Back to Journals » Eye and Brain » Volume 6 » Issue 1
Parasitic diseases of zoonotic importance in humans of northeast India, with special reference to ocular involvement
Authors Das D , Islam S, Bhattacharjee H, Deka A, Yambem D, Tahiliani P, Deka P, Bhattacharyya P, Deka S, Das K, Bharali G, Deka A, Paul R
Received 28 May 2014
Accepted for publication 29 July 2014
Published 22 September 2014 Volume 2014:6(1) Pages 1—8
DOI https://doi.org/10.2147/EB.S64404
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Margaret Wong-Riley
Dipankar Das,1 Saidul Islam,2 Harsha Bhattacharjee,1 Angshuman Deka,1 Dinakumar Yambem,1 Prerana Sushil Tahiliani,1 Panna Deka,1 Pankaj Bhattacharyya,1 Satyen Deka,1 Kalyan Das,1 Gayatri Bharali,1 Apurba Deka,1 Rajashree Paul1
1Sri Sankaradeva Nethralaya, Guwahati, 2Department of Parasitology, College of Veterinary Science, Assam Agricultural University, Guwahati, Assam, India
Abstract: Parasitic zoonotic diseases are prevalent in India, including the northeastern states. Proper epidemiological data are lacking from this part of the country on zoonotic parasitic diseases, and newer diseases are emerging in the current scenario. Systemic manifestation of such diseases as cysticercosis, paragonimiasis, hydatidosis, and toxoplasmosis are fairly common. The incidence of acquired toxoplasmal infection is showing an increasing trend in association with acquired immunodeficiency syndrome. Among the ocular parasitic diseases, toxoplasmosis, cysticercosis, toxocariasis, dirofilariasis, gnathostomiasis, hydatidosis, amebiasis, giardiasis, etc, are the real problems that are seen in this subset of the population. Therefore, proper coordination between various medical specialities, including veterinary science and other governing bodies, is needed for better and more effective strategic planning to control zoonoses.
Keywords: zoonoses, regional infections, toxoplasmosis, cysticercosis, toxocariasis, hydatidosis
Introduction
India is the second-most densely inhabited and seventh-largest country in the world, and there is a lot of variation in the geographic areas, ethnicity, religion, food habits, personal behavior, level of education, and standard of living.1 The recent census of the country showed that 72.2% of the population lived in rural areas, with over 70% of this population owning livestock, which play an important role in the causation of zoonotic diseases in humans. Parasitic zoonoses affect human as well as animal health directly and indirectly, which may affect the socioeconomic condition of the country as a whole. Poor economic conditions, sanitation, water supply, and personal habits are some of the causes on the Indian subcontinent of harboring the zoonoses, particularly in the northeast part of India.1 This paper focuses on important parasitic zoonoses in Assam and other northeastern states of India.
Toxoplasmosis
Toxoplasmosis is a common disease in both mammals and birds.2 The disease is caused by the obligate intracellular protozoan Toxoplasma gondii. Intermediate hosts include sheep, goats, pigs, and humans. Oocysts of T. gondii are uniquely found in the intestinal mucosa of cats (definitive hosts). Once they are released, they spread to humans and other animals through a variety of vectors. Toxoplasmosis in the central nervous system is nowadays seen in acquired immunodeficiency syndrome (AIDS) patients in India.3
Serum IgA titers in enzyme-linked immunosorbent assays (ELISAs) are positive in chronic toxoplasmosis, whereas IgM is positive in acute infection. Therefore, the titers for both IgG and IgM are important, and need to be strongly correlated with clinical findings. In a national serological survey in India,4 a total of 23,094 serum samples were tested for antibodies to the parasite. IgG and IgM antibodies were found to be positive in 24.3% and 2% of the samples, respectively. The lowest seroprevalence value was obtained in north India, while it was highest in the south.4 The data probably indicate the effect of significantly drier conditions in the north, leading to reduced survivability of T. gondii oocysts.
Our previous study,5 conducted in a tertiary eye-care center in one of the northeast states (Assam) of India, showed 37 cases of toxoplasmal uveitis (40.21%) in 1 year compared to 27.87% by Biswas et al6 from south India. Out of the 37 cases, 14 were acquired, while 23 cases were congenital toxoplasmal retinochoroidal lesions. Serum IgG only was positive in 30 of 37 cases and IgM only in three cases, while both serum IgG and IgM positivity was observed in four cases. Chorioretinitis was the most common presentation, seen in about 95.6% of cases. T. gondii tends to affect the macula due to the high oxygen content owing to the end-arterial system of the retinal circulation. Retinochoroiditis is a more appropriate term for acute inflammation, as the organism first lodges in the retina followed by the choroid.
The prevalence of toxoplasmosis in India shows a wide variation. One study showed as high as 77% in women in the reproductive age-group.7 There is a report of this parasitic infection among pregnant women in northeast India. In a study done by Borkakoty et al, the seroprevalence of T. gondii infection was observed to be 44.6% and 36.8% among pregnant women with and without history of pregnancy wastage, respectively.8 A higher prevalence of T. gondii infection has been observed in women belonging to the lower socioeconomic class.8 It has also been observed that the seroprevalence of T. gondii in humans in India is lower compared to their Western counterparts. This may be due to a preference for dogs as pets rather than cats in the Western population.
The majority of reported cases of ocular toxoplasmosis are congenital. The clinical manifestations of congenital ocular toxoplasmosis in infants include microphthalmia, enophthalmos, ptosis, nystagmus, choroidal coloboma, and strabismus.9,10 Posterior uveitis is the most common manifestation, and presents as necrotizing retinitis usually adjacent to a larger atrophic retinochoroidal scar, which is often located in the macula in congenital cases. The critical sign of acquired active toxoplasmosis is a whitish yellow retinal lesion associated with hazy vitreous as a result of the presence of vitreous cells (“headlight in the fog” appearance). Old retinochoroidal scars can often be seen adjacent to the new whitish yellow lesion. The areas of retinitis are the result of tissue-cyst bursting and releasing bradyzoites that transform into tachyzoites, which in turn invade the neighboring cells. These destructive lesions are usually larger than one disk diameter, and appear as soft, white, fluffy infiltrates surrounded by retinal edema with choroiditis.9–11 When the tachyzoites come under increasing attack by the host’s immune response, they gradually transform back into inflammatory exudates, and are frequently present around the retinal vessels adjacent to an area of active inflammation. Chorioretinal scars are occasionally found in uninvolved areas. Patients often complain of blurred vision, floaters, photophobia, and pain in the affected eye. Occasionally, the initial presentation would be severe unilateral papillitis, macular hard exudates distributed in star-like fashion, and vitreal inflammation simulating neuroretinitis. Multifocal active toxoplasmosis simultaneously involving both the retina and the optic nerve is unusual, but was documented in an immunocompromised patient with human immunodeficiency virus (HIV) infection. Differential diagnosis of toxoplasmal lesions could be tuberculous granuloma, fungal endophthalmitis, sarcoid, syphilitic retinitis, cytomegalovirus infection, intraocular lymphoma, and toxocariasis.
Histopathologically, the toxoplasmal lesion shows necrosis of the involved retina, with destruction of retinal architecture and underlying choroid. Since the parasite has a propensity for attacking the neural tissue, the trophozoites and the cysts are usually found in the superficial layers of the retina within the areas of necrosis. The infiltrate consists predominantly of lymphocytes, macrophages, and epithelioid cells, with plasma cells found in the periphery of the lesion.9,11,12 Cell-mediated immunity is said to be the major defense mechanism against toxoplasmal infection. In patients with ocular toxoplasmosis, the cellular immune responses appear to be directed predominantly against surface protein P22. However, some evidence also indicates that part of the disease may be mediated by autoimmune mechanism directed against certain retinal antigens.12 The role of humoral response in toxoplasmosis remains unclear.
Ocular toxoplasmosis can be frequently diagnosed clinically on the basis of its characteristic retinochoroidal scar and inflammation. Serological testing confirms the clinical diagnosis. There may be a history of eating undercooked or raw meat or being exposed to cats (source of acquired infection). The most common presentations of acquired toxoplasmosis are lymphadenopathy, fever, headache, malaise, pharyngitis, fatigue, and night sweats.13
In the case of congenital toxoplasmosis, inquiry about previous maternal abortion, birth history, history of seizures, etc, are important. Complete ocular examinations, including retinal evaluation, are also of paramount importance in these cases. All patients are required to be seen by a physician for any remote lesion elsewhere in the body.
Laboratory investigations include ELISA for toxoplasmosis (IgG, IgM, and IgA). This should show a positive titer from the current or any previous infection. However, a negative titer on any dilution does not exclude the diagnosis. IgM is found within 2–6 months of initial infection, after which only IgG remains positive. A 1:1 dilution should be done, as only a positive result is necessary in the setting of classical fundus findings. An HIV test should be considered in any of the atypical cases or when the patient is a high-risk candidate for AIDS. Anterior-chamber paracentesis for toxoplasmosis may be done where a polymerase chain reaction (PCR) facility is available. This can be a useful tool in the diagnosis of atypical toxoplasmosis cases, including the determination of local antibody production in aqueous humor. To demonstrate intraocular anti-T. gondii antibody production, total IgG amount and specific anti-T. gondii IgG titers are determined with each serum and aqueous humor sample. Total IgG concentrations are measured by nephelometry, and specific T. gondii IgG titers by highly sensitive agglutination tests. Recently, amplification of ribosomal ribonucleic acid gene of T. gondii from aqueous samples with PCR was found to be an important tool for the diagnosis of toxoplasmosis.9
The main aim of treatment is to stop the multiplication of tachyzoites during episodes of active retinochoroiditis. The disease is self-limited in immunocompetent patients. Mid-peripheral retinochoroiditis may not require treatment. Some congenital cases with bilateral macular healed toxoplasmal scars require squint clinic evaluation, low-vision aid, and regular follow-up. If there are complaints of headache or a suspicion of a neurological problem, skull X-ray or a computed tomography scan of brain is advised so as to look for any intracranial calcification, and further neurologist consultation and follow-up may be required.9
Measures for the prevention of toxoplasmosis are primarily directed toward the prevention of primary infection. Meat should be cooked up to 60°C for at least 15 minutes or frozen to below −20°C for at least 24 hours to destroy the cysts. Any contact with cat feces should be avoided. Hand washing after touching uncooked meat and after contact with cats or soil that could be contaminated with cat feces should be practiced to avoid infection. Consumption of raw eggs and nonpasteurized milk, particularly goat’s milk, should be avoided. Fruits and vegetables should be adequately washed before ingestion. Daily cleaning of the cat-litter box is also essential in order to prevent sporulation. For females, only nonpregnant women should perform this duty. Blood transfusions and organ transplants from seropositive individuals should be avoided if the recipient is seronegative.9–12
Treatment should be considered if a lesion is at or near the optic disk (within two disk diameters), a lesion is within the temporal arcade, a lesion threatens the large retinal vessel, a lesion has induced a substantial hemorrhage, a lesion with an intense inflammatory reaction, extensive chronic exudative lesions regardless of location, and in cases of severe vitreous haze, loss of two lines in visual acuity, persistence of inflammation for more than a month, congenital toxoplasmal retinochoroiditis in the first year of life, newborn diagnosed as congenital toxoplasmosis regardless of the presence of ocular lesions, and any lesion in an immunocompromised host.9
Presently, newer drugs with fewer side effects are being tried in ocular toxoplasmosis, but their long-term results need to be further evaluated. Treatment options include a combination of pyrimethamine, sulfadiazine, and folinic acid with or without clindamycin. Trimethoprim and sulfamethoxazole combination is also another combination that is being tried in developing countries.9–11 Monotherapy with azithromycin and clindamycin is another good therapeutic option, though recurrences have been seen with it.9–11 Oral prednisolone is added to these regimens to counter the inflammatory reaction. Topical corticosteroids are used for anterior uveitis, but periocular injections are contraindicated to avoid local immunosuppression and uncontrollable disease. Laser photocoagulation, cryotherapy, and vitrectomy are used as adjunctive treatment modalities. Spiramycin is the treatment of choice in pregnant women with toxoplasmosis.9–11
Cysticercosis
The prevalence of taeniasis in humans varies from 0.75% to 1.0% in certain communities, particularly in rural areas, where there is more contact with pigs.1,14 Taeniasis is more common in areas with poor hygiene. In India, neurocysticercosis is an important emerging disease of the central nervous system, second to tuberculosis.1 Neurocysticercosis is also emerging as one of the principal causes of epilepsy. The occurrence of Cysticercus cellulosae in muscles of pigs is very well established, ranging from 3% to 26%.1,15,16 In India, little information on Taenia saginata is available, due to a ban on the slaughter of cows and the majority of the population not consuming beef on religious grounds. Cysticercosis can also infect the eye. Intraocular cysticercosis management is somewhat different from the neurocysticercosis and orbital or ocular adnexal cysticercosis.12
Humans are definitive hosts, and pigs are the intermediate hosts of Taenia solium.17 In cysticercosis, humans become intermediate hosts by ingesting the eggs of T. solium from contaminated food and water. After entering the intestinal wall, the embryo enters the bloodstream and can lodge in various organs, such as the central nervous system, eye, skeletal muscles, and subcutaneous tissue.18,19 Autoinfections can also occur in some cases. Ocular involvement can be destructive, because cysticercus lesions gradually increase in size, leading to blindness in 3–5 years. The death of the parasite causes the release of toxic substances, leading to intense inflammatory reactions and eye damage.17–19 Initial medical therapy of intraocular cysticercosis with antihelminthic drugs like albendazole or praziquantel is not known to be very effective. Early surgical removal of the parasite is the treatment of choice.17–19 Neurocysticercosis is also a worldwide problem, and thus it needs serious attention.17 Clinical, imaging (Figure 1), and pathological findings of cysticercosis patients presenting in our center (a tertiary eye care center in Assam) have been documented. Orbital cysticercosis is also seen in patients from northeast India.
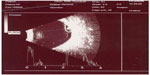  | Figure 1 B-scan ultrasound showing cysticercus lesion with a scolex inside the cyst. |
Toxocariasis
Toxocara canis infection is typically found in children. The average age of diagnosis is 7.5 years.2 T. canis is an ascarid that can only complete its life cycle in dogs. It is a ubiquitous parasite found worldwide, including India. Ocular involvement can present as retinal peripheral granuloma (Figure 2), posterior-pole granuloma, or endophthalmitis.2 Its systemic manifestations include fever, dry or hacking cough, hepatosplenomegaly, and neurological symptoms like convulsions. ELISA is the most reliable test, but is not commonly available in India. Intraocular manifestations respond well to steroids.2 Vitreoretinal surgery has also been suggested as an effective method by which the secondary effects of toxocariasis can be managed. Various authors have tried to address the retinal complications of this disorder by reattaching the retina subsequently.2 Often, granulomas removed completely show atypical toxocaral lesions.2 Results of ELISAs performed on the vitreous samples are strongly positive. Photocoagulation and cryotherapy have also been tried in some cases to kill the organisms.2
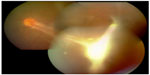  | Figure 2 Toxocara lesion in the left fundus. |
Gnathostomiasis
Gnathostomiasis is a rare parasitic disease in humans, who become incidental hosts after the intake of undercooked or raw meat of definitive hosts, such as cats, dogs and wild animals.20 Second intermediate hosts may be freshwater fish, chickens, snails, frogs, or paratenic hosts like birds. After ingestion, the larvae cross the gastric and intestinal wall and migrate indiscriminately throughout the body.20 Clinical manifestations of gnathostomiasis occur due to migration of parasitic larvae and host response to the toxins. We have come across two cases of intraocular gnathostomiasis in our center, one of which was in the vitreous cavity, while the other was seen in the iris tissue. The first was published, and in both the cases the live worm was retrieved (Figure 3).20
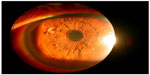  | Figure 3 Parasite (Gnathostoma spp.) in the iris of the right eye. |
Gnathostomiasis is most prevalent in Southeast Asia and Latin America, with an increasing global trend. Travel patterns, food habits, and aquaculture for tourism involving hunting and fishing may be responsible for this rise in the global incidence. Ocular gnathostomiasis may involve the eyelids, conjunctiva, cornea, anterior chamber, uvea, or vitreous.20–22 The commonest ocular clinical manifestation is anterior uveitis.20,23 Consumption of smoked or undercooked fish may have contributed to the occurrence of the disease in this particular region of India.20
Dirofilariasis
Dirofilariasis is a zoonotic disease seen in different parts of the world.24–26 The worm may get lodged in various tissues of the human body, including the eye and ocular adnexa.24–32 There may be deep-seated infection, such as in the lungs, where early and proper diagnosis can save a patient from major complications.24–27 Dirofilariasis infections are often reported in European, Mediterranean, and Asian countries.24–27 There are few reports of ocular dirofilariasis from India.30 We have had five cases of unpublished ocular dirofilariasis. In three cases, the worm was extracted from the conjunctiva, and once each from the lower lid and the anterior chamber (Figure 4).
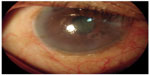  | Figure 4 Live parasite in the anterior chamber of the right eye (Dirofilaria). |
Hydatidosis
Echinococcosis or hydatidosis is an infection caused by the larval form of Echinococcus granulosus, and is a significant public health problem. In India, there is an alarming increase of this infection, mostly in north India.13 The presence of stray dogs and dead carcasses plays an important role in the transmission of this disease in our country.1,33 However, it has been observed that ocular involvement of hydatidosis is not very common in this part of the country. Nicholson et al reported seven cases of hydatid cysts of the orbit.34 We have also seen and documented cases of hydatid disease in the eye and ocular adnexa. Three cases of orbital hydatidosis have been seen in our center. An interesting observation in the histopathology of the cyst wall was made. It showed multiple pores in the laminated structures of the cyst wall. We believe these pores to be the route of passage of fluid in and out of the cyst.
Thelaziasis
Ocular involvement of thelaziasis infection is rare. The first human case of Thelazia callipaeda infection was reported from Vietnam.35 This nematode is a zoonotic parasite in the eyes, as implied by its name “Oriental eye worm” or “eye worm”.35 It was reported for the first time in a dog from Pakistan in 1910. Subsequently, thelaziasis cases have been reported from the People’s Republic of China, France, Germany, India, Indonesia, Italy, Japan, South Korea, the Netherlands, Russia, Switzerland, Taiwan, Myanmar, and Thailand.36 The adult worm is parasitic in the conjunctival sac of a host, and produces larvae continuously. When flies lick the tear in the eye of the final host, such as humans, the larvae enter the conjunctival sac and become adult in 1 month. Symptoms of T. callipaeda infection include excessive watering, visual impairment, ulcers, and scarring of the cornea.37,38 We have also seen cases of ocular adnexal thelaziasis infection (Figures 5 and 6) in our center.
  | Figure 5 Thelazia retrieved from the conjunctival sac. |
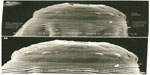  | Figure 6 Scanning electron microscopy ([A] 1400× and [B] 1900×) showing the proximal part of Thelazia. |
Amebiasis and giardiasis
Human infection due to Entamoeba histolytica has been reported in India. A study showed many animals to be potential reservoirs of infection. Although it mainly affects the gastrointestinal tract, intraocular inflammatory disease has also been associated with it.39 It is diagnosed by identifying the cysts and the trophozoites. Acanthamoeba spp. are also known to affect the eyes, particularly corneas. Giardiasis intestinalis is common in children and occasionally in HIV-infected individuals. Ocular manifestations of giardiasis include iridocyclitis,40 chorioretinitis,41 and “salt and pepper” fundus.41 Diagnosis of giardiasis can be made from the stool sample, while in some cases jejunal biopsy may be required. Quinacrine hydrochloride or metronidazole is effective for the treatment for giardiasis.
Larva migrans, diffuse unilateral subacute neuroretinitis
More than 200 million people are affected by hookworms in India.1,2 Both cutaneous larva migrans and visceral larva migrans have been reported throughout the country. They are caused by Ancylostoma caninum and A. braziliense. However, A. ceylanicum was detected in dogs living among the tea-growing community. This may lead to an endemicity of the parasite in northeast India.1,2,42 A rare eye condition called diffuse unilateral subacute neuroretinitis has been described, which is very rarely seen in the retina and choroid. A. caninum and Baylisascaris procyonis (a nematode found in raccoon) were found to be associated with it.43
Cryptosporidiosis
Cryptosporidiosis has been reported worldwide. Cryptosporidium parvum, which is associated with diarrhea in children, has also been reported from various parts of India.44,45 Ocular manifestation, which is rare, occurs in the form of conjunctivitis. It is seen in urban slum areas in patients with diarrhea. It is also an opportunistic infection in HIV-infected individuals.
Conclusion
There are various factors that favor zoonoses in northeastern states of India. Increased vector populations, poverty, food habits, large numbers of stray animals, poor personal hygiene, and lack of awareness of the diseases are some of the factors responsible. It has been observed that certain zoonotic diseases are showing a rising trend, such as toxoplasmosis and gnathostomiasis. A systematic study on zoonoses is required to make the final comment on these observations. Health education, vector control, control of animal movements, control over roadside slaughter of animals, safe water supply, food hygiene, and socioeconomic development are essential in order to prevent these zoonotic diseases. Proper diagnosis in time may even save vision and lives. Therefore, proper coordination between various medical specialities, including veterinary science and other governing bodies, is needed for better and more effective strategic plans to control zoonoses.
Acknowledgments
Sri Kanchi Sankara Health and the Educational Foundation provided financial support for this article. The authors would like to thank the Department of Oculoplasty and Medical Retina, Sri Sankaradeva Nethralaya and Drs Himadri Shekar Das and Pradip Hatimota, radiologists, Matrix Imaging, Guwahati, India. This was presented as a lead paper at the National Symposium and XXIII National Congress of Veterinary Parasitology, December 12–14, 2012, in Guwahati, India.
Disclosure
The authors report no conflicts of interest in this work.
References
Singh BB, Sharma R, Sharma JK, Juyal PD. Parasitic zoonoses in India: an overview. Rev Sci Tech. 2010;29:629–637. | |
Nussenblatt RB, Whitcup SM. Uveitis: Fundamentals and Clinical Practice. 3rd ed. Amsterdam: Elsevier; 2004. | |
Mathew MJ, Chandy MJ. Central nervous system toxoplasmosis in acquired immunodeficiency syndrome: an emerging disease in India. Neurol India. 1999;47:182–187. | |
Dhumne M, Sengupta C, Kadival G, Rathinaswamy A, Velumani A. National seroprevalence of Toxoplasma gondii in India. J Parasitol. 2007;93:1520–1521. | |
Das D, Bhattacharjee H, Bhattacharyya PK, et al. Pattern of uveitis in North East India: a tertiary eye care centre study. Indian J Ophthalmol. 2009;57:144–146. | |
Biswas J, Narain S, Das D, Ganesh SK. Pattern of uveitis in a referral uveitis clinic in India. Int Ophthalmol. 1996–1997;20:223–228. | |
Singh S, Nautiyal BL. Prevalence of toxoplasmosis in Kumaon region of India. Indian J Med Res. 1991;93:247–249. | |
Borkakoty BJ, Borthakur AK, Gohain M. Prevalence of Toxoplasma gondii infection amongst pregnant women in Assam, India. Indian J Med Microbiol. 2007;25:431–432. | |
Jain L, Das D, Bhattacharjee H, Jain G. Ocular toxoplasmosis. Delhi J Ophthalmol. 2009;15:19–23. | |
De Jong PT. Ocular toxoplasmosis: common and rare symptoms and signs. Int Ophthalmol. 1989;13:391–397. | |
Jabs DA. Ocular toxoplasmosis. Int Ophthalmol Clin. 1990;30:264–270. | |
Nussenblatt RB, Mittal KK, Fuhrman S, Sharma SD, Palestine AG. Lymphocyte proliferative responses of patients with ocular toxoplasmosis to parasite and retinal antigens. Am J Ophthalmol. 1989;107:632–641. | |
Zimmerman LE. Ocular pathology of toxoplasmosis. Surv Ophthalmol. 1961;6:832–856. | |
Singh GS, Prabhakar S, Cho SY, et al. Taenia solium taeniasis and cysticercosis in Asia. In: Singh GS, Prabhakar S, editors. Taenia solium Cysticercosis: From Basic to Clinical Science. Oxford: CABI; 2002:111–128. | |
D’Souza P. Studies on porcine cysticercosis with special reference to serodiagnosis. J Vet Parasitol. 1998;12:64. | |
Prasad KN, Chawla S, Jain D, et al. Human and porcine Taenia solium in rural north India. Trans R Soc Trop Med Hyg. 2002;96:515–516. | |
Das D, Deka S, Islam S, et al. Neuro and intraocular cysticercosis: a clinicopathological case report. Eye Brain. 2010;2:39–42. | |
Sharma T, Sinha S, Shah N, et al. Intraocular cysticercosis: clinical characteristics and visual outcome after vitreoretinal surgery. Ophthalmology. 2003;110:996–1004. | |
Gemolotto G. A contribution to surgical treatment of intraocular cysticercosis. Arch Ophthalmol. 1955;59:365–368. | |
Bhattacharjee H, Das D, Medhi J. Intravitreal gnathostomiasis and review of literature. Retina. 2007;27:67–73. | |
Bathrick ME, Mango CA, Mueller JF. Intraocular gnathostomiasis. Ophthalmology. 1981;88:1293–1295. | |
Biswas J, Gopal L, Sharma T, Badrinath SS. Intraocular Gnathostoma spinigerum: clinicopathologic study of two cases and review of literature. Retina. 1994;14:438–444. | |
Basak SK, Sinha TK, Bhattacharya D, Hazra TK, Parikh S. Intravitreal live Gnathostoma spinigerum. Indian J Ophthalmol. 2004;52:57–58. | |
Mittal M, Sathish KR, Bhatia PG, Chidamber BS. Ocular dirofilariasis in Dubai, UAE. Indian J Ophthalmol. 2008;56:325–326. | |
Logar J, Novsak V, Rakovec S, Stanisa O. Subcutaneous infection caused by Dirofilaria repens imported to Slovenia. J Infect. 2001;42:72–74. | |
Stringfellow GJ1, Francis IC, Coroneo MT, Walker J. Orbital dirofilariasis. Clin Experiment Ophthalmol. 2002;30:378–380. | |
Hira PR, Madda JP, al-Shamali MA, Eberhard ML. Dilofilariasis in Kuwait. first report of human infection due to Dirofilaria repens in the Arabian Gulf. Am J Trop Med Hyg. 1994;51:590–592. | |
Beaver PC. Intraocular filariasis: a brief review. Am J Trop Med Hyg. 1989;40:40–45. | |
Pampiglione S, Rivasi F. Human dirofilariasis due to Dirofilaria (Nochtiella) repens: an update of world literature from 1995 to 2000. Parassitologica. 2000;42:231–254. | |
Sekhar HS, Srinivasa H, Batru RR, Mathai E, Shariff S, Macaden RS. Human ocular dirofilariasis in Kerala Southern India. Indian J Pathol Microbiol. 2000;43:77–79. | |
Font RL, Neafie RC, Perry HD. Subcutaneous dirofilariasis of the eyelid and ocular adnexa: report of 6 cases. Arch Ophthalmol. 1980;98:1079–1082. | |
Jariya P, Sucharit S. Dirofilaria repens from the eyelid of a woman in Thailand. Am J Trop Med Hyg. 1983;32:1456–1457. | |
Khurana S, Das A, Malla N. Increasing trends in seroprevalence of human hydatidosis in North India: a hospital-based study. Trop Doct. 2000;37:100–102. | |
Nicholson M, Kashyap VG, Honavar SG, Somasheila MI. Hydatid disease of the orbit – lesson forgotten!. Poster presented at: 70th All India Ophthalmological Conference; February 2–5, 2012; Cochin, India. | |
Otranto D, Lia RP, Buono V, Traversa D, Giangaspero A. Biology of Thelazia callipaeda (Spirurida, Thelaziidae) eyeworms in naturally infected definitive hosts. Parasitology. 2004;129:627–633. | |
Nguyen VD, Thanh HL, Chai JY. The first human case of Thelazia callipaedia infection in Vietnam. Korean J Parasitol. 2012;50:221–223. | |
Zakir R, Zhong-Xia Z, Chioddini P, Canning CR. Intraocular infestation with the worm, Thelazia callipaeda. Br J Ophthalmol. 1999;83:1194–1195. | |
Mahanta J, Alger J, Bordoloi P. Eye infestation with Thelazia species. Indian J Ophthalmol. 1996;44:99–101. | |
Rodger FC, Chir PK, Hosain AT. Night blindness in the tropics. Arch Ophthalmol. 1960;63:927–935. | |
Knox DL, King JJ. Retinal arteritis, iridocyclitis, and giardiasis. Ophthalmology. 1982;89:1303–1308. | |
Pettoello Mantovani M, Giardino I, Magli A, di Martino L, Guandalini S. Intestinal giardiasis associated with ophthalmologic changes. J Pediatr Gastroenterol Nutr. 1990;11:196–200. | |
Traub RJ, Robertson ID, Irwin P, Mencke N, Monis P, Thompson RC. Humans, dogs, and parasitic zoonoses – unravelling the relationship in remote endemic community in northeast India using molecular tools. Parasitol Res. 2003;90 Suppl 3:S156–S157. | |
Kazacos KR, Vestre WA, Kazacos EA, Raymond LA. Diffuse unilateral subacute neuroretinitis syndrome: a probable cause. Arch Ophthalmol. 1984;102:967–968. | |
Das P, Roy SS. Molecular characterization of Cryptosporodium spp. from children in Kolkata. India J Clin Microbiol. 2006;44:4246–4249. | |
Panja SC. Emerging parasitic zoonoses in India. Poster presented at: 5th All India Annual Conference of the Association of Public Health Veterinarians; December 21–23, 2002; Andhra Pradesh, India. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
