Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 9 » Issue 1
Paradoxical association between body mass index and in-hospital mortality in elderly patients with chronic obstructive pulmonary disease in Japan
Authors Yamauchi Y, Hasegawa W, Yasunaga H , Sunohara M, Jo T , Takami K, Matsui H, Fushimi K, Nagase T
Received 30 September 2014
Accepted for publication 17 November 2014
Published 9 December 2014 Volume 2014:9(1) Pages 1337—1346
DOI https://doi.org/10.2147/COPD.S75175
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Richard Russell
Yasuhiro Yamauchi,1,2 Wakae Hasegawa,1 Hideo Yasunaga,3 Mitsuhiro Sunohara,1 Taisuke Jo,1,2 Kazutaka Takami,1 Hiroki Matsui,3 Kiyohide Fushimi,4 Takahide Nagase1
1Department of Respiratory Medicine, Graduate School of Medicine, The University of Tokyo, Tokyo, Japan; 2Division for Health Service Promotion, The University of Tokyo, Tokyo, Japan; 3Department of Clinical Epidemiology and Health Economics, School of Public Health, The University of Tokyo, Tokyo, Japan; 4Department of Health Policy and Informatics, Tokyo Medical and Dental University, Graduate School of Medicine, Tokyo, Japan
Background and objective: The prevalence and mortality of chronic obstructive pulmonary disease (COPD) in elderly patients are increasing worldwide. Low body mass index (BMI) is a well-known prognostic factor for COPD. However, the obesity paradox in elderly patients with COPD has not been well elucidated. We investigated the association between BMI and in-hospital mortality in elderly COPD patients.
Methods: Using the Diagnosis Procedure Combination database in Japan, we retrospectively collected data for elderly patients (>65 years) with COPD who were hospitalized between July 2010 and March 2013. We performed multivariable logistic regression analysis to compare all-cause in-hospital mortality between patients with BMI of <18.5 kg/m2 (underweight), 18.5–22.9 kg/m2 (low–normal weight), 23.0–24.9 kg/m2 (high–normal weight), 25.0–29.9 kg/m2 (overweight), and ≥30.0 kg/m2 (obesity) with adjustment for patient backgrounds.
Results: In all, 263,940 eligible patients were identified. In-hospital mortality was 14.3%, 7.3%, 4.9%, 4.3%, and 4.4%, respectively, in underweight, low–normal weight, high–normal weight, overweight, and obese patients. Underweight patients had a significantly higher mortality than low–normal weight patients (odds ratio [OR]: 1.55, 95% confidence interval [CI]: 1.48–1.63), whereas lower mortality was associated with high–normal weight (OR: 0.76, CI: 0.70–0.82), overweight (OR: 0.73, CI: 0.66–0.80), and obesity (OR: 0.67, CI: 0.52–0.86). Higher mortality was significantly associated with older age, male sex, more severe dyspnea, lower level of consciousness, and lower activities of daily living.
Conclusion: Overweight and obese patients had a lower mortality than low–normal weight patients, which supports the obesity paradox.
Keywords: mortality, obesity paradox, COPD
Introduction
Chronic obstructive pulmonary disease (COPD) is a life-threatening lung disease that interferes with normal breathing and is not fully reversible. Worldwide, an estimated 64 million people had moderate-to-severe COPD in 2004, and it caused the deaths of over 3 million individuals in 2005.1 Prevalence and mortality in COPD are higher in older patients,2 and there is an independent association between older patients with COPD and higher mortality.3,4
Low body mass index (BMI) is a potential prognostic factor for short- and long-term mortality in COPD.5–9 However, the relationship between obesity and mortality of COPD is controversial. The obesity paradox, which is based on a protective effect of adipose tissue against mortality, has been observed in various chronic diseases, including cardiovascular disease,10 chronic heart failure,11 stroke,12 chronic kidney disease,13 type 2 diabetes mellitus,14 and pulmonary hypertension.15 Further, the obesity paradox has been reported in respiratory diseases,16 and the possibility of an obesity paradox in COPD has been discussed.17,18 However, the obesity paradox in patients with COPD has not been adequately examined. Further, most studies demonstrating the association between low BMI and higher mortality in chronic diseases have been conducted in Western populations. It has been found that Asian populations have a different association between BMI and health risks to Western populations; this is because Asians have a lower mean BMI than non-Asians and Asians have a higher percentage of body fat than non-Asians with a similar BMI.19
Using a nationwide inpatient database, we aimed to evaluate the association between BMI and mortality in elderly patients with COPD in Japan.
Methods
Data source
The Diagnosis Procedure Combination database is a nationwide inpatient database in Japan. The database includes administrative claims data and discharge abstract data. Main diagnosis, comorbidities present on admission, and complications occurring during hospitalization are coded using the International Classification of Disease and Related Health Problems, 10th Revision (ICD-10) codes accompanied by text data in Japanese. The database also contains the following details: type of admission (emergent or non-emergent), patient’s age, sex, body height and weight, smoking index (defined as the number of cigarettes smoked per day multiplied by the number of years smoked), severity of dyspnea based on the Hugh-Jones dyspnea scale,20 levels of consciousness based on the Japan Coma Scale,21,22 on admission, activities of daily life on admission converted to the Barthel index,23 intensive care unit admission during hospitalization, use of mechanical ventilation, and discharge status. The grading of dyspnea severity was based on the Hugh-Jones classification20 and defined as follows: 1) the patient’s breathing was as good as that of other people of their age and build when working, walking, and climbing hills or stairs; 2) the patient could walk at the same pace as healthy people of their age and build on level ground but was unable to maintain that pace on hills or stairs; 3) the patient was unable to maintain the pace of healthy people of their age and build on level ground but could walk about 1.6 kilometers or more at their own speed; 4) the patient was unable to walk more than about 50 meters on level ground without a rest; 5) the patient was breathless when talking or undressing or was unable to leave their home because of breathlessness; and (unspecified) the patient could not be classified into the above grades because of their bedridden status. The numbers of participating hospitals were 980, 1,075, and 1,057, respectively, for July 2010 to March 2011, April 2011 to March 2012, and April 2012 to March 2013.
This study was approved by the Institutional Review Board of The University of Tokyo. It waived the requirement for patient informed consent because of the anonymous nature of the data.
Patient selection
We retrospectively collected data for patients aged over 65 years who had been admitted to hospital because of COPD (ICD-10 codes J41, J42, J43, J44) as the main diagnosis, or who had been admitted for any cause but had COPD as comorbidity on admission, and were discharged between July 1, 2010 and March 31, 2013. COPD and other comorbidities were based on physician diagnosis.
Comorbidities on admission
The following comorbidities were identified using ICD-10 codes: pneumonia caused by pathogenic microbes (J10–J18); asthma (J45, J46); aspiration pneumonia (J69); interstitial pneumonia (J84); pulmonary embolism (I26); respiratory failure (J96); lung cancer (C34); heart failure (I50); ischemic heart disease (IHD; I20–I22, I25); cardiac arrhythmia (I44, I45, I47–I49); cerebral vascular diseases (I60–I69); chronic liver disease (K70, K71, K73, K74, K76); chronic renal failure (N18); anxiety (F40–F41); depression (F30–F33); and bone fracture (S02, S12, S22, S32, S42, S52, S62, S72, S82, S92, T02, T10, T12).
BMI categories
BMI categories were assigned based on World Health Organization classifications of underweight (BMI <18.5 kg/m2), normal weight (18.5–24.9 kg/m2), overweight (25.0–29.9 kg/m2), and obese (≥30.0 kg/m2) individuals. Normal weight was further divided into low–normal (18.5–22.9 kg/m2) and high–normal (23.0–24.9 kg/m2).24,25
Outcome
The primary outcome was all-cause in-hospital mortality.
Statistical analysis
We used chi-square tests to examine differences in categorical variables. We performed a multivariable logistic regression for in-hospital mortality to analyze patient-level factors associated with the outcome after adjustment for within-hospital clustering by means of a generalized estimating equation.26 For the BMI categories, we defined the low–normal weight group as the reference category. The threshold for significance was P<0.05. We performed all statistical analyses using SPSS software (v20; IBM, Armonk, NY, USA).
Results
Among 19 million patients during the 33 months between July 2010 and March 2013, we found 263,940 patients with COPD who were aged over 65 years. The mean age was 77.8 years (standard deviation [SD]: 7.2), and the proportions of males and females were 80.0% (n=211,057) and 20.0% (n=52,883), respectively. The mean number of smoking pack-years was 55.6 (SD: 35.6). Detailed clinical characteristics and primary diagnoses on admission appear in Table S1. Table 1 shows the patient characteristics divided by BMI category. We observed that the mean age increased with decreasing BMI. The proportion of patients aged over 85 years was higher in the underweight group, but lower in the overweight and obese groups. The percentage of males was higher in the underweight group. The proportion of patients with severe dyspnea (Hugh-Jones class V) was lowest (14.6%) in the overweight group and highest (27.1%) in the underweight group. Disability scores, consciousness levels, and types of admission showed similar patterns to the dyspnea grades.
The percentage of the following comorbidities was higher in the lower BMI categories: pneumonia caused by pathogenic microbes, aspiration pneumonia, respiratory failure, heart failure, and bone fracture. In contrast, the percentage of the following comorbidities was higher in the higher BMI categories: asthma, interstitial pneumonitis, IHD, liver disease, and chronic renal failure. Figure 1 shows all-cause in-hospital mortality for each BMI category. In-hospital mortality was 14.3%, 7.3%, 4.9%, 4.3%, and 4.4%, respectively, in the underweight, low–normal weight, high–normal weight, overweight, and obese groups.
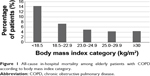 
|
Figure 1 All-cause in-hospital mortality among elderly patients with COPD according to body mass index category. |
Table 2 shows the results of the logistic regression analysis for all-cause in-hospital mortality. The underweight group had a significantly higher mortality than the reference low–normal weight group. The overweight and obese groups had significantly lower mortality rates than the reference group. Older age, male sex, more severe dyspnea scores, lower activities of daily life scores, and lower levels of consciousness were also significantly associated with higher mortality. Emergency admission in elderly COPD patients was likewise associated with high mortality. Regarding pulmonary comorbidities, higher mortality was significantly associated with pneumonia caused by pathogenic microbes, aspiration pneumonia, interstitial pneumonitis, respiratory failure, and lung cancer. With extrapulmonary comorbidities, higher mortality was associated with heart failure, liver disease, chronic renal failure, and bone fracture. Conversely, lower mortality was associated with asthma, IHD, and cerebrovascular diseases. Emergency admission in elderly COPD patients was also associated with high mortality.
 
|
Table 2 Multivariable logistic regression analysis for all-cause in-hospital mortality |
Discussion
This study used a national inpatient database in Japan to investigate the association between BMI and mortality in 263,940 elderly inpatients with COPD. Underweight patients had a significantly higher mortality than low–normal weight patients, whereas overweight and obese patients showed significantly lower mortality rates. The present study is the first to demonstrate the obesity paradox in all-cause in-hospital mortality in elderly patients with COPD in a nationwide setting.
Previous studies on BMI and COPD mortality have found low BMI to be associated with high mortality in COPD patients.5–9 One limited investigation determined that higher mortality was associated with lower BMI and that mortality decreased with increasing BMI in severe COPD; however, in mild and moderate COPD, the lowest mortality was found in patients with normal BMI.7 Most studies evaluating BMI and mortality in COPD have not examined the association between being overweight or obese and lower mortality in COPD patients. In the present study, higher mortality was associated with underweight, which concurs with the results of previous studies,5–9 which found lower BMI to be associated with higher mortality in COPD. Moreover, we found that lower mortality was associated with overweight and obesity, which supports the obesity paradox in elderly patients with COPD.
Generally, overweight and obesity are well-known risk factors for several chronic diseases and all-cause mortality in healthy individuals. However, one systematic review of cohort studies examined the association between BMI and total mortality in patients with coronary artery disease; it found that compared with normal BMI patients, those with low BMI (<20 kg/m2) had an increased risk for all-cause mortality and that overweight patients (BMI 25–29.9 kg/m2) had the lowest mortality risk. Obese patients (BMI ≥30 kg/m2) showed no increased risk for total mortality.10 These findings appear to be at variance with the above-mentioned prevailing orthodoxy. Similarly, this paradoxical association between obesity and mortality has been reported for chronic heart failure,11 stroke,12 chronic kidney disease,13 type 2 diabetes mellitus,14 and pulmonary hypertension.15 This phenomenon is referred to as the obesity paradox. However, this paradoxical association between mortality and BMI has not been previously evaluated in COPD patients.
Even though overweight and obesity have not been well-elucidated in COPD patients, there is evidence in the literature that overweight and obesity may be related to poor outcomes with COPD. Overweight and obesity have been reported to be associated with reduced forced expiratory volume in 1 second (FEV1), and weight gain has been shown to reduce pulmonary function longitudinally.27,28 COPD patients are at increased risk of developing obesity since they have decreased physical activity owing to exertional dyspnea and long-term use of systemic glucocorticosteroids to prevent exacerbation;17 this suggests a potential link between obesity and COPD. Epidemiologically, obesity has been found to be more prevalent in COPD patients than in the general population, and the prevalence of obesity was more frequent in early-stage COPD.17 Thus, the harmful long-term effects related to obesity may result in poor outcomes with COPD. In addition, it has been suggested that obesity is related to systemic inflammation in COPD by releasing pro-inflammatory cytokines from adipose tissue and that it contributes to the development of comorbidities and exacerbations in COPD.17,29 Further, insulin resistance, which is a common underlying pathophysiological finding in metabolic syndrome, is reportedly increased in COPD, and it may contribute to systemic extrapulmonary complications, which are linked to systemic low-grade inflammation in COPD patients.30 Therefore, obesity and overweight have been recognized as being associated with the severity of COPD, and obesity and overweight may exert undesirable effects in COPD patients. However, the present study demonstrated that overweight and obese patients had lower mortality rates than those with low–normal weight, which supports the existence of an obesity paradox in elderly patients with COPD.
Some studies have reported a favorable effect of obesity in COPD.31–33 It is reasonable to predict that obese COPD patients are more likely to experience greater dyspnea and exercise intolerance and may have poor prognosis; however, recent evidence suggests that obese COPD patients have similar or better dyspnea scores during exercise and do not have diminished exercise capacity compared with normal-weight COPD patients.32,33 In pulmonary function tests, obese COPD patients had both a reduced end-expiratory lung volume and a preserved inspiratory capacity;31 these are related to dyspnea scores and are favorable prognostic implications for patients with COPD.34 In addition, the annual decline in FEV1 in obese men with COPD was lower than in males with normal BMI; however, this effect was not observed in women, which indicates that obesity and overweight may exert protective effects in the progressive airflow limitation in COPD.35 The precise physiological mechanism of the obesity paradox in COPD remains unknown, though.
IHD is well-known to be a major comorbidity in COPD owing to shared risk factors of smoking and systemic low-grade inflammation.36,37 The prevalence of IHD in COPD patients has been reported to vary between 4.7% and 60%.38,39 In a population-based study, coronary artery disease was found in 7%–13% of patients diagnosed with COPD.40,41 The prevalence of IHD in the present study was 10.9% in elderly COPD patients, and this figure is comparable with that reported in previous studies.40,41 Further, since both IHD and COPD are leading causes of death, the coexistence of IHD and COPD worsens the prognosis for both diseases. It has been reported that COPD is a predictor of cardiovascular disease mortality; in particular, the association between COPD and cardiovascular disease was stronger in adults aged under 65 years.42 This suggests that cardiovascular disease should be evaluated and treated with particular care in younger adults with COPD.
Asthma often coexists with COPD,43 and asthma–COPD overlap syndrome has been investigated.44 Hardin et al demonstrated that patients with that syndrome frequently underwent COPD exacerbation, showed worse health-related quality of life,45 and could have a poorer prognosis than COPD patients without asthma. However, Fu et al found that COPD patients had a poorer prognosis than patients with asthma–COPD overlap syndrome: the former showed decreased lung function and reduced performance in the 6-minute walk test.46 Fu et al discussed the possibility that the overlap syndrome had a protective effect on disease activity; they suggested that less airway reversibility at baseline is associated with the development of irreversible airflow obstruction, which contributes to a poor prognosis.46,47 The present study also demonstrated that elderly COPD patients with asthma had a better prognosis than those with COPD alone; this confirms the better prognosis in elderly patients with asthma–COPD overlap syndrome. The use of inhaled glucocorticosteroids is effective for treating obstructive inflammatory airway diseases and reduced COPD exacerbation; it is recommended that patients with asthma–COPD overlap syndrome be treated with inhaled corticosteroids,44 and thus may have a better prognosis than patients with COPD alone.
Several limitations in the present study should be acknowledged. Since we based the diagnosis of COPD on physician-diagnosed COPD, the accuracy of COPD diagnosis was not confirmed by specialists. However, in actual practice, COPD is not always diagnosed by specialists; thus, data relating to physician-diagnosed COPD, such as those in epidemiological studies and the present study, are a valuable source of information. In addition, since the Diagnosis Procedure Combination database does not include the stages of COPD severity or details of pulmonary function tests, including FEV1 and other indices, the severity of COPD could not be precisely evaluated in this study.
In conclusion, this study demonstrates that higher mortality was associated with underweight; lower mortality was associated with overweight and obesity in elderly COPD patients. This supports the existence of the obesity paradox in elderly patients with COPD.
Acknowledgments
This work was supported by grants from the Ministry of Health, Labour and Welfare, Japan (Research on Policy Planning and Evaluation and grant to the Respiratory Failure Research Group), and Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Disclosure
The authors report no conflicts of interest in this work.
References
World Health Organization [webpage on the Internet]. Chronic respiratory disease: Burden of COPD. World Health Organization; 2014. Available from: http://www.who.int/respiratory/copd/burden/en/index.html. Accessed September 30, 2014. | ||
Halbert RJ, Natoli JL, Gano A, Badamgarav E, Buist AS, Mannino DM. Global burden of COPD: systematic review and meta-analysis. Eur Respir J. 2006;28(3):523–532. | ||
Patil SP, Krishnan JA, Lechtzin N, Diette GB. In-hospital mortality following acute exacerbations of chronic obstructive pulmonary disease. Arch Intern Med. 2003;163(10):1180–1186. | ||
Singanayagam A, Schembri S, Chalmers JD. Predictors of mortality in hospitalized adults with acute exacerbation of chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2013;10(2):81–89. | ||
Connors AF Jr, Dawson NV, Thomas C, et al. Outcomes following acute exacerbation of severe chronic obstructive lung disease. The SUPPORT investigators (Study to Understand Prognoses and Preferences for Outcomes and Risks of Treatments). Am J Respir Crit Care Med. 1996;154(4 Pt 1):959–967. | ||
Gray-Donald K, Gibbons L, Shapiro SH, Macklem PT, Martin JG. Nutritional status and mortality in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1996;153(3):961–966. | ||
Landbo C, Prescott E, Lange P, Vestbo J, Almdal TP. Prognostic value of nutritional status in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1999;160(6):1856–1861. | ||
Celli BR, Cote CG, Marin JM, et al. The body-mass index, airflow obstruction, dyspnea, and exercise capacity index in chronic obstructive pulmonary disease. N Engl J Med. 2004;350(10):1005–1012. | ||
Tsimogianni AM, Papiris SA, Stathopoulos GT, Manali ED, Roussos C, Kotanidou A. Predictors of outcome after exacerbation of chronic obstructive pulmonary disease. J Gen Intern Med. 2009;24(9):1043–1048. | ||
Romero-Corral A, Montori VM, Somers VK, et al. Association of bodyweight with total mortality and with cardiovascular events in coronary artery disease: a systematic review of cohort studies. Lancet. 2006;368(9536):666–678. | ||
Oreopoulos A, Padwal R, Kalantar-Zadeh K, Fonarow GC, Norris CM, McAlister FA. Body mass index and mortality in heart failure: a meta-analysis. Am Heart J. 2008;156(1):13–22. | ||
Andersen KK, Olsen TS. The obesity paradox in stroke: lower mortality and lower risk of readmission for recurrent stroke in obese stroke patients. Int J Stroke. Epub 2013 Mar 12. | ||
Kovesdy CP, Anderson JE, Kalantar-Zadeh K. Paradoxical association between body mass index and mortality in men with CKD not yet on dialysis. Am J Kidney Dis. 2007;49(5):581–591. | ||
Carnethon MR, De Chavez PJ, Biggs ML, et al. Association of weight status with mortality in adults with incident diabetes. JAMA. 2012;308(6):581–590. | ||
Zafrir B, Adir Y, Shehadeh W, Shteinberg M, Salman N, Amir O. The association between obesity, mortality and filling pressures in pulmonary hypertension patients; the “obesity paradox”. Respir Med. 2013;107(1):139–146. | ||
Jee SH, Sull JW, Park J, et al. Body-mass index and mortality in Korean men and women. N Engl J Med. 2006;355(8):779–787. | ||
Franssen FM, O’Donnell DE, Goossens GH, Blaak EE, Schols AM. Obesity and the lung: 5. Obesity and COPD. Thorax. 2008;63(12):1110–1117. | ||
Chittal P, Babu AS, Lavie CJ. Obesity paradox: does fat alter outcomes in chronic obstructive pulmonary disease? COPD. Epub 2014 Jun 19. | ||
WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363(9403):157–163. | ||
Hugh-Jones P, Lambert AV. A simple standard exercise test and its use for measuring exertion dyspnoea. Br Med J. 1952;1(4749):65–71. | ||
Ohta T, Waga S, Handa W, Saito I, Takeuchi K. [New grading of level of disordered consiousness (author’s transl)]. No Shinkei Geka. [Neurological surgery]. 1974;2(9):623–627. Japanese. | ||
Todo T, Usui M, Takakura K. Treatment of severe intraventricular hemorrhage by intraventricular infusion of urokinase. J Neurosurg. 1991;74(1):81–86. | ||
Mahoney FI, Barthel DW. Functional evaluation: the Barthel index. Md State Med J. 1965;14:61–65. | ||
World Health Organization [webpage on the Internet]. Global Database on Body Mass Index. World Health Organization; 2014. Available from: http://apps.who.int/bmi/index.jsp. Accessed October 26, 2014. | ||
Hasegawa W, Yamauchi Y, Yasunaga H, et al. Factors affecting mortality following emergency admission for chronic obstructive pulmonary disease. BMC Pulm Med. 2014;14(1):151. | ||
Hubbard AE, Ahern J, Fleischer NL, et al. To GEE or not to GEE: comparing population average and mixed models for estimating the associations between neighborhood risk factors and health. Epidemiology. 2010;21(4):467–474. | ||
Bottai M, Pistelli F, Di Pede F, et al. Longitudinal changes of body mass index, spirometry and diffusion in a general population. Eur Respir J. 2002;20(3):665–673. | ||
Wise RA, Enright PL, Connett JE, et al. Effect of weight gain on pulmonary function after smoking cessation in the Lung Health Study. Am J Respir Crit Care Med. 1998;157(3 Pt 1):866–872. | ||
Fabbri LM, Rabe KF. From COPD to chronic systemic inflammatory syndrome? Lancet. 2007;370(9589):797–799. | ||
Bolton CE, Evans M, Ionescu AA, et al. Insulin resistance and inflammation – A further systemic complication of COPD. COPD. 2007;4(2):121–126. | ||
Ora J, Laveneziana P, Ofir D, Deesomchok A, Webb KA, O’Donnell DE. Combined effects of obesity and chronic obstructive pulmonary disease on dyspnea and exercise tolerance. Am J Respir Crit Care Med. 2009;180(10):964–971. | ||
Guenette JA, Jensen D, O’Donnell DE. Respiratory function and the obesity paradox. Curr Opin Clin Nutr Metab Care. 2010;13(6):618–624. | ||
Laviolette L, Sava F, O’Donnell DE, et al. Effect of obesity on constant workrate exercise in hyperinflated men with COPD. BMC Pulm Med. 2010;10:33. | ||
Casanova C, Cote C, de Torres JP, et al. Inspiratory-to-total lung capacity ratio predicts mortality in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2005;171(6):591–597. | ||
Watson L, Vonk JM, Löfdahl CG, et al; European Respiratory Society Study on Chronic Obstructive Pulmonary Disease. Predictors of lung function and its decline in mild to moderate COPD in association with gender: results from the Euroscop study. Respir Med. 2006;100(4):746–753. | ||
Vestbo J, Hurd SS, Agusti AG, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2013;187(4):347–365. | ||
Sin DD, Man SF. Why are patients with chronic obstructive pulmonary disease at increased risk of cardiovascular diseases? The potential role of systemic inflammation in chronic obstructive pulmonary disease. Circulation. 2003;107(11):1514–1519. | ||
Mullerova H, Agusti A, Erqou S, Mapel DW. Cardiovascular comorbidity in COPD: systematic literature review. Chest. 2013;144(4):1163–1178. | ||
Roversi S, Roversi P, Spadafora G, Rossi R, Fabbri LM. Coronary artery disease concomitant with chronic obstructive pulmonary disease. Eur J Clin Invest. 2014;44(1):93–102. | ||
Eriksson B, Lindberg A, Müllerova H, Rönmark E, Lundbäck B. Association of heart diseases with COPD and restrictive lung function – results from a population survey. Respir Med. 2013;107(1):98–106. | ||
Schnell K, Weiss CO, Lee T, et al. The prevalence of clinically-relevant comorbid conditions in patients with physician-diagnosed COPD: a cross-sectional study using data from NHANES 1999–2008. BMC Pulm Med. 2012;12:26. | ||
Sidney S, Sorel M, Quesenberry CP Jr, DeLuise C, Lanes S, Eisner MD. COPD and incident cardiovascular disease hospitalizations and mortality: Kaiser Permanente Medical Care Program. Chest. 2005;128(4):2068–2075. | ||
Gibson PG, Simpson JL. The overlap syndrome of asthma and COPD: what are its features and how important is it? Thorax. 2009;64(8):728–735. | ||
Global Initiative for Asthma [webpage on the Internet]. Asthma, COPD and Asthma-COPD Overlap Syndrome (ACOS). Global Initiative for Asthma; 2014. Available from: http://www.ginasthma.org/documents/14. Accessed July 27, 2014. | ||
Hardin M, Silverman EK, Barr RG, et al; COPD Gene Investigators. The clinical features of the overlap between COPD and asthma. Respir Res. 2011;12:127. | ||
Fu JJ, Gibson PG, Simpson JL, McDonald VM. Longitudinal changes in clinical outcomes in older patients with asthma, COPD and asthma-COPD overlap syndrome. Respiration. 2014;87(1):63–74. | ||
Vonk JM, Jongepier H, Panhuysen CI, Schouten JP, Bleecker ER, Postma DS. Risk factors associated with the presence of irreversible airflow limitation and reduced transfer coefficient in patients with asthma after 26 years of follow up. Thorax. 2003;58(4):322–327. |
Supplementary material
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


