Back to Journals » Patient Preference and Adherence » Volume 10
Patient-reported preferences for oral versus intravenous administration for the treatment of cancer: a review of the literature
Authors Eek D , Krohe M, Mazar I, Horsfield A, Pompilus F, Friebe R, Shields A
Received 18 February 2016
Accepted for publication 18 April 2016
Published 24 August 2016 Volume 2016:10 Pages 1609—1621
DOI https://doi.org/10.2147/PPA.S106629
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Johnny Chen
Daniel Eek,1 Meaghan Krohe,2 Iyar Mazar,2 Alison Horsfield,3 Farrah Pompilus,2 Rachel Friebe,2 Alan L Shields2
1AstraZeneca, Gothenburg, Sweden; 2Adelphi Values, Boston, MA, USA; 3AstraZeneca, Macclesfield, UK
Objective: The emergence of various modes of administration for cancer treatment, including oral administration, brings into focus the importance of patient preference for administration. The purpose of this research was to evaluate the administration preferences of cancer patients, specifically between oral and intravenous (IV) treatment, as well as the factors contributing to preference.
Methods: A literature search was conducted in OvidSP to identify research in which the preferences of cancer patients for oral or IV treatment have been evaluated. Data were analyzed in two stages: 1) those articles that directly compared preference between modes of administration were tallied to determine explicit preference for oral or IV treatment; and 2) all attributes associated with patient preference were documented.
Results: Of the 48 abstracts identified as part of the initial OvidSP search, eight articles were selected for full-text review. One article was removed following full-text review, and seven additional articles were identified through a gray literature search, yielding a total of 14 articles for evaluation. In Stage 1, 13 of the 14 articles compared preference, of which eleven articles (84.6%) reported that patients preferred oral treatment over IV, while two (15.4%) stated that cancer patients preferred IV treatment over oral. In Stage 2, the most frequently reported attributes contributing to preference included convenience, ability to receive treatment at home, treatment schedule, and side effects.
Discussion: Evidence suggests that oncology patients prefer oral treatment to IV. Rationale for preference was due to a number of factors, including convenience, perception of efficacy, and past experience. Further evaluation should be conducted, given the limited data on patient preference in oncology.
Keywords: oncology, patient preference, mode of administration, literature review, mode of administration, oncology, treatment
Introduction
Advances in the detection and treatment of cancer over the last four decades have resulted in growing numbers of cancer survivors in both Europe and the US. It is estimated that the 5-year survival rates for adults have increased up to 50% in both regions.1 Survivorship can mean complete recovery for a patient in some cases, but in others, patients may experience recurrence, develop another form of cancer, or require intermittent treatment when the disease becomes active, among other trajectories.2 An important consideration associated with increased survivorship and continued treatments are the adverse events that can be associated with cancer drugs, which, in turn, negatively affect patients’ quality of life. This has led oncologists to focus more closely on the patient’s overall treatment experience, taking into account the benefits of a therapy as well as its side effects to inform treatment decisions.3
One component of the patient treatment experience is the way in which therapy is administered. Although oral treatment options may not be available in all cancer types or to all patients based on their treatment needs,4 the development of cancer drugs that can be administered efficaciously through oral as well as traditional intravenous (IV) methods is becoming increasingly common.5,6 In 2014, 40% of the five oncology drugs approved by European Medicines Agency with associated European Public Assessment Reports were orally administered treatments,7 indicative of the continued relevance of both IV and oral modes of administration for cancer care.
Scholars have argued that the reduction of the stress and discomfort associated with IV treatments, coupled with the convenience of oral oncology drugs, are benefits of an oral mode of administration.8,9 Orally administered cancer drugs are perceived to afford patients greater flexibility than IV treatment, in that the former may allow patients to forgo hospital visits.9,10 Research suggests that, when the efficacy and side effects of orally administered cancer treatments are similar to those of IV treatments, patients with incurable malignancies prefer the former, possibly because oral drugs are perceived to afford a greater level of quality of life than their IV counterparts.8
However, concerns exist regarding absorption8 associated with oral treatments, patient adherence to self-administered medication,8,10 and misconceptions regarding their convenience10 as well as their side effects.10 Therefore, the most appropriate mode of administration may not be the same in all contexts, but rather is dependent on a patient’s needs and preferences. As oncologists become more attuned to patient preferences and their quality of life during treatment, there may be a trend toward prescribing a medication that is most convenient and appropriate for each patient.11
The emergence of various modes of administration, including oral administration, for the treatment of cancer brings into focus the importance of patient preference and the factors that contribute to that preference. The purpose of this paper is to evaluate findings from peer-reviewed literature on patient preference within oncology, and to determine if evidence exists regarding treatment preferences for oral versus IV administration.
Materials and methods
Search strategy
The databases Embase, MEDLINE®, and PsycINFO were searched through the OvidSP platform to identify studies published from January 2010 through January 2015. The following search terms were used to identify peer-reviewed literature regarding the preferences of patients for oral versus IV administration of cancer treatment: orals or oral by mouth or per os or per oral route or oral route or oral route of drug; intravenous or injection or injecting or intravenous route or intravenous route of drug; preference or prefer or preferred or choice or select or selection; and cancer or oncology or oncology field or oncologies or growth or tumor or malignancy or malignance or melanoma or sarcoma or malignant cells.
The following limitations were set for the search: limit to the English language; limit to human subjects; limit to peer-reviewed journals; and limit to adult subjects.
Finally, reference lists from identified articles were reviewed and a gray literature search was conducted to identify additional articles that may have been missed in the initial search. Gray literature has been defined as materials that may be held by libraries and other institutions but which are not under the control of commercial publishers, or which were not intended to be submitted for publication, such as government reports or conference proceedings.12 As part of this research, sources meeting this definition as well as published articles identified outside of the databases searched via OvidSP were considered for review.
The gray literature search served as a supplementary search to address any gaps in the OvidSP database search. The gray literature search was conducted by entering similar keywords to those used in the OvidSP database search into an online search engine (Google) and manually screening the hits from the first three pages for sources most relevant to the topic of patient preference for oral versus IV cancer treatment (ie, empirical research directly assessing patient preference for either mode of administration). The limitations outlined earlier were not applied to the gray literature search (ie, non-peer-reviewed sources before January 2010 were considered).
Article selection
The resulting abstracts were uploaded to the Abstrackr (Brown University, Providence, RI, USA) platform for screening. Abstrackr is a software program developed to assist with screening abstracts for systematic reviews, available from: http://abstrackr.cebm.brown.edu/. Codes were developed to organize the abstracts into categories to help identify the articles’ eligibility for full-text review. For example, abstracts considered for exclusion may have been coded as pediatric population or case study data. Two trained researchers screened and coded all abstracts, and once all citations were screened, they were exported to Microsoft Excel for an additional level of review. Any discrepancies with respect to eligibility were discussed and resolved. An article was retrieved for full review if the abstract met each of the following criteria: referenced patient-reported preference for oral versus IV administration in a cancer population; derived from a peer-reviewed journal; reported on English-language studies published in the last 5 years; and referenced an adult patient population.
Articles were excluded from full review if the abstract met at least one of the following criteria: concerned patient preference in a pediatric population; focus of article was on case study data; were conference abstracts; or reported on non-patient preference (eg, physician, consensus panels).
Articles that all reviewers agreed upon were retrieved for full-text review, as were relevant articles identified through reference lists and gray literature searches. Following this, some articles were deemed irrelevant after full-text review and excluded from data extraction.
Data analysis
An approach consistent with thematic content analysis informed the review of the selected articles. This allows researchers to provide detailed descriptions of qualitative data to address a targeted research question.13 An inductive (“bottom-up”) or deductive (“top-down”) approach can be employed. This analysis relied on a primarily top-down approach in that data most relevant to the research question (ie, patient preference for either mode of administration) were extracted and evaluated; however, the analysis aimed to remain grounded in the data, allowing for concepts deemed important in the literature to be considered, as well (eg, predictors of preference based on prior treatment experience).
Articles selected for full-text review were evaluated using this approach, and salient information pertaining to the study design, sample demographics, therapeutic area, and any notable results was recorded in Table 1. Relevant information relating to patient preference was analyzed in two stages:
- The first stage consisted of documenting data that reported preference between the two modes of administration (ie, oral treatment versus IV treatment) by patients (eg, the number of patients in a sample stating that they preferred to receive an oral treatment rather than an IV treatment). In this stage, all articles that directly measured preference between modes of administration were reviewed in detail and were tallied to determine how many sources listed explicit preference for oral or IV treatment when the two were compared.
- The second stage aimed to document all attributes associated with patients’ preferences (eg, what patients liked or disliked about oral or IV treatments regardless of their attitudes toward the alternative mode of administration) identified in the detailed review of each article conducted in the first stage.
Results
Search results
The initial OvidSP literature search identified 48 abstracts as potentially relevant, from which eight articles were selected for full-text review. Following full-text review, one article was removed as failing to meet inclusion criteria. Next, a gray literature search was conducted. The review of abstracts from the OvidSP search demonstrated the need for additional data on patient preference in oncology, specifically additional sources assessing preference in a more diverse oncology patient population, as the majority of the studies focused on, or were predominantly composed of, patients with breast cancer. The gray literature search led to the identification of an additional seven unique sources. These were added to the original seven full-text articles, for a final total of 14 articles.
Article selection
Of the 14 peer-reviewed articles identified for full-text review, eleven8,14–23 articles reported patient preference for oral treatment, two articles reported preference for IV treatment,6,24 and one article did not explicitly state patient preference for either mode of administration.25 The cancer populations studied in these articles included: breast (n=9), lung (n=3), colorectal (n=4), lymphoma (n=2), other (n=2), bowel (n=1), cholangiocarcinoma (n=1), colon (n=1), gastrointestinal (n=1), genital (n=1), gynecologic (n=1), leukemia (n=1), multiple myeloma (n=1), ovarian (n=3), stomach (n=1), and renal cell (n=1). These conditions are not mutually exclusive, as several studies sampled patients with various cancer types.
Stage 1: preference for oral versus IV mode of administration
Stage 1 of this study evaluated reports of patients’ preference between oral treatments and IV treatments. Of the 14 articles reviewed as part of this study, 13 directly measured patient preference for either mode of administration. Among these, eleven articles (84.6%) reported that patients preferred oral over IV administration and two (15.4%) reported that patients preferred IV over oral administration.
Table 1 provides a summary of results from the articles reviewed. The “study results” column reports Stage 1 and Stage 2 results and includes findings on patients’ preferences for mode of administration, details reported by the sample population, quantitative results, and attributes associated with preference.
Amongst the studies reviewed, six included patients with breast cancer only and an additional three articles included patients with breast cancer, among other cancers. Two articles studied patients with colorectal cancer only. With the exception of four studies, most include a large sample size of >50 patients. Seven studies stratified findings by predictors, such as previous treatment experience, age, and sex.
While there was a clear trend depicting patient preference for oral versus IV treatment, some findings were inconclusive across studies. Both Borner et al23 and Pfeiffer et al24 studied patient preference using the Treatment Preference Questionnaire in colorectal cancer using a similar study design (a crossover study where patients were first treated with either oral or IV chemotherapy then with the other mode of administration). Borner et al reported that patients preferred oral chemotherapy over IV, whereas Pfeiffer et al reported that patients preferred IV over oral. Pfeiffer et al attributed this to the fact that more side effects were experienced with oral capecitabine than the intravenous Nordic fluorouracil/leucovorin taken by patients in their sample, leading patients to prefer reduced toxicity over convenience.
Some studies reported differences in treatment preference or perceptions of treatment by demographics, such as age. For example, Liu et al8 reported that convenience was more important to younger male patients than older female patients, whereas other studies did not find significant differences when evaluating results by age. Specifically, Schott et al22 compared views on oral and IV treatment by age in a German breast cancer population. The authors reported that there were no significant differences between older (those over the age of 50 years) and younger patients in terms of their views on how daily life is impacted due to hospital visits for IV treatment, and no differences in terms of level of concern about taking oral medication incorrectly. That is, both age groups reported that IV treatment had a medium-to-strong impact on daily life and neither group was worried about taking oral treatments incorrectly.
Stage 2: attributes associated with patient preference
This section reports all attributes identified in the published literature that were associated with patients’ mode of administration preferences.
Twelve of the 14 articles reported attributes associated with patient preference. Among the articles evaluating preference, “convenience”8,16,21 and ability to receive treatment at home8,16 were the most frequently reported reasons for preferring an oral mode of administration. Time required to stand upright (specifically, the need to remain standing for 30 minutes after taking oral bisphosphonate treatment),15 the inability to eat or drink,15 and forgetfulness8 were the only negative aspects of oral treatments reported by patients.
The most frequently reported rationale for patient preference for IV treatment among cancer patients included concepts related to treatment schedule, specifically “completion of treatment in 1 day”,8 and “treatment duration”.17 Side effects19 and impact on daily life22 were the only negative aspects of IV treatment reported in the reviewed articles.
Three articles23–25 provide patient preference attributes, but did not directly relate them to either oral or IV modes of administration. The attributes included the following: the treatment does not lead to side effects (infection, vomiting, diarrhea, painful mouth sores, nausea, tiredness),23,24 can be taken at home,23,24 is a pill,23,24 does not affect mood or daily activities,23,24 is taken at hospital,23,24 is an injection,24 does not cause pain,24 cancer got better,23 others (family, friends) preferred the treatment,23 efficacy,25 side effects,25 cost,25 and dosing regimen.25
Tables 2–5 provide a description of the positive and negative attributes associated with oral and IV modes of administration as reported by cancer patients.
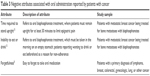  | Table 3 Negative attributes associated with oral administration reported by patients with cancer |
  | Table 5 Negative attributes associated with IV administration reported by patients with cancer |
Discussion
The results of this literature review suggest that, in patients with cancer, a preference for oral treatment administration over IV has been reported. In this selected sample of studies, convenience and ability to receive treatment at home were the most frequently reported factors associated with patient preference for oral treatment, possibly relating to the fact that oral treatments may allow patients to forgo or reduce the number of hospital visits in comparison to IV treatment.10 In those studies where preference was reported for IV administration, the treatment schedule, specifically treatment duration, was the most frequently reported factor regarded as a positive attribute associated with receiving IV treatment.
It is important to note that while cancer patients in these studies preferred oral over IV treatment, it was also reported that patients were generally unwilling to accept reduced efficacy8,19 or greater treatment toxicity24 in favor of other treatment attributes, such as convenience. However, when patients rated efficacy as less important than other treatment factors such as convenience, researchers speculated that this was because patients were made aware that the oral and IV formulations were equally effective.23 Therefore, patients’ assessments about the efficacy and toxicity of a treatment, whether known or perceived, appear to continue to drive preference for mode of administration over other potential personal benefits that a patient may value.
Additionally, there was some evidence in the literature reviewed that treatment preferences for mode of administration may relate to factors such as line of treatment or demographic characteristics. For instance, one article reported that the percentage of breast cancer patients who cited convenience as a reason for choosing oral treatment increased by 20 percentage points between the first compared to second and third lines of treatment, from 52% to 73% and 72%, respectively.16 Another study reported that convenience was a more important treatment attribute to younger men than women or patients in other age groups, and that women were more likely to report that starting an IV line was a painful and/or difficult experience.8
Scholars have noted that patients’ treatment experiences and preferences are more salient than ever for two reasons: not only are cancer patients living longer due to improved cancer therapies, but their treatments are also often associated with similar survival benefits.3 Therefore, patients are receiving medical care for longer and are able to choose between therapies that may be associated with different degrees of side effects and/or impacts to their functional ability. Therefore, clinicians have been prompted to evaluate how treatments affect patients’ outcomes both in terms of disease control and quality of life.
Research also suggests that experts may not value oral cancer treatments as strongly as their patients do, and that they have concerns about its appropriate administration. A survey of 400 oncologists found that while the most commonly reported recommendation for a first-line, single-agent therapy for the treatment of metastatic colorectal was oral therapy (42%), twice as many oncologists reported that their patients would prefer this mode of administration. Just 8% of clinicians reported that their patients would prefer IV treatment over oral treatment alternatives. While most oncologists (77%) reported that patients and their clinicians should make treatment decisions jointly, 16% felt that clinicians alone should make the decision. Main reasons for not prescribing oral chemotherapy included issues surrounding efficacy, compliance, income, and side effects. Therefore, both clinicians and patients alike recognize the advantages and disadvantages associated with oral and IV modes of administration, although patients appear to support the use of oral chemotherapy more so than their clinicians.
Studies regarding patient preference in other disease areas, such as schizophrenia and diabetes, have reported that IV administration is preferred over oral treatments, suggesting that the findings produced in studies of cancer patients may not be applicable to other conditions. For example, Caroli et al reported that patients with schizophrenia preferred IV to oral treatment, for reasons such as the treatment schedule, perceived efficacy, and the decreased risk of forgetting to take the treatment, despite the fact that some patients reported disliking injections. Schizophrenia preference attributes were characterized by nonadherence to oral medications and the refusal of some patients to take the medications at home, issues that may not be experienced by oncology patients.26 A further example from Casciano et al suggests that prior experience with IV treatment relates to greater acceptance of this mode of administration for type 2 diabetes patients, a condition associated with arguably greater exposure to IV treatment than cancer.27
There are some limitations to this study. First, the search terms and limitations may have prevented identification of articles addressing the substantive topics (eg, articles in non-English journals). Second, the predominance of articles that included breast cancer populations (nine of the 14 reviewed articles) may hinder the ability to extrapolate the findings reported in this paper to other cancer types. Additionally, the exclusive focus on the peer-reviewed literature may provide a limited understanding regarding how treatment choices are made by patients outside of hypothetical and/or clinical trial study contexts, and of their perceptions of these modes of administration in a real-world setting. Referring to patient blogs and personal accounts on social media, data from patient and/or expert interviews, and other sources that rely on open-ended or spontaneous methods to elicit patients’ actual experience with different modes of administration for oncology treatment would supplement findings from the literature. Lastly, it should be reiterated that not all cancer patients may have the opportunity to choose between orally or intravenously administered treatment for their disease, for various reasons such as oral formulations not being available in their indication4 or needing to receive both IV as well as oral treatment rather than being able to choose between the two.
The conclusions presented in this paper were drawn from a small sample of articles that included a wide range of cancer types and possible treatment regimens. Patient preference may vary by cancer type and treatment offerings, which warrants future research that assesses preference in one cancer type in a larger patient sample, ideally in a population other than breast cancer, as breast cancer appears to have been the focus of much of the literature on mode of administration preference thus far. The findings from this review can serve as the foundation for understanding attributes that may relate to oncology patients’ treatment preferences and can inform measurement of patient preference in a larger empirical study.
Despite these limitations, this review provides several important conclusions and avenues for further research. The results from recent clinical trials in ovarian cancer have pointed to the importance of personalized treatment plans for patients depending on their type and stage of disease, demonstrating the importance of conducting research aimed at better understanding preference for different modes of administration.28 However, the majority of articles reviewed as part of this study did not thoroughly evaluate preference by line of treatment or stage of disease, both of which may be associated with valuing certain aspects of a treatment over others.
In conclusion, among the articles reviewed, support was found to suggest that cancer patients prefer oral over IV treatment. Preference for a mode of administration can be associated with numerous factors, including convenience, perception of efficacy, and past treatment experience. However, as caveated earlier, further research is needed to determine whether this holds true in cancer patient populations other than breast cancer. Although prior research may have broadly addressed the factors associated with treatment that are of importance to cancer patients, a targeted evaluation of how these relate to preference for oral versus IV treatment is needed.
Acknowledgments
The search and review of the literature, generation of results, and drafting of the manuscript were financially supported by AstraZeneca, Macclesfield, UK.
Disclosure
DE and AH are employees of AstraZeneca. IM, MK, FP, RF, and ALS are employees of Adelphi Values. The authors report no other conflicts of interest.
References
Rowland JH, Kent EE, Forsythe LP, et al. Cancer survivorship research in Europe and the United States: where have we been, where are we going, and what can we learn from each other? Cancer. 2013;119(Suppl 11):2094–2108. | ||
American Cancer Society. Cancer Treatment and Survivorship Facts & Figures 2014–2015. Atlanta, GA: American Cancer Society; 2014. | ||
Zagadailov E, Fine M, Shields A. Patient-reported outcomes are changing the landscape in oncology care: challenges and opportunities for payers. Am Health Drug Benefits. 2013;6(5):264–274. | ||
Batlle JF, Arranz EE, de Castro Carpeño J, et al. Oral chemotherapy: potential benefits and limitations. Rev Oncol. 2004;6(6):335–340. | ||
European Medicines Agency. European Medicines Agency recommends approval of 44 medicines for human use and six medicines for veterinary use in first half 2013. Available from: http://www.ema.europa.eu/ema/index.jsp?curl=pages/news_and_events/news/2013/07/news_detail_001860.jsp&mid=WC0b01ac058004d5c1. Accessed June 2, 2015. | ||
Mahner S, Harter P, Fuxius S, et al. Präferenz study – Patients’ individual choice for oral vs. intravenous Treosulfan in elderly patients with ovarian cancer: analysis of tolerability – for the North-Eastern German Society of Gynecological Oncology (NOGGO) study group. J Cancer Res Clin Oncol. 2012;138(Suppl 1):82. | ||
European Medicines Agency. European public assessment reports. 2015; Available from: http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/landing/epar_search.jsp. Accessed September 2, 2015. | ||
Liu G, Franssen E, Fitch MI, Warner E. Patient preferences for oral versus intravenous palliative chemotherapy. J Clin Oncol. 1997;15(1):110–115. | ||
Borner M, Scheithauer W, Twelves C, Maroun J, Wilke H. Answering patients’ needs: oral alternatives to intravenous therapy. Oncologist. 2001;6(Suppl 4):12–16. | ||
Weingart SN, Brown E, Bach PB, et al. NCCN Task Force Report: Oral chemotherapy. J Natl Compr Cancer Netw. 2008;6(Suppl 3):S1–S14. | ||
Partridge AH, Avorn J, Wang PS, Winer EP. Adherence to therapy with oral antineoplastic agents. J Natl Cancer Inst. 2002;94(9):652–661. | ||
Schopfel J. Towards a Prague Definition of Grey Literature. Available from: http://greynet.org/images/GL12_S1P,_Sch_pfel.pdf. Accessed May 6, 2015. | ||
Braun V, Clarke V. Using thematic analysis in psychology. Qual Res Psychol. 2006;3(2):77–101. | ||
Beusterien K, Grinspan J, Kuchuk I, et al. Use of conjoint analysis to assess breast cancer patient preferences for chemotherapy side effects. Oncologist. 2014;19(2):127–134. | ||
Fallowfield L, Stebbing J, Braybrooke J, Langridge C, Jenkins V. The preferences and experiences of different bisphosphonate treatments in women with breast cancer. Psychooncology. 2011;20(7):755–761. | ||
Gornas M, Szczylik C. Oral treatment of metastatic breast cancer with capecitabine: what influences the decision-making process? Eur J Cancer Care (Engl). 2010;19(1):131–136. | ||
Ishitobi M, Shibuya K, Komoike Y, Koyama H, Inaji H. Preferences for oral versus intravenous adjuvant chemotherapy among early breast cancer patients. Patient Prefer Adherence. 2013;7:1201–1206. | ||
Peeters L, Sibille A, Anrys B, et al. Maintenance therapy for advanced non-small-cell lung cancer: a pilot study on patients’ perceptions. J Thorac Oncol. 2012;7(8):1291–1295. | ||
Seaman S, Nelson A, Noble S. Cancer-associated thrombosis, low-molecular-weight heparin, and the patient experience: a qualitative study. Patient Prefer Adherence. 2014;8:453–461. | ||
Teuffel O, Cheng S, Ethier MC, et al. Health-related quality of life anticipated with different management strategies for febrile neutropenia in adult cancer patients. Support Care Cancer. 2012;20(11):2755–2764. | ||
Calhoun EA, Roland PY. Convenience is the main reason why ovarian cancer patients prefer oral regimen: ISPOR fifth annual international meeting, Arlington, VA. Value Health. 2000;3(2):134–135. | ||
Schott S, Schneeweiss A, Reinhardt J, et al. Acceptance of oral chemotherapy in breast cancer patients – a survey study. BMC Cancer. 2011;11:129. | ||
Borner MM, Schoffski P, de Wit R, et al. Patient preference and pharmacokinetics of oral modulated UFT versus intravenous fluorouracil and leucovorin: a randomised crossover trial in advanced colorectal cancer. Eur J Cancer. 2002;38(3):349–358. | ||
Pfeiffer P, Mortensen JP, Bjerregaard B, et al. Patient preference for oral or intravenous chemotherapy: a randomised cross-over trial comparing capecitabine and Nordic fluorouracil/leucovorin in patients with colorectal cancer. Eur J Cancer. 2006;42(16):2738–2743. | ||
DiBonaventura MD, Copher R, Basurto E, Faria C, Lorenzo R. Patient preferences and treatment adherence among women diagnosed with metastatic breast cancer. Am Health Drug Benefits. 2014;7(7):386–396. | ||
Caroli F, Raymondet P, Izard I, Plas J, Gall B, Delgado A. Opinions of French patients with schizophrenia regarding injectable medication. Patient Prefer Adherence. 2011;5:165–171. | ||
Casciano R, Malangone E, Ramachandran A, Gagliardino JJ. A quantitative assessment of patient barriers to insulin. Int J Clin Pract. 2011;65(4):408–414. | ||
Ledermann J, Harter P, Gourley C, et al. Olaparib maintenance therapy in patients with platinum-sensitive relapsed serous ovarian cancer: a preplanned retrospective analysis of outcomes by BRCA status in a randomised phase 2 trial. Lancet Oncol. 2014;15(8):852–861. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.





