Back to Journals » International Journal of Women's Health » Volume 11
Ovarian cancer in the world: epidemiology and risk factors
Authors Momenimovahed Z, Tiznobaik A, Taheri S , Salehiniya H
Received 9 December 2018
Accepted for publication 18 March 2019
Published 30 April 2019 Volume 2019:11 Pages 287—299
DOI https://doi.org/10.2147/IJWH.S197604
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Elie Al-Chaer
Zohre Momenimovahed,1,2 Azita Tiznobaik,2,3 Safoura Taheri,4 Hamid Salehiniya5,6
1Department of Midwifery and Reproductive Health, School of Nursing and Midwifery, Qom University of Medical Sciences, Qom, Iran; 2Department of Midwifery and Reproductive Health, School of Nursing and Midwifery, Tehran University of Medical Sciences, Tehran, Iran; 3Department of Midwifery and Reproductive Health, School of Nursing and Midwifery, Hamedan University of Medical Sciences, Hamedan, Iran; 4Department of Midwifery and Reproductive Health, School of Nursing and Midwifery, Ilam University of Medical Sciences, Ilam, Iran; 5Social Determinants of Health Research Center, Birjand University of Medical Sciences, Birjand, Iran; 6Epidemiology and Biostatistics Department, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran
Aim: Ovarian cancer is one of the most common gynecologic cancers that has the highest mortality rate. Considering the fact that knowledge on the incidence, mortality of ovarian cancer, as well as its risk factors is necessary for planning and preventing complications, this study was conducted with the aim of examining the epidemiology and risk factors of ovarian cancer in the world.
Materials and methods: In order to access the articles, Medline, Web of Science Core Collection, and Scopus databases were searched from their start to the year 2018. Full-text, English observational studies that referred to various aspects of ovarian cancer were included in the study.
Results: In total, 125 articles that had been published during the years 1925–2018 were entered into the study. Ovarian cancer is the seventh most common cancer among women. Increased risk factors of cancer have led to an upward trend in the incidence of cancer around the world. In 2018, 4.4% of entire cancer-related mortality among women was attributed to ovarian cancer. Although the incidence of cancer is higher among high Human Development Index (HDI) countries, the trend of mortality rate tends to be reversing. Various factors affect the occurrence of ovarian cancer, from which genetic factor are among the most important ones. Pregnancy, lactation, and oral contraceptive pills play a role in reducing the risk of this disease.
Conclusion: This study provides significant evidence about ovarian cancer. Considering the heavy burden of ovarian cancer on women’s health, preventive measures as well as health education and early detection in high risk groups of women are highly recommended. Although some risk factors cannot be changed, a focus on preventable risk factors may reduce the risk of ovarian cancer. More studies are needed to explore the role of unclear risk factors in ovarian cancer occurrence.
Keywords: Ovarian cancer, epidemiology, risk factor
Introduction
Cancer is the most common cause of mortality in most parts of the world,1 and currently is the most common impediment to achieving desirable life expectancy in most countries.2 Ovarian cancer is one of the most common gynecologic cancers that rank third after cervical and uterine cancer.2 It also has the worst prognosis and the highest mortality rate.3 Although ovarian cancer has a lower prevalence in comparison with breast cancer, it is three times more lethal,4 and it is predicted that, by the year 2040, the mortality rate of this cancer will rise significantly.2 The high mortality rate of ovarian cancer is caused by asymptomatic and secret growth of the tumor, delayed onset of symptoms, and lack of proper screening that result in its diagnosis in the advanced stages. Thus, silent killer is a name that has been given to this cancer.4–6
Like many cancers, the incidence of ovarian cancer varies across the world.7 The epidemiological diversity of ovarian cancer in different regions can be attributed to the risk factors that account for the occurrence of ovarian cancer.8 The highest prevalence of ovarian cancer is seen in non-Hispanic white women (12.0 per 100,000), followed by Hispanic (10.3 per 100,000), non- Hispanic black (9.4 per 100,000), and Asian/Pacific Islander women (9.2 per 100,000).9 However, due to differences in access to diagnostic and therapeutic services, the mortality of ovarian cancer has a different pattern, and the highest mortality rate is seen in African populations.10 The statistics show that between one third to two fifths of the total cancer cases can be prevented by eliminating and reducing risk factors.2 Considering the fact that knowledge on the incidence, mortality, and geographical diversity of ovarian cancer as well as its risk factors is necessary for planning and preventing complications, and since we could find no comprehensive study on the risk factors of ovarian cancer in the world, the aim of this study was to examine the trends in incidence and mortality across the world and to present all possible factors associated with OC.
Materials and methods
Search engines
In order to access the articles, Medline, Web of Science Core Collection (Indexes = SCI-EXPANDED, SSCI, A & HCI Timespan) and Scopus databases were searched from their start to the year 2018.
Search strategy
At first, the search strategies were defined to increase validity of the review. In order to search the articles, a systematic and accurate review of the published articles was carried out by two researchers independently, and a list of potential articles was prepared. The views of a specialist and expert librarian were used to ensure the comprehensiveness of searching strategies and reviewing resources. Searching for articles was carried out with no time limit using keywords such as; ovarian cancer, incidence, mortality, risk factor, and a combination of them in English language. All keywords were checked within PubMed Medical Subject Heading (MeSH). Then, to ensure the adequacy of the search, a manual search was also done in valid journals, followed by a manual search for the references of full-text articles and systematic reviews. All retrieved articles were entered into a database in the Endnote X7.
Inclusion criteria
The criteria for entering the study included the following: Full-text articles (retrospective and prospective cohort and case-control studies), English language articles, the use of keywords in the title or abstract (ovarian cancer, incidence, mortality, risk factors, and a combination of these terms).
Exclusion criteria
Case reports, case series, systematic reviews, and animal studies were excluded.
Results
Characteristics of the selected studies
In total, 145 articles that had been published during the years 1925–2018 were entered into the study. In the initial search, 588 articles were obtained from databases, and 46 articles were extracted by manual search. After removing repetitive articles using EndNote software, 493 articles were selected for review. After reviewing the titles and abstracts, 322 articles that were unrelated to the purpose of study and were not consistent with the criteria of the study were removed. Furthermore, 26 articles were also removed for scientific reasons (editorial: 5, qualitative: 6, duplicate: 9, not available full text: 6). Finally, 145 articles that had been published during the years 1925–2018 were entered into the study (Figure 1).
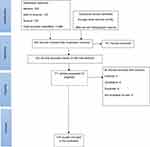 | Figure 1 Flowchart of the included eligible studies in review. |
The types of ovarian cancer
Different subtypes of ovarian cancer were discussed in nine studies. Studies show that up to 90% of all OC have epithelial origin and the remaining OC have non-epithelial origin.11–13 Among epithelial OC, 3% are mucinous and others are non-mucinous.14 Non-mucinous are further found to have serous (70% of non-mucinous), endomterioid (10%), clear cell (10%), and unspecified subtypes (5%).14–16 According to recent studies, serous carcinomas are divided into two separate subtypes: high grade and low grade.17,18 Compared to epithelial cancers, non-epithelial cancers are less invasive.19
Incidence
Population growth, increased risk factors of cancer, decreased pregnancy and duration of lactation, as well as tube ligation have led to an upward trend in the incidence of cancer around the world.2,20,21 Ovarian cancer is the seventh most common cancer among women2 and, in the absence of protective factors, the lifetime risk of ovarian cancer is about 2.7%.21 According to Globoccan, 295,414 cases of ovarian cancer have been identified in 2018, accounting for 3.4% of all cancer cases in women.2 The Age Standardized Rate (ASR) of ovarian cancer is estimated to be 6.6 in 2018.2 The incidence of epithelial ovarian cancers varies in different age and race groups.9 The incidence of this cancer is higher among transitioned countries,2 and approximately 30% of ovarian cancer cases occur in European countries.22 In 2012, the highest rates of ovarian cancer occurred in China (14.60% of all cases), India (11.33% of all cases), and the US (81.8% of all cases).22 In that year, 22,240 cases of ovarian cancer were detected in the USalone.9 Among the Asian countries, Singapore, Kazakhstan, and Brunei have the highest standardized incidence rate of ovarian cancer.23
Mortality
In 2018, 184,799 deaths occurred due to ovarian cancer, accounting for 4.4% of the entire cancer-related mortality among women.2 Based on Globoccan 2018, the ASR of ovarian cancer mortality is 3.9.2 Although the incidence of cancer is higher among high Human Development Index (HDI) countries, the trend of mortality rate tends to be reversing.2 The highest mortality rate in Asia is seen in India, and the mortality rate has decreased in Europe and North America in recent years, especially among young people.24 The mortality-to-incidence ratio is high among African women, indicating their lack of access to suitable treatment.10 Two-thirds of ovarian cancer mortality is attributable to high-grade serous carcinoma.12 The FIGO’s high stage at diagnosis and surgery,25 and the presence of comorbidity26 are among the most important predictors of high mortality in ovarian cancer.
Risk factors
Table 1 shows factors related to ovarian cancer.
 | Table 1 Factors related to ovarian cancer in the world |
Demographic factor
Age
The epithelial ovarian cancer is an age-related disease, and is considered mainly a postmenopausal disease.10,27 Increased incidence of this cancer is more pronounced in women over 65 years of age.28 According to previous studies, median age at diagnosis is 50–79 years.27,29,30 The relationship between age and the outcome of ovarian cancer is uncertain. Although many researchers have pointed out that the younger age of ovarian cancer is associated with the improved outcome,29,31,32 other stated age is not an independent prognostic factor.33 Older age in this disease is associated with more advanced disease and lower survival rate.27,34 Older women are treated less aggressively in contrast with younger ovarian cancer patients, and, thus, survival is lower in these group.35 An age of over 64 years is one of the predictors of mortality in people with ovarian cancer.25
Reproductive factors
Menstrual-related factors
Tung et al36 stated that non-mucinous tumors are strongly associated with menstrual periods (odds ratio=1.5 for the highest vs the lowest quartile) and ovulation cycles (odds ratio=2.8 for the highest vs the lowest quartile). In numerous studies, researchers indicated an inverse relationship between ovulation cycles and the risk of ovarian cancer.37,38 The result of a case-control study showed that, in women who have not had an ovulation cycle for 8.7 years, the risk of ovarian cancer was reduced by 4-times (OR=0.23 [0.10–0.50]).39 These findings support the theory of “incessant ovulation”. Based on this theory, ovulation without interruption can contribute to the incidence of ovarian cancer by damaging the epithelium of ovaries; therefore, any factor that contributes to the reduction of ovulation can have a protective effect against ovarian cancer.40 However, Moorman et al41 believed that, contrary to the lack of ovulation due to pregnancy or the use of oral contraceptives, the lack of ovulation caused by menstrual disorders is associated with an increased risk of ovarian cancer.
Age of menarche and menopause
Although the result of some studies showed a relationship between the early onset of menarche and risk of ovarian cancer,42,43 other researchers reported that age of menarche and menopause has no effect on the risk of ovarian cancer.44–49
Parity
Results of several studies suggest that pregnancy has a protective role against ovarian cancer.38,45,49–52 Based on the results of a case-control study, the risk of ovarian cancer is reduced in women with live birth (P<0.001) or induced abortion (P<0.05), and this risk decreases with an increase in the number of live birth cases (P<0.001). The result of a case-control study showed that, for every full-term pregnancy, OR is equal to 0.76 [0.69–0.85] for non-mucinous tumors and 1.03 [0.88–1.21] for mucinous tumors.53 The result of a study showed that increased pregnancy is associated with a consistent reduction in the relative risk of invasive ovarian cancer (odds ratio for each additional birth=0.81 [0.75–0.85]), epithelial cancer (0.81 [0.77–0.86]), stromal cancer (0.84 [0.72–0.98]), and germ-cell cancer (0.71 [0.48–1.05]).54 However, Poole et al34 stated that pregnancy decreases the risk of less aggressive disease compared to an advanced disease.
Pregnancy characteristics
Jordan et al,55 in a case-control study, showed that preterm labor increases the risk of ovarian cancer (OR=1.48 [1.02–2.15]). This finding has been confirmed in Skold et al’s56 study. The results of a study showed that the delivery of a male infant is associated with a 2-times increase in the risk of mucinous ovarian cancer (OR=2.19 [1.15–4.17]).55 Mucci et al57 concluded that low birth weight among term infants has a protective effect on the mother’s ovarian cancer, while Skold et al56 did not find any relationship between infant’s weight and ovarian cancer. Skold et al56 also rejected the role of pre-eclampsia and multiple pregnancies in the occurrence of cancer in the mother. However, Calderon-Margalit et al,58 in a cohort study, concluded that pre-eclampsia increases the risk of ovarian cancer by more than 2-times (HR=2.59 [1.35–4.94]).
Age at childbirth
Results of a case-control study indicated that older age in pregnancy is associated with a decreased risk of ovarian cancer59 relative to the number of pregnancies. This result has been confirmed in other studies.51,52,54 Adami et al54 stated that, for every 5-year increase in the age at first childbirth, the risk of ovarian cancer would be reduced by up to 10% (OR=0.89 [0.84–0.94] epithelial cancer, 0.92 [0.77–1.10] stromal cancer, 0.92 [0.65–1.32] germ-cell cancer, 0.93 [0.80–1.09] borderline tumors).
Gynecologic factors
Pelvic inflammatory disease
The role of inflammation and pelvic inflammatory disease in the occurrence of ovarian cancer is controversial among experts. Ness et al,60 in a case-control study, supported the hypothesis that suggests inflammation contributes to the onset of ovarian cancer. On the other hand, Jia et al61 concluded that events associated with inflammation in the ovary (such as repairing the damaged ovary) are associated with an increase in the release of cancer cells in the tissues around the ovary. Thus, they stated that ovulation and other events associated with inflammation in the ovary contribute to an increased risk of ovarian cancer. In line with this, a case-control study referred to the role of chlamydia trachomatis infection in the development of ovarian cancer.62 Wong et al63 stated that, although chlamydia trachomatis inflammation may contribute to the development of ovarian cancer, chlamydia is a common pathogen in the genital area, and it is difficult to determine the exact relationship between the two. Merritt et al,64 in a case-control study in 2008, had an opposite view, and stated that chronic inflammation has no role in the development of ovarian cancer. Lin et al65 reported that there is a link between PID and ovarian cancer (HR=1.92 [1.27–2.92]). In a cohort study, Rusmussen et al66 reported that inflammation associated with serous ovarian cancer is not associated with the risk of other types of ovarian cancer. The result of another case-control study suggested that, although there is a relationship between pelvic inflammatory disease (PID) and ovarian cancer, this risk is higher in the cases of recurrent PID (OR=1.88 [1.13–3.12], P=0.014).67
Endometriosis
The relationship between endometriosis and ovarian cancer has been shown in various studies through various mechanisms. The results of a cohort study showed that, in people with endometrium, aging, living in urban areas, low or high income, depression, pelvic infection, and lack of childbearing increase the risk of ovarian cancer.68 In 1925, Sampson69 proposed a link between endometriosis and ovarian cancer based on the theory of malignant changes of endometriosis. Inflammation and the PTEN, CTNNB1 (β-catenin), KRAS, microsatellite instability, and ARID1A genes are involved in the occurrence of endometriosis-associated ovarian cancer.70 THe results of a cohort study showed that endometriosis increases the risk of ovarian cancer (SIR=1.34 [1.16–1.55]) and this risk is higher in endometrioid (SIR=1.64 [1.09–2.37]) and clear cells (SIR=3.64 [2.36–5.38]).71 Compared to other types of ovarian cancer, endometriosis-associated ovarian cancer is detected at a younger age and lower stages.70 Melin et al72 stated that, although endometriosis increases the risk of ovarian cancer (SIR=1.43 [1.19–1.71]), hysterectomy may have a protective effect against ovarian cancer before or at the time of endometriosis diagnosis. Stewart et al,73 in a cohort study, stated that nulliparous women with endometriosis are 3-times more likely to develop ovarian cancer (HR=3.11 [1.13–8.57]). They stated that, although hysterectomy plays a protective role against ovarian cancer, unilateral oophorectomy/salpingo-oophorectomy without hysterectomy increases the risk of ovarian cancer by 4-times (HR=4.23 [1.30–13.77]). Erzen and Kovacic74 believed that the relationship between endometriosis and ovarian cancer is affected by the age of the patient. The incidence of epithelial ovarian cancer in women with endometriosis rises from 4.99 in less than 30 years to 35.81 in more than 50 years per 10,000 people per year.75 In a case-control study, researchers concluded that hyperestrogenism or exogenous is a risk factor for the onset of ovarian cancer after endometriosis.76 Cottreau et al77 believed that the use of danazol for the treatment of endometriosis is associated with a 3.2-times increased risk of ovarian cancer [1.2–8.5]. They stated that there is no such a risk in the consumers of leuprolide/nafarelin.
Ovarian cysts
Some types of benign ovarian cysts may act as a precursor of malignant ovarian tumors. According to a case-control study, ovarian cyst is associated with increased risk of borderline ovarian tumors (OR=1.3 [0.9–1.8]), and this risk increased among women who were undergoing surgery.78 In addition, complex ovarian cysts significantly increase the risk of malignancy in postmenopause.79 However in another study, complex cysts in postmenopausal women were not the immediate precursors of ovarian cancer.80Crayfold,81 in a cohort of women, indicated that removal of ovarian cysts was not associated with a decrease in ovarian cancer related mortality.
Tubal ligation
The risk of ovarian cancer was reduced in women with tubal ligation.82–85 In a cohort study, tubal ligation was associated with a 20% reduction in risk of high‐grade serous carcinoma.86 Women with tubal ligation have a decreased risk of invasive serous cancer (19%), invasive mucinous cancer (32%), clear cell cancer (42%), and endometrioid cancer (52%).82,85 No association was found between tubal ligation and low-grade serous tumors.82,86 Younger age at tubal ligation was not associated with increased protective effects of this method.82 A mechanical barrier for carcinogenic agents can reduce ovarian cancer after tubal ligation.87
Hormonal factors
Contraceptive methods
Results of most studies indicate that the use of oral contraceptive methods is associated with a reduced risk of all histological types of ovarian cancer.36,51,88,89 Results of a case-control study in Canada indicated that the use of hormonal contraceptive pills is associated with a significant reduction in all histological types of epithelial ovarian cancer, except for mucinous tumors. According to the findings of this study, OR for each year of use of these pills was 0.89 [0.85–0.93] for non-mucinous tumors and 0.98 [0.93–1.04] for mucinous tumors.53 The result of a case-control study showed that oral contraceptive pill (OCP) decreases the risk of fatal and advanced ovarian cancer compared to less advanced cases.34 Royar et al89 stated that, each year, use of combined oral contraceptive pills reduces the risk of ovarian cancer by 7% (OR=0.93 [0.90–0.96]), and this reduction is more pronounced during the first use at the age of less than 25 years. Although there is an inverse relationship between the time of using hormonal contraceptive pill, the age of its use, and the risk of ovarian cancer, the duration of consumption is more important.51,59 This risk reduction can persist for up to 10–15 years after the discontinuation of pills,90 however the protective effect of oral contraception has not been proven in many studies.50 In a case-control study, no relationship was found between the use of contraceptive methods (except for tube ligation) and the risk of ovarian cancer.37
Hormone replacement therapy (HRT)
The result of a case-control study showed that combined estrogen-progesterone therapy after menopause does not increase the risk of ovarian cancer.91 Hempling et al92 examined the effect of exposure to post-menopausal hormone therapy, and stated that HRT is not associated with ovarian cancer, even in long term use. However, Glud et al93 stated that oral hormone therapy is associated with an increased risk of ovarian cancer in people who have not previously had a hysterectomy. Researchers believe that the use of estrogenic methods, especially for 10 years or more, is associated with an increased risk of ovarian cancer.94 The result of a case-control study showed that, although hormone therapy with estrogen alone increases the risk of ovarian cancer, it has no significant effect on the survival of the patient.95 Rossing et al,91 in a case-control study, stated that the progesterone component of a combined hormone therapy reduces the risk of ovarian cancer. Mørch et al96 believed that, regardless of the duration of use, formulation, estrogen dose, type of regimen, type of progesterone, and method of use, hormone therapy is associated with an increased risk of ovarian cancer.
Infertility treatments
Ovarian cancer is a rare and, at the same time, a fatal disease. Regardless of infertility treatments, nulliparity itself97 and infertility are risk factors of ovarian cancer, so it is difficult to investigate the relationship between infertility treatment and ovarian cancer. The “incessant ovulation theory” states that ovulation without interruption can contribute to the development of ovarian cancer by damaging the ovary epithelium, and, therefore, any factor that contributes to the reduction of ovulation can have a protective effect against ovarian cancer.40 Several studies have indicated an association between the increased risk of ovarian cancer and the use of clomiphene citrate98 and gonadotropin.99 The results of a cohort study showed an increase in ovarian cancer after exposure to clomiphene citrate, and indicated that the risk of ovarian cancer increases with increasing dosage of clomiphene citrate among nulliparous women.98 The results of a case-control study indicated that the use of ovulation-inducing drugs, especially hMG, increases the risk of epithelial ovarian tumors.100 Although several studies have suggested a relationship between the risk of ovarian cancer and the use of ovulation-inducing drugs, this risk was not significant in many studies.46,101–105 In a cohort study of 54,362 women, Jensen et al106 reported that the risk of ovarian cancer does not increase with the use of clomiphene citrate, gonadotropins, human chorionic gonadotrophin, and gonadotrophin releasing hormone, and there is no relationship between duration of use, duration of follow-ups, or pregnancy. Brinton et al (2001)99 suggested that an increased risk of ovarian cancer among people who take ovulation-inducing drugs requires a more attention to the choice of individuals.
Genetic factors
Family history
The most important risk factor for ovarian cancer is a family history of breast or ovarian cancer.9 Personal history of breast cancer is associated with an increase in the risk of ovarian cancer (OR=3.7 [1.8–7.7]).107 The results of a case-control study showed that the risk of ovarian cancer increases in women with a family history of breast, uterine, or ovarian cancer in their mother or sister (P<0.001).37
BRCA mutations
More than one-fifth of ovarian cancers are due to mutations in tumor suppressor genes,108 and 65–85% of inherited ovarian tumors result from germline mutations in BRCA genes.109 Although the risk of ovarian cancer in carriers of BRCA1 and BRCA2 mutations is less than 3% by the age of 40, this risk increases to 10% by the age of 50.12 The 10-year risk of developing ovarian cancer in individuals with breast cancer is 12.7% and 6.8% in the carriers of BRCA1 and BRCA2 mutation.12 Cumulative risk of ovarian cancer up to the age of 80 is 49% in BRCA1 mutation carriers and 21% in BRCA2 mutation carriers.110 About 25% of breast cancer deaths in stage I are due to the occurrence of ovarian cancer.111 Salpingo-oophorectomy in BRCA-positive individuals reduces the risk of ovarian cancer by 75%.112 Since, most epithelial cancers originate from the fallopian tube, salpingectomy decreases the risk of ovarian cancer by 35–50%.113
Lynch syndrome
Lynch syndrome is an autosomal dominant cancer predisposition syndrome that is responsible for 1–3% of all colorectal cancer.114 Lynch syndrome is responsible for 10–15% of the total inherited ovarian cancer cases,115 and the lifetime risk of this cancer in individuals with a family history of Lynch syndrome is 6–8%.116 Most of the ovarian cancers associated with Lynch syndrome are non-mucinous, and 82–84% of them are in stage I or II.115 Lynch occurs due to a hereditary mutation in one of the four mismatch repair genes (MHL1, MSH2, MSH6, and PMS2),12 and MSH2 and MLH1 are the most common mutations in these individuals.117 The most common types of ovarian cancer in these individuals are endometrioid and clear cell ovarian cancers.116
Lifestyle factors
Nutrition and diet
According to the findings of a case-control study, there is a positive correlation between daily intake of fish and the risk of ovarian cancer (P<0.05), and this correlation is negative for daily intake of milk (P=0.05).37,118 Results of a case-control study showed that the risk of ovarian cancer is associated with a higher cholesterol intake (OR=1.42 [1.03–1.97]), and this risk is reduced by consumption of vegetables (OR=0.77 [0.60–1.04]), vitamin supplement (OR=0.49 [0.30–0.81]), beta-carotene (OR=0.31 [0.11–0.91]), and B-complex vitamins (OR=0.61 [0.36–1.05]).119 McCann et al120 refer to the protective role of phytoestrogens in the development of ovarian cancer, and believe that a plant-based diet plays an important role in the reduction of hormone-related cancers.120,121 The results of a case-control study showed that saturated fat is associated with an increased risk of ovarian mucinous tumors.53 Ong et al122 revealed that an increased concentration of vitamin D in plasma may reduce the risk of ovarian cancer. This risk reduction is also seen in the case of calcium and lactose consumption.118
Obesity and physical activity
The results of a study showed that obesity reduces the risk of survival in ovarian cancer (HR=3.40 [1.16–9.99]), and increases the risk of death caused by the disease (HR=0.58 [0.35–0.96]).123 Central adiposity is associated with an increased risk of ovarian cancer, indicating the conversion of androgen in the peripheral tissues.124 Rodriguez et al125 reported a 36% increase in the risk of ovarian cancer among obese people who have never used postmenopausal estrogen treatment, and stated that obesity and tallness increase the mortality of ovarian cancer. Anderson et al,126 in a cohort study, reported that waist–hip ratio is associated with an increased risk of ovarian cancer (RR=1.59 for high quartile vs low quartile [1.05–2.40]). However, Kotsopoulos et al127 stated that height, weight, and adiposity are not related to the prognosis of ovarian cancer. Beehler et al128 believed that the relationship between obesity and risk of ovarian cancer is related to menopause condition. Beehler et al,128 in a case-control study, showed that obesity before menopause is associated with an increased risk of ovarian cancer (adjusted OR=2.19 [1.49–4.04]), although it is not associated with the risk of ovarian cancer at post-menopausal age. Leitzmann et al129 suggested that obesity, with its hormonal mechanism, increases the risk of ovarian cancer and, in addition to that, increases the mortality of affected individuals.38 On the other hand, researchers in a case-control study concluded that physical activity is associated with a reduction in the risk of ovarian cancer.130 However, this result has not been confirmed by other studies,131,132 Anderson et al126 stated that leisure-time physical activity is associated with an increase in the incidence of ovarian cancer (RR=:1.42 for high activity vs low activity [1.03–1.97]).
Alcohol, caffeine, and cigarettes
Several researchers around the world believe that alcohol does not increase the risk of ovarian cancer,46,133–137 but Goodman and Tung138 argue that alcohol’s relation to ovarian cancer is related to the type of alcohol. Schouten et al139 believed that drinking alcohol in the form of wine, beer, or liquor is not associated with an increased risk of ovarian cancer. However, the result of a case-control study showed that caffeine and coffee consumption may increase the risk of ovarian cancer in women before menopause.135
Although many researchers believe that cigarette smoking does not change the risk of ovarian cancer in women before and after menopause,135,140 Jordan et al141 state that smoking a pack of cigarettes daily for 20 years is associated with a doubled risk of benign mucinous tumors, borderline tumors, and malignant tumors (OR=2.7 [1.6–4.4] for benign tumors, OR=2.7 [1.7–4.4] for borderline tumors, and OR=2.1 [0.9–5.0] for malignant tumors). Gram et al,142 in a cohort study, showed that the length and amount of smoking increases the risk of borderline tumors. Kim et al38 believed that smoking increases the risk of death in people with ovarian cancer by up to 25% (HR=1.25 [1.01–1.54]). Marchbanks et al143 stated that, although smoking increases the risk of epithelial mucinous tumors (OR=2.9 [1.7–4.9]), it is not associated with other histological types of ovarian cancer. This result has also been confirmed in other studies.46,52,136,144–146 The age at smoking onset is not associated with the risk of ovarian cancer.147
Other
Lactation
Researchers have reported an inverse relationship between the duration of breastfeeding, the number of breast-fed children, and the risk of ovarian cancer.50,148 Results of a case-control study showed that lactation reduces the risk of ovarian cancer by 22% (OR=0.78 [0.64–0.96]), and this risk decreases with longer lactation period (OR=0.56 [0.32–0.98] for 18 months average duration of breastfeeding vs none). In this study, the most risk reduction was related to endometrioid and clear cell ovarian cancers.149 Tung et al36 were also in agreement with the views of other researchers, and stated that the duration of lactation reduces the risk of non-mucinous tumors (OR=: 0.4 for the highest vs the lowest quartile). However, there was no such a protective effect against mucinous tumors in this study.
Socioeconomic status
The socioeconomic status is one of the predictors of incidence and survival of ovarian cancer.150 Access to healthcare,151 patient awareness about the symptoms of ovarian cancer, timely response to symptoms, lifestyle, and underlying illnesses justify the link between socioeconomic status and ovarian cancer.152 The result of a case-control study showed a negative relationship between educational level and the risk of ovarian cancer.153 In the Brewster et al’s 154 study, a weaker social status was associated with the more advanced illness.
Conclusion
The aim of this study was to review the epidemiological aspects and the risk factors of ovarian cancer in the world. Ovarian cancer, as one of the major gynecological cancers, kills many women around the world, and this mortality varies from country to country. The findings of this study showed that various factors affect the occurrence of ovarian cancer, from which genetic, environmental and lifestyle factors are among the most important ones. Many factors such as pregnancy, lactation, and oral contraceptive pills play a role in reducing the risk of this disease.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Momenimovahed Z, Ghoncheh M, Pakzad R, Hasanpour H, Salehiniya H. Incidence and mortality of uterine cancer and relationship with human development index in the world. Cukurova Med J. 2017;42(2):233–240. doi:10.17826/cutf.322865
2. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
3. Coburn S, Bray F, Sherman M, Trabert B. International patterns and trends in ovarian cancer incidence, overall and by histologic subtype. Int J Cancer. 2017;140(11):2451–2460. doi:10.1002/ijc.30676
4. Yoneda A, Lendorf ME, Couchman JR, Multhaupt HA. Breast and ovarian cancers: a survey and possible roles for the cell surface heparan sulfate proteoglycans. J Histochem Cytochem. 2012;60(1):9–21. doi:10.1369/0022155411428469
5. Jacobs IJ, Menon U. Progress and challenges in screening for early detection of ovarian cancer. Mol Cell Proteomics. 2004;3(4):355–366. doi:10.1074/mcp.R400006-MCP200
6. Badgwell D, Bast JRC. Early detection of ovarian cancer. Dis Markers. 2007;23(5–6):397–410.
7. Lowe KA, Chia VM, Taylor A, et al. An international assessment of ovarian cancer incidence and mortality. Gynecol Oncol. 2013;130(1):107–114. doi:10.1016/j.ygyno.2013.03.026
8. Hunn J, Rodriguez GC. Ovarian cancer: etiology, risk factors, and epidemiology. Clin Obstet Gynecol. 2012;55(1):3–23.
9. Torre LA, Trabert B, DeSantis CE, et al. Ovarian cancer statistics, 2018. CA Cancer J Clin. 2018;68(4):284–296.
10. Chornokur G, Amankwah EK, Schildkraut JM, Phelan CM. Global ovarian cancer health disparities. Gynecol Oncol. 2013;129(1):258–264. doi:10.1016/j.ygyno.2012.12.016
11. Bell DA. Origins and molecular pathology of ovarian cancer. Modern Pathol. 2005;18(S2):S19. doi:10.1038/modpathol.3800306
12. Andrews L, Mutch DG. Hereditary ovarian cancer and risk reduction. Best Pract Res Clin Obstet Gynaecol. 2017;41:31–48. doi:10.1016/j.bpobgyn.2016.10.017
13. Reid BM, Permuth JB, Sellers TA. Epidemiology of ovarian cancer: a review. Cancer Biol Med. 2017;14(1):9. doi:10.20892/j.issn.2095-3941.2016.0084
14. Prat J. Ovarian carcinomas: five distinct diseases with different origins, genetic alterations, and clinicopathological features. Virchows Archiv. 2012;460(3):237–249. doi:10.1007/s00428-012-1203-5
15. Lukanova A, Kaaks R. Endogenous hormones and ovarian cancer: epidemiology and current hypotheses. Cancer Epidemiol Prev Biomarkers. 2005;14(1):98–107.
16. McCluggage WG. Morphological subtypes of ovarian carcinoma: a review with emphasis on new developments and pathogenesis. Pathology. 2011;43(5):420–432. doi:10.1097/PAT.0b013e328348a6e7
17. Vang R, Shih I-M, Kurman RJ. Ovarian low-grade and high-grade serous carcinoma: pathogenesis, clinicopathologic and molecular biologic features, and diagnostic problems. Adv Anat Pathol. 2009;16(5):267. doi:10.1097/PAP.0b013e3181b4fffa
18. Kurman R, Carcangiu M, Herrington C, Young R. WHO Classification of Tumours of Female ReSroductive Organs. Lyon: IARC; 2014.
19. Kurman RJ, Shih I-M. The origin and pathogenesis of epithelial ovarian cancer-a proposed unifying theory. Am J Surg Pathol. 2010;34(3):433. doi:10.1097/PAS.0b013e3181cf3d79
20. Isherwood A, Bamford J, Dupuy AV. Ovarian cancer in select EU countries: incidence, treatable populations and sales by key drug class. Rev Epidemiol Sante Publique. 2018;66:S286. doi:10.1016/j.respe.2018.05.131
21. Sopik V, Iqbal J, Rosen B, Narod SA. Why have ovarian cancer mortality rates declined? Part I. Incidence. Gynecol Oncol. 2015;138(3):741–749. doi:10.1016/j.ygyno.2015.06.017
22. Rahmani K, Moradi-Lakeh M, Mansori K, Bidokhti F, Asadi-Lari M. global inequalities in incidence and mortality of ovarian cancer and associated factors: an ecological study. Alzheimers Park Res Ther. 2018;1:1.
23. Razi S, Ghoncheh M, Mohammadian-Hafshejani A, Aziznejhad H, Mohammadian M, Salehiniya H. The incidence and mortality of ovarian cancer and their relationship with the human development index in Asia. Cancermedicalscience. 2016;10:628.
24. Malvezzi M, Carioli G, Rodriguez T, Negri E, La Vecchia C. Global trends and predictions in ovarian cancer mortality. Ann Oncol. 2016;27(11):2017–2025. doi:10.1093/annonc/mdw306
25. Ørskov M, Iachina M, Guldberg R, Mogensen O, Nørgård BM. Predictors of mortality within 1 year after primary ovarian cancer surgery: a nationwide cohort study. BMJ Open. 2016;6(4):e010123.
26. Tetsche MS, Dethlefsen C, Pedersen L, Sorensen HT, Norgaard M. The impact of comorbidity and stage on ovarian cancer mortality: a nationwide Danish cohort study. BMC Cancer. 2008;8(1):31. doi:10.1186/1471-2407-8-172
27. Chan J, Urban R, Cheung M, et al. Ovarian cancer in younger vs older women: a population-based analysis. Br J Cancer. 2006;95(10):1314. doi:10.1038/sj.bjc.6603457
28. Mohammadian M, Ghafari M, Khosravi B, et al. Variations in the incidence and mortality of ovarian cancer and their relationship with the human development index in European Countries in 2012. Biomed Res Ther. 2017;4(08):1541–1557. doi:10.15419/bmrat.v4i08.228
29. Arora N, Talhouk A, McAlpine JN, Law MR, Hanley GE. Long-term mortality among women with epithelial ovarian cancer: a population-based study in British Columbia, Canada. BMC Cancer. 2018;18(1):1039. doi:10.1186/s12885-018-4242-8
30. Zheng G, Yu H, Kanerva A, Försti A, Sundquist K, Hemminki K. Familial risks of ovarian cancer by age at diagnosis, proband type and histology. PLoS One. 2018;13(10):e0205000. doi:10.1371/journal.pone.0205000
31. Chan JK, Loizzi V, Lin YG, et al. IV invasive epithelial ovarian carcinoma in younger versus older women: what prognostic factors are important? Obstet Gynecol. 2003;102(1):156–161.
32. Chan JK, Loizzi V, Magistris A, et al. Differences in prognostic molecular markers between women over and under 45 years of age with advanced ovarian cancer. Clin Cancer Res. 2004;10(24):8538–8543. doi:10.1158/1078-0432.CCR-04-0626
33. Massi D, Susini T, Savino L, Boddi V, Amunni G, Colafranceschi M. Epithelial ovarian tumors in the reproductive age group: age is not an independent prognostic factor. Cancer. 1996;77(6):1131–1136.
34. Poole EM, Merritt MA, Jordan SJ, et al. Hormonal and reproductive risk factors for epithelial ovarian cancer by tumor aggressiveness. Cancer Epidemiol Prev Biomarkers. 2013. doi:10.1158/1055-9965.EPI-12-1183-T
35. Ries LA. Ovarian cancer. Survival and treatment differences by age. Cancer. 1993;71(2 Suppl):524–529.
36. Tung K, Goodman M, Wu A, et al. Reproductive factors and epithelial ovarian cancer risk by histologic type: a multiethnic case-control study. Am J Epidemiol. 2003;158(7):629–638.
37. Mori M, Harabuchi I, Miyake H, Casagrande JT, Henderson BE, Ross RK. Reproductive, genetic, and dietary risk factors for ovarian cancer. Am J Epidemiol. 1988;128(4):771–777.
38. Kim SJ, Rosen B, Fan I, et al. Epidemiologic factors that predict long-term survival following a diagnosis of epithelial ovarian cancer. Br J Cancer. 2017;116(7):964. doi:10.1038/bjc.2017.35
39. Salazar-Martinez E, Lazcano-Ponce EC, Gonzalez Lira-Lira G, Escudero-De Los Rios P, Salmeron-Castro J, Hernandez-Avila M. Reproductive factors of ovarian and endometrial cancer risk in a high fertility population in Mexico. Cancer Res. 1999;59(15):3658–3662.
40. Fathalla MF. Incessant ovulation–a factor in ovarian neoplasia? Lancet. 1971;2(7716):163. doi:10.1016/S0140-6736(71)92335-X
41. Moorman P, Schildkraut J, Calingaert B, Halabi S, Vine M, Berchuck A. Ovulation and ovarian cancer: a comparison of two methods for calculating lifetime ovulatory cycles (United States). Cancer Causes Control. 2002;13(9):807–811. doi:10.1023/A:1020678100977
42. Fujita M, Tase T, Kakugawa Y, et al. Smoking, earlier menarche and low parity as independent risk factors for gynecologic cancers in Japanese: a case-control study. Tohoku J Exp Med. 2008;216(4):297–307.
43. Jordan SJ, Webb PM, Green AC. Height, age at menarche, and risk of epithelial ovarian cancer. Cancer Epidemiol Biomarkers Prev. 2005;14(8):2045–2048.
44. Titus-Ernstoff L, Perez K, Cramer D, Harlow B, Baron J, Greenberg E. Menstrual and reproductive factors in relation to ovarian cancer risk. Br J Cancer. 2001;84(5):714. doi:10.1054/bjoc.2000.1596
45. Kvale G, Heuch I, Nilssen S, Beral V. Reproductive factors and risk of ovarian cancer: a prospective study. Int J Cancer. 1988;42(2):246–251.
46. Kurian AW, Balise RR, McGuire V, Whittemore AS. Histologic types of epithelial ovarian cancer: have they different risk factors? Gynecol Oncol. 2005;96(2):520–530. doi:10.1016/j.ygyno.2004.10.037
47. Franceschi S, La Vecchia C, Booth M, et al. Pooled analysis of 3 European case‐control studies of ovarian cancer: II. Age at menarche and at menopause. Int J Cancer. 1991;49(1):57–60.
48. Weiderpass E, Sandin S, Inoue M, et al. Risk factors for epithelial ovarian cancer in Japan - results from the Japan Public Health Center-based Prospective Study cohort. Int J Oncol. 2012;40(1):21–30. doi:10.3892/ijo.2011.1194
49. Hankinson SE, Colditz GA, Hunter DJ, et al. A prospective study of reproductive factors and risk of epithelial ovarian cancer. Cancer. 1995;76(2):284–290.
50. Riman T, Dickman PW, Nilsson S, et al. Risk factors for epithelial borderline ovarian tumors: results of a Swedish case-control study. Gynecol Oncol. 2001;83(3):575–585. doi:10.1006/gyno.2001.6451
51. Soegaard M, Jensen A, Hogdall E, et al. Different risk factor profiles for mucinous and nonmucinous ovarian cancer: results from the Danish MALOVA study. Cancer Epidemiol Biomarkers Prev. 2007;16(6):1160–1166. doi:10.1158/1055-9965.EPI-07-0089
52. Huusom LD, Frederiksen K, Hogdall EV, et al. Association of reproductive factors, oral contraceptive use and selected lifestyle factors with the risk of ovarian borderline tumors: a Danish case-control study. Cancer Causes Control. 2006;17(6):821–829. doi:10.1007/s10552-006-0022-x
53. Risch HA, Marrett LD, Jain M, Howe GR. Differences in risk factors for epithelial ovarian cancer by histologic type: results of a case-control study. Am J Epidemiol. 1996;144(4):363–372.
54. Adami H-O, Lambe M, Persson I, et al. Parity, age at first childbirth, and risk of ovarian cancer. Lancet. 1994;344(8932):1250–1254. doi:10.1016/S0140-6736(94)90749-8
55. Jordan SJ, Green AC, Nagle CM, et al. Beyond parity: association of ovarian cancer with length of gestation and offspring characteristics. Am J Epidemiol. 2009;170(5):607–614. doi:10.1093/aje/kwp185
56. Sköld C, Bjørge T, Ekbom A, et al. Preterm delivery is associated with an increased risk of epithelial ovarian cancer among parous women. Int J Cancer. 2018;143:1858–1867. doi:10.1002/ijc.31581
57. Mucci LA, Dickman PW, Lambe M, et al. Gestational age and fetal growth in relation to maternal ovarian cancer risk in a Swedish cohort. Cancer Epidemiol Prev Biomarkers. 2007;16(9):1828–1832. doi:10.1158/1055-9965.EPI-06-0962
58. Calderon-Margalit R, Friedlander Y, Yanetz R, et al. Preeclampsia and subsequent risk of cancer: update from the Jerusalem Perinatal Study. Am J Obstet Gynecol. 2009;200(1):
59. Moorman PG, Calingaert B, Palmieri RT, et al. Hormonal risk factors for ovarian cancer in premenopausal and postmenopausal women. Am J Epidemiol. 2008;167(9):1059–1069. doi:10.1093/aje/kwn006
60. Ness RB, Grisso JA, Cottreau C, et al. Factors related to inflammation of the ovarian epithelium and risk of ovarian cancer. Epidemiology. 2000;11(2):111–117.
61. Jia D, Nagaoka Y, Katsumata M, Orsulic S. Inflammation is a key contributor to ovarian cancer cell seeding. Sci Rep. 2018;8(1):12394. doi:10.1038/s41598-018-30261-8
62. Ness RB, Goodman MT, Shen C, Brunham RC. Serologic evidence of past infection with Chlamydia trachomatis, in relation to ovarian cancer. J Infect Dis. 2003;187(7):1147–1152. doi:10.1086/368380
63. Wong A, Maclean AB, Furrows SJ, Ridgway GL, Hardiman PJ, Perrett CW. Could epithelial ovarian cancer be associated with chlamydial infection? Eur J Gynaecol Oncol. 2007;28(2):117–120.
64. Merritt MA, Green AC, Nagle CM, Webb PM. Talcum powder, chronic pelvic inflammation and NSAIDs in relation to risk of epithelial ovarian cancer. Int J Cancer. 2008;122(1):170–176. doi:10.1002/ijc.23017
65. Lin H-W, Tu -Y-Y, Lin SY, et al. Risk of ovarian cancer in women with pelvic inflammatory disease: a population-based study. Lancet Oncol. 2011;12(9):900–904. doi:10.1016/S1470-2045(11)70165-6
66. Rasmussen CB, Jensen A, Albieri V, Andersen KK, Kjaer SK. Is pelvic inflammatory disease a risk factor for Ovarian cancer? Cancer Epidemiol Biomarkers Prev. 2017;26(1):104–109. doi:10.1158/1055-9965.EPI-16-0459
67. Risch HA, Howe GR. Pelvic inflammatory disease and the risk of epithelial ovarian cancer. Cancer Epidemiol Biomarkers Prev. 1995;4(5):447–451.
68. Chiang AJ, Chang C, Huang C-H, Huang W-C, Kan Y-Y CJ. Risk factors in progression from endometriosis to ovarian cancer: a cohort study based on medical insurance data. J Gynecol Oncol. 2018;29:3. doi:10.3802/jgo.2018.29.e28
69. Sampson JA. Endometrial carcinoma of the ovary, arising in endometrial tissue in that organ. Arch Surg. 1925;10(1):1–72. doi:10.1001/archsurg.1925.01120100007001
70. Pavone ME, Lyttle BM. Endometriosis and ovarian cancer: links, risks, and challenges faced. Int J Womens Health. 2015;7:663. doi:10.2147/IJWH.S66824
71. Mogensen JB, Kjær SK, Mellemkjær L, Jensen A. Endometriosis and risks for ovarian, endometrial and breast cancers: a nationwide cohort study. Gynecol Oncol. 2016;143(1):87–92. doi:10.1016/j.ygyno.2016.07.095
72. Melin A, Sparen P, Persson I, Bergqvist A. Endometriosis and the risk of cancer with special emphasis on ovarian cancer. Hum Reprod. 2006;21(5):1237–1242. doi:10.1093/humrep/dei462
73. Stewart LM, Holman CAJ, Aboagye-Sarfo P, Finn JC, Preen DB, Hart R. In vitro fertilization, endometriosis, nulliparity and ovarian cancer risk. Gynecol Oncol. 2013;128(2):260–264. doi:10.1016/j.ygyno.2012.10.023
74. Erzen M, Kovacic J. Relationship between endometriosis and ovarian cancer. Eur J Gynaecol Oncol. 1998;19(6):553–555.
75. Wang K-C, Chang W-H, Lee W-L, et al. An increased risk of epithelial ovarian cancer in Taiwanese women with a new surgico-pathological diagnosis of endometriosis. BMC Cancer. 2014;14(1):831.
76. Zanetta GM, Webb MJ, Li H, Keeney GL. Hyperestrogenism: a relevant risk factor for the development of cancer from endometriosis. Gynecol Oncol. 2000;79(1):18–22.
77. Cottreau CM, Ness RB, Modugno F, Allen GO, Goodman MT. Endometriosis and its treatment with danazol or lupron in relation to ovarian cancer. Clin Cancer Res. 2003;9(14):5142–5144.
78. Rossing MA, Cushing-Haugen KL, Wicklund KG, Doherty JA, Weiss NS. Risk of epithelial ovarian cancer in relation to benign ovarian conditions and ovarian surgery. Cancer Causes Control. 2008;19(10):1357.
79. Caserta R, Nesti E, Caserta L, Guerriero V, Di Francesco D, Panariello S. [Small ovarian cysts in postmenopause: assessment of their malignant potential with vaginal ultrasonography and tumor marker Ca125 titration]. Minerva Ginecol. 2001;53(1 Suppl 1):120–124.
80. Hartge P, Hayes R, Reding D, et al. Complex ovarian cysts in postmenopausal women are not associated with ovarian cancer risk factors: preliminary data from the prostate, lung, colon, and ovarian cancer screening trial. Am J Obstet Gynecol. 2000;183(5):1232–1237.
81. Crayford TJ, Campbell S, Bourne TH, Rawson HJ, Collins WP. Benign ovarian cysts and ovarian cancer: a cohort study with implications for screening. Lancet. 2000;355(9209):1060–1063.
82. Sieh W, Salvador S, McGuire V, et al. Tubal ligation and risk of ovarian cancer subtypes: a pooled analysis of case-control studies. Int J Epidemiol. 2013;42(2):579–589.
83. Ness RB, Dodge RC, Edwards RP, Baker JA, Moysich KB. Contraception methods, beyond oral contraceptives and tubal ligation, and risk of ovarian cancer. Ann Epidemiol. 2011;21(3):188–196.
84. Ness RB, Grisso JA, Vergona R, Klapper J, Morgan M, Wheeler JE. Oral contraceptives, other methods of contraception, and risk reduction for ovarian cancer. Epidemiology. 2001;12(3):307–312.
85. Madsen C, Baandrup L, Dehlendorff C, Kjaer SK. Tubal ligation and salpingectomy and the risk of epithelial ovarian cancer and borderline ovarian tumors: a nationwide case-control study. Acta Obstet Gynecol Scand. 2015;94(1):86–94.
86. Gaitskell K, Green J, Pirie K, Reeves G, Beral V. Collaborators obotMWS. Tubal ligation and ovarian cancer risk in a large cohort: substantial variation by histological type. Int J Cancer. 2016;138(5):1076–1084.
87. Cibula D, Widschwendter M, Zikan M, Dusek L. Underlying mechanisms of ovarian cancer risk reduction after tubal ligation. Acta Obstet Gynecol Scand. 2011;90(6):559–563.
88. Tsilidis K, Allen N, Key T, et al. Oral contraceptive use and reproductive factors and risk of ovarian cancer in the European Prospective investigation into cancer and nutrition. Br J Cancer. 2011;105(9):1436.
89. Royar J, Becher H, Chang-Claude J. Low-dose oral contraceptives: protective effect on ovarian cancer risk. Int J Cancer. 2001;95(6):370–374.
90. La Vecchia C, Franceschi S. Oral contraceptives and ovarian cancer. Eur J Cancer Prev. 1999;8(4):297–304.
91. Rossing MA, Cushing-Haugen KL, Wicklund KG, Doherty JA, Weiss NS. Menopausal hormone therapy and risk of epithelial ovarian cancer. Cancer Epidemiol Biomarkers Prev. 2007;16(12):2548–2556.
92. Hempling RE, Wong C, Piver MS, Natarajan N, Mettlin CJ. Hormone replacement therapy as a risk factor for epithelial ovarian cancer: results of a case-control study. Obstet Gynecol. 1997;89(6):1012–1016.
93. Glud E, Kjaer S, Thomsen B, et al. Hormone therapy and the impact of estrogen intake on the risk of ovarian cancer. Arch Intern Med. 2004;164(20):2253–2259.
94. Lacey JV
95. Wernli KJ, Newcomb PA, Hampton JM, Trentham-Dietz A, Egan KM. Hormone therapy and ovarian cancer: incidence and survival. Cancer Causes Control. 2008;19(6):605–613.
96. Mørch LS, Løkkegaard E, Andreasen AH, Krüger-Kjær S, Lidegaard Ø. Hormone therapy and ovarian cancer. Jama. 2009;302(3):298–305.
97. Liat L-G, Jaron R, Liraz O, Shlomo M, Bruno L. Are infertility treatments a potential risk factor for cancer development? Perspective of 30 years of follow-up. Gynecol Endocrinol. 2012;28(10):809–814.
98. Reigstad MM, Storeng R, Myklebust TÅ, et al. Cancer risk in women treated with fertility drugs according to parity status—A registry-based cohort study. Cancer Epidemiol Biomarkers Prev. 2017;26(6):953–962.
99. Brinton LA, Lamb EJ, Moghissi KS, et al. Ovarian cancer risk after the use of ovulation-stimulating drugs. Obstet Gynecol. 2004;103(6):1194–1203. doi:10.1097/01.AOG.0000128139.92313.74
100. Shushan A, Paltiel O, Iscovich J, Elchalal U, Peretz T, Schenker JG. Human menopausal gonadotropin and the risk of epithelial ovarian cancer. Fertil Steril. 1996;65(1):13–18.
101. Dos Santos Silva I, Wark P, McCormack V, et al. Ovulation-stimulation drugs and cancer risks: a long-term follow-up of a British cohort. Br J Cancer. 2009;100(11):1824. doi:10.1038/sj.bjc.6605086
102. Calderon-Margalit R, Friedlander Y, Yanetz R, et al. Cancer risk after exposure to treatments for ovulation induction. Am J Epidemiol. 2008;169(3):365–375. doi:10.1093/aje/kwn318
103. Potashnik G, Lerner-Geva L, Genkin L, Chetrit A, Lunenfeld E, Porath A. Fertility drugs and the risk of breast and ovarian cancers: results of a long-term follow-up study. Fertil Steril. 1999;71(5):853–859.
104. Venn A, Watson L, Lumley J, Gilles G, King C, Healy D. Breast and ovarian cancer incidence after infertility and in vitro fertilisation. Lancet. 1995;346(8981):995–1000. doi:10.1016/S0140-6736(95)91687-3
105. Franceschi S, Vecchia CL, Negri E, et al. Infertility: fertility drugs and risk of epithelial ovarian cancer in Italy. Hum Reprod. 1994;9(9):1673–1675.
106. Jensen A, Sharif H, Frederiksen K, Kjær SK. Use of fertility drugs and risk of ovarian cancer: danish population based cohort study. BMJ. 2009;338:b249. doi:10.1136/bmj.b902
107. Kazerouni N, Greene MH, Lacey JV
108. Walsh T, Casadei S, Lee MK, et al. Mutations in 12 genes for inherited ovarian, fallopian tube, and peritoneal carcinoma identified by massively parallel sequencing. Proc Natl Acad Sci U S A. 2011;108(44):18032–18037. doi:10.1073/pnas.1115052108
109. Toss A, Tomasello C, Razzaboni E, et al. Hereditary ovarian cancer: not only BRCA 1 and 2 genes. Biomed Res Int. 2015;2015:11.
110. Kotsopoulos J, Gronwald J, Karlan B, et al. Age-specific ovarian cancer risks among women with a BRCA1 or BRCA2 mutation. Gynecol Oncol. 2018;150(1):85–91. doi:10.1016/j.ygyno.2018.05.011
111. Metcalfe KA, Lynch HT, Ghadirian P, et al. The risk of ovarian cancer after breast cancer in BRCA1 and BRCA2 carriers. Gynecol Oncol. 2005;96(1):222–226. doi:10.1016/j.ygyno.2004.09.039
112. Kauff ND, Satagopan JM, Robson ME, et al. Risk-reducing salpingo-oophorectomy in women with a BRCA1 or BRCA2 mutation. N Engl J Med. 2002;346(21):1609–1615. doi:10.1056/NEJMoa020119
113. Falconer H, Yin L, Gronberg H, Altman D. Ovarian cancer risk after salpingectomy: a nationwide population-based study. J Natl Cancer Inst. 2015;107:2. doi:10.1093/jnci/dju410
114. Iordache PD, Mates D, Gunnarsson B, et al. Identification of Lynch syndrome risk variants in the Romanian population. J Cell Mol Med. 2018;22(12):6068–6076. doi:10.1111/jcmm.13881
115. Nakamura K, Banno K, Yanokura M, et al. Features of ovarian cancer in Lynch syndrome. Mol Clin Oncol. 2014;2(6):909–916. doi:10.3892/mco.2014.397
116. Lu KH, Daniels M. Endometrial and ovarian cancer in women with Lynch syndrome: update in screening and prevention. Fam Cancer. 2013;12(2):273–277. doi:10.1007/s10689-013-9664-5
117. Helder-Woolderink J, Blok E, Vasen H, Hollema H, Mourits M, De Bock G. Ovarian cancer in Lynch syndrome; a systematic review. Eur J Cancer. 2016;55:65–73. doi:10.1016/j.ejca.2015.12.005
118. Goodman MT, Wu AH, Tung KH, et al. Association of dairy products, lactose, and calcium with the risk of ovarian cancer. Am J Epidemiol. 2002;156(2):148–157.
119. Pan SY, Ugnat A-M, Mao Y, Sw W, Johnson KC;
120. McCann SE, Freudenheim JL, Marshall JR, Graham S. Risk of human ovarian cancer is related to dietary intake of selected nutrients, phytochemicals and food groups. J Nutr. 2003;133(6):1937–1942. doi:10.1093/jn/133.6.1937
121. McCann SE, Moysich KB, Mettlin C. Intakes of selected nutrients and food groups and risk of ovarian cancer. Nutr Cancer. 2001;39(1):19–28. doi:10.1207/S15327914nc391_3
122. Ong J-S, Cuellar-Partida G, Lu Y, et al. Association of vitamin D levels and risk of ovarian cancer: a Mendelian randomization study. Int J Epidemiol. 2016;45(5):1619–1630. doi:10.1093/ije/dyw207
123. Bandera EV, Lee VS, Qin B, Rodriguez-Rodriguez L, Powell CB, Kushi LH. Impact of body mass index on ovarian cancer survival varies by stage. Br J Cancer. 2017;117(2):282–289. doi:10.1038/bjc.2017.162
124. Delort L, Kwiatkowski F, Chalabi N, Satih S, Bignon YJ, Bernard-Gallon DJ. Central adiposity as a major risk factor of ovarian cancer. Anticancer Res. 2009;29(12):5229–5234.
125. Rodriguez C, Calle EE, Fakhrabadi-Shokoohi D, Jacobs EJ, Thun MJ. Body mass index, height, and the risk of ovarian cancer mortality in a prospective cohort of postmenopausal women. Cancer Epidemiol Biomarkers Prev. 2002;11(9):822–828.
126. Anderson JP, Ross JA, Folsom AR. Anthropometric variables, physical activity, and incidence of ovarian cancer: the Iowa Women’s Health Study. Cancer. 2004;100(7):1515–1521. doi:10.1002/cncr.20146
127. Kotsopoulos J, Moody JR, Fan I, et al. Height, weight, BMI and ovarian cancer survival. Gynecol Oncol. 2012;127(1):83–87. doi:10.1016/j.ygyno.2012.05.038
128. Beehler GP, Sekhon M, Baker JA, et al. Risk of ovarian cancer associated with BMI varies by menopausal status. J Nutr. 2006;136(11):2881–2886. doi:10.1093/jn/136.11.2881
129. Leitzmann MF, Koebnick C, Danforth KN, et al. Body mass index and risk of ovarian cancer. Cancer. 2009;115(4):812–822. doi:10.1002/cncr.24086
130. Cottreau CM, Ness RB, Kriska AM. Physical activity and reduced risk of ovarian cancer. Obstet Gynecol. 2000;96(4):609–614.
131. Moorman PG, Jones LW, Akushevich L, Schildkraut JM. Recreational physical activity and ovarian cancer risk and survival. Ann Epidemiol. 2011;21(3):178–187. doi:10.1016/j.annepidem.2010.10.014
132. Huang T, Eliassen AH, Hankinson SE, et al. A prospective study of leisure‐time physical activity and risk of incident epithelial ovarian cancer: impact by menopausal status. Int J Cancer. 2016;138(4):843–852. doi:10.1002/ijc.29834
133. Webb PM, Purdie DM, Bain CJ, Green AC. Alcohol, wine, and risk of epithelial ovarian cancer. Cancer Epidemiol Biomarkers Prev. 2004;13(4):592–599.
134. Chang ET, Canchola AJ, Lee VS, et al. Wine and other alcohol consumption and risk of ovarian cancer in the California Teachers Study cohort. Cancer Causes Control. 2007;18(1):91–103. doi:10.1007/s10552-006-0083-x
135. Kuper H, Titus-Ernstoff L, Harlow BL, Cramer DW. Population based study of coffee, alcohol and tobacco use and risk of ovarian cancer. Int J Cancer. 2000;88(2):313–318.
136. Tworoger SS, Gertig DM, Gates MA, Hecht JL, Hankinson SE. Caffeine, alcohol, smoking, and the risk of incident epithelial ovarian cancer. Cancer. 2008;112(5):1169–1177. doi:10.1002/cncr.23275
137. Riman T, Dickman PW, Nilsson S, Nordlinder H, Magnusson CM, Persson IR. Some life-style factors and the risk of invasive epithelial ovarian cancer in Swedish women. Eur J Epidemiol. 2004;19(11):1011–1019.
138. Goodman MT, Tung K-H. Alcohol consumption and the risk of borderline and invasive ovarian cancer. Obstet Gynecol. 2003;101(6):1221–1228.
139. Schouten LJ, Zeegers MP, Goldbohm RA, van den Brandt PA. Alcohol and ovarian cancer risk: results from the Netherlands Cohort Study. Cancer Causes Control. 2004;15(2):201–209. doi:10.1023/B:CACO.0000019512.71560.2b
140. Goodman MT, Tung KH. Active and passive tobacco smoking and the risk of borderline and invasive ovarian cancer (United States). Cancer Causes Control. 2003;14(6):569–577. doi:10.1023/A:1024828309874
141. Jordan SJ, Green AC, Whiteman DC, Webb PM. Risk factors for benign, borderline and invasive mucinous ovarian tumors: epidemiological evidence of a neoplastic continuum? Gynecol Oncol. 2007;107(2):223–230. doi:10.1016/j.ygyno.2007.06.006
142. Gram IT, Braaten T, Adami HO, Lund E, Weiderpass E. Cigarette smoking and risk of borderline and invasive epithelial ovarian cancer. Int J Cancer. 2008;122(3):647–652. doi:10.1002/ijc.23108
143. Marchbanks PA, Wilson H, Bastos E, Cramer DW, Schildkraut JM, Peterson HB. Cigarette smoking and epithelial ovarian cancer by histologic type. Obstet Gynecol. 2000;95(2):255–260.
144. Pan SY, Ugnat AM, Mao Y, Wen SW, Johnson KC. Association of cigarette smoking with the risk of ovarian cancer. Int J Cancer. 2004;111(1):124–130. doi:10.1002/ijc.20242
145. Modugno F, Ness RB, Cottreau CM. Cigarette smoking and the risk of mucinous and nonmucinous epithelial ovarian cancer. Epidemiology. 2002;13(4):467–471.
146. Gram IT, Lukanova A, Brill I, et al. Cigarette smoking and risk of histological subtypes of epithelial ovarian cancer in the EPIC cohort study. Int J Cancer. 2012;130(9):2204–2210. doi:10.1002/ijc.26235
147. FRANKS AL LEE, Kendrick NC, Rubin JS, et al. Cigarette smoking and the risk of epithelial ovarian cancer. Am J Epidemiol. 1987;126(1):112–117.
148. Su D, Pasalich M, Lee AH, Binns CW. Ovarian cancer risk is reduced by prolonged lactation: a case-control study in southern China. Am J Clin Nutr. 2013;97(2):354–359. doi:10.3945/ajcn.112.044719
149. Jordan SJ, Siskind V, Green AC, Whiteman DC, Webb PM. Breastfeeding and risk of epithelial ovarian cancer. Cancer Causes Control. 2010;21(1):109–116. doi:10.1007/s10552-009-9440-x
150. Booth CM, Li G, Zhang-Salomons J, Mackillop WJ. The impact of socioeconomic status on stage of cancer at diagnosis and survival: a population-based study in Ontario, Canada. Cancer. 2010;116(17):4160–4167. doi:10.1002/cncr.25427
151. Morris CR, Sands MT, Smith LH. Ovarian cancer: predictors of early-stage diagnosis. Cancer Causes Control. 2010;21(8):1203–1211. doi:10.1007/s10552-010-9547-0
152. Praestegaard C, Kjaer SK, Nielsen TS, et al. The association between socioeconomic status and tumour stage at diagnosis of ovarian cancer: A pooled analysis of 18 case-control studies. Cancer Epidemiol. 2016;41:71–79. doi:10.1016/j.canep.2016.01.012
153. Alberg AJ, Moorman PG, Crankshaw S, et al. Socioeconomic status in relation to the risk of Ovarian cancer in African-American Women: a population-based case-control study. Am J Epidemiol. 2016;184(4):274–283. doi:10.1093/aje/kwv450
154. Brewster DH, Thomson CS, Hole DJ, Black RJ, Stroner PL, Gillis CR. Relation between socioeconomic status and tumour stage in patients with breast, colorectal, ovarian, and lung cancer: results from four national, population based studies. BMJ. 2001;322(7290):830–831.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
