Back to Journals » International Journal of Nanomedicine » Volume 15
Pharmacology and Pharmacokinetics of Vitamin E: Nanoformulations to Enhance Bioavailability
Authors Mohd Zaffarin AS , Ng SF , Ng MH , Hassan H , Alias E
Received 10 August 2020
Accepted for publication 12 October 2020
Published 8 December 2020 Volume 2020:15 Pages 9961—9974
DOI https://doi.org/10.2147/IJN.S276355
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Phong A Tran
Video abstract presented by Anis Syauqina Mohd Zaffarin and Ekram Alias.
Views: 869
Anis Syauqina Mohd Zaffarin,1 Shiow-Fern Ng,2 Min Hwei Ng,3 Haniza Hassan,4 Ekram Alias1
1Department of Biochemistry, Faculty of Medicine, Universiti Kebangsaan Malaysia, Kuala Lumpur 56000, Malaysia; 2Faculty of Pharmacy, Universiti Kebangsaan Malaysia, Kuala Lumpur 50300, Malaysia; 3Centre for Tissue Engineering and Regenerative Medicine, Universiti Kebangsaan Malaysia, Kuala Lumpur, 56000, Malaysia; 4Department of Human Anatomy, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, Serdang 43400, Selangor, Malaysia
Correspondence: Ekram Alias
Department of Biochemistry, Faculty of Medicine, Universiti Kebangsaan Malaysia, Jalan Yaacob Latif, Bandar Tun Razak, W.P., Kuala Lumpur 56000, Malaysia
Tel +60 3 9145 9559
Fax +60 3 9145 9546
Email [email protected]
Haniza Hassan
Department of Human Anatomy, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, Serdang, Selangor 43400, Malaysia
Email [email protected]
Abstract: Vitamin E belongs to the family of lipid-soluble vitamins and can be divided into two groups, tocopherols and tocotrienols, with four isomers (alpha, beta, gamma and delta). Although vitamin E is widely known as a potent antioxidant, studies have also revealed that vitamin E possesses anti-inflammatory properties. These crucial properties of vitamin E are beneficial in various aspects of health, especially in neuroprotection and cardiovascular, skin and bone health. However, the poor bioavailability of vitamin E, especially tocotrienols, remains a great limitation for clinical applications. Recently, nanoformulations that include nanovesicles, solid-lipid nanoparticles, nanostructured lipid carriers, nanoemulsions, and polymeric nanoparticles have shown promising outcomes in improving the efficacy and bioavailability of vitamin E. This review focuses on the pharmacological properties and pharmacokinetics of vitamin E and current advances in vitamin E nanoformulations for future clinical applications. The limitations and future recommendations are also discussed in this review.
Keywords: tocopherols, tocotrienols, nanoparticles, drug delivery, bioavailability
Introduction
Vitamin E was first discovered in 1922 in green leafy vegetables and was thought to be an important nutrient for human reproduction.1 It was then proven to be effective in preventing lipid peroxidation and scavenging free radicals; hence, it is recognised as a potent antioxidant.2,3 Since then, many studies have been performed to explore other therapeutic effects of vitamin E and its pharmacokinetics.4
Vitamin E is a family of lipid-soluble compounds consisting of two major groups, namely, tocopherols and tocotrienols. Each group can be further divided into four different isomers, that is, alpha, beta, gamma, and delta, depending on the presence and position of the methyl group(s) as the side chain. Generally, vitamin E is made up of a chromanol ring and an isoprenoid or phytyl side chain. Tocopherols have a long and saturated side chain, while tocotrienols differ from tocopherols by the presence of unsaturated double bonds on the side chain (Figure 1). This also explains the higher affinity of tocotrienols to the lipid membrane compared to tocopherols.5
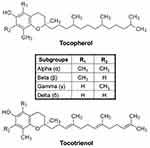 |
Figure 1 Chemical structures of tocopherol and tocotrienol. The different vitamin E isoforms are determined based on the presence and position of methyl group(s) as side chains on the chromanol ring. |
Vitamin E, particularly α-tocopherol, can easily be obtained and consumed by the vast majority of the world’s population. It is ubiquitously found in various staple foods, such as vegetable oils, palm oil, rice bran, wheat germ, olive, barley, soybean, nuts and grains. Meanwhile, tocotrienols are also found in many different natural sources. Similar to tocopherols, the percentages of tocotrienol content are different from one source to another, ie, rice bran (50%), palm oil (75%) and annatto (99.9%).6
Both tocopherols and tocotrienols are well known for their potent antioxidant properties. They are capable of scavenging reactive oxygen species (ROS) and free radicals. The presence of hydroxyl group on the chromanol ring contributes to this antioxidant activity by readily donating the hydrogen atom to reduce free radicals.7 In addition, vitamin E also possesses anti-inflammatory effects apart from its antioxidant properties.8 These properties of vitamin E have attracted great interest among researchers to investigate its therapeutic potential for the prevention and treatment of various diseases.
Despite a rising number of studies on tocotrienols, their application in the field of nutraceutical therapy is still limited due to poor oral bioavailability. A previous study reported that the absolute bioavailability of tocotrienols in rats administered with the 50% tocotrienol-rich fraction (TRF) was relatively low for γ-, δ- and α-tocotrienols (9.1%, 8.5% and 27.7%, respectively).9 Therefore, this review aimed to discuss the pharmacological and pharmacokinetic properties of vitamin E, as well as the development of potential nanoformulation strategies for improved delivery and bioavailability of vitamin E. The limitations and recommendations for future studies will also be discussed in this review.
Vitamin E
Pharmacodynamics: Vitamin E is Much More Than Just an Antioxidant
Vitamin E has gained researchers’ attention as a potential adjuvant therapy for various disorders due to its excellent antioxidant and anti-inflammatory properties.10 Inflammation is a result of the enhanced body immune response and is characterised by the increased production of free radicals and pro-inflammatory mediators, which may lead to excessive damage to host tissues.11 Its occurrence is indicated by the presence of markers such as cytokines and C-reactive protein (CRP). Furthermore, the secretion of substances such as eicosanoids and pro-inflammatory cytokines is damaging, as they not only result in inflammation but may subsequently trigger tumour growth and invasiveness, leading to cancer.12 With regard to the anti-inflammatory properties of vitamin E, several studies have demonstrated that vitamin E could inhibit the secretion of inflammation-mediating molecules such as eicosanoids and cyclooxygenase-2 enzyme (COX-2).13,14 Vitamin E also suppresses pro-inflammatory signalling pathways, such as nuclear factor kappa beta (NF-kβ) and signal transducer and activator of transcription 3 (STAT-3)-mediated pathways.15
The antioxidant and anti-inflammatory properties of vitamin E are highly beneficial for future therapeutic use. A number of studies have been performed to demonstrate the benefits of vitamin E in various aspects of health, including neuroprotection and cardiovascular, skin and bone health, as well as for treating cancers (Figure 2A). Different routes of administration for vitamin E may be applied for different therapeutic uses (Figure 2B).
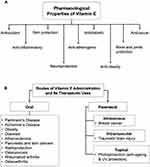 |
Figure 2 (A) Various pharmacological properties of vitamin E. (B) The different routes of vitamin E administration and its therapeutic uses. |
Vitamin E possesses neuroprotective effects, and this has been proven in clinical trials and models of various neurological disorders, including traumatic brain injury (TBI), Alzheimer’s disease (AD), and Parkinson’s disease (PD). A randomised double-blind controlled trial involving adults with severe head injury (n=100) reported that intramuscular injection of vitamin E (400 IU/day) had a significantly lower mortality rate compared to the vitamin C and placebo-treated groups.16 An in vivo study involving albino rats subjected to TBI showed low malondialdehyde (MDA) levels and mortality rates when administered α-tocopherol (alone or in combination with vitamin C) compared to the non-TBI group.17
In animal models of AD, vitamin E was found to reduce the progression of the disease by inhibiting p38 signalling pathway activation, which prevents amyloid beta (Aβ)-induced tau hyperphosphorylation.18 On the other hand, in another study using a transgenic mouse model of AD, it was demonstrated that vitamin E supplementation (200 mg/kg) significantly reduced the high level of DNA damage. Together, these results suggest that vitamin E could be employed as an alternative treatment for AD.19 Meanwhile, in the context of PD, a two-population-based cohort study reported that dietary intake of vitamin E and β-carotene was associated with a low risk of the disease.20
Vitamin E, especially tocotrienols, has also been studied thoroughly for its potential in preventing metabolic syndrome and cardiovascular diseases (CVDs).21,22 These effects are mediated through its antioxidant mechanisms, anti-inflammatory effects, inhibition of the HMG-CoA reductase enzyme and reduced expression of adhesion molecules.
Studies using obese animal models indicated that vitamin E may act as a potential anti-obesity agent.23,24 Supplementation with γ-tocotrienol (50 mg/kg) significantly reversed the increased fasting blood glucose, insulin and fat content, body weight, liver weight and secretion of pro-inflammatory cytokines in high-fat diet-induced obese mice.24 A study using diabetic rats reported that TRF supplementation (200 mg/kg/day) reduced fasting blood glucose and oxidative stress, as well as improved vascular walls.25 Meanwhile, vitamin E supplementation (100 mg/kg) in an animal model of atherosclerosis also attenuated atherosclerotic lesions, although this was only seen in the early stages.26
In addition, vitamin E is also widely used in the field of dermatology for its antioxidant and ultraviolet (UV) radiation protective effects, which provide photoprotection and delay skin ageing.14 A study performed on human gluteal skin showed that pre-treatment with a newly formulated vitamin E topical agent (10% tocopherols and 0.3% tocotrienols) resulted in improved photoprotection against minimal UVB radiation.27 Vitamin E also exhibited anti-ageing effects through its ability to enhance collagen synthesis and prevent collagen degradation.28 In addition, supplementation with palm TRF (500 µg/mL) was also shown to suppress excessive melanin production in primary human skin melanocytes in vitro.29 This is due to the increased expression of endosome docking/fusion proteins, which contributes to the degradation of melanosomes by docking to lysosomes.30
Vitamin E has been proven to provide excellent skin protection when used in combination with other antioxidants. A single-blind controlled trial by Mireles-Rocha et al involving 45 healthy volunteers reported that combination treatment of vitamin C and E provided the best protective effect against UVB radiation compared to individual treatments.31 In human keratinocytes, it was shown that single or combination treatment of α-tocopherol with coenzyme Q10, krill oil, lipoic acid, resveratrol, grape seed oil and selenium reduced ROS generation and cytokine secretion, which may contribute to the photoprotective effect.32
With regard to its potential application for cancer treatments, vitamin E possesses promising anti-cancer properties such as suppressing cancer cell proliferation, preventing angiogenesis, modulating growth factors, promoting cell cycle arrest, and inducing apoptosis.33 These special properties of tocotrienols have been reported in various in vitro studies using different cell lines (breast, lung, liver, pancreas, skin, and bladder cancers).34,35 Several in vivo studies have also shown that tocotrienols successfully accumulate in cancer cells but not in normal tissues.36,37 Previous studies demonstrated that δ-tocotrienol (200 mg/kg) significantly enhanced the efficacy of gemcitabine to inhibit pancreatic cancer growth and survival in vitro and in vivo by suppressing NF-kB activity.37,38 Another study on mouse melanoma xenografts showed that δ-tocotrienol treatment (100 mg/kg) significantly reduced the tumour volume and delayed tumour progression.39
Vitamin E, especially tocotrienols, is beneficial in improving bone strength by inducing bone formation and osteoblast (bone-forming cells) activity, as well as suppressing osteoclasts (cells mediating the bone resorption process).40,41 A study using a combined therapy of annatto-derived tocotrienols (60 mg/kg) and lovastatin (750 µg/kg) in an animal model of post-menopausal osteoporosis showed that the treatment caused increased osteoblast surface, decreased osteoclast surface, reduced eroded surface, and higher bone volume compared to the untreated group.42,43 Another study using the same animal model demonstrated that TRF supplementation (60 mg/kg) reduced lipid peroxidation during early fracture healing through a reduction in the levels of free radicals responsible for the delayed healing process.44
Tocotrienols also greatly benefit inflammation-mediated bone loss, such as rheumatoid arthritis (RA) and osteoarthritis (OA). A recent study by Zainal et al using an animal model of RA reported that oral TRF supplementation (30 mg/kg) was able to reduce the joint inflammation seen at the paws and reduced the plasma levels of pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) and CRP in the treated rats compared to the untreated controls.45 In an animal model of OA, it was shown that supplementation with annatto-derived tocotrienol (100 mg/kg) prevented cartilage degradation, which may prevent the progression of OA.46
Bioavailability and Pharmacokinetics of Vitamin E: Recent Progress and Challenges
Although the plethora of effects of vitamin E are promising for future therapeutic use, its application is greatly hindered due to its poor oral bioavailability, especially for tocotrienols. Bioavailability, as defined by the Food and Drug Administration (FDA), is the rate and extent that a drug or active pharmaceutical compound is absorbed and becomes available at the site of action. The pharmacokinetic limitations of tocotrienols result from a variety of factors, including their solubility, absorption, distribution, and rate of elimination.
Similar to other lipid-soluble vitamins, vitamin E absorption is highly dependent on dietary fat in the body, bile salts, and pancreatic enzymes. During oral administration, vitamin E is packed into chylomicrons (a large triglyceride-rich lipoprotein produced from lipids, mainly fatty acids and cholesterol) in the intestines and enters the circulation via lymph nodes. Once vitamin E is packed into chylomicrons, it can be sent either to tissues or the liver. The hydrolysis of chylomicrons by lipoprotein lipase enzymes in the circulation allows the transfer of lipids and vitamin E to tissues.47 The chylomicron remnants will also be taken up by the liver. Most of the vitamin E in chylomicron remnants will be incorporated with very-low-density lipoproteins (VLDL), while the rest will be excreted in bile. Vitamin E is then metabolised through the hydroxylation and oxidation of its side chains by cytochrome P450. The final metabolites, which are carboxyethyl hydroxychromans (CEHC) and carboxymethylbutyl hydroxychromans (CMBHC), will then be excreted in urine and faeces.48,49
In a previous study involving healthy human volunteers, Yap et al showed that the 24-hour area under the curve (AUC0-∞) of TRF was increased by approximately 2-fold in the fed state compared to the fasting state.50 A shorter time to reach peak plasma concentration and a higher concentration of TRF recorded by the volunteers in the fed state indicated that food consumption increased the absorption of vitamin E, thus improving its bioavailability.50 More recent studies have shown that the time taken to achieve the peak plasma concentration (Tmax) of tocotrienols via oral supplementation was between 3 and 4 hours after a meal, unlike tocopherol, which achieved its Tmax at approximately 6 hours post-meal.51,52 The findings of these studies also indicated that α-tocopherol had a much higher peak plasma concentration (Cmax) than tocotrienols (1.82–2.92 µM and 0.89–1.92 µM, respectively). The elimination half-life (t1/2) of α-tocopherol in humans was reported to be approximately 20 hours;6 however, for different tocotrienol isomers, the t1/2 was between 2.3 and 4.4 hours.53 Thus, the frequency of tocotrienol supplementation is normally recommended as twice daily to maintain its bioactive levels.
Apart from oral administration, vitamin E can also be administered parenterally through intravenous, subcutaneous, intramuscular, and intraperitoneal injection. Parenteral administration of tocotrienols provides a much faster rate of absorption than oral administration. Comparison between studies showed that the oral administration of TRF (300 mg) provides a Tmax value within 3–4 hours,50 while a single subcutaneous injection of tocotrienol (δ-tocotrienol at a dose of 300 mg/kg) exhibited a Tmax value at 1 hour post-injection.54 Intramuscular and intraperitoneal injections of tocotrienols in rats resulted in lower plasma concentrations when compared with oral administration,9 suggesting minimal absorption of tocotrienol following these routes of administration. It is known that intramuscular and intraperitoneal injections do not involve the formation of mixed micelles for enhanced absorption, unlike the oral route.
Compared to the other lipid-soluble vitamins, vitamin E was shown to be more evenly distributed in the body, especially in the plasma, liver, and adipose tissues.55,56 In an in vivo study, a group of mice treated with a tocopherol-stripped diet (AIN76m) containing 0.05% γ-tocotrienol demonstrated that tocotrienol and its metabolites could also be detected in other organs, including the lung, spleen, and colon.49 In another study, tocotrienols were found to be preferably absorbed by adipose tissues in rats; however, this uptake was attenuated in the presence of tocopherol.57 In the presence of tocopherol, tocotrienols compete to bind to alpha-tocopherol transfer protein (α-TTP), the main vitamin E transport protein in the circulation. α-TTP is widely known to have a higher affinity towards α-tocopherol than the other isoforms of vitamin E. However, another study by Khanna et al discovered that oral supplementation with α-tocotrienol (5 mg/kg) led to successful transport to several vital organs.58 This suggests that the distribution of tocotrienols can still be achieved by a different mechanism other than α-TTP, albeit to a lesser extent.
In the last decade, a number of strategies have been proposed and carried out with the intention of increasing the bioavailability of vitamin E. One of the strategies was through adjusting the composition of tocotrienol isomers in a fraction. In a previous study using human subjects, supplementation with the tocotrienol fraction containing solely γ and δ isomers (75% and 25%, respectively) resulted in superior bioavailability of γ- but not δ-tocotrienol isomers when compared with conventional TRF.59 The higher bioavailability of γ-tocotrienol isomer in subjects taking the γ- and δ-only-containing tocotrienols was represented by higher AUC0-∞ values of 41,091.37 compared to 24,256.29 µg/L*h in the conventional TRF-supplemented group.
Other studies have also explored the complexation of vitamin E emulsions with cyclodextrin, a cyclic oligosaccharide, as a strategy to increase its bioavailability by improving its solubility.60,61 Cyclodextrin is an FDA-approved substance commonly employed as a solubilising agent for various drugs. In a study by Ikeda et al, it was found that rats administered 72.8 mg of γ-tocotrienol/β-cyclodextrin (γ-T3/β-CD) complex had higher plasma concentrations of tocotrienols when compared with those administered 13.9 mg of tocotrienol alone or a combination of 13.9 mg of tocotrienol and 62.2 mg of β-CD (Cmax values of 0.09, 0.05 and 0.04 nmol/mL, respectively).60 Similarly, another study that explored the potential of cyclodextrin in improving the bioavailability of tocotrienols showed that mice treated with the TRF/γ-cyclodextrin (TRF/γ-CD) complex (14.5 mg diluted in 200 µL of corn oil) had a higher plasma concentration of tocotrienols (Cmax value of 11.4) than the TRF-treated control group (2.79 mg diluted in 200 µL of corn oil), with a Cmax value of 7.9 µM.61 There was also an approximately 1.4-fold increase in the AUC0-∞ of the TRF/γ-CD complex compared to TRF alone. Taken together, the complexation of cyclodextrin with vitamin E could improve the bioavailability due to the enhancement in solubility of the formulation. However, this approach possessed some limitations, such as possibility that the formulation being unstable after one hour post-preparation,60 as well as the suitability of cyclodextrin as a solubilising agent for vitamin E. The glycaemic index of cyclodextrin should be taken into consideration since it is a form of carbohydrate residue, which may elevate the blood sugar level, especially in patients with diabetes.
Another strategy that can be proposed to improve the bioavailability of tocotrienols is through the application of nanoformulations. Nanoformulations offer many advantages, including improved efficacy of active pharmaceutical ingredients, biocompatibility, reduced toxicity and enhanced bioavailability due to the small size and large surface area.62 Compared to the other approaches mentioned above, nanoformulations appear to offer a wider range of strategies to be explored considering that nanoformulations appear in many forms.
Nanoformulations and Applications
Nanoformulations have been widely studied for the application of drug delivery. These involve the use of nanomaterials with sizes ranging between 1 and 100 nm. Due to their small size and large surface area, nanoparticle-incorporated compounds are superior in terms of their solubility, efficacy, safety, and pharmacokinetics.63 The application of nanoformulations for the delivery of lipophilic drugs and/or active compounds offers several benefits, including protection from gastrointestinal degradation, prolonged systemic circulation, controlled drug release and improved absorption in the intestine.64 These in turn improve the bioavailability and enhance the efficacy of administered drugs or active pharmaceutical compounds.
To overcome the poor bioavailability of vitamin E, different nanoformulation strategies have been used to address the issue for potential therapeutic applications.65 These strategies include loading vitamin E in nanovesicles, solid-lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs), nanoemulsions and polymeric nanoparticles (Figure 3). The findings from different studies involving vitamin E nanoformulations are summarised in Table 1.
 |
Table 1 Summary on the Findings for the Different Studies on Vitamin E Nanoformulation |
Lipid-Based Nanoparticle Systems – Niosomes, Liposomes, Solid-Lipid Nanoparticles, Nanostructured Lipid Carriers and Nanoemulsions
Niosomes and Liposomes
Niosomes and liposomes are lipid-based nanovesicles that are highly promising for the delivery of lipophilic drugs and active compounds. Niosomes are lamellar structures made up of non-ionic surfactants and cholesterol. On the other hand, liposomes are structures that consist of an aqueous core that is surrounded by one or more phospholipid bilayers. Although they have almost similar physical and chemical properties by comparison, nonetheless, niosomes are shown to have a higher permeability to small solutes and ions than liposomes.66 Similar to liposomes, the application of niosomes as a drug delivery vehicle is also suitable because they are non-toxic, less expensive and more stable.67
It is widely known that tocotrienols exhibit anti-cancer properties in vitro against many types of cancer including liver, lung, pancreatic, and breast cancers.38,68,69 Nonetheless, the tumour-suppressing effects of tocotrienols are not always observed in vivo, which could be explained by the poor bioavailability. To date, a limited number of studies have started to explore the potential use of nanovesicles in enhancing the anti-cancer properties of tocotrienols.65 However, research on the pharmacokinetic profile of this system, including its bioavailability and safety, has yet to be explored.
As part of a strategy to enhance the tumour-suppressing effect of tocotrienols in vivo, Fu et al first developed a D-α-tocopheryl polyethylene glycol 1000 succinate (TPGS)-based niosome (consisting of Span 60, TPGS, cholesterol, and dioleoylphosphatidylethanolamine (DOPE)) linked with the transferrin receptor to encapsulate TRF. TRF was entrapped in the vesicle via probe sonication before cross-linking with the transferrin receptor, followed by ultracentrifugation to remove the unentrapped TRF. This system appeared to significantly increase tocotrienol uptake in vitro (using A431, B16F10 and T98G cell lines) and hence improved the therapeutic efficacy compared to the unentrapped TRF.70 In a more recent study, Tan et al developed 6-O-palmitoyl-ascorbic acid (AP)-based niosomes (made up of AP, TPGS, cholesterol, and 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[carboxy(polyethylene glycol)-2000] (DSPE-PEG(2000)-carboxylic acid)) targeting transferrin receptors for intravenous administration of γ-tocotrienol aimed at treating breast cancer.71 The γ-tocotrienol-loaded niosomes in this study were prepared using a solvent evaporation method, where the solvents (hexane and methanol) were removed from the γ-tocotrienol mixture using a rotary evaporator under reduced pressure.71 Both in vitro and in vivo studies have proven that tumour-targeted niosomes significantly improve the therapeutic efficacy of γ-tocotrienol.71 The authors also reported that no adverse effects were observed after administration of γ-tocotrienol-loaded niosomes for 10 days, which indicates that the nanovesicle formulation is safe for therapeutic use. Thus, the aforementioned studies suggested that nanovesicles can be a suitable carrier for improved delivery and enhanced efficacy of vitamin E.
Solid-Lipid Nanoparticles and Nanostructured Lipid Carriers
SLNs and NLCs are alternatives for liposomes, as they use different lipid components that offer better drug-loading efficiency and are suitable for large-scale manufacturing processes.72 SLNs are made up of a lipid monolayer enclosing a hydrophobic solid-lipid core that permits incorporation of lipid-soluble compounds, while NLCs are known as second-generation SLNs. NLCs differ from SLNs in the composition of the lipid core, which is made up of amorphous solid and liquid oil matrix. The presence of this unique lipophilic core in both SLNs and NLCs makes them an ideal carrier for various lipid-soluble compounds, as it reduces substance mobility, thereby allowing controlled drug release. Both vitamin E-encapsulated SLNs and NLCs were prepared using the solvent-evaporation method, which usually involves high-shear homogenisation or hot high-pressure homogenisation techniques followed by ultrasonication to obtain a stable nanoparticle formulation.73–77 A previous study found that NLCs are superior to SLNs in terms of their stability and loading capacity.78
The use of SLNs has been proven to enhance the intestinal permeability of γ-tocotrienol, which subsequently led to increased oral bioavailability.79 SLNs containing γ-tocotrienol were found to increase the intestinal permeability in situ by approximately 10-fold compared to the control, which was prepared in micellar solution.79 The same study also reported that the SLN formulation (10 mg/kg) had higher Cmax and AUC0-∞ values (938 ng/mL and 12.1 µg/h/mL, respectively) than the micellar solution of the same dose (212 ng/mL and 3.9 µg/h/mL, respectively).
There was also evidence showing that NLCs also enhance the anti-cancer properties of tocotrienol. In a study assessing the anti-cancer effect on neoplastic +SA epithelial mammary cells, it was discovered that TRF-loaded NLCs (IC50: 2.12 ± 0.21 µM) have a more potent anti-cancer activity than free TRF (IC50: 2.73 ± 0.11 µM).76 Moreover, vitamin E-loaded SLNs and NLCs were also reported as safe since both formulations did not trigger skin irritation upon topical application.80,81 Taken together, these studies suggest that the use of SLN or NLC formulations is beneficial in improving the bioavailability and efficacy of tocotrienols.
Nanoemulsions
Nanoemulsions are a kinetically stable dispersion that contains two immiscible phases, aqueous and oil, in the presence of surfactants. The preparation methods generally involve high-energy processes, such as high-pressure homogenisation, microfluidisation, and phase inversion temperature (PIT) emulsification, for the production of stable nanoemulsions. Preparation of vitamin E using these methods produced more stable nanoparticles, as characterised by their low viscosity and long-term colloidal stability against creaming and sedimentation compared to conventional methods.82,83 Vitamin E nanoemulsions prepared using high-pressure homogenisation have been shown to produce smaller droplets (size between 80 and 300 nm), resulting in a faster rate of lipid digestion.83,84
Previous studies demonstrated that tocotrienols prepared in the form of a self-emulsifying drug delivery system (SEDDS) have improved oral bioavailability.85,86 Alqahtani et al successfully developed δ- and γ-tocotrienol SEDDS formulations (0.05, 0.25 and 2.5 mg/kg) and compared their oral bioavailability with the equivalent dosage of commercially available δ- and γ-tocotrienol soft gel capsules in rats.86 It was discovered that the groups treated with SEDDS formulations had a higher Cmax value (δ-tocotrienol: 192, 255 and 544 ng/mL; γ-tocotrienol: 395, 60 and 102 ng/mL) when compared to the soft gel capsules (δ-tocotrienol: 38, 69 and 460 ng/mL; γ-tocotrienol: 282, 47 and 71 ng/mL).86 The absolute bioavailability value determined from the AUC0-∞ of δ-tocotrienol SEDDS (0.5 mg/kg) was approximately seven-fold higher than that of the soft gel capsule.86 In a separate study, the absolute bioavailability of γ-tocotrienol SEDDS (10, 25, and 50 mg/kg) was two-fold higher compared to the commercial TRF administered with the same doses.87 Furthermore, an in vitro study also showed that SEDDS formulations have a significantly higher cellular uptake (regardless of the composition of tocotrienols in the formulation), indicating that the SEDDS formulation may enhance the intestinal absorption of tocotrienols.86
The efficacy of vitamin E is also improved when formulated as nanoemulsions. A previous study has shown that the administration of α-tocopherol nanoemulsion (30 mg/kg) in streptozotocin (STZ)-induced diabetic rats significantly reduced the level of the oxidative stress marker thiobarbituric acid reactive substance (TBARS) in the liver compared to those treated with blank nanoemulsion and control α-tocopherol micellar solution.88 Another study by Ledet et al investigated the efficacy of oral γ-tocotrienol nanoemulsion as a radioprotective agent.89 It was discovered that mice treated with γ-tocotrienol nanoemulsion (300 mg/kg) 24 hours prior to exposure to radiation had a higher survival rate compared to the mice treated with blank nanoemulsion and γ-tocotrienol alone (200 mg/kg and 400 mg/kg, respectively).89
Apart from oral delivery, vitamin E nanoemulsions have also been tested for transdermal delivery. For example, Pham et al developed a TRF nanoemulsion prototype to test the anti-cancer properties against human epidermoid carcinoma, A431, and human tongue squamous cell carcinoma, SCC-4, in vitro. It was reported that TRF nanoemulsion showed a dose-dependent and more prolonged anti-cancer effect compared to the cells treated with TRF in propylene glycol mixture (control).90 Previous in vitro and in vivo studies also confirmed that TRF prepared as nano- or microemulsions was safe for topical delivery, as there were no ocular or dermal irritations observed.91,92 Therefore, nanoemulsions may act as a potential vehicle for the safe and improved delivery of vitamin E.
Other Nanoformulations – Polymeric Nanoparticles
The synthesis of polymeric nanoparticles involves the incorporation of one or more polymer chains of varying hydrophobicity, which contributes to their ability to form self-assembled micelles in aqueous solutions. Polymeric nanoparticles are usually prepared by nanoprecipitation and solvent-evaporation methods.93–96 Briefly, the active compounds and polymers used are dissolved with water-immiscible organic solvents and mixed under constant stirring. Techniques such as probe sonication or microfluidisation are applied to further reduce the particle size (nanoparticle size between 10 and 170 nm) prior to the removal of organic solvents via evaporation.93–96 Examples of polymeric nanoparticles include poly(lactic-co-glycolic) acid (PLGA), polyethylene glycol (PEG), and chitosan. Previous studies have demonstrated that the use of polymers could offer many benefits, especially in improving the solubility and bioavailability of different lipophilic compounds, including vitamin E.
A study by Simon et al aimed to improve the oral bioavailability of α-tocopherol found that degradation of α-tocopherol in the gastric environment and intestinal conditions was prevented when α-tocopherol was incorporated into PLGA or a combination of PLGA and chitosan (PLGA/chitosan) nanoparticles.93 This subsequently led to a higher plasma bioavailability of α-tocopherol. Both PLGA and PLGA/chitosan nanoparticles containing α-tocopherol have higher Cmax and AUC0-∞ values (Cmax: 3.81 and 3.92 µg/mL, respectively; AUC0-∞: 99 and 80.9 µg/h/mL, respectively) than free α-tocopherol (Cmax: 2.91 µg/mL; AUC0-∞: 36.64 µg/mL).93 Incorporation of α-tocopherol into PLGA and PLGA/chitosan nanoparticles (at a dose of 1.5 mg/mL) also successfully improved the bioavailability by 170% and 121% compared to free α-tocopherol of the same dose.
Encapsulation of α-tocopherol and TRF into either PLGA or PLGA/chitosan nanoparticles also appeared to enhance their cellular uptake, antioxidant activity and anti-cancer activity.94 These nanoparticles were found to be non-toxic and did not induce apoptosis in Caco-2 cells, with over 95% average cell viability achieved.94 Interestingly, the same study also showed that the PLGA and PLGA/chitosan nanoparticles had antioxidant activity even in the absence of α-tocopherol and TRF. These findings suggested that the use of these polymers as a vehicle could further enhance the antioxidant properties of vitamin E.
Similar to NLC, the use of polymeric nanoparticles also enhanced the anti-cancer effect of tocotrienol, as demonstrated in previous studies. It was discovered that tocotrienol-loaded polymeric nanoparticles inhibited breast cancer cell growth in a concentration-dependent manner.94–96 Taken together, the findings from these studies may suggest that polymeric nanoparticles could be a good and highly promising vehicle to improve the delivery of vitamin E.
Challenges and Future Directions
There are some limitations and challenges for the clinical application of vitamin E nanoformulations. First, there is a limited number of studies reporting the bioavailability of vitamin E nanoformulations to date, despite the promising outcomes on its efficacy. There have also been very few clinical studies performed thus far on the bioavailability of vitamin E itself.50,97 The toxicity profiles for vitamin E nanoformulations were also not clearly outlined, and current knowledge only depends on animal behaviour and wellness from pre-clinical studies. Therefore, more in vivo and human studies are needed to provide important insights into various applications, the toxicity, and the pharmacokinetics of vitamin E nanoformulations.
This review mainly highlighted the potential application of vitamin E nanoformulations in cancer treatments. Given the various pharmacological properties of vitamin E, future studies should explore the potential application of vitamin E nanoformulations for other critical illnesses, such as heart diseases. As an antioxidant, vitamin E is very prone to oxidation in the presence of oxygen, light, and heat.98 Therefore, future studies on vitamin E nanoformulations should consider taking measures to minimise oxidation. In addition, it would be better if further tests on the total antioxidant capacity of each vitamin E nanoformulation were performed. These tests would ensure that the vitamin E nanoformulation does not lose its antioxidant properties during the development process.
Conclusion
In summary, vitamin E has been proven to have a wide range of therapeutic effects beyond its well-known antioxidant properties. Despite these promising effects, vitamin E, especially tocotrienol, is not well recognised for therapeutic interventions due to its poor bioavailability. In vivo studies have shown that the concentration of tocotrienols in plasma is lower in the presence of alpha-tocopherol due to the lower binding affinity towards α-TTP. Furthermore, tocotrienols have a relatively shorter t1/2 than tocopherols, which also contributes to poor bioavailability. In vitro and in vivo studies have demonstrated that nanoformulations improve the delivery and efficacy of vitamin E by enhancing its absorption, cellular uptake, solubility, and stability. These promising findings suggest that nanoformulations should be applied as carriers of vitamin E, particularly tocotrienols, to achieve better therapeutic applications.
Abbreviations
ROS, reactive oxygen species; TRF, tocotrienol-rich fraction; CRP, C-reactive protein; COX-2, cyclooxygenase-2 enzyme; NF-kB, nuclear factor kappa B; STAT-3, signal transducer and activator of transcription 3; TBI, traumatic brain injury; AD, Alzheimer’s disease; PD, Parkinson’s disease; MDA, malondialdehyde; Aβ, amyloid beta; CVDs, cardiovascular diseases; UV, ultraviolet; RA, rheumatoid arthritis; OA, osteoarthritis; FDA, Food and Drug Administration; VLDL, very-low-density lipoprotein; CEHC, carboxyethyl hydroxychroman; CMBHC, carboxymethylbutyl hydroxychroman; AUC0-∞, area under the curve; Tmax, time taken to achieve peak plasma concentration; Cmax, peak plasma concentration; t1/2, half-life; α-TTP, alpha-tocopherol transfer protein; TPGS, D-α-tocopheryl polyethylene glycol 1000 succinate; DOPE, dioleoylphosphatidylethanolamine; AP, 6-O-palmitoyl-ascorbic acid; DSPE-PEG(2000)-carboxylic acid, 1,2-distearoyl-sn-glycero-3-phosphoetanolamine-N-[carboxy(polyethylene glycol)-2000]; SLNs, solid-lipid nanoparticles; NLCs, nanostructured lipid carriers; PIT, phase inversion temperature; SEDDS, self-emulsifying drug delivery system; TBARS, thiobarbituric acid reactive substance; PLGA, poly(lactic-co-glycolic) acid; PEG, polyethylene glycol.
Acknowledgment
This work was supported by the Fundamental Research Grant Scheme [FRGS/1/2019/STG05/UKM/02/1] by the Ministry of Education Malaysia.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Evans HM, Bishop KS. On the existence of a hitherto unrecognized dietary factor essential for reproduction. Science. 1922;56(1458):650–651. doi:10.1126/science.56.1458.650
2. Esterbauer H, Dieber-Rotheneder M, Striegl G, Waeg G. Role of vitamin E in preventing the oxidation of low-density lipoprotein. Am J Clin Nutr. 1991;53(1):314S–321S. doi:10.1093/ajcn/53.1.314S
3. Tappel AL. Vitamin E as the biological lipid antioxidant. In: Harris RS, Wool IG, Marrian GF, Thimann KV, eds. Vitamins & Hormones. Vol 20: Academic Press; 1962:493–510.
4. Brigelius-Flohe R, Traber MG. Vitamin E: function and metabolism. FASEB J. 1999;13(10):1145–1155. doi:10.1096/fasebj.13.10.1145
5. Sen CK, Khanna S, Roy S. Tocotrienols: vitamin E beyond tocopherols. Life Sci. 2006;78(18):2088–2098. doi:10.1016/j.lfs.2005.12.001
6. Aggarwal BB, Sundaram C, Prasad S, Kannappan R. Tocotrienols, the vitamin E of the 21st century: its potential against cancer and other chronic diseases. Biochem Pharmacol. 2010;80(11):1613–1631. doi:10.1016/j.bcp.2010.07.043
7. Peh HY, Tan WSD, Liao W, Wong WSF. Vitamin E therapy beyond cancer: tocopherol versus tocotrienol. Pharmacol Ther. 2016;162:152–169.
8. Chin K-Y, Pang K-L, Soelaiman I-N. Tocotrienol and its role in chronic diseases. In: Gupta SC, Prasad S, Aggarwal BB, editors. Anti-Inflammatory Nutraceuticals and Chronic Diseases. Switzerland: Springer International Publishing; 2016:97–130.
9. Yap SP, Yuen KH, Lim AB. Influence of route of administration on the absorption and disposition of α,- γ- and δ-tocotrienols in rats. J Pharm Pharmacol. 2003;55(1):53–58. doi:10.1111/j.2042-7158.2003.tb02433.x
10. Wong SK, Chin K-Y, Hj Suhaimi F, Ahmad F, Ima-Nirwana S. Vitamin E as a potential interventional treatment for metabolic syndrome: evidence from animal and human studies. Front Pharmacol. 2017;8(444). doi:10.3389/fphar.2017.00444
11. Belardelli F. Role of interferons and other cytokines in the regulation of the immune response. APMIS. 1995;103(1‐6):161–179. doi:10.1111/j.1699-0463.1995.tb01092.x
12. Greene ER, Huang S, Serhan CN, Panigrahy D. Regulation of inflammation in cancer by eicosanoids. Prostaglandins Other Lipid Mediat. 2011;96(1–4):27–36. doi:10.1016/j.prostaglandins.2011.08.004
13. Jiang Q. Natural forms of vitamin E: metabolism, antioxidant and anti-inflammatory activities and the role in disease prevention and therapy. Free Radic Biol Med. 2014;72:76–90. doi:10.1016/j.freeradbiomed.2014.03.035
14. Kanchi MM, Shanmugam MK, Rane G, Sethi G, Kumar AP. Tocotrienols: the unsaturated sidekick shifting new paradigms in vitamin E therapeutics. Drug Discov Today. 2017;22(12):1765–1781. doi:10.1016/j.drudis.2017.08.001
15. Zingg J-M. Vitamin E: a role in signal transduction. Annu Rev Nutr. 2015;35(1):135–173. doi:10.1146/annurev-nutr-071714-034347
16. Razmkon A, Sadidi A, Sherafat-Kazemzadeh E, et al. Administration of vitamin C and vitamin E in severe head injury: a randomized double-blind controlled trial. Neurosurgery. 2011;58(CN_suppl_1):133–137. doi:10.1227/NEU.0b013e3182279a8f
17. Ishaq GM, Saidu Y, Bilbis LS, Muhammad SA, Jinjir N, Shehu BB. Effects of α‑tocopherol and ascorbic acid in the severity and management of traumatic brain injury in albino rats. J Neurosci Rural Prac. 2013;04(03):292–297. doi:10.4103/0976-3147.118784
18. Giraldo E, Lloret A, Fuchsberger T, Viña J. Aβ and tau toxicities in Alzheimer’s are linked via oxidative stress-induced p38 activation: protective role of vitamin E. Redox Biol. 2014;2:873–877. doi:10.1016/j.redox.2014.03.002
19. Damanhuri H, Rahim NA, Nasri W, et al. Tocotrienol-rich fraction supplementation modulates antioxidant enzymes activity and reduces DNA damage in APPswe/PS1dE9 Alzheimer’s disease mouse model. Sains Malays. 2016;45:1363–1370.
20. Yang F, Wolk A, Håkansson N, Pedersen NL, Wirdefeldt K. Dietary antioxidants and risk of Parkinson’s disease in two population-based cohorts. Mov Disord. 2017;32(11):1631–1636. doi:10.1002/mds.27120
21. Ahsan H, Ahad A, Iqbal J, Siddiqui WA. Pharmacological potential of tocotrienols: a review. Nutr Metab. 2014;11:22. doi:10.1186/1743-7075-11-52
22. Prasad K. Tocotrienols and cardiovascular health. Curr Pharm Des. 2011;17(21):2147–2154. doi:10.2174/138161211796957418
23. Alcalá M, Sánchez-Vera I, Sevillano J, et al. Vitamin E reduces adipose tissue fibrosis, inflammation, and oxidative stress and improves metabolic profile in obesity. Obesity. 2015;23(8):1598–1606. doi:10.1002/oby.21135
24. Zhao L, Kang I, Fang X, et al. Gamma-tocotrienol attenuates high-fat diet-induced obesity and insulin resistance by inhibiting adipose inflammation and M1 macrophage recruitment. Int J Obes. 2015;39(3):438–446. doi:10.1038/ijo.2014.124
25. Budin SB, Othman F, Louis SR, Bakar MA, Das S, Mohamed J. The effects of palm oil tocotrienol-rich fraction supplementation on biochemical parameters, oxidative stress and the vascular wall of streptozotocin-induced diabetic rats. Clinics. 2009;64(3):235–244. doi:10.1590/S1807-59322009000300015
26. Tang F, Lu M, Zhang S, et al. Vitamin E conditionally inhibits atherosclerosis in ApoE knockout mice by anti-oxidation and regulation of vasculature gene expressions. Lipids. 2014;49(12):1215–1223. doi:10.1007/s11745-014-3962-z
27. Pedrelli VF, Lauriola MM, Pigatto PD. Clinical evaluation of photoprotective effect by a topical antioxidants combination (tocopherols and tocotrienols). J Eur Acad Dermatol Venereol. 2012;26(11):1449–1453. doi:10.1111/j.1468-3083.2011.04219.x
28. Makpol S, Jam FA, Khor SC, Ismail Z, Mohd Yusof YA, Wan Ngah WZ. Comparative effects of biodynes, tocotrienol-rich fraction, and tocopherol in enhancing collagen synthesis and inhibiting collagen degradation in stress-induced premature senescence model of human diploid fibroblasts. Oxid Med Cell Longev. 2013;2013:1–8. doi:10.1155/2013/298574
29. Makpol S, Mohd Arifin NN, Ismail Z, Chua KH, Mohd Yusof YA, Wan Ngah WZ. Modulation of melanin synthesis and its gene expression in skin melanocytes by palm tocotrienol rich fraction. Afr J Biochem Res. 2009;3(12):385–392.
30. Choi B, Heo JH, Kwon HJ, Lee ES, Sohn S. Tocotrienols enhance melanosome degradation through endosome docking/fusion proteins in B16F10 melanoma cells. Food Funct. 2013;4(10):1481–1488. doi:10.1039/c3fo60289c
31. Mireles-Rocha H, Galindo I, Huerta M, Trujillohernández B, Cortés-Franco R. UVB photoprotection with antioxidants: effects of oral therapy with d-α-tocopherol and ascorbic acid on the minimal erythema dose. Acta Derm Venereol. 2002;82(1):21–24.
32. Fasano E, Serini S, Mondella N, et al. Antioxidant and anti-inflammatory effects of selected natural compounds contained in a dietary supplement on two human immortalized keratinocyte lines. Biomed Res Int. 2014;2014.
33. Abraham A, Kattoor AJ, Saldeen T, Mehta JL. Vitamin E and its anticancer effects. Crit Rev Food Sci Nutr. 2019;59(17):2831–2838. doi:10.1080/10408398.2018.1474169
34. Jiang Q. Natural forms of vitamin E as effective agents for cancer prevention and therapy. Adv Nutr. 2017;8(6):850–867. doi:10.3945/an.117.016329
35. Wong SK, Kamisah Y, Mohamed N, et al. Potential role of tocotrienols on non-communicable diseases: a review of current evidence. Nutrients. 2020;12(1):259.
36. Hiura Y, Tachibana H, Arakawa R, et al. Specific accumulation of γ- and δ-tocotrienols in tumor and their antitumor effect in vivo. J Nutr Biochem. 2009;20(8):607–613. doi:10.1016/j.jnutbio.2008.06.004
37. Husain K, Francois RA, Hutchinson SZ, et al. Vitamin E delta-tocotrienol levels in tumor and pancreatic tissue of mice after oral administration. Pharmacology. 2009;83(3):157–163. doi:10.1159/000190792
38. Husain K, Francois RA, Yamauchi T, Perez M, Sebti SM, Malafa MP. Vitamin E δ-tocotrienol augments the antitumor activity of gemcitabine and suppresses constitutive NF-κB activation in pancreatic cancer. Mol Cancer Ther. 2011;10(12):2363–2372. doi:10.1158/1535-7163.MCT-11-0424
39. Montagnani Marelli M, Marzagalli M, Moretti RM, et al. Vitamin E δ-tocotrienol triggers endoplasmic reticulum stress-mediated apoptosis in human melanoma cells. Sci Rep. 2016;6(1):30502. doi:10.1038/srep30502
40. Brooks R, Kalia P, Ireland DC, Beeton C, Rushton N. Direct inhibition of osteoclast formation and activity by the vitamin E isomer gamma-tocotrienol. Int J Vitam Nutr Res. 2011;81(6):358–367. doi:10.1024/0300-9831/a000087
41. Ha H, Lee J-H, Kim H-N, Lee ZH. α-Tocotrienol inhibits osteoclastic bone resorption by suppressing RANKL expression and signaling and bone resorbing activity. Biochem Biophys Res Commun. 2011;406(4):546–551. doi:10.1016/j.bbrc.2011.02.085
42. Abdul-Majeed S, Mohamed N, Soelaiman I-N. The use of delta-tocotrienol and lovastatin for anti-osteoporotic therapy. Life Sci. 2015;125:42–48. doi:10.1016/j.lfs.2014.12.012
43. Abdul-Majeed S, Mohamed N, Soelaiman I-N. Effects of tocotrienol and lovastatin combination on osteoblast and osteoclast activity in estrogen-deficient osteoporosis. Evid Based Complement Alternat Med. 2012;2012:1–9. doi:10.1155/2012/960742
44. Shuid AN, Mohamad S, Mohamed N, Mokhtar SA, Muhammad N, Soelaiman IN. Bone oxidative changes during early fracture healing of postmenopausal osteoporosis rat model. Asian J Anim Vet Adv. 2011;6(12):1193–1203. doi:10.3923/ajava.2011.1193.1203
45. Zainal Z, Rahim AA, Radhakrishnan AK, Chang SK, Khaza’ai H. Investigation of the curative effects of palm vitamin E tocotrienols on autoimmune arthritis disease in vivo. Sci Rep. 2019;9:11. doi:10.1038/s41598-019-53424-7
46. Chin KY, Wong SK, Sidik FZJ, et al. The effects of annatto tocotrienol supplementation on cartilage and subchondral bone in an animal model of osteoarthritis induced by monosodium iodoacetate. Int J Environ Res Public Health. 2019;16(16):13. doi:10.3390/ijerph16162897
47. Lodge JK. Vitamin E bioavailability in humans. J Plant Physiol. 2005;162(7):790–796. doi:10.1016/j.jplph.2005.04.012
48. Birringer M, Pfluger P, Kluth D, Landes N, Brigelius-Flohé R. Identities and differences in the metabolism of tocotrienols and tocopherols in HepG2 cells. J Nutr. 2002;132(10):3113–3118. doi:10.1093/jn/131.10.3113
49. Yang Z, Lee M-J, Zhao Y, Yang CS. Metabolism of tocotrienols in animals and synergistic inhibitory actions of tocotrienols with atorvastatin in cancer cells. Genes Nutr. 2012;7(1):11–18. doi:10.1007/s12263-011-0233-y
50. Yap SP, Yuen KH, Wong JW. Pharmacokinetics and bioavailability of α-, γ- and δ-tocotrienols under different food status. J Pharm Pharmacol. 2001;53(1):67–71. doi:10.1211/0022357011775208
51. Qureshi AA, Khan DA, Mahjabeen W, Trias AM, Silswal N, Qureshi N. Impact of δ-tocotrienol on inflammatory biomarkers and oxidative stress in hypercholesterolemic subjects. J Clin Exp Cardiolog. 2015;6(367):2.
52. Qureshi AA, Khan DA, Silswal N, Saleem S, Qureshi N. Evaluation of pharmacokinetics, and bioavailability of higher doses of tocotrienols in healthy fed humans. J Clin Exp Cardiolog. 2016;7(4):434. doi:10.4172/2155-9880.1000434
53. Mahipal A, Klapman J, Vignesh S, et al. Pharmacokinetics and safety of vitamin E δ-tocotrienol after single and multiple doses in healthy subjects with measurement of vitamin E metabolites. Cancer Chemother Pharmacol. 2016;78(1):157–165. doi:10.1007/s00280-016-3048-0
54. Satyamitra M, Ney P, Graves J, Mullaney C, Srinivasan V. Mechanism of radioprotection by δ-tocotrienol: pharmacokinetics, pharmacodynamics and modulation of signalling pathways. Br J Radiol. 2012;85(1019):e1093–e1103. doi:10.1259/bjr/63355844
55. Baxter LL, Marugan JJ, Xiao J, et al. Plasma and tissue concentrations of α-tocopherol and δ-tocopherol following high dose dietary supplementation in mice. Nutrients. 2012;4(6):467–490. doi:10.3390/nu4060467
56. Kawakami Y, Tsuzuki T, Nakagawa K, Miyazawa T. Distribution of tocotrienols in rats fed a rice bran tocotrienol concentrate. Biosci Biotechnol Biochem. 2007;71(2):464–471. doi:10.1271/bbb.60524
57. Uchida T, Abe C, Nomura S, Ichikawa T, Ikeda S. Tissue distribution of α- and γ-tocotrienol and γ-tocopherol in rats and interference with their accumulation by α-tocopherol. Lipids. 2012;47(2):129–139. doi:10.1007/s11745-011-3620-7
58. Khanna S, Patel V, Rink C, Roy S, Sen CK. Delivery of orally supplemented α-tocotrienol to vital organs of rats and tocopherol-transport protein deficient mice. Free Radic Biol Med. 2005;39(10):1310–1319. doi:10.1016/j.freeradbiomed.2005.06.013
59. Meganathan P, Jabir RS, Fuang HG, et al. A new formulation of gamma delta tocotrienol has superior bioavailability compared to existing tocotrienol-rich fraction in healthy human subjects. Sci Rep. 2015;5(1):13550. doi:10.1038/srep13550
60. Ikeda S, Uchida T, Ichikawa T, et al. Complexation of tocotrienol with γ-cyclodextrin enhances intestinal absorption of tocotrienol in rats. Biosci Biotechnol Biochem. 2010;74:1452–1457. doi:10.1271/bbb.100137
61. Miyoshi N, Wakao Y, Tomono S, Tatemichi M, Yano T, Ohshima H. The enhancement of the oral bioavailability of γ-tocotrienol in mice by γ-cyclodextrin inclusion. J Nutr Biochem. 2011;22(12):1121–1126. doi:10.1016/j.jnutbio.2010.09.011
62. Desai N. Challenges in development of nanoparticle-based therapeutics. AAPS J. 2012;14(2):282–295. doi:10.1208/s12248-012-9339-4
63. Ventola CL. Progress in nanomedicine: approved and investigational nanodrugs. Pharm Ther. 2017;42(12):742.
64. Patra JK, Das G, Fraceto LF, et al. Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnology. 2018;16(1):71.
65. Maniam G, Mai CW, Zulkefeli M, Dufès C, Tan DMY, Fu JY. Challenges and opportunities of nanotechnology as delivery platform for tocotrienols in cancer therapy. Front Pharmacol. 2018;9(1358). doi:10.3389/fphar.2018.01358
66. Bartelds R, Nematollahi MH, Pols T, et al. Niosomes, an alternative for liposomal delivery. PLoS One. 2018;13(4):e0194179. doi:10.1371/journal.pone.0194179
67. Ge X, Wei M, He S, Yuan W-E. Advances of non-ionic surfactant vesicles (niosomes) and their application in drug delivery. Pharmaceutics. 2019;11(2):55. doi:10.3390/pharmaceutics11020055
68. Shun M-C, Yu W, Gapor A, et al. Pro-apoptotic mechanisms of action of a novel vitamin E analog (α-TEA) and a naturally occurring form of vitamin E (δ-tocotrienol) in MDA-MB-435 human breast cancer cells. Nutr Cancer. 2004;48(1):95–105. doi:10.1207/s15327914nc4801_13
69. Wada S, Satomi Y, Murakoshi M, Noguchi N, Yoshikawa T, Nishino H. Tumor suppressive effects of tocotrienol in vivo and in vitro. Cancer Lett. 2005;229(2):181–191. doi:10.1016/j.canlet.2005.06.036
70. Fu JY, Zhang W, Blatchford DR, Tetley L, McConnell G, Dufès C. Novel tocotrienol-entrapping vesicles can eradicate solid tumors after intravenous administration. J Control Release. 2011;154(1):20–26. doi:10.1016/j.jconrel.2011.04.015
71. Tan DMY, Fu JY, Wong FS, Er HM, Chen YS, Nesaretnam K. Tumor regression and modulation of gene expression via tumor-targeted tocotrienol niosomes. Nanomedicine. 2017;12(20):2487–2502. doi:10.2217/nnm-2017-0182
72. Puri A, Loomis K, Smith B, et al. Lipid-based nanoparticles as pharmaceutical drug carriers: from concepts to clinic. Crit Rev Ther Drug Carrier Syst. 2009;26(6):523–580. doi:10.1615/CritRevTherDrugCarrierSyst.v26.i6.10
73. Abuasal BS, Qosa H, Sylvester PW, Kaddoumi A. Comparison of the intestinal absorption and bioavailability of γ-tocotrienol and α-tocopherol: in vitro, in situ and in vivo studies. Biopharm Drug Dispos. 2012;33(5):246–256. doi:10.1002/bdd.1790
74. Ali H, El-Sayed K, Sylvester PW, Nazzal S. Molecular interaction and localization of tocotrienol-rich fraction (TRF) within the matrices of lipid nanoparticles: evidence studies by Differential Scanning Calorimetry (DSC) and Proton Nuclear Magnetic Resonance spectroscopy (1H NMR). Colloids Surf B Biointerfaces. 2010;77(2):286–297. doi:10.1016/j.colsurfb.2010.02.003
75. Ali H, Shirode AB, Sylvester PW, Nazzal S. Preparation, characterization, and anticancer effects of simvastatin–tocotrienol lipid nanoparticles. Int J Pharm. 2010;389(1):223–231. doi:10.1016/j.ijpharm.2010.01.018
76. Ali H, Shirode AB, Sylvester PW, Nazzal S. Preparation and in vitro antiproliferative effect of tocotrienol loaded lipid nanoparticles. Colloids Surf a Physicochem Eng Asp. 2010;353(1):43–51. doi:10.1016/j.colsurfa.2009.10.020
77. de Carvalho SM, Noronha CM, Floriani CL, et al. Optimization of α-tocopherol loaded solid lipid nanoparticles by central composite design. Ind Crops Prod. 2013;49:278–285. doi:10.1016/j.indcrop.2013.04.054
78. Naseri N, Valizadeh H, Zakeri-Milani P. Solid lipid nanoparticles and nanostructured lipid carriers: structure, preparation and application. Adv Pharm Bull. 2015;5(3):305–313. doi:10.15171/apb.2015.043
79. Abuasal BS, Lucas C, Peyton B, et al. Enhancement of intestinal permeability utilizing solid lipid nanoparticles increases γ-tocotrienol oral bioavailability. Lipids. 2012;47(5):461–469. doi:10.1007/s11745-012-3655-4
80. Abla MJ, Banga AK. Formulation of tocopherol nanocarriers and in vitro delivery into human skin. Int J Cosmet Sci. 2014;36(3):239–246. doi:10.1111/ics.12119
81. Saez V, Souza IDL, Mansur CRE. Lipid nanoparticles (SLN & NLC) for delivery of vitamin E: a comprehensive review. Int J Cosmet Sci. 2018;40(2):103–116. doi:10.1111/ics.12452
82. Date AA, Desai N, Dixit R, Nagarsenker M. Self-nanoemulsifying drug delivery systems: formulation insights, applications and advances. Nanomedicine. 2010;5(10):1595–1616. doi:10.2217/nnm.10.126
83. McClements D, Decker E, Weiss J. Emulsion-based delivery systems for lipophilic bioactive components. J Food Sci. 2007;72(8):R109–R124.
84. Parthasarathi S, Muthukumar SP, Anandharamakrishnan C. The influence of droplet size on the stability, in vivo digestion, and oral bioavailability of vitamin E emulsions. Food Funct. 2016;7(5):2294–2302. doi:10.1039/C5FO01517K
85. Xu F, Pandya JK, Chung C, McClements DJ, Kinchla AJ. Emulsions as delivery systems for gamma and delta tocotrienols: formation, properties and simulated gastrointestinal fate. Food Res Int. 2018;105:570–579. doi:10.1016/j.foodres.2017.11.033
86. Alqahtani S, Alayoubi A, Nazzal S, Sylvester PW, Kaddoumi A. Nonlinear absorption kinetics of Self-Emulsifying Drug Delivery Systems (SEDDS) containing tocotrienols as lipophilic molecules: in vivo and in vitro studies. AAPS J. 2013;15(3):684–695. doi:10.1208/s12248-013-9481-7
87. Alqahtani S, Alayoubi A, Nazzal S, Sylvester PW, Kaddoumi A. Enhanced solubility and oral bioavailability of gamma-tocotrienol using a Self-Emulsifying Drug Delivery System (SEDDS). Lipids. 2014;49(8):819–829.
88. Hatanaka J, Chikamori H, Sato H, et al. Physicochemical and pharmacological characterization of alpha-tocopherol-loaded nano-emulsion system. Int J Pharm. 2010;396(1–2):188–193. doi:10.1016/j.ijpharm.2010.06.017
89. Ledet GA, Biswas S, Kumar VP, et al. Development of orally administered gamma-tocotrienol (GT3) nanoemulsion for radioprotection. Int J Mol Sci. 2017;18(1):15.
90. Pham J, Nayel A, Hoang C, Elbayoumi T. Enhanced effectiveness of tocotrienol-based nano-emulsified system for topical delivery against skin carcinomas. Drug Deliv. 2016;23(5):1514–1524.
91. Hasan ZAA, Idris Z, Abd Gani SS, Basri M. In vitro safety evaluation of palm tocotrienol-rich fraction nanoemulsion for topical application. J Oil Palm Res. 2018;30(1):150–162.
92. Hasan ZAA, Ismail R, Ahmad S. Does the palm tocotrienol-rich fraction induce irritant contact dermatitis. J Oil Palm Res. 2008;20:508–515.
93. Simon LC, Stout RW, Sabliov C. Bioavailability of orally delivered alpha-tocopherol by poly (lactic-co-glycolic) acid (PLGA) nanoparticles and chitosan covered PLGA nanoparticles in F344 rats. Nanobiomedicine. 2016;3(2016):3–8. doi:10.5772/63305
94. Alqahtani S, Simon L, Astete CE, et al. Cellular uptake, antioxidant and antiproliferative activity of entrapped alpha-tocopherol and gamma-tocotrienol in poly (lactic-co-glycolic) acid (PLGA) and chitosan covered PLGA nanoparticles (PLGA-Chi). J Colloid Interface Sci. 2015;445:243–251. doi:10.1016/j.jcis.2014.12.083
95. Abu-Fayyad A, Behery F, Salim AA, et al. PEGylated gamma-tocotrienol isomer of vitamin E: synthesis, characterization, in vitro cytotoxicity, and oral bioavailability. Eur J Pharm Biopharm. 2015;96:185–195. doi:10.1016/j.ejpb.2015.07.022
96. Abu-Fayyad A, Nazzal S. Synthesis, characterization, and in-vitro antitumor activity of the polyethylene glycol (350 and 1000) succinate derivatives of the tocopherol and tocotrienol isomers of vitamin E. Int J Pharm. 2017;519(1–2):145–156. doi:10.1016/j.ijpharm.2017.01.020
97. Borel P, Preveraud D, Desmarchelier C. Bioavailability of vitamin E in humans: an update. Nutr Rev. 2013;71(6):319–331. doi:10.1111/nure.12026
98. Sabliov CM, Fronczek C, Astete CE, Khachaturyan M, Khachatryan L, Leonardi C. Effects of temperature and UV light on degradation of α-tocopherol in free and dissolved form. J Am Oil Chem Soc. 2009;86(9):895. doi:10.1007/s11746-009-1411-6
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

