Back to Journals » Neuropsychiatric Disease and Treatment » Volume 15
Patient-reported depression severity and cognitive symptoms as determinants of functioning in patients with major depressive disorder: a secondary analysis of the 2-year prospective PERFORM study
Authors Haro JM , Hammer-Helmich L , Saragoussi D, Ettrup A, Larsen KG
Received 26 February 2019
Accepted for publication 17 June 2019
Published 13 August 2019 Volume 2019:15 Pages 2313—2323
DOI https://doi.org/10.2147/NDT.S206825
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Roger Pinder
Josep Maria Haro,1 Lene Hammer-Helmich,2 Delphine Saragoussi,3 Anders Ettrup,4 Klaus Groes Larsen5
1Research and Teaching Unit, Parc Sanitari Sant Joan De Deu, CIBERSAM, University of Barcelona, Sant Boi De Llobregat, Barcelona, Spain; 2Medical Affairs, Real World Evidence, H. Lundbeck A/S, Valby, Denmark; 3Real World Evidence and Epidemiology, Lundbeck SAS, Issy-les-Moulineaux, France; 4Medical Affairs, Vortioxetine, H. Lundbeck A/S, Valby, Denmark; 5Biometrics, H. Lundbeck A/S, Valby, Denmark
Purpose: To investigate the temporal interrelationship between depression severity, cognitive symptoms, and functioning in patients with major depressive disorder (MDD) in the PERFORM study (NCT01427439).
Patients and methods: PERFORM was a 2-year, multicenter, prospective, noninterventional cohort study in outpatients with MDD who were either initiating antidepressant monotherapy or undergoing their first switch of antidepressant. Patients were enrolled by a general practitioner or psychiatrist. Structural equation model (SEM) analysis was used to explore temporal associations between patient-reported depression severity (9-item Patient Health Questionnaire score), cognitive symptoms (5-item Perceived Deficits Questionnaire score), and functional impairment (Sheehan Disability Scale total score). Standardized regression coefficients (SRCs) were used to evaluate the relationship between each outcome and scores from the most recent prior visit over the 2 years of follow-up.
Results: Between February 25, 2011, and February 19, 2015, 1,159 eligible patients with MDD completed the baseline and ≥1 follow-up visit at 194 sites in five European countries (France, Germany, Spain, Sweden, and the UK). Overall, 1,090 patients had assessments for ≥1 outcome measure at two consecutive visits. Severity of cognitive symptoms at baseline and Months 2 and 18 predicted functional impairment at Months 2, 6, and 24, respectively (SRC: 0.18, 0.15, and 0.22; P<0.001). Depression severity at Months 2, 6, and 12 predicted functional impairment at Months 6, 12, and 18, respectively (SRC: 0.17, 0.25, and 0.22; P<0.001). Severity of cognitive symptoms at baseline and Month 18 predicted depression severity at Months 2 and 24, respectively (SRC: 0.19 and 0.22; P<0.001). Functional impairment did not significantly predict the severity of depression or cognitive symptoms, and depression severity did not significantly predict the severity of cognitive symptoms at any time point.
Conclusion: Patient-reported severity of cognitive symptoms appears to be an independent and significant determinant of subsequent functional impairment and depression severity in patients with MDD.
Keywords: major depressive disorder, cognitive symptoms, functional impairment, structural equation model
Introduction
Major depressive disorder (MDD) is a common and debilitating condition that affects more than 120 million people worldwide.1 MDD is characterized by emotional, physical, and cognitive symptoms that significantly affect patient functioning.2 Compared with the general population, patients with MDD report substantial deficits in family, work, and social functioning.3–8 Functional impairment imposes a significant burden in MDD.9–12 Assessment of functional impairment in patients with MDD provides important information on disease impact and remission from the patient’s own perspective. Functional recovery should be considered an important treatment goal if patients are to return to productive and fulfilling daily lives.8,13,14
Functional impairment has been shown to persist even after remission of mood symptoms in patients with MDD,6,15–18 preventing a full return to social and professional life. In a large US study designed to assess the efficacy of sequential acute treatments for MDD (Sequenced Treatment Alternatives to Relieve Depression [STAR*D]), only 7% of patients reported within-normal functioning before initiation of antidepressant therapy.6 In STAR*D, patients who achieved remission of depressive symptoms showed greatest improvement in functional impairment; however, 20–40% of these remitted patients continued to experience ongoing deficits in functioning.6 Residual functional impairment following remission of depressive symptoms has also been shown to be a predictor of subsequent relapse in patients with MDD.19 This lack of synchronicity between symptomatic improvement and functioning highlights an unmet clinical need in patients with MDD.8
Cognitive symptoms, such as disturbances in attention, memory, processing speed and executive functioning, are a core feature of MDD that are gaining attention as a major contributor to disease burden and an appropriate concomitant treatment target.7,14,20–23 Indeed, addressing cognitive symptoms of MDD may be necessary to achieve functional recovery. Systematic reviews have concluded that there is evidence for an association between cognitive functioning and functional impairment in individuals with MDD, but that the quality of evidence is weak and the evidence for a causal link is limited.21,24
Recent real-world evidence suggests an association between cognitive symptoms and functional impairment in patients with MDD, independent of the severity of depression.25 These data stem from the PERFORM (Prospective Epidemiological Research on Functioning Outcomes Related to Major depressive disorder) study, which was a European prospective observational cohort study undertaken to better understand the course of a depressive episode and its impact on patient functioning over a 2-year period in outpatients with MDD who were either initiating or undergoing their first switch of antidepressant monotherapy.25 Functional impairment in this population was not only associated with severity of depressive symptoms, but also with severity of cognitive symptoms consistently throughout the 2 years of follow-up. However, the time-dependent interrelationships between severity of depressive symptoms, cognitive symptoms, and functional impairment in patients with MDD remain to be fully elucidated. In particular, it is not known which, if any, of these factors precede the others and whether the causal relationships between these factors change over time.
This analysis was undertaken to further investigate the interrelationship of depression severity, cognitive symptoms, and functioning in patients treated for MDD in the PERFORM study using the structural equation model (SEM) approach.26 This statistical method allows for simultaneous estimation of associations between groups of variables, permitting elucidation of the time dependency of changes and potential causal relationships. This analysis aims to provide further insight into the role of cognitive symptoms in patients with MDD and the potential impact of these cognitive symptoms in terms of depression severity and functional impairment.
Methods
Study design
PERFORM comprised a 2-year, prospective, multicenter, noninterventional cohort study in outpatients with MDD who were enrolled by either a psychiatrist or a general practitioner (GP) at 194 clinical sites in five European countries (France, Germany, Spain, Sweden, and the UK) (NCT01427439). Study design and inclusion/exclusion criteria have been reported in detail previously.25,27 In brief, study participants were 18–65 years of age, were clinically diagnosed with MDD (Diagnostic and Statistical Manual of Mental Disorders 4th Edition, Text Revision criteria; confirmed by the Mini International Psychiatric Interview questionnaire [depression module]), and were either starting antidepressant monotherapy or switching antidepressant for the first time. The treating physician determined which antidepressant was prescribed, and the decision was not influenced by study participation.
Ethical approval and consent to participate
Ethical approval was obtained from the necessary authorities for each study site in accordance with national regulations regarding observational studies (see Supplementary Appendix 1). The study was conducted in accordance with the Declaration of Helsinki. All patients provided written informed consent to participate in the study.
Study assessments
Study assessments were conducted at patients’ routine clinic visits at baseline and 2, 6, 12, 18, and 24 months. Patients assessed their depression severity using the 9-item Patient Health Questionnaire (PHQ-9);28 cognitive symptoms using the 5-item Perceived Deficit Questionnaire (PDQ-5);29,30 and functional impairment using the Sheehan Disability Scale (SDS).31,32 In this analysis, SDS total scores were included only if patients reported scores in all three domains. Further information on each of these scales can be found in Supplementary Appendix 2.
Statistical analysis
The population for each structural equation comprised all patients who met the study inclusion criteria and provided data for the parameters at the appropriate time points. Descriptive summary statistics are shown for the three outcomes of interest by time point.
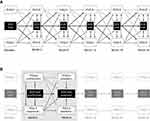
The SEM approach was used to explore the temporal association between PHQ-9, PDQ-5, and SDS total score.26 An SEM is a series of multivariate linear regressions modeled in a single analysis. Figure 1A shows all the effects that were estimated in the SEM; the three outcome measures were allowed to depend on the scores of all outcomes at the most recent prior visit, but not at any earlier visits. Outcome measures from the same visit were allowed to be correlated.
Each of the three outcome measures was assessed at five post-baseline time points. A joint model for the 15 post-baseline measurements was estimated by full information maximum likelihood (FIML) in R (version 3.3.1) using the “lavaan” package (version 0.5–20).33 FIML produces unbiased parameter estimates when data are missing at random. For model evaluation, the root mean square error of approximation (RMSEA; good fit generally accepted as <0.08) and comparative fit index (CFI; good fit generally accepted as >0.95) were used, while chi-squared tests were used to evaluate the statistical significance of specific parameters in the model. Standardized regression coefficients (SRCs) were reported to facilitate direct comparison of effect size between PHQ-9, SDS, and PDQ-5 scores at the different time points.
As well as the model using SDS total score, similar models were fitted with each of the three SDS subscales in three separate SEM analyses to explore consistency across the different functionality items and to explore whether results were mainly driven by one or two of the subscales. Alternative and more complex SEMs that also included effects of scores earlier than the prior time point were used to explore longer dependences in the trajectories of the three patient-reported outcomes.
G-computation was used as a sensitivity analysis.34–36 This method provides estimates with causal interpretations from nonrandomized data under certain conditions, such as when unmeasured confounders may be present (Figure 1B). In G-computation, confounding effects are removed by creating new datasets of counterfactuals, where the “exposure” is uncorrelated with the potential confounders and then estimating the mean difference between two different exposures. Further details of the G-computation methodology are provided in Supplementary Appendix 3.
In this analysis, P-values were not corrected for multiple comparisons and should be considered nominal.
Results
Study population
A total of 1,895 patients were screened for inclusion in the PERFORM study, 1,402 of whom met all study inclusion/exclusion criteria and were enrolled. The first patient was screened on February 25, 2011, and the last patient completed the study on February 19, 2015. A total of 1,159 patients who completed the baseline visit with at least one follow-up visit without any violation of inclusion or exclusion criteria were included in this analysis. The study population has been described in detail previously.25,27 Baseline demographic and clinical characteristics are shown in Table S1. In brief, most patients were enrolled and followed up by GPs (83.6%), 56.6% had experienced a previous depressive episode, 78.7% were initiating antidepressant therapy, and 21.3% were switching antidepressant therapy for the first time. The main reasons for switching were lack of efficacy (77.3%), adverse events (9.3%), patient decision (6.9%), and lack of compliance (3.2%). The most frequent antidepressants prescribed at the initial study visit were citalopram (24.0%), escitalopram (20.2%), fluoxetine (10.5%), sertraline (9.5%), and duloxetine (9.0%); all other antidepressants were used in less than 5% of patients. Mean (SD) age was 44.3 (12.0) years and 73.2% of patients were female.
A total of 1,090 patients (94% of the study population) had assessments for at least one outcome measure at two consecutive visits. Summary characteristics of the three patient-reported outcomes of interest at the six time points over the 2 years of follow-up are shown in Table 1 and Figure 2. As shown, there were more patients with a PHQ-9 score and fewer with an SDS score than with a PDQ-5 score at all time points (Table 1). A gradual decrease in mean scores over time was seen for all three outcome measures (Figure 2), suggesting general improvement in depressive symptoms, cognitive symptoms, and functioning over the 2 years of follow-up. A slower rate of change was observed for cognitive symptoms (PDQ-5 score) than for the other outcomes; however, no formal statistical comparisons were performed.
 |
Table 1 Summary statistics of PDQ-5 score, SDS total score, and PHQ-9 score by visit |
The number of patients included in the SEM and the G-computation analyses ranged from 207 to 446.
Relationship between depression severity, cognitive symptoms, and functional impairment (SEM analysis)
The fit of the SEM produced a CFI of 0.941 and RMSEA of 0.077, suggesting an acceptable (CFI) or good (RMSEA) fit of the model. More complex models were explored with more dependencies on earlier time points. These models produced marginally better fits of the data according to the fit indices; however, relationships between outcomes at the consecutive visits were considered to be most relevant in terms of potential implications for routine practice.
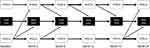
Figure 3 shows paths that were statistically significant at the P<0.001 level in the SEM; the thickness of each path is proportional to its standardized effect size. The strict confidence level of 0.001 in the SEM analysis is chosen to disregard statistically significant effects that likely have no clinical importance. As expected, patient-rated depression severity (PHQ-9 score), functional impairment (SDS total score), and cognitive symptoms (PDQ-5 score) depended moderately to strongly on the value of the previous assessment on the same scale at each time point over the 2 years of follow-up, with SRCs of 0.37–0.71 for PHQ-9, 0.44–0.50 for SDS total score, and 0.60–0.84 for PDQ-5 (Table S2). Depression severity at Months 2, 6, and 12 predicted the degree of functional impairment at Months 6, 12, and 18, respectively (SRC: 0.17, 0.25, and 0.22). Severity of cognitive symptoms at baseline, Month 2, and Month 18 predicted the degree of functional impairment at Months 2, 6, and 24, respectively (SRC: 0.18, 0.15, and 0.22). Severity of cognitive symptoms at baseline and Month 18 also predicted depression severity at Months 2 and 24, respectively (SRC: 0.19 and 0.22). However, the degree of functional impairment did not predict depression severity or the severity of cognitive symptoms at the P<0.001 level at the subsequent visit at any time point. Similarly, depression severity did not predict the severity of cognitive symptoms at the P<0.001 level at the subsequent visit at any time point.
The SEM analysis was repeated, substituting each of the three SDS subscale scores for the SDS total score. There were no notable differences between the three analyses, and no ambiguities were observed between the analysis using the SDS total score and the analyses based on the individual subscales (data not shown).
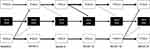
Figure 4 provides a graphical representation of the standardized effects based on the G-computation, including effects that were significant at the P<0.05 level; the thickness of each effect is proportional to its standardized effect size. Results of the G-computation generally confirmed those of the SEM analysis. Depression severity at Months 2, 6, and 12 predicted the degree of functional impairment at Months 6, 12, and 18, respectively (SRC: 0.19, 0.30, and 0.26). Severity of cognitive symptoms at baseline and Month 18 predicted the degree of functional impairment at Months 2 and 24, respectively (SRC: 0.15 and 0.21). The severity of cognitive symptoms at baseline and Month 18 predicted depression severity at Months 2 and 24, respectively (SRC: 0.19 and 0.21). As in the SEM analysis, the degree of functional impairment did not predict depression severity or the severity of cognitive symptoms at the subsequent visit at any time point, and depression severity did not predict the severity of cognitive symptoms at the subsequent visit at any time point.
Figure S1 shows the SRCs from the SEM plotted against the G-computed effect estimates. No systematic differences were observed between estimates based on the two approaches, suggesting robustness of the findings. In general, the standard errors were 40–70% greater for the G-computed estimates (data not shown), owing partly to the complexity of the SEM that modeled 15 variables simultaneously. This is reflected in the fact that fewer effects were statistically significant using G-computation compared with the SEM. Nevertheless, the two methods were in excellent agreement, and all effects that were statistically significant at the P<0.05 level in the G-computation were statistically significant at the P<0.001 level in the SEM.
Discussion
This analysis of data from the PERFORM study shows that severity of cognitive symptoms is an important determinant of both subsequent functional impairment and depression severity throughout the 2 years of follow-up, but particularly at baseline and at later time points. These results are in keeping with previous findings concerning the potential impact of cognitive symptoms on functioning and health-related quality of life in patients with MDD,25,37–41 and also highlight the influence of cognitive symptoms on the course of mood symptoms in this population. As expected, the results of this analysis also show that depression severity is a major determinant of functional impairment in patients with MDD, which is consistent with the results of previous studies.7,20,25,42–44 In contrast, functional impairment was not found to predict the severity of depression or cognitive symptoms at any subsequent visit. Similarly, depression severity did not predict cognitive symptoms at any subsequent visit. These findings highlight the impact of cognitive symptoms on functioning in patients with MDD. The results of the SEM analysis based on total SDS score were confirmed in separate analyses for each of the three SDS functioning subscales and using G-computation in a causal inference model as a sensitivity analysis, suggesting robustness of the findings and implying a causal relationship between both depressive and cognitive symptoms and everyday functioning in patients with MDD.
It is well documented that functional impairment can persist in patients with MDD even after remission of mood symptoms.6,15–18 Other studies have shown cognitive symptoms to persist longer than depressive symptoms in MDD and that these residual cognitive symptoms can persist even in patients who achieve clinical remission.23,45,46 In the PERFORM study, residual perceived cognitive symptoms in patients who achieved remission of depression at Month 2 were also found to be associated with an increased risk of relapse at Month 6.47 Collectively, these findings indicate that cognitive symptoms are an important and independent treatment target in patients with MDD and suggest that treatment interventions targeting both initial and residual cognitive symptoms may improve functional recovery in this population, both directly and/or via improvement of clinical outcomes. Functional recovery is critical for patients with MDD to enable them to return to productive and fulfilling daily lives.8,13,14
Both pharmacological and nonpharmacological antidepressant treatments may improve cognitive symptoms to some extent when improving mood symptoms; however, to date, only vortioxetine has been shown to have robust and direct effects on cognitive function.48,49 In a recent interventional, open-label, real-world Canadian study undertaken to examine the association between patient-reported cognitive symptoms and workplace productivity in working patients with MDD treated with vortioxetine (Assessment in Work productivity and the Relationship with Cognitive symptoms [AtWoRC] study), a highly significant positive correlation was seen between improvements in cognitive symptoms and improvements in functioning and workplace productivity after 12 and 52 weeks of vortioxetine treatment.50,51 As in the present study, SEM analyses confirmed that improvements in cognitive symptoms predicted long-term improvements in functional outcomes even after adjusting for severity of depressive symptoms.51
A major strength of this study is that it was performed in a real-world setting with long-term longitudinal follow-up of a large cohort of patients, most of whom were enrolled and followed up by GPs. Study sites were balanced with the national proportions of these clinicians treating patients with depression to ensure findings were applicable to routine practice. All outcome measures were self-reported and therefore represent disease severity and impact from the patient’s own perspective, which appears particularly valid in patients with mental health disorders such as MDD.52 Although the use of objective measures can provide important information for the clinician, use of patient-reported outcomes is in keeping with awareness of the limitations of clinical symptom-based measures in assessing recovery from mental illness in a way that is meaningful to patients.53–55 Furthermore, it has recently been reiterated that while both objective measures of cognitive function and subjectively rated cognitive symptoms may be clinically relevant, the overlap and correlation between the two approaches are limited.50,51,56 In this study, cognitive symptoms were assessed using the PDQ-5. Despite depression often being associated with cognitive symptoms, there is limited awareness for recognition and assessment of cognitive symptoms in patients with MDD during routine care. The PDQ was initially developed in order to provide a self-report measure of cognitive dysfunction in patients with multiple sclerosis.29,30 The PDQ scores of patients with multiple sclerosis have been found to be correlated with scores on depression scales, but not with neuropsychological test scores.57 In addition, the antidepressants vortioxetine and duloxetine have demonstrated significant improvements in PDQ scores relative to placebo.58 The results of the present study suggest that self-perceived cognitive symptoms are more relevant in patients with depression than objective measures of cognitive function. The full-length PDQ consists of 20 items;29,30,59 the abbreviated 5-item version was used in this study because it is more convenient to administer in routine practice settings. Indeed, use of neuropsychological tests to assess cognitive function is limited by a lack of availability and time constraints in general practice.
Considering the length of the study, the decrease over time in the number of patients reporting the outcomes of interest was modest. The long duration of study also enables characterization of long-term functional outcomes, which may lag behind improvements on other measures, such as depressive symptoms. In terms of the methodology used, the SEM uses all the repeated measures of the three variables at all time points in a single model, allowing evaluation of all the potential associations at the same time instead of focusing on each time point and direction in separate models. By simultaneously taking a range of associations into account, the significant predictions of long-term functional outcomes found in the present analysis can be regarded as being independent of changes in other symptom domains. Direct adjustments for the impact of factors such as illness duration, type of antidepressant, educational level, etc. were not made in the SEM because the analyses were adjusted for the immediately preceding SDS, PHQ-9, and PDQ-5 outcomes, which would reflect any effect of other factors. Only a potential interaction between these factors and SDS, PHQ-9, or PDQ-5 remained unadjusted. Such an interaction would imply that one of the factors has a different impact on SDS, PHQ-9, and PDQ-5 outcomes at different time points; however, this is unlikely to be the case for most of the factors. The agreement between the results of the SEM and the G-computation, which controls for potential confounding factors, supports the robustness of our findings.
A potential limitation is that the study recruited only outpatients who were initiating antidepressant monotherapy or switching antidepressant monotherapy for the first time, which may somewhat limit generalization of the study findings. The potential for negative response bias in patients with depression when completing self-report scales must also be recognized. Furthermore, as one of the SDS subscores was not completed by patients who did not work or study for reasons unrelated to the disorder, analyses of SDS total scores are limited to patients who provided scores for all three SDS domains, ie, to the working population only. Regarding the statistical methodology used, more complex SEMs that explored dependencies between the three outcomes from earlier time points than the immediately prior visit generally produced better fit of the data according to the fit indices, but at the expense of clear clinical interpretation and relevance. In terms of clinical utility, it is reasonable to assume that the relationships between depression severity or cognitive symptoms and functioning at the subsequent visit are the most appropriate in terms of relevance for routine practice.
In summary, beyond confirming that depression severity is a major predictor of functional impairment in MDD, this analysis shows that the severity of patient-reported cognitive symptoms is also an independent and important predictor of later functional impairment and generally predicts depression severity at subsequent clinic visits. These findings highlight the importance of recognizing cognitive symptoms in patients with MDD in daily practice and suggest that treatment interventions targeting emotional, physical, and cognitive symptoms could improve functional recovery in this population.
Data availability
The authors declare that the data supporting the findings of this study are available within the article. The authors may be contacted for further data sharing.
Acknowledgments
The authors would like to express their gratitude to all the patients and physicians who participated in the study. The authors also thank Professor Bengt Jönsson (Department of Economics, Stockholm School of Economics, Stockholm, Sweden) and Professor Martin Knapp (Personal Social Services Research Unit, London School of Economics and Political Science, London, UK) for their advice regarding the PERFORM study design and interpretation of the study findings; Inferential for assistance with statistical analyses; and Mapi for logistical management of the study. Writing and editorial assistance was provided by Jennifer Coward of Anthemis Consulting Ltd, funded by H. Lundbeck A/S. The PERFORM study was funded by Lundbeck SAS.
Author contributions
JMH and DS conceived and designed the study, JMH was involved in the acquisition of data, KGL was responsible for data analysis, and JMH, LHH, DS, AE, and KGL contributed to data interpretation. The authors are entirely responsible for the scientific content of the article and all authors contributed substantially to drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
JMH has received honoraria for being an advisor or providing educational talks for Lundbeck, Otsuka, Roche, and Eli Lilly and Company. LHH, AE, and KGL are full-time employees of H. Lundbeck A/S. DS was a full-time employee of Lundbeck SAS at the time the study was conducted. The authors report no other conflicts of interest in this work.
References
1. Kessler RC, Birnbaum HG, Shahly V, et al. Age differences in the prevalence and co-morbidity of DSM-IV major depressive episodes: results from the WHO World Mental Health Survey Initiative. Depress Anxiety. 2010;27:351–364. doi:10.1002/da.20634
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). Washington, DC: American Psychiatric Association; 2013.
3. Kessler RC, Berglund P, Demler O, et al. The epidemiology of major depressive disorder: results from the National Comorbidity Survey Replication (NCS-R). JAMA. 2003;289:3095–3105. doi:10.1001/jama.289.23.3095
4. Srisurapanont M, Hong JP, Tian-Mei S, et al. Clinical features of depression in Asia: results of a large prospective, cross-sectional study. Asia Pac Psychiatry. 2013;5:259–267. doi:10.1111/j.1758-5872.2012.00209.x
5. Lin CH, Yen YC, Chen MC, Chen CC. Depression and pain impair daily functioning and quality of life in patients with major depressive disorder. J Affect Disord. 2014;166:173–178. doi:10.1016/j.jad.2014.03.039
6. IsHak WW, James DM, Mirocha J, et al. Patient-reported functioning in major depressive disorder. Ther Adv Chronic Dis. 2016;7:160–169. doi:10.1177/2040622316639769
7. Kim JM, Chalem Y, di Nicola S, et al. A cross-sectional study of functional disabilities and perceived cognitive dysfunction in patients with major depressive disorder in South Korea: the PERFORM-K study. Psychiatry Res. 2016;239:353–361. doi:10.1016/j.psychres.2016.01.022
8. Sheehan DV, Nakagome K, Asami Y, Pappadopulos EA, Boucher M. Restoring function in major depressive disorder: a systematic review. J Affect Disord. 2017;215:299–313. doi:10.1016/j.jad.2017.02.029
9. Katon W. The impact of depression on workplace functioning and disability costs. Am J Manag Care. 2009;15(11 Suppl):S322–S327.
10. Birnbaum HG, Kessler RC, Kelley D, Ben-Hamadi R, Joish VN, Greenberg PE. Employer burden of mild, moderate, and severe major depressive disorder: mental health services utilization and costs, and work performance. Depress Anxiety. 2010;27:78–89. doi:10.1002/da.v27:1
11. Gelenberg AJ. The prevalence and impact of depression. J Clin Psychiatry. 2010;71:e06. doi:10.4088/JCP.8001tx17c
12. Kessler RC. The costs of depression. Psychiatr Clin North Am. 2012;35:1–14. doi:10.1016/j.psc.2011.11.005
13. Zimmerman M, McGlinchey JB, Posternak MA, et al. How should remission from depression be defined? The depressed patient’s perspective. Am J Psychiatry. 2006;163:148–150. doi:10.1176/appi.ajp.163.12.2189
14. Lam RW, McIntosh D, Wang J, et al; CANMAT Depression Work Group. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder: section 1. Disease burden and principles of care. Can J Psychiatry. 2016;61:510–523. doi:10.1177/0706743716659416
15. Israel JA. Remission in depression: definition and initial treatment approaches. J Psychopharmacol. 2006;20(3 Suppl):5–10. doi:10.1177/1359786806064306
16. Kennedy N, Foy K, Sherazi R, McDonough M, McKeon P. Long-term social functioning after depression treated by psychiatrists: a review. Bipolar Disord. 2007;9:25–37. doi:10.1111/bdi.2007.9.issue-1-2
17. McKnight PE, Kashdan TB. The importance of functional impairment to mental health outcomes: a case for reassessing our goals in depression treatment research. Clin Psychol Rev. 2009;29:243–259. doi:10.1016/j.cpr.2009.01.005
18. Trivedi MH, Morris DW, Wisniewski SR, et al. Increase in work productivity of depressed individuals with improvement in depressive symptom severity. Am J Psychiatry. 2013;170:633–641. doi:10.1176/appi.ajp.2012.12020250
19. IsHak WW, Greenberg JM, Cohen RM. Predicting relapse in major depressive disorder using patient-reported outcomes of depressive symptom severity, functioning, and quality of life in the Individual Burden of Illness Index for Depression (IBI-D). J Affect Disord. 2013;151:59–65. doi:10.1016/j.jad.2013.08.002
20. Naismith SL, Longley WA, Scott EM, Hickie IB. Disability in major depression related to self-rated and objectively-measured cognitive deficits: a preliminary study. BMC Psychiatry. 2007;7:32. doi:10.1186/1471-244X-7-32
21. Lam RW, Kennedy SH, Mclntyre RS, Khullar A. Cognitive dysfunction in major depressive disorder: effects on psychosocial functioning and implications for treatment. Can J Psychiatry. 2014;59:649–654. doi:10.1177/070674371405901206
22. Gonda X, Pompili M, Serafini G, Carvalho AF, Rihmer Z, Dome P. The role of cognitive dysfunction in the symptoms and remission from depression. Ann Gen Psychiatry. 2015;14:27. doi:10.1186/s12991-015-0068-9
23. Conradi HJ, Ormel J, de Jonge P. Presence of individual (residual) symptoms during depressive episodes and periods of remission: a 3-year prospective study. Psychol Med. 2011;41:1165–1174. doi:10.1017/S0033291710001911
24. Evans VC, Iverson GL, Yatham LN, Lam RW. The relationship between neurocognitive and psychosocial functioning in major depressive disorder: a systematic review. J Clin Psychiatry. 2014;75:1359–1370. doi:10.4088/JCP.13r08939
25. Hammer-Helmich L, Haro JM, Jönsson B, et al. Functional impairment in patients with major depressive disorder: the 2-year PERFORM study. Neuropsychiatr Dis Treat. 2018;14:239–249. doi:10.2147/NDT.S146098
26. Bollen KA. Structural Equations with Latent Variables. Hoboken, NJ: John Wiley & Sons, Inc; 1989.
27. Haro JM, Lamy F-X, Jönsson B, et al. Characteristics of patients with depression initiating or switching antidepressant treatment: baseline analyses of the PERFORM cohort study. BMC Psychiatry. 2018;18:80. doi:10.1186/s12888-018-1657-3
28. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606–613. doi:10.1046/j.1525-1497.2001.016009606.x
29. Sullivan MJ, Edgley K, Dehoux E. A survey of multiple sclerosis. Part 1. Perceived cognitive problems and compensatory strategy use. Can J Rehabil. 1990;4:99–105.
30. National Multiple Sclerosis Society. Multiple Sclerosis Quality of Life Inventory: A User’s Manual. New York, NY: The Consortium of Multiple Sclerosis Centers Health Services Research Subcommittee, The National Multiple Sclerosis Society; 1997.
31. Sheehan DV, Harnett-Sheehan K, Raj BA. The measurement of disability. Int Clin Psychopharmacol. 1996;11(Suppl 3):89–95. doi:10.1097/00004850-199606003-00015
32. Sheehan KH, Sheehan DV. Assessing treatment effects in clinical trials with the Discan metric of the Sheehan Disability Scale. Int Clin Psychopharmacol. 2008;23:70–83. doi:10.1097/YIC.0b013e3282f2b4d6
33. Rosseel Y. lavaan: an R package for structural equation modeling. J Stat Softw. 2012;48:1–36. doi:10.18637/jss.v048.i02
34. Robins JM. A new approach to causal inference in mortality studies with sustained exposure periods – application to control of the healthy worker survivor effect. Mathematical Modelling. 1986;7:1393–1512. doi:10.1016/0270-0255(86)90088-6
35. Pearl J. Causal diagrams for empirical research. Biometrika. 1995;82:669–688. doi:10.1093/biomet/82.4.669
36. Snowden JM, Rose S, Mortimer KM. Implementation of G-computation on a simulated data set: demonstration of a causal inference technique. Am J Epidemiol. 2011;173:731–738. doi:10.1093/aje/kwq472
37. Jaeger J, Berns S, Uzelac S, Davis-Conway S. Neurocognitive deficits and disability in major depressive disorder. Psychiatry Res. 2006;145:39–48. doi:10.1016/j.psychres.2005.11.011
38. McIntyre RS, Cha DS, Soczynska JK, et al. Cognitive deficits and functional outcomes in major depressive disorder: determinants, substrates, and treatment interventions. Depress Anxiety. 2013;30:515–527. doi:10.1002/da.22063
39. Bortolato B, Carvalho AF, McIntyre RS. Cognitive dysfunction in major depressive disorder: a state-of-the-art clinical review. CNS Neurol Disord Drug Targets. 2014;13:1804–1818. doi:10.2174/1871527313666141130203823
40. Woo YS, Rosenblat JD, Kakar R, Bahk W-M, McIntyre RS. Cognitive deficits as a mediator of poor occupational function in remitted major depressive disorder patients. Clin Psychopharmacol Neurosci. 2016;14:1–16. doi:10.9758/cpn.2016.14.1.1
41. Saragoussi D, Christensen MC, Hammer-Helmich L, Rive B, Touya M, Haro JM. Long-term follow-up on health-related quality of life in major depressive disorder: a 2-year European cohort study. Neuropsychiatr Dis Treat. 2018;14:1339–1350. doi:10.2147/NDT
42. Adler DA, McLaughlin TJ, Rogers WH, et al. Job performance deficits due to depression. Am J Psychiatry. 2006;163:1569–1576. doi:10.1176/appi.ajp.163.12.2189
43. Rytsälä HJ, Melartin TK, Leskelä US, et al. Determinants of functional disability and social adjustment in major depressive disorder: a prospective study. J Nerv Ment Dis. 2006;194:570–576. doi:10.1097/01.nmd.0000230394.21345.c4
44. Iosifescu DV. The relation between mood, cognition and psychosocial functioning in psychiatric disorders. Eur Neuropsychopharmacol. 2012;22(Suppl 3):S499–S504. doi:10.1016/j.euroneuro.2012.08.002
45. Hasselbalch BJ, Knorr U, Kessing LV. Cognitive impairment in the remitted state of unipolar depressive disorder: a systematic review. J Affect Disord. 2011;134:20–31. doi:10.1016/j.jad.2010.11.011
46. Ladegaard N, Videbech P, Lysaker PH, Larsen ER. The course of social cognitive and metacognitive ability in depression: deficits are only partially normalized after full remission of first episode major depression. Br J Clin Psychol. 2016;55:269–286. doi:10.1111/bjc.2016.55.issue-3
47. Saragoussi D, Touya M, Haro JM, et al. Factors associated with failure to achieve remission and with relapse after remission in patients with major depressive disorder in the PERFORM study. Neuropsychiatr Dis Treat. 2017;13:2151–2165. doi:10.2147/NDT.S136343
48. McIntyre RS, Harrison J, Loft H, Jacobson W, Olsen CK. The effects of vortioxetine on cognitive function in patients with major depressive disorder: a meta-analysis of three randomized controlled trials. Int J Neuropsychopharmacol. 2016;10:pyw055. doi:10.1093/ijnp/pyw055
49. Baune BT, Brignone M, Larsen KG. A network meta-analysis comparing effects of various antidepressant classes on the digit symbol substitution test (DSST) as a measure of cognitive dysfunction in patients with major depressive disorder. Int J Neuropsychopharmacol. 2018;21:97–107. doi:10.1093/ijnp/pyx070
50. Chokka P, Bougie J, Rampakakis E, Proulx J. Assessment in Work Productivity and the Relationship with Cognitive Symptoms (AtWoRC): primary analysis from a Canadian open-label study of vortioxetine in patients with major depressive disorder (MDD). CNS Spectr. 2019;24:338–347. doi:10.1017/S1092852918000913
51. Chokka P, Bougie J, Proulx J, Holmegaard Tvistholm A, Ettrup A. Long-term functioning outcomes are predicted by cognitive symptoms in working patients with major depressive disorder treated with vortioxetine: results from the AtWoRC study. CNS Spectr. 2019;1–12. [Epub ahead of print]. doi:10.1017/S1092852919000786
52. Andresen R, Caputi P, Oades LG. Do clinical outcome measures assess consumer-defined recovery? Psychiatry Res. 2010;177:309–317. doi:10.1016/j.psychres.2010.02.013
53. IsHak WW, Mirocha J, Pi S, et al. Patient-reported outcomes before and after treatment of major depressive disorder. Dialogues Clin Neurosci. 2014;16:171–183.
54. Slade M, Longden E. Empirical evidence about recovery and mental health. BMC Psychiatry. 2015;15:285. doi:10.1186/s12888-015-0678-4
55. Oliveira-Maia AJ, Mendonça C, Pessoa MJ, Camacho M, Gago J. The mental health recovery measure can be used to assess aspects of both customer-based and service-based recovery in the context of severe mental illness. Front Psychol. 2016;7:1679. doi:10.3389/fpsyg.2016.01679
56. Fava M, Mahableshwarkar AR, Jacobson W, et al. What is the overlap between subjective and objective cognitive impairments in MDD? Ann Clin Psychiatry. 2018;30:176–184.
57. Lovera J, Bagert B, Smoot KH, et al. Correlations of perceived deficits questionnaire of multiple sclerosis quality of life inventory with Beck depression inventory and neuropsychological tests. J Rehabil Res Dev. 2006;43:73–82. doi:10.1682/JRRD.2004.09.0118
58. Mahableshwarkar AR, Zajecka J, Jacobson W, Chen Y, Keefe RS. A randomized, placebo-controlled, active-reference, double-blind, flexible-dose study of the efficacy of vortioxetine on cognitive function in major depressive disorder. Neuropsychopharmacology. 2015;40:2025–2037. doi:10.1038/npp.2015.52
59. Lam RW, Lamy FX, Danchenko N, et al. Psychometric validation of the Perceived Deficits Questionnaire-Depression (PDQ-D) instrument in US and UK respondents with major depressive disorder. Neuropsychiatr Dis Treat. 2018;14:2861–2877. doi:10.2147/NDT.S175188
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.