retinopexy alone. In a series described by Chang et al,10 4 of the 11 patients who achieved success had already undergone scleral buckling (1 patient) and 1 had undergone scleral buckling with vitrectomy. In Mansour’s11 series of 17 patients, all had previously undergone scleral buckling surgery. For the case series reported by Cheng et al,66 a short-duration external scleral buckle was used to flatten the retina in addition to a gas bubble.
Therefore, pneumatic retinopexy surgery as a primary procedure to manage retinal detachments secondary to inferior retinal breaks has only been reported in very small number of patients, and in most of these patients, previous scleral buckle and vitrectomy surgery was used. In the vast majority of cases, the gas bubble was used for additional internal tamponade after the vitreous traction had been relieved by the previous or concomitant scleral buckling surgery.
Breaks >1 clock hour apart
Retinal breaks within 1 clock hour in extent are more amenable to pneumatic retinopexy because the gas bubble can be positioned over the breaks with appropriate posturing. However, alternate head posturing has been described as a method of successfully tamponading breaks >1 clock hour apart.59
Giant tears or dialyses
The vast majority of giant retinal tears are managed by pars plana vitrectomy.67 However, small studies have demonstrated good results with pneumatic retinopexy. Irvine and Lahey reported an 80% reattachment rate in five eyes with acute giant tears with mobile flaps and <6 clock hours in extent.68 It was noted that subretinal fluid flows rapidly from the vitreous to the aqueous humors in these patients, and therefore, the authors theorize that subretinal fluid can be drained via a paracentesis rather than by a transscleral route.
Four patients with tears larger than 1 clock hour were also included in Tornambe’s study of expanded indications for pneumatic retinopexy, and the pooled group of patients achieved a success rate of 75%.59
A case series of four patients with traumatic retinal dialysis showed 100% primary reattachment following pneumatic retinopexy.69 All retinal tears were at least 3 clock hours in size and were located in the superior 8 clock hours of the retina. The authors theorize that the broad area of vitreous attachment to the posterior border of the torn retina may facilitate reattachment following pneumatic retinopexy.
Recurrent retinal detachments in the superior retina without identified breaks
The role of pneumatic retinopexy in recurrent retinal detachments without a detectable break is controversial, but the results of a small case series suggest it may have a useful role in these cases. McGimpsey et al described a technique where the limits of retinal detachment were first demarcated with argon laser followed by gas injection, in cases of superior recurrent retinal detachment without an identifiable break.70 Once the retina was reattached, argon laser was applied from the demarcation line to the ora serrata, paying particular attention to the area where a retinal break was suspected according to Lincoff’s rules.71 Six of six eyes in this series had successful reattachment, with a minimum follow-up of 497 days.
Pneumatic retinopexy to preserve macular attachment in macula-on retinal detachment
This is another controversial indication for pneumatic retinopexy that has not been reported in the peer-reviewed literature. In these cases, pneumatic retinopexy can be used to keep the macula attached in a macula-on retinal detachment, so that vitrectomy or scleral buckling surgery can be delayed to a more appropriate time when the necessary surgical team and resources are available.72 By injecting the gas bubble and positioning the patient face down, the macula will remain attached while awaiting a definitive procedure.
Young patients
Retinal detachment in young patients is rare, and may be associated with trauma, myopia, aphakia and retinopathy of prematurity.73 Pars plana vitrectomy is challenging in these cases because of firm vitreoretinal adhesion of the posterior hyaloid, and poses a significant risk of presenile cataract. A study of 19 eyes of 19 patients younger than 20 years of age revealed a primary success rate of 84% for pneumatic retinopexy, which is comparable to outcomes in adults.74
Retinal detachment associated with macular hole
The presence of a macular hole can complicate a retinal detachment, particularly in myopic eyes. Pneumatic retinopexy followed by prone posturing can be used to successfully repair these detachments.75,76 The application of laser to the macular hole rim is contentious77,78 and we do not routinely use laser in these cases.
Miscellaneous
Bilateral retinal detachments can be treated with pneumatic retinopexy.79–81 These patients can undergo simultaneous bilateral pneumatic retinopexy, and the possibility of faster visual rehabilitation may offer a particular advantage in these patients.
In some cases of retinal detachment, particularly in pseudophakic eyes, it may not be possible to visualize the causative break despite thorough examination. However, such cases were included in a study of 141 eyes undergoing pneumatic retinopexy and the absence of a visible break did not affect the success rate.7
Successful pneumatic retinopexy for a case of retinal detachment associated with a progressive superotemporal retinoschisis has been described.82
Pneumatic retinopexy may be a useful adjunct in cases of retinal detachment which are complicated by hypotony and choroidal detachment.83 Restoration of the IOP and reduction of the extent of detachment facilitate sooner definitive surgery than would usually be feasible.
Pneumatic retinopexy has been used to manage retinal detachments with several other retinal comorbidities, including vitelliform macular dystrophy, cytomegalovirus retinitis, Marfan syndrome and choroidal coloboma.84–87
Conclusion
Pneumatic retinopexy is recognized by most vitreoretinal surgeons as an effective technique for the initial management of rhegmatogenous retinal detachments, but is currently underutilized. When choosing this surgical technique, vitreoretinal surgeons and their patients must accept a greater chance of re-operation compared to scleral buckling or pars plana vitrectomy, and have a low threshold for proceeding to further surgery if pneumatic retinopexy fails. However, as a minimally invasive procedure, pneumatic retinopexy remains an excellent option in the management of retinal detachment in carefully selected patients.
Disclosure
The authors report no conflicts of interest in this work.
References
Hwang JC. Regional practice patterns for retinal detachment repair in the United States. Am J Ophthalmol. 2012;153(6):1125–1128. | ||
Assi AC, Chateris DG, Gregor ZJ. Practice patterns of pneumatic retinopexy in the United Kingdom. Br J Ophthalmol. 2001;85(2):244. | ||
Hilton GF, Grizzard WS. Pneumatic retinopexy. A two-step outpatient operation without conjunctival incision. Ophthalmology. 1986;93(5):626–641. | ||
Tornambe PE, Hilton GF. Pneumatic retinopexy. A multicenter randomized controlled clinical trial comparing pneumatic retinopexy with scleral buckling. The Retinal Detachment Study Group. Ophthalmology. 1989;96(6):772–783. | ||
Schaal S, Sherman MP, Barr CC, Kaplan HJ. Primary retinal detachment repair: comparison of 1-year outcomes of four surgical techniques. Retina. 2011;31(8):1500–1504. | ||
Rosengren B. Results of treatment of detachment of the retina with diathermy and injection of air into the vitreous. Acta Ophthalmol. 1938;16(4):573–579. | ||
Goldman DR, Shah CP, Heier JS. Expanded criteria for pneumatic retinopexy and potential cost savings. Ophthalmology. 2014;121(1):318–326. | ||
Feng H, Adelman R. Cataract formation following vitreoretinal procedures. Clin Ophthalmol. 2014;8:1957–1965. | ||
Chan CK, Lin SG, Nuthi AS, Salib DM. Pneumatic retinopexy for the repair of retinal detachments: a comprehensive review (1986–2007). Surv Ophthalmol. 2008;53(5):443–478. | ||
Chang TS, Pelzek CD, Nguyen RL, Purohit SS, Scott GR, Hay D. Inverted pneumatic retinopexy: a method of treating retinal detachments associated with inferior retinal breaks. Ophthalmology. 2003;110(3):589–594. | ||
Mansour AM. Pneumatic retinopexy for inferior retinal breaks. Ophthalmology. 2005;112(10):1771–1776. | ||
Muni RH. Randomized trial comparing pneumatic retinopexy vs. vitrectomy in the management of primary rhegmatogenous retinal detachment (PIVOT): 1-year results. Presented at American Society of Retina Specialists 35th Annual Meeting, August 11–15; 2017; Boston, MA, USA. | ||
Chang JS, Smiddy WE. Cost-effectiveness of retinal detachment repair. Ophthalmology. 2014;121(4):946–951. | ||
Tornambe PE. Pneumatic retinopexy: the evolution of case selection and surgical technique. A twelve-year study of 302 eyes. Trans Am Ophthalmol Soc. 1997;95:551–578. | ||
Muni R, Kertes PJ. Marking of retinal breaks in detached retina with laser photocoagulation before pneumatic retinopexy: a prospective case series. Retina. 2009;29(3):405–408. | ||
Ai E, Gardner TW. Current patterns of intraocular gas use in North America. Arch Ophthalmol. 1993;111(3):331–332. | ||
Yee KMP, Sebag J. Long-term results of office-based pneumatic retinopexy using pure air. Br J Ophthalmol. 2011;95(12):1728–1730. | ||
Sebag J, Tang M. Pneumatic retinopexy using only air. Retina. 1993;13(1):8–12. | ||
Algvere PV, Gjötterberg M, Olivestedt G, Fituri S. Results of pneumatic retinopexy with air. Acta Ophthalmol (Copenh). 1992;70(5):632–636. | ||
Juzoji H, Iwasaki T, Usui M, Hasemi M, Yamakawa N. Histological study of intraocular changes in rabbits after intravitreal gas injection. Jpn J Ophthalmol. 1997;41(5):278–283. | ||
Bochow TW, Olk RJ, Hershey JM. Pneumatic retinopexy perfluoroethane (C2F6) in the treatment of rhegmatogenous retinal detachment. Arch Ophthalmol. 1992;110(12):1723–1724. | ||
Bourla DH, Gupta A, Hubschman JP, Bourla N, Yu F, Schwartz SD. The slower the better: on the instability of gas jets in a model of pneumatic retinopexy. Invest Ophthalmol Vis Sci. 2007;48(6):2734–2737. | ||
Tornambe PE. Pneumatic retinopexy. Surv Ophthalmol. 1988;32(4):270–281. | ||
Mikhail MA, Mangioris G, Casalino G, et al. Outcome of primary rhegmatogenous retinal detachment surgery in a tertiary referral centre in Northern Ireland – A regional study. Ulster Med J. 2017;86(1):15–19. | ||
Abe T, Nakajima A, Nakamura H, Ishikawa M, Sakuragi S. Intraocular pressure during pneumatic retinopexy. Ophthalmic Surg Lasers. 1998;29(5):391–396. | ||
Coden DJ, Freeman WR, Weinreb RN. Intraocular pressure response after pneumatic retinopexy. Ophthalmic Surg. 1988;19(9):667–669. | ||
Steinmetz RL, Kreiger AE, Sidikaro Y. Previtreous space gas sequestration during pneumatic retinopexy. Am J Ophthalmol. 1989;107(2):191–192. | ||
Hilton GF, Das T, Majji AB, Jalali S. Pneumatic retinopexy: principles and practice. Indian J Ophthalmol. 1996;44(3):131–143. | ||
Hilton GF, Tornambe PE. Pneumatic retinopexy. An analysis of intraoperative and postoperative complications. The Retinal Detachment Study Group. Retina. 1991;11(3):285–294. | ||
Baker SR, Hainsworth DP. Suprachoroidal gas as a complication of pneumatic retinopexy. Retina. 2000;20(2):224–225. | ||
Jun AS, Pieramici DJ, Bridges WZ. Clear corneal cataract wound dehiscence during pneumatic retinopexy. Arch Ophthalmol. 2000;118(6):847–848. | ||
Zaidi AA, Alvarado R, Irvine A. Pneumatic retinopexy: success rate and complications. Br J Ophthalmol. 2006;90(4):427–428. | ||
Mudvari SS, Ravage ZB, Rezaei KA. Retinal detachment after primary pneumatic retinopexy. Retina. 2009;29(10):1474–1478. | ||
Gorovoy IR, Eller AW, Friberg TR, Coe R. Characterization of pneumatic retinopexy failures and the pneumatic pump: a new complication of pneumatic retinopexy. Retina. 2014;34(4):700–704. | ||
Smiddy WE, Flynn HW Jr, Nicholson DH, et al. Results and complications in treated retinal breaks. Am J Ophthalmol. 1991;112(6):623–631. | ||
Holz ER, Mieler WF. View 3: the case for pneumatic retinopexy. Br J Ophthalmol. 2003;87(6):787–789. | ||
Sinkar SN, Simon SJ, Gilhotra JS. Giant retinal tear after pneumatic retinopexy. Retin Cases Brief Rep. 2012;6(2):151–152. | ||
Yam JC, Liu DT, Lee VY, Lam PT, Lam DS. Giant retinal tear after pneumatic retinopexy. Acta Ophthalmol. 2008;86(2):232–233. | ||
Hilton GF, Tornambe PE, Brinton DA, et al. The complication of pneumatic retinopexy. Trans Am Ophthalmol Soc. 1990;88:191–210. | ||
Kim RY, D’Amico DJ. Postoperative complications of pneumatic retinopexy. Int Ophthalmol Clin. 2000;40:165–173. | ||
Mougharbel M, Koch FH, Böker T, Spitznas M. No cataract two years after pneumatic retinopexy. Ophthalmology. 1994;101(7):1191–1194. | ||
Tornambe PE, Hilton GF, Brinton DA, et al. Pneumatic retinopexy. A two-year follow-up study of the multicenter clinical trial comparing pneumatic retinopexy with scleral buckling. Ophthalmology. 1991;98(7):1115–1123. | ||
Feng H, Adelman RA. Cataract formation following vitreoretinal procedures. Clin Ophthalmol. 2014;8:1957–1965. | ||
Taher RM, Haimovici R. Anterior chamber gas entrapment after phakic pneumatic retinopexy. Retina. 2001;21(1):681–682. | ||
Tan CS, Wee K, Zaw MD, Lim TH. Anterior chamber gas bubble following pneumatic retinopexy in a young, phakic patient. Clin Exp Ophthalmol. 2011;39(3):276–277. | ||
McDonald HR, Abrams GW, Irvine AR, et al. The management of subretinal gas following attempted pneumatic retinal reattachment. Ophthalmology. 1987;94(4):319–326. | ||
Eckardt C. Staphylococcus epidermidis endophthalmitis after pneumatic retinopexy. Am J Ophthalmol. 1987;103(5):720–721. | ||
Sharma T. Post-pneumatic retinopexy endophthalmitis: management of infection and persistent retinal detachment. Ophthalmic Surg Lasers Imaging. Epub 2010 Mar 9. | ||
Avins LR, Krummenacher TR. Macular holes after pneumatic retinopexy. Case reports. Arch Ophthalmol. 1988;106(6):724–725. | ||
Runge PE, Wyhinny GJ. Macular hole secondary to pneumatic retinopexy. Case report. Arch Ophthalmol. 1988;106(5):586–587. | ||
Hejny C, Han DP. Vitrectomy for macular hole after pneumatic retinopexy. Retina. 1997;17(4):356–357. | ||
Moshfeghi AA, Salam GA, Deramo VA, et al. Management of macular holes that develop after retinal detachment repair. Am J Ophthalmol. 2003;136(5):895–899. | ||
Tunc M, Lahey JM, Kearney JJ, Lewis JM, Francis R. Cystoid macular oedema following pneumatic retinopexy vs scleral buckling. Eye (Lond). 2007;21(6):831–834. | ||
Chen JC, Robertson JE, Coonan P, et al. Results and complications of pneumatic retinopexy. Ophthalmology. 1988;95(5):601–606. | ||
Lee HN, Lin KH, Tsai HY, Shen YC, Wang CY, Wu R. Aniseikonia following pneumatic retinopexy for rhegmatogenous retinal detachment. Am J Ophthalmol. 2014;158(5):1056–1061. | ||
Algvere P, Hallnäs K, Palmqvist BM. Success and complications of pneumatic retinopexy. Am J Ophthalmol. 1988;106(4):400–404. | ||
Abecia E, Pinilla I, Olivan JM, Larrosa JM, Polo V, Honrubia FM. Anatomic results and complications in a long-term follow-up of pneumatic retinopexy cases. Retina. 2000;20(2):156–161. | ||
Assi AC, Charteris DG, Pearson RV, Gregor ZJ. Pneumatic retinopexy in the treatment of primary rhegmatogenous retinal detachment. Eye (Lond). 1999;13(Pt 6):725–728. | ||
Tornambe PE, Hilton GF, Kelly NF, Salzano TC, Wells JW, Wendel RT. Expanded indications for pneumatic retinopexy. Ophthalmology. 1988;95(5):597–600. | ||
Friberg TR, Eller AW. Laser pneumatic retinopexy for repair of recurrent retinal detachment after failed scleral buckle-ten years experience. Ophthalmic Surg Lasers. 2001;32(1):13–18. | ||
Modi YS, Townsend J, Epstein AE, Smiddy WE, Flynn HW Jr. Pneumatic retinopexy for retinal detachment occurring after prior scleral buckle or pars plana vitrectomy. Ophthalmic Surg Lasers Imaging Retina. 2014;45(5):409–413. | ||
Petrushkin HJ, Elgohary MA, Sullivan PM. Rescue pneumatic retinopexy in patients with failed primary retinal detachment surgery. Retina. 2015;35(9):1851–1859. | ||
Mansour AM, Feghali JG, To’mey K, Jaroudi N. Increased intraocular pressure with head-down position. Am J Ophthalmol. 1984;98(1):114–115. | ||
Mansour AM. Inverted pneumatic retinopexy. Ophthalmology. 2004;111:1435; author reply 1435. | ||
Hwang JF, Chen SN, Lin CJ. Treatment of inferior rhegmatogenous retinal detachment by pneumatic retinopexy technique. Retina. 2011;31(2):257–261. | ||
Cheng HC, Lee SM, Lee FL, Liu JH, Kuan CH, Lin PK. Short-term external buckling with pneumatic retinopexy for retinal detachment with inferior retinal breaks. Am J Ophthalmol. 2013;155(4):750.e1–756.e1. | ||
Shunmugam M, Ang GS, Lois N. Giant retinal tears. Surv Ophthalmol. 2014;59(2):192–216. | ||
Irvine AR, Lahey JM. Pneumatic retinopexy for giant retinal tears. Ophthalmology. 1994;101(3):524–528. | ||
Melgen SE, Michels M. Pneumatic retinopexy for the treatment of giant retinal dialyses. Am J Ophthalmol. 1994;118(6):762–765. | ||
McGimpsey SJ, Cooke CA, Best RM. Pneumatic retinopexy. Ophthalmology. 2007;114:401–402. | ||
Saxena S, Lincoff H. Finding the retinal break in rhegmatogenous retinal detachment. Indian J Ophthalmol. 2001;49(3):199–202. | ||
Mandelcorn ED, Manusow JS, Mandelcorn MS. Pearls for performing pneumatic retinopexy. Retin Spec. 2015: November (13):18–21. | ||
Winslow RL, Tasman W. Juvenile rhegmatogenous retinal detachment. Ophthalmology. 1978;85(6):607–618. | ||
Chen SN, Hwang JF. Treatment of rhegmatogenous retinal detachment in teenagers by pneumatic retinopexy technique. Am J Ophthalmol. 2007;143(2):217–221. | ||
Menchini U, Scialdone A, Visconti C, Brancato R. Pneumoretinopexy in the treatment of retinal detachment with macular hole. Int Ophthalmol. 1988;12(4):213–215. | ||
Rashed O, Sheta S. Evaluation of the functional results after different techniques for treatment of retinal detachment due to macular holes. Graefes Arch Clin Exp Ophthalmol. 1989;227(6):508–512. | ||
Lu L, Li Y, Cai S, Yang J. Vitreous surgery in highly myopic retinal detachment resulting from a macular hole. Clin Exp Ophthalmol. 2002;30(4):261–265. | ||
Kwok AK, Cheng LL, Gopal L, Sharma T, Lam DS. Endolaser around macular hole in the management of associated retinal detachment in highly myopic eyes. Retina. 2000;20(5):439–444. | ||
Kerimoglu H, Ozkagnici A, Okudan S. Bilateral rhegmatogenous retinal detachment repaired with simultaneous bilateral pneumatic retinopexy. Can J Ophthalmol. 2009;44(2):210. | ||
Tornambe PE. Bilateral retinal detachment repaired with bilateral pneumatic retinopexy. Case report. Arch Ophthalmol. 1987;105(11):1489. | ||
Rubin U, De Jager C, Zakour M, Gonder JT. A second case of bilateral rhegmatogenous retinal detachments repaired with simultaneous bilateral pneumatic retinopexy. Retin Cases Brief Rep. 2017;11(3):255–257. | ||
Vrabec TR. Pneumatic retinopexy in a progressive rhegmatogenous retinoschisis retinal detachment. Arch Ophthalmol. 2000;118(5):720–721. | ||
Yeung L, Kokame GT, Brod RD, Lightman DA, Lai JC. Pneumatic retinopexy for retinal detachment associated with severe choroidal detachment. Retina. 2011;31(1):87–92. | ||
Glacet-Bernard A, Coscas G. Full-thickness macular hole and retinal detachment complicating Best’s disease. Eur J Ophthalmol. 1993;3(1):53–54. | ||
McAuliffe PF, Heinemann MH. The use of pneumatic retinopexy to delay surgical repair of a retinal detachment associated with the ganciclovir intraocular device. Ophthalmic Surg Lasers. 1998;29(3):244–246. | ||
Greco GM, Ambrosino L. Treatment of retinal detachment in Marfan syndrome. Ann Ophthalmol. 1993;25(2):72–76. | ||
Giansanti F, Giuntoli M, Mazzini C, Pieretti G, Abbruzzese G, Menchini U. Pneumatic retinopexy for retinal detachment associated with choroidal coloboma. Eur J Ophthalmol. 2012;22(4):680–682. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
