Back to Journals » Journal of Multidisciplinary Healthcare » Volume 13
Physicians’ Attitudes and Ethical Obligations to Pharmacogenetic Testing
Authors Muflih S , Al-Husein BA , Karasneh R , Alzoubi KH
Received 9 January 2020
Accepted for publication 27 February 2020
Published 10 March 2020 Volume 2020:13 Pages 249—258
DOI https://doi.org/10.2147/JMDH.S245369
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Scott Fraser
Suhaib Muflih, 1 Belal A Al-Husein, 1 Reema Karasneh, 2 Karem H Alzoubi 1
1Department of Clinical Pharmacy, Jordan University of Science and Technology, Irbid, Jordan; 2Department of Basic Medical Sciences, Yarmouk University, Irbid, Jordan
Correspondence: Suhaib Muflih
Department of Clinical Pharmacy, Jordan University of Science and Technology, Irbid 22110, Jordan
Tel +962-79-7868603
Email [email protected]
Introduction: Despite the increased utilization of pharmacogenetic (PGt) testing to guide drug therapy, little is known about the ethical challenges posed by the use of these genetic tools.
Methods: This cross-sectional study aimed to address ethical issues related to ancillary genetic information, consent forms, and potential confidentiality breaches from physicians’ perspectives. A questionnaire was administered to all practicing physicians working in KAUH.
Results: Almost 49% and 65% of physicians were willing to recommend PGt testing for adult and pediatric patients, respectively. The findings showed that physicians attitudes towards the clinical utility of PGt testing became more preceptive. The majority (73.7%) indicated that PGt testing should not be treated as other routine laboratory tests. The finding also focused on potential conflicts regarding ancillary genetic information, in which 78.8% indicated that they would like to preserve the confidentiality and privacy of the patients and only 14.4% of physicians did not feel obligated to let patients know about any future risk that might be uncovered using PGt testing. The findings showed that collecting both verbal and written consents was imperative prior to testing. Seriousness and predictability of the diseases were reported to be legitimate circumstances that allow disclosure of genetic information.
Discussion: Unless the field of PGt testing addresses the ethical challenges that might be encountered during PGt treatment, these issues might influence its acceptance in routine clinical settings. Establishing a minimal set of ethical standards may help emphasize the role of physicians and thus facilitate the implementation of PGt tests.
Keywords: PGt testing, ethical issues, physicians, informed consent form, ancillary information
Introduction
Pharmacogenetic (PGt) testing as a relatively new tool for the personalized treatment and prevention of chronic diseases, is a significant medical advancement.1 While PGt studies investigate individuals’ responses to medications based on single-gene mutations, pharmacogenomics (PGx) approaches imply the study of many genes or whole sets of genes.2 Although PGt has potential to improve drug safety and efficacy, its acceptance in medical practice depends heavily on the ethical issues that need to be addressed to ensure the appropriate use of PGt testing.1 Some ethical challenges arise from the application of ethical principles that support patient autonomy and confidentiality on one hand,3 and what genetic information can be revealed on the other hand.4 After the successful completion of Human Genomic Project, the availability of DNA sequences has rapidly advanced the diagnosis of genetic diseases by identifying genetic markers that link inherited genetic diseases with the responsible genes.5 Another field of genetic testing application that evaluates the safety and efficacy of medication is drug-gene testing or PGt testing that can be used to assess how genetic variations influence individuals’ response to medications.1 Genetic variations in CYPs P450-mediated drug metabolism can significantly affect the rate of drug metabolism among different individuals. For instance, polymorphism in the CYP2C9 gene is responsible for about a 30% to 90% reduction in the enzymatic activity and subsequently the inter-individual unpredictability of warfarin clearance from the body.6 Thus, using PGt testing may guide drug dose and drug choices by providing pharmacokinetics information and recognizing drug effectiveness.
Further, PGt tests have the potential to reveal ancillary or secondary medical information unrelated to test purposes,4 in which additional information pertaining to a tendency towards developing future diseases can be estimated. For example; APOE profiles that may help to predict statin medications treatment response can provide extra information about Alzheimer’s disease.4 When the patient refuses to warn relatives, the health-care professional has to choose between obligations to protect and respect patients’ right to privacy or to prevent the harm and warn at-risk relatives.7 Haga et al reported that many health professionals felt that ancillary information would scare patients, and reported that it was their duty to disclose it; particularly if the condition was treatable. Further, they believed that it would be challenging to communicate the genetic ancillary risk information, especially for patients with low knowledge and given the shortage of time of a typical appointment.4 Additionally, informed consent for PGt testing is another area that raises a number of ethical issues. Many providers believed that informed consent was necessary while others believed it was unnecessary or warranted when testing may reveal secondary information.8
Despite revision to more than 250 prescribed drugs regarding the impact of genetic variation on medication safety and efficacy,9 there is a paucity of studies that have been conducted to assess some of ethical issues associated with the utilization of PGt testing among a cohort of physicians in Jordan. The contribution of this research is expected to significantly help develop possible solutions that are both reasonable and ethically acceptable to promote the use of PGt testing and maximize the potential benefit health outcome of these tools even if some ethical road blockers remain unresolved. This proposed study will focus primarily on investigating physicians’ attitudes towards PGt testing and the ethical obligations arising from its clinical implementation, as it is important for ethicists and health-care providers to work together to clarify the ethical challenges that will likely arise in clinical settings.
Methods
A cross-sectional, descriptive design was implemented in a tertiary hospital clinical setting in the northern province of Jordan. The study protocol was submitted in order to obtain approval by the Jordan University of Science and Technology’s (JUST) Institutional Review Board (IRB). The population was physicians working in King Abdulla University Hospital’s (KAUH) Clinics affiliated by JUST. Physicians’ currently involved in medication therapy decision-making were eligible to participate in the study. Potential participants received a self-administered questionnaire with a covering letter introducing the study and explaining its objectives. The physician survey collected data on demographic characteristics and opinions regarding PGt testing-related ethical issues.
The study protocol, the 3-page survey, and the informed consent form have been submitted and approved by Jordan University of Science and Technology’s (JUST) Institutional Review Board (IRB) before the implementation of the study in compliance with the Health Insurance Portability and Accountability Act (HIPAA). The population was physicians working in King Abdullah University Hospital’s (KAUH) Clinics affiliated by JUST. Physicians currently involved in medication therapy decision-making were included in the study. Eligible participants received a self-administered paper-based questionnaire along with the informed consent form, which stated the main objectives of the study, risks, benefits, and other important information necessary to help participants make voluntary and informed decisions regarding their participation in this study. The informed consent form was read, signed and dated by each participant agreed to partake in the study. The participating physicians were given a copy of the signed and dated informed consent form to keep. The physician survey collected data on demographic characteristics and opinions regarding PGt testing-related ethical issues. The data collected did not include direct identifiers of participating physicians.
Questionnaire Development
Scales for measuring attitudes towards PGt testing have been developed, for the most part, in studies relating physicians’ knowledge, opinions, and attitudes to implementing PGt testing in routine clinical practice.10–14 We developed a set of main ideas and primary items directly relevant to our topic from the scientific literature on physicians’ attitudes towards PGt and published studies on physicians’ ethical obligations to respect patients’ privacy, maintain the confidentiality of their genetic information, and warn at-risk families.3,7,15-17 The primary items were reviewed by five experts in the field of pharmacogenetics required to provide feedback and suggest necessary changes in order to establish both face and content validity of the questionnaire. Afterwards, the reliability of the questionnaire was established using a pilot test by collecting data from 15 subjects not included in the study sample. The final survey was three pages long and contained 25 items, including both Likert scale and multiple-choice formats.
The 5-point Likert scale was used to measure physicians’ attitudes towards PGt testing by giving each item of the questionnaire one point (Strongly Disagree=1, Disagree=2, Neutral=3, Agree=4, and Strongly Agree=5), to analyze and rank the results as follows:
- (2.33) Low score on Likert scale
- (2.34–3.67) Medium score on Likert scale
- (3.68–5) High score on Likert scale
Sample Size and Statistical Analysis
A large, representative sample was planned for this study. Due to the lack of studies that consider the effect size required for this type of research, the necessary sample size required to detect a significant effect with 0.08 statistical power and a significance level of 0.05 was estimated to be 200 participants. Univariate analysis was conducted to describe and summarize nominal, ordinal, and continuous data. Measures of central tendency, variation, and normality were utilized for continuous variables, while counts and percentages were used for categorical variables. Statistical packages IBM® SPSS version 24.0 as utilized to check all the assumptions of the One-way analysis of variance (ANOVA) and the linear regression models. One-way ANOVA analysis was performed to address physicians’ attitudes and opinions based on gender, duration of practice, and specialty. The Chi-square statistic was performed for testing relationships between categorical variables. Further, before using the items collected from available studies to measure attitudes, they were grouped into construct subscales by performing factor analysis that aim to reduce number of items that capturing same variance.18
Research Questions
To achieve the goal of the study, the following research questions were answered:
- Do physicians have favorable attitudes towards PGt testing?
- Should physicians obtain PGt testing, if recommended, for adult and pediatric patients when there is a possibility of disclosing a future disease and inquiring specific therapies, which might affect their health insurance coverage?
- Do you think that PGt tests should be treated as other routine laboratory tests?
- How to deal with ancillary genetic information when an individual is not currently seeking treatment for?
- How to proceed with certain PGt test results that might also predict future risk of disease as well as drug response? For example, the apolipoprotein E4 (APOE4) gene variant is associated with a reduced response to statins, and an increased risk of Alzheimer’s disease.
Results
Sample Characteristics
A total of 200 physicians successfully completed the paper-based survey. The majority of participating physicians were female and more likely to be employed by only governmental hospitals. The majority of the participants had active practice in internal and family medicine, and most of the participants practiced medicine for a medium duration of 6–10 years in overall medical practice (see Table 1).
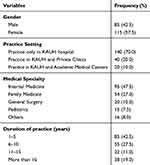 |
Table 1 Percentage Distribution of Selected Demographic Variables of Prescribers in the Sample |
First Question: Do Physicians Have Favorable Attitudes Towards PGt Testing?
The attitude-related aspects of the survey were administered to 15 medical students to be able to perform exploratory factor analysis on the data. The Keiser-Meyer-Olkin (KMO), which measures sampling adequacy was 0.926, and the Bartlett’s test of sphericity found significant at the 0.001 level. The determinant of the R-matrix was 0.038 which was greater than 0.000001. These results indicated that the data set was appropriate for factor analysis.19 We tested the two-factor solution of 12 items on the collected sample that consisted of 200 physicians as a part of the confirmatory factor analysis, we arrived at a two-factor model of nine items as the finalized scale, which measures: (a) physician attitudes towards the clinical utility of PGt testing and (b) physician attitudes towards risks and limitations of PGt testing. Cronbach Alpha values were 0.83 and 0.76, respectively. The majority of participants assigned higher ratings in the attitude component of the clinical utility of PGt testing (see Table 2). Gender and type of specialty were significantly associated with physicians’ preferences towards PGt testing. Male physicians and those who were practicing internal medicine had higher attitudes towards the clinical utility of PGt testing than female physicians and those who were practicing in other branches of medicine.
 |
Table 2 Mean and Standard Deviations of Physicians’ Attitudes Towards the Use of PGt Testing |
Second Question: Should Physicians Obtain PGt Testing, if Recommended, for Adult and Pediatric Patients When There Is a Possibility of Disclosing a Future Disease and Inquiring Specific Therapies, Which Might Affect Their Health Insurance Coverage?
When the participants were asked about their willingness to obtain PGt testing for adult patients, almost one-half (49.4%) answered “Yes.” However, when participants were asked about their willingness to obtain PGt testing for pediatric patients, almost 65% agreed by answering “Yes.” Physicians who agreed to administer PGt testing for both adult and pediatric patients regardless the potential of disclosing future diseases scored higher only on the attitudes towards the clinical utility of PGt testing scale than those who were rejected or were not sure about their decision-making (p < 0.05).
Third Question: Do You Think That PGt Tests Should Be Treated as Other Routine Laboratory Tests?
When participants were asked whether they think that PGt tests should be treated as other routine laboratory tests, in which verbal consent or minimal explanation of testing suffice, only 26.3% answered “Yes,” while the majority disagreed or were not sure about their clinical decision. There were no significant differences in the responses of male and female physicians to the question. However, physicians who did not agree to deal with PGt testing as other routine clinical tests expressed more positive attitudes towards the confidentiality PGt testing results than those thought PGt testing is very similar to other routine clinical tests (p < 0.05). Variability in requesting informed consents PGt testing also existed in this study. The majority indicated that no specific consent forms were required if testing offers lower risks and higher benefits (56.3%), or when there is a potential risk of discrimination or stigmatization for some infectious diseases (55.6%) such as tuberculosis and HIV/AIDS (see Table 3).
 |
Table 3 Frequency Distribution of Physicians’ Responses to the Need of Informed Consent |
Fourth Question: How to Deal with Ancillary Genetic Information When an Individual Is Not Currently Seeking Treatment for?
When participants were asked about how to proceed with general secondary or ancillary genomic information, 78.8% felt the need to restrict sharing the genetic information to minimize harm to the patients and their families, while 21.2% indicated that they would disclose the results of genomic research to the patients but not to families. When patients refused to accept responsibility to share PGt testing results with their family members, most of the participants (45%) indicated that they would take reasonable steps to directly warn at-risk family members and only 5.6% indicated that they would protect the framework of the physician–patient relationship by not disclosing patient’s information to family (see Table 4). More than 70% of participants reported that seriousness and predictability of the diseases, the ability to identify at-risk patients, and when early monitoring could reduce risk or prevent harm were legitimate circumstances that allow them to warn at-risk individuals. Participants who agreed to share secondary genetic information only with patients expressed higher attitudes towards the risk and limitations of PGt testing than those who were not interested in sharing secondary generic findings with patients and families (p < 0.05). Gender, duration of practice, and medical specialty were not significantly associated with physicians’ decisions regarding disclosing genetic information.
 |
Table 4 Frequency Distribution of Physicians’ Responses Regarding Disclosure of Ancillary Information |
Fifth Question: How to Proceed with Certain Pharmacogenetic Test Results That Might Also Predict Future Risk of Disease as Well as Drug Response? For Example, the Apolipoprotein E4 (APOE4) Gene Variant Is Associated with a Reduced Response to Statins and an Increased Risk of Alzheimer’s Disease
A factual statement of the apolipoprotein E4 (APOE4) gene variant, which is associated with a reduced response to statins and an increased risk of Alzheimer’s disease,20 was used to illustrate potential clinical and ethical issues that may arise with PGt testing. The majority of participants (53.8%) indicated that patients should be forewarned about the possibility of carrying high-risk gene that may cause a disease in the future (see Table 4). Moreover, participants who indicated that patients should agree to opt out of learning secondary findings prior to genetic sequencing were less likely to have ever participated in the implementation (eg, ordering testing or translating results) of PGt testing in their practice than those who preferred to disclose future risks. Physicians who were willing to inform patients about the possibility of carrying high-risk gene that may cause a disease in the future had significantly higher attitudes towards the clinical utility of PGt testing (p < 0.05) and were more willing to accept PGt testing in practice (p < 0.05) than those who did not feel obligated to let patients know about future risk. Male physicians and those who were practicing family medicine were less likely to inform patients about the possibility of carrying high-risk gene; however, the association was not significant.
Discussion
First Question
The overall attitudes of physicians towards PGt testing were positive, as the majority agreed or strongly agreed about the potential benefits of PGt testing in decreasing the number of adverse drug events, prescribing correct medication, and potentially improving the safety and efficacy of medications. However, concerns included vulnerability to unauthorized access to genetic information, risk of discrimination in health insurance and employability were also expressed by participants (see Table 2). The findings of this study were consistent with other previously published studies.21,22 Participants of this study were more perceptive for the risks and limitations associated with PGt testing compared to Rogausch et al study when they considered the potential of discrimination by health insurance companies and employers.12 The increasing availability of genetic markers and direct-to-consumer advertisements about genetic testing may explain our findings. The significant relationships between patients’ attitudes and both gender and specialty could be explained by a higher percentage of male and internal medicine physicians having attained higher levels of genetic education. The lack of statistical significance of duration of practice might reflect similar comfort levels of physicians’ knowledge about PGt tests, across years of practice, due to experiencing similar exposure with PGt testing.
Second Question
Almost 49.4% and 65% of participants were willing to obtain PGt testing for adult and pediatric patients, respectively. This finding could emphasize the importance of raising awareness about genetic-based treatment which allows integrating genetic information with other patient factors to maximize the benefits of medications that patients expect. A greater number of physicians agreed to administer PGt testing for children expressed slightly more positive attitudes towards the confidentiality PGt testing results that those who refused to do so. This finding could be partly explained by their extensive knowledge of PGt testing. Similar findings were reported by O’Donnell et al who implemented PGt testing to 868 adult patients receiving at least one prescription medication. Authors reported that almost 14% of the most prevalent medications taken by patients including aspirin, omeprazole, atorvastatin, hydrochlorothiazide, lisinopril, and amlodipine, flagged cautionary or high-risk medication alerts, and required changes based on PGt information.23 Physicians need to be continuously educated about other factors that necessitate testing. For example, the American College of Medical Genetics (ACMG) and National Society of Genetic Counselors (NSGC) recommend against testing asymptomatic children at risk for adult-onset disease when no reasonable medical intervention is available.16,24 Further, Crews et al suggested that implementing a tailored intervention based on the findings of genetic variants might be challenging among different ethnic and racial groups.25
Third Question
This finding may reflect physicians’ awareness and knowledge of PGt tests, which are conducted in speciated laboratories at higher costs than routine tests and require health-care providers to consistently update their genetic knowledge. This finding of this study also suggests that physicians must be familiar with current ethical issues that could be associated with its use to allow them to convey reliable information. A cross-sectional multi-country survey conducted in the USA, Canada, the UK, and Australia examined the public’s views on DNA information versus other medical information, further support our previous findings. The study reported that more than half of participants held exceptionalist views since they believed that genetic information has special properties than other types of health and medical information. Although a higher percentage of participants holding genetic exceptionalist views perceived the risk of genetic information (eg, linking personally identifiable information to DNA, being discriminated against by health insurance companies), they more willing to donate their DNA for research than other counterparts, reflecting more perceived benefits regarding PGt testing.26
Although PGt tests might still reveal personal information that could adversely affect patients’ insurability and employability, the findings of this study revealed that informed consent for PGt may not be essential for all clinical uses of PGt drugs that could be attributed to the lack of physicians’ experience regarding ethical questions that are linked to disease-specific predictive genetic testing. In contrast to our findings, several studied revealed that the potential risk of secondary findings with PGt tests would warrant informed consent.15,17,27 The discrepancy might be explained by participants’ belief that informed consent may not provide complete protection of their rights and some patients do not fully understand the potential risks and benefits associated with PGt procedures. However, before the performance of PGt testing, the patient should be given a full explanation of the aim and the possible results of the test with a clear explanation for all medical terms to help patients make appropriate therapeutic options towards their health.28 A clear informed consent process would also provide an opportunity for both patients and health-care providers to set the specific testing options. Further, as the participants of this study indicated that they did not require requesting informed consent when there is a potential risk of discrimination or stigmatization for some infectious diseases, it could be explained by the lack of standardized process of consenting that might deprive patients’ from receiving treatment only based on theoretical genetic results.
Fourth Question
Although health professionals expressed positive attitudes and interest in the use of PGt testing, their willingness to disclose secondary genetic information was limited likely due to their concerns about causing psychological harm to patients, the lack of evidence of clinical utility of PGt testing, as well as their limited ability to interpret and communicate ancillary disease risk information to their patients. This finding reinforces the need for additional educational resources and well-defined clinical guidelines about the use of PGt testing. Although the majority of participating physicians in this study refrained from sharing secondary genetic information with patients’ family members, it is their duty to disclose genetic information, especially if the condition is treatable. The findings of this research were in consistent with previous studies; a series of focus groups designed to identify the attitudes of health professionals toward PGt testing and disease-risk information, revealed that disclosing ancillary information would scare patients.4 Importantly, Haga and Burke believed that disclosing ancillary information to patients may result into undesirable or stigmatizing feelings that could be a reason to avoid testing or excluding certain gene variants from being tested.29
In contrast to the public’s expectations in the USA and Israel, there is large public support for a legal duty to share patients’ genetic information with their relatives and vice versa in the UK.30 The lack of clear guidelines regarding the ethical and legal issues surrounding the disclosure of patients’ confidentiality to warn at-risk families put physicians in the thick of debate. A few participants in this study believed that directly warning at-risk family members would be the ideal approach as then the family members would be able to decide whether or not to undergo further testing and examination in order to take preventative measures at early stages; however, this approach could not be free of harm and ethical dilemma. The disclosure of genetic risk information directly to at-risk family members shows disregard for patients’ autonomy and confidentiality that might cause mental and emotional distress for patients as well as undermine patients’ trust in their healthcare system. An ongoing court case concerning the rule of confidentiality introduces an ethical dilemma faced by physicians in the UK (ABC VRS St George’s Healthcare NHS Trust).31 According to the Joint Committee on Genomics in Medicine (JCGM), the avoidance of harm inflicted on the daughter of a man with Huntington’s disease exceeds the patients’ claim to confidentiality, thus physicians’ duty to forewarn at-risk family members about an inherited disease may be justified.31,32 However, to better respect patients’ autonomy and avoid ethical dilemma physicians can inform patients prior to testing that under certain circumstances, which should be mentioned in an informed consent, they would disclose relevant genetic information to family members if the patient refuses to do so. Participants, however, indicated that they would encourage patients about the importance of sharing genetic information with other family members who might get the most benefits once genetic information is shared with them. Similar findings were reported by Lacroix et al40.
Fifth Question
PGt testing can help physicians provide their patients with information about their probability to respond to a common medication such as simvastatin, besides, their increased risk of Alzheimer’s disease, which is associated with polymorphism of apolipoprotein E4. Despite the fact that PGt discipline is in the beginning stage, given the currently available data on PGt, it seems that physicians strongly perceived its clinical utility and felt ethically obligated to let patients know about their future risk of such diseases. According to geneticists, there are several disease-related factors such as the type of the diseases and disease severity that could influence physicians' treatment decision-making.4 A lack of practice models and advanced informatics tools for handling and prioritizing incidental findings and returning them to patients, could further delay the clinical acceptance of PGt testing.33 A qualitative study showed that the majority of patients expressed an imperative need to participate in decision-making about receiving incidental findings.34 As incidental findings pose additional concerns for health-care providers and patients, reporting incidental findings for genetic variants of identified clinical significance during subsequent analysis of the resulting genetic data, could help mitigate these concerns and identify which incidental findings should be further assessed.35 The American College of Medical Genetics and Genomics (ACMG) developed recommendations for reporting secondary findings whenever considered clinically meaningful or actionable, for example, the association of malignant hyperthermia with pathologic variants of RYR1 gene.36
As a general agreement, medically actionable incidental findings should be offered to patients or routinely reported to them; however, more studies are needed to support physicians’ ethical decision-making and promote evidence-based practices regarding the extent of disclosing different incidental information.37 Further, several studies identified several approached that could guide physicians through different ethical issues regarding PGt testing results such as (a) strict confidentiality to maintain patient confidentiality and autonomy and not sharing any information without their consent; (b) duty to warn, where physicians can disclose genetic information to patients and relatives as long as there is no clear policies that imply otherwise; (c) informed consent, which would maintain patients’ autonomy by inform them prior to testing about different circumstances that may result in disclosing of genetic information.38–40
Study Limitations
Some limitations of this study should be mentioned. Analysis of different ethical issues was limited to only those physicians who work in one large tertiary hospital in Jordan. The lack of actual implementation of PGt testing by the majority of hospital departments may influence physicians self-reporting of their ethical obligations. Participants in this study may have other means of communicating information to patients and their families due to sharing similar norms and values with the society. Also, underrepresentation in some medical specialties was a limitation.
Conclusion
In an early step towards searching for complex ethical issues associated with the use of PGt testing in clinical practice, this study carefully assessed physicians’ attitudes towards PGt testing and their views regarding the relevance of ancillary genetic information, the need for disclosing genetic information, the importance of consent forms, and circumstances that may permit confidentiality breaches to prevent harm to other at-risk individuals. Consequently, this study provided a significant insight into physicians’ ethical obligations pertaining to the implementation of PGt testing that may influence its rate of acceptance in routine clinical settings. However, transforming PGt testing into clinical practices should be accompanied with increased knowledge and more optimistic attitudes among physicians to help them establish ethical standards and practice guidelines needed to emphasize their role testing and facilitate the implementation of PGt tests. Future research should be focused on recruiting a larger number of different physicians working in medical settings in which PGt testing is available to better measure their ethical obligations regarding PGt testing. Furthermore, if ethical issues pertinent to PGt testing continue to limit the number of patients who might benefit from such testing, the drug companies might become less interested in incorporating PGt testing results into drug production and development, which might impose considerable ethical concern to society.
Acknowledgment
This project was carried out as part of “The Research Ethics Education Program in Jordan” and has been supported by NIH (grant number 1R25TW010026-01).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Franceschini N, Frick A, Kopp JB. Genetic testing in clinical settings. Am J Kidney Dis. 2018;72(4):569–581. doi:10.1053/j.ajkd.2018.02.351
2. Kalow W. Pharmacogenetics and pharmacogenomics: origin, status, and the hope for personalized medicine. Pharmacogenomics J. 2006;6(3):162–165. doi:10.1038/sj.tpj.6500361
3. Offit K, Groeger E, Turner S, Wadsworth E, Weiser M. The “duty to warn” a patient’s family members about hereditary disease risks. Am J Ophthalmol. 2005;139(3):584. doi:10.1016/j.ajo.2004.12.097
4. Haga SB, Burke W, Ginsburg GS, Mills R, Agans R. Primary care physicians’ knowledge of and experience with pharmacogenetic testing. Clin Genet. 2012;82(4):388–394. doi:10.1111/cge.2012.82.issue-4
5. Stavljenić-rukavina A. 1. Prenatal diagnosis of chromosomal disorders-molecular aspects. EJIFCC. 2008;19(1):2.
6. Lee CR, Goldstein JA, Pieper JA. Cytochrome P450 2C9 polymorphisms: a comprehensive review of the in-vitro and human data. Pharmacogenet Genomics. 2002;12(3):251–263. doi:10.1097/00008571-200204000-00010
7. Dugan RB, Wiesner GL, Juengst ET, O’riordan M, Matthews AL, Robin NH, editors. Duty to warn at‐risk relatives for genetic disease: genetic counselors’ clinical experience.
8. Haga SB, Mills R. A review of consent practices and perspectives for pharmacogenetic testing. Pharmacogenomics. 2016;17(14):1595–1605. doi:10.2217/pgs-2016-0039
9. US Food and Drug Administration. Table of pharmacogenomic biomarkers in drug labeling. Available from:https://www.fda.gov/drugs/science-and-research-drugs/table-pharmacogenomic-biomarkers-drug-labeling. Accessed February 16, 2020.
10. Dodson C. Diffusion of Innovation: Knowledge and Attitudes of Oncology Nurses Regarding Pharmacogenomic Testing. The University of North Carolina at Chapel Hill; 2012.
11. Roederer MW, Van Riper M, Valgus J, Knafl G, McLeod H. Knowledge, attitudes and education of pharmacists regarding pharmacogenetic testing. Per Med. 2012;9(1):19–27. doi:10.2217/pme.11.87
12. Rogausch A, Prause D, Schallenberg A, Brockmöller J, Himmel W. Patients’ and physicians’ perspectives on pharmacogenetic testing. Pharmacogenomics. 2006;7:49–59. doi:10.2217/14622416.7.1.49
13. Shaw K, Amstutz U, Carleton BC. Using pharmacogenetics to understand adverse drug reactions in children. Paediatr Child Health. 2011;16(9):537–538. doi:10.1093/pch/16.9.537
14. Taber KAJ, Dickinson BD. Pharmacogenomic knowledge gaps and educational resource needs among physicians in selected specialties. Pharmgenomics Pers Med. 2014;7:145. doi:10.2147/PGPM
15. Netzer C, Biller‐andorno N. Pharmacogenetic testing, informed consent and the problem of secondary information. Bioethics. 2004;18(4):344–360. doi:10.1111/j.1467-8519.2004.00401.x
16. Counselors N. Prenatal and Childhood testing for adult-onset disorders. Perspect Genet Couns. 1995;17:5.
17. Bioethics N. Pharmacogenetics: Ethical Issues: a Guide to the Report: Nuffield Council on Bioethics; 2003.
18. Pett MA, Lackey NR, Sullivan JJ. Making Sense of Factor Analysis: The Use of Factor Analysis for Instrument Development in Health Care Research. Sage; 2003.
19. Tabachnick BG, Fidell LS. Using Multivariate Statistics.
20. Corder EH, Saunders AM, Strittmatter WJ, et al. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science. 1993;261(5123):921–923. doi:10.1126/science.8346443
21. Dressler LG, Deal AM, Patel J, Markey J, Riper MV, McLeod HL. Cancer pharmacogenomics, adoption by oncologists and patient benefit. Per Med. 2014;11(2):143–153. doi:10.2217/pme.14.1
22. Walden LM, Brandl EJ, Changasi A, et al. Physicians’ opinions following pharmacogenetic testing for psychotropic medication. Psychiatry Res. 2015;229(3):913–918. doi:10.1016/j.psychres.2015.07.032
23. O’donnell PH, Danahey K, Jacobs M, et al., editors. Adoption of a clinical pharmacogenomics implementation program during outpatient care–initial results of the University of Chicago “1,200 Patients Project”.
24. Botkin JR, Belmont JW, Berg JS, et al. Points to consider: ethical, legal, and psychosocial implications of genetic testing in children and adolescents. Am J Hum Genet. 2015;97(1):6–21. doi:10.1016/j.ajhg.2015.05.022
25. Crews KR, Hicks JK, Pui CH, Relling MV, Evans WE. Pharmacogenomics and individualized medicine: translating science into practice. Clin Pharmacol Ther. 2012;92(4):467–475.
26. Middleton A, Milne R, Howard H, et al. Members of the public in the USA, UK, Canada and Australia expressing genetic exceptionalism say they are more willing to donate genomic data. Eur J Hum Genet. 2019;1–11.
27. Robertson JA. Consent and privacy in pharmacogenetic testing. Nat Genet. 2001;28(3):207. doi:10.1038/90032
28. Directors A. Clinical utility of genetic and genomic services: a position statement of the American College of Medical Genetics and Genomics. Genet Med. 2015;17(6):505.
29. Haga SB, Burke W. Pharmacogenetic testing: not as simple as it seems. Genet Med. 2008;10(6):391. doi:10.1097/GIM.0b013e31817701d4
30. Dheensa S, Fenwick A, Shkedi-rafid S, Crawford G, Lucassen A. Health-care professionals’ responsibility to patients’ relatives in genetic medicine: a systematic review and synthesis of empirical research. Genet Med. 2016;18(4):290–301. doi:10.1038/gim.2015.72
31. Middleton A, Milne R, Robarts L, Roberts J, Patch C. Should doctors have a legal duty to warn relatives of their genetic risks? Lancet. 2019;394(10215):2133–2135. doi:10.1016/S0140-6736(19)32941-1
32. Royal College of Physicians RCoP, British Society for, Medicine, G. Consent and confidentiality in genomic medicine. Guidance on the use of genetic and genomic information in the clinic. A report of the Joint Committee on Genomics in Medicine; 2019. Available from: https://www.rcplondon.ac.uk/projects/outputs/consentand-confidentiality-genomic-medicine.
33. Brothers KB, Langanke M, Erdmann P. Implications of the incidentalome for clinical pharmacogenomics. Pharmacogenomics. 2013;14(11):1353–1362. doi:10.2217/pgs.13.119
34. Clift KE, Halverson CM, Fiksdal AS, Kumbamu A, Sharp RR, McCormick JB. Patients’ views on incidental findings from clinical exome sequencing. App Trans Genom. 2015;4:38–43. doi:10.1016/j.atg.2015.02.005
35. Hippman C, Nislow C. Pharmacogenomic testing: clinical evidence and implementation challenges. J Pers Med. 2019;9(3):40. doi:10.3390/jpm9030040
36. Green RC, Berg JS, Grody WW, et al. ACMG recommendations for reporting of incidental findings in clinical exome and genome sequencing. Genet Med. 2013;15(7):565–574. doi:10.1038/gim.2013.73
37. Lohn Z, Adam S, Birch P, Friedman J. Incidental findings from clinical genome-wide sequencing: a review. J Genet Couns. 2014;23(4):463–473. doi:10.1007/s10897-013-9604-4
38. Statement A. Professional disclosure of familial genetic information. The American Society of Human Genetics Social Issues Subcommittee on Familial Disclosure. Am J Hum Genet. 1998;62:474–483.
39. Godard B, Hurlimann T, Letendre M, Egalite N. Guidelines for disclosing genetic information to family members: from development to use. Fam Cancer. 2006;5(1):103–116. doi:10.1007/s10689-005-2581-5
40. Lacroix M, Godard B, Knoppers BM. Warning Patients’ Relatives of Genetic Risks: Policy Approaches; 2005.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
