Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
Oral administration of encapsulated bovine lactoferrin protein nanocapsules against intracellular parasite Toxoplasma gondii
Authors Anand N, Sehgal R, Kanwar R, Dubey M, Vasishta RK, Kanwar J
Received 23 March 2015
Accepted for publication 18 July 2015
Published 8 October 2015 Volume 2015:10(1) Pages 6355—6369
DOI https://doi.org/10.2147/IJN.S85286
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Thomas Webster
Namrata Anand, 1 Rakesh Sehgal, 1 Rupinder Kaur Kanwar, 2 Mohan Lal Dubey, 1 Rakesh Kumar Vasishta, 3 Jagat Rakesh Kanwar 2
1Department of Medical Parasitology, Postgraduate Institute of Medical Education and Research, Chandigarh, India; 2Nanomedicine-Laboratory of Immunology and Molecular Biomedical Research, School of Medicine, Centre for Molecular and Medical Research, Faculty of Health, Deakin University, Geelong, VIC, Australia; 3Department of Histopathology, Postgraduate Institute of Medical Education and Research, Chandigarh, India
Abstract: Toxoplasma gondii is a deadly intracellular parasite known to reside in every nucleated cell and known to cause severe complications in immunocompromised host. Standard drugs are cost effective and cause side effects, therefore, there is a necessity for a new drug molecule with immunomodulatory potential. Lactoferrin (Lf) is a natural milk protein, which has shown antimicrobial properties in its nanoformulation using alginate chitosan calcium phosphate bovine lactoferrin nanocapsules (AEC-CCo-CP-bLf-NCs). The present study was aimed to analyze and compare the effect of bovine Lf (bLf) in its native as well as nanoformulation (AEC-CCo-CP-bLf-NC) against coccidian parasite T. gondii. In vitro analysis has shown a significant increase in nitric oxide production and low parasitemia in in vitro cell culture model. In vivo BALB/c mice model have been used to develop human toxoplasmosis model. After treatment with NCs it has substantially increased the bioavailability of the protein and showed comparatively increased levels of reactive oxygen species, nitric oxide production, and Th1 cytokine which helped in parasite clearance. The mechanism of action of NCs has been clarified by immunoreactivity analysis, which showed accumulation of Lf in macrophages of various visceral organs, which is the site of parasite multiplication. Effect of NCs has significantly decreased (P< 0.05) the parasite load in various organs and helped survival of mice till day 25 postinfection. Fe metabolism inside the mice has been found to be maintained even after administration of mono form of Lf, this indicates novelty of Lf protein. From the present study we concluded that nanoformulation did not reduce the therapeutic potential of Lf protein; however, nanoformulation has enhanced the stability of the protein and shown anti-toxoplasmal activity. Our study presents for the first time nanoformulation of Lf protein against Toxoplasma, which has advantages over the standard drug therapy without any side effects.
Keywords: nanocapsules, oral delivery, cytokines, Toxoplasma gondii, ceramic nanocapsules and reactive oxygen species, immunoreactivity, parasite load
Corrigendum for this paper has been published
Introduction
Toxoplasma gondii is one of the most important intracellular coccidian parasites known to cause toxoplasmosis in human and animals. T. gondii is a deadly parasite as it has the capacity to invade all nucleated cells and highly fatal during pregnancy. It is generally assumed that approximately 25%–30% of the world’s human population is infected by this disease. Toxoplasma infection in human is most common in immunocompromised and immunocompetent situation like in case of HIV-infected person, pregnant women, and transplant patients. The newborn gets infection from infected mother in later stages of pregnancy.
High levels of interleukin-10 (IL), IL-4, and IL-8 may lead to rapid multiplication of parasite and progression of disease in immunocompromised patients, which lead to ocular infection, schizophrenia1,2 like complication. Treatment of toxoplasmosis includes routine old drugs such as sulfadiazine and pyrimethamine,3 which are good for the treatment but all of them show adverse side effects leading to gastric problem, weight loss, headache, blurred vision, etc. Side effects of these drugs can be overcome by using natural potential protein/drug molecule, which directly targets on the infected site without causing any side effects. Therefore, there is a need of immunomodulatory drug that boost up the immune response against infection and help in recovery. In the field of new technology and advancement, we need a natural potent molecule that has already been documented against many microbes, acts without any side effects, and also does not show any resistance. Various studies have been reported on immune regulatory, antimicrobial, and antioxidant capacities of natural herbal and dairy products. Among all the milk proteins, Lactoferrin (Lf) has been found to be the most potent one. There are several studies that have documented the role of Lf molecules against many microbes like bacteria, viruses, and fungal infections.4
Lf is a 78–80 kDa molecule known to be present in many body fluids like milk, colostrum, serum, semen, and tears, therefore being a part of the body it does not cause any side effects5 and show immunomodulatory activity.6 Antiparasitic activity has also been documented against parasites like Entamoeba,7 Giardia,8 Cryptosporidium,9 and Trypanosoma10 but clear mechanism of action has not been detailed out in these studies. The effect of natural Lf protein is limited in these studies and results are not so promising. Due to the bioavailability of this protein, it has given a lot of curiosity for its use in advance drug treatment for better drug delivery and target therapy. Among the various routes of drug administration, oral drug delivery is marked as the safest. But, for development of novel drug molecule there are a lot of issues that has to be faced when drugs are being passed through gastrointestinal tract of human. These include enzymatic action and gut pH, which may lead to degradation of this protein. Recent advances in nanotechnology have improved the onsite drug therapy as well as oral delivery system for better efficacy of loaded drug molecules. Nanotechnology has offered various benefits such as slow drug delivery, target specificity, better efficacy, and less side effects, which are indispensible properties for drug design.
Various nanoparticles have been used to load Lf molecules for target delivery,11 against many infections.12 Lipid nanoparticles have been used against cancer and for brain carcinoma delivery. Kanwar et al and group has prepared alginate chitosan calcium phosphate nanocapsules (AEC-CCo-CP-NCs), which have been extensively used as drug carriers and as drug therapy. Alginate and chitosan are biodegradable polymers, and calcium phosphate is naturally present in teeth therefore are safe to use as NCs. These NCs have been loaded with bovine Lf (bLf) molecule to protect it from degradation of gut environment. Therefore, bioavailability of Lf protein at the site of infection does not alter and has shown maximum efficacy.13–15
Earlier successful studies have shown efficacy of these NCs against various tumors16 and osteoarthritis,17 and have shown promising results against diseased mice models. Not only Lf protein has been incorporated in these NCs, but also herbal products have been tried against arthritis like diseases,18 which has divulged multiple availability of these NCs for any drug/molecule/peptide to be loaded. These success stories have initiated the idea to use NCs in treatment of parasitic infections. These NCs loaded with Lf have never been tried against parasitic diseases like toxoplasmosis; therefore, its potential effects need utmost evaluation. The present study is therefore the first study that has tried to scrutinize the effect of these NCs against Toxoplasma. So, the present study proposes to analyze the effect of native bLf protein along with its nanoformulation (AEC-CCo-CP-bLf-NC) against intracellular parasite T. gondii. This study is the pioneer one to establish the antiparasitic effect against T. gondii infection using bLf nanoformulation.
Materials and methods
Toxoplasma gondii
T. gondii was routinely maintained in Department of Medical Parasitology, PGIMER, Chandigarh, India in Swiss albino mice through intraperitoneal (I/P) route and passage was done every 3–4 days.
In vitro study
Cell lines and cell culture conditions
The J7741 mouse macrophage cell line was collected from National Cell Culture Science (NCCS) Pune, India and was grown in tissue culture flasks using RPMI medium containing high glucose and L-glutamine, supplemented with 20% heat-inactivated fetal bovine serum (Sigma-Aldrich, St Louis, MO, USA). Penicillin/streptomycin was added at a final concentration of 0.1 mg/mL to prevent contamination of microbes, and the culture was maintained at 37°C in 5% CO2 to ensure a saturated humid atmosphere. Culture media were changed every 2–3 days and cells were passaged at 90% confluency.
In vitro cell culture model
J7741 mouse macrophages were taken as a source of host cells for growth of intracellular parasites and were infected with RH strain of T. gondii tachyzoites. Tachyzoites were incubated with macrophage cells at a ratio of 0.5:1 and examined after 24 hours for calculating mean number of tachyzoites present inside each macrophage. After obtaining optimum parasitemia, treatment of Lf protein was started.
Lactoferrin
bLf was obtained from Australia. Various iron saturated forms of bLf such as apo, mono, and holo forms were prepared and used for in vitro inhibition assay. Fe-saturated Lf was prepared by the method previously described by Bates and Wernicke.19 Apo Lf was prepared by the method previously described by Feng et al20 with minor modifications. Iron estimation of all forms of Lf was done by using previous method with minor modification.21 Apo Lf was found to be 8%–10% saturated with Fe, mono form of Lf was found to be 40%–65% saturated, and holo form of Lf was found to be 85%–95% saturated.
In vitro inhibition study
After obtaining optimum parasitemia in J7741 macrophage cells as described above, bLf in its various Fe formulations was incubated at different doses of 10, 20, and 40 μg/mL for 24 hours. Sulphadiazine, a standard drug was kept as positive control and used at a concentration of 20 μg/mL. Cells with only media were kept as an untreated group.
Nitrite production
Nitrite production from Nitrate is a measurement of nitric oxide (NO) within cells and was measured by using Griess reagent (Sigma-Aldrich) as per standard protocol.22 Briefly, cell culture supernatant of different groups were taken in 96-well plates and mixed with Griess reagent. Samples were allowed to keep at room temperature for 15 minutes and reading was taken at 595 nm using spectrophotometer. Amount of NO production was calculated by plotting standard curve against the reading obtained from culture supernatant of different groups. The results were expressed as the mean nitrite concentration (μM/mL) ± standard error of the mean.
In vivo study
Preparation of diet containing AEC-CCo-CP-bLf NCs for oral therapy
The bLf dose for oral therapy was standardized in earlier studies,15 where we found that 1.2% (w/w) or 12 g Fe-bLf/kg of diet showed no side effects and helped in clearing various tumors. The same dose was then used in our recently published study with a nanoformulated Fe-bLf diet13 with similar findings. But, in present study instead of Fe-Lf, native form of bLf has been used along with its nanoformulation. The diet was mixed with the appropriate amount of autoclaved milli Q water and made into pellets of suitable sizes, to be able to give as a treatment diet. An equal quantity of this diet was given to mice each time to normalize the dosage in metabolic cages. The detailed structure and release profile of NCs has been studied previously13 and after the success stories of these NCs they have been now targeted against intracellular parasite T. gondii.
Animal model of human toxoplasmosis and in vivo studies
A human toxoplasmosis disease model was developed by inoculating 100 tachyzoites of T. gondii through I/P route inside Balb/c mice. All animal procedures were done in accordance with the guidelines from institutional animal ethics committee (IAEC), PGIMER, Chandigarh, India. All animal experiments included five mice per treatment group and 4- to 5-week-old female mice (Mus musculus mice-BALB/c) were used in the present study. The mice were fed either control AIN 93G diet or AIN 93G supplemented with either of the following treatments: bLf; AEC-CCo-CP-bLf NCs and sulfadiazine as standard drug. Mice were weighed thrice weekly and assessed for signs of any physical or physiological distress. Sulfadiazine standard drug was administrated through oral route at a concentration of 40 mg/kg/day. Parasitemia was monitored routinely and the presence of tachyzoite in blood was confirmed as a marker for infection. As infection appeared in the blood, different drugs were given as described previously. The parasite started multiplying and appeared in blood after day 5 postinfection. So, the effect of different formulations was studied at days 10 and 15 postinfection and different parameters were assessed. Details of the experiment have been mentioned in Table 1.
Histopathological analysis
In terms of tissue sample preparation, mice were sacrificed under euthanasia by cervical dislocation, liver and spleen were removed aseptically and fixed in 10% buffered formalin and processed for histological examination. Histopathological examination of parasite was assessed by observing the presence of inflammation and the presence of parasite with the use of Hematoxylin and Eosin stain (H&E) as per standard protocol.23
Parasite load determination – absolute quantification (real time–polymerase chain reaction)
Parasite load determination was done by using real time-polymerase chain reaction (RT-PCR) using absolute quantification. DNA was extracted from the blood of Toxoplasma-infected mice and quantification of extracted DNA was done using Nano drop (Thermo Fischer, Waltham, MA, USA). From the extracted DNA, B1 gene of T. gondii was amplified using specific primers as described earlier24 and PCR product was purified using Roche kit (Roche, Basel, Switzerland). The number of copies per unit volume (mL) was calculated using the formula described elsewhere.25 Serial dilutions of these stock solutions served as quantification standards to plot the standard curves. Following formula was used to calculate the copy number.
Number of copies/mL =6.023×1023×C×OD260/Mwt |
where 6.023×1023= Avogadro’s number, C =5×10−5 g/mL for DNA, Mwt = bp ×6.58×102 g OD260 = absorbance at 260 nm.
When assaying the samples for Toxoplasma B1 gene, the corresponding standard series was run under the same conditions and the copy number of the samples was determined by reading off the standard series with the crossing point (Cp) values of the samples.
Quantification of reactive oxygen species and NO production in different organs
Reactive oxygen species (ROS) are produced intracellularly through multiple mechanisms depending upon the cell and tissue type. ROS production helps in host defense as it kills intracellular microbes, therefore its production was observed in liver and spleen cells of mice using the dihydro-fluorescein diacetate dye (DCFH). The spleen and the liver were aseptically removed and chopped with the help of sterile surgical blade into single cell suspension, which was washed with 1× phosphate buffer saline. Cells were stained with dihydro-fluorescein diacetate dye as previously mentioned26 and acquired with flow cytometry using fluorescence activated cell sorting (FACS Caliber, BD Bioscience, Oxford, UK).
NO production was estimated as described previously using Griess reagent. Mice were sacrificed, blood was collected from heart, and serum was separated by centrifugation, and levels of NO were measured as per standard protocol.
Immunoreactivity for Lf localization
Mice were sacrificed from different groups after the treatment period, spleen and liver tissues were collected aseptically. For immunohistochemical staining, the sections were fixed in formalin as described earlier. The sections were rehydrated with tris-buffered saline (TBS)–Tween-20 and blocked with 3% hydrogen peroxide in 3% bovine serum albumin to overcome endogenous peroxidase activity. Goat anti-bLf antibody HRP conjugate (Bethyl Labs, Montgomery, TX, USA) (1:1,000) was used for the detection of exogenous bLf. The sections were then washed in TBS–Tween-20, developed with 3,3′-diaminobenzidine substrate (Sigma-Aldrich) and incubated for 10 minutes in the dark. After counterstaining with hematoxylin, slides were fixed with permanent mounting media and observed under 20× and 40× oil immersion objectives using bright field microscope (Nikon Y1000).
Cytokine profile
Cytokines expressed in mouse serum were determined using the Single Analyte ELISA Kits (QIAGEN) for IL-1β, IL-6, IL-18, IL-12, IL-10, IL-15, IFN-1α, IFN-1β, IL-2, IL-4, IL-5, IL-17, IL-13, IFN-γ, IL-19, GM-CSF, IL-7, IL-11, IL-3, and tumor growth factor-beta (TGF-β), and followed as per manufacturer’s instructions as described earlier.13,14 In brief, after euthanizing mice, blood was collected via cardiac puncture and transferred into unheparinized tubes. The blood was allowed to clot by incubating the tubes at RT for 20 minutes. Blood clot was removed by centrifugation at 2,000× g for 10 minutes at 4°C and the supernatant was carefully transferred into a fresh tube and stored immediately at −80°C.
Biodistribution
Tissues (small intestine, spleen, liver, large intestine, brain, kidney, lung, heart, and stomach) from treated group of mice were removed and stored at −80°C until use. bLf was measured using ELISA with antibodies to bLf and amount in ng/mL of Lf was plotted using a histogram.
Regulation of miRNA and genes involved in iron metabolism by AEC-CCo-CP-bLf-NCs
Gene expression of iron regulation of nano-bLf diet was analyzed using RNA isolated from mice tissues using the Trizol method. miRNA involved in iron metabolism and expression in different mice tissues were analyzed using miRNA isolation kit. Tissues stored in RNA later were homogenized thoroughly on ice before isolation of miRNA was performed as per manufacturer’s instructions. Samples were run in triplicates and the mean Ct value used to calculate regulation of genes using the 2−ΔΔCt method.
Survival curve
Survival curve was drawn from different groups. Mortality and survival of mice in different groups were observed till the completion of the experiment and survival curve was plotted till day 25 postinfection by using Graph pad prism software.
Statistical analysis
All the statistical analysis were done using Graph pad Prism version 5.0. Parasite copy number was calculated using Box plot assay. Measurement of in vitro growth inhibition, ROS, NO production, copy number, cytokine assay, and biodistribution were analyzed by unpaired t-test. One-way ANOVA test was applied for analysis of parasitemia and cytokine response between more than two group. Results were presented as SD ± or SEM. Any P>0.05 is presented as non-significant values and *P<0.05 is considered as significant, **P<0.01, ***P<0.001 are highly significant.
Results
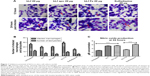
Efficacy of native bLf on in vitro cell culture model of T. gondii
Antiparasitic activity of different forms of bLf protein was assessed by using different concentrations. Out of all three protein forms, native bLf has shown maximum inhibitory effect (IC50 value) at 20 μg/mL concentration as shown in Giemsa smears showing least number of infected macrophages and number of tachyzoites per macrophage (Figure 1A), whereas Fe-bLf supported the parasite growth (large number of infected macrophages) compared with untreated and apo form of protein when observed at 12 and 24 hour treatment (Figure 1A). Apo form (iron free) of the protein has inhibited the parasites to a certain extend but due to less iron (Fe) content, few macrophages showed deformation and degradation. Among all the treatment groups used, native bLf showed best results followed by sulfadiazine and apo bLf, which have shown very few number of intracellular parasites as well as number of infected macrophages (Figure 1B). The native form of the protein was found to be non-toxic to the cells as described previously in our study and therefore chosen for in vivo study.26
NO levels
Nitrite production is a measurement of NO production and was found to be significantly elevated in all the treatment groups compared with untreated groups. The highest level of NO production was found to be present in bLf and bLf apo-treated group, which can be co-related with less number of infected macrophages as well as few number of intracellular tachyzoites mentioned in microscopy results. Similarly, NO levels were found to be higher when treated with standard drug sulfadiazine (20 μg/mL) (Figure 1C). These results indicate that NO production has helped in reducing the number of intracellular tachyzoites significantly in the above-mentioned group.
Inhibitory effect of bLf and AEC-CCo-CP-bLf against Toxoplasma gondii in vivo
All the parameters were studied at days 10 and 15 postinfection. To reduce the cost of the study, few parameters were studied at day 15 postinfection.
Histopathological analysis
Histopathological studies were performed in liver and spleen sections to assess the effect of different formulations, standard drugs and various parameters were studied at days 10 and 15 postinfection.
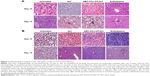
Liver
Liver sections of untreated control group showed the presence of inflammation due to accumulation of lymphocytes and histiocytes along with actively multiplying parasites (tachyzoite forms) (Figure 2Ai). Whereas in all treatment groups including bLf, AEC-CCo-CP-bLf and sulfadiazine, no histopathological abnormalities like inflammation or tissue degradation were found. Moreover, no sign of parasitic infection was observed at day 10 postinfection (Figure 2Aii–Aiv). When histopathological liver sections were observed a day 15 postinfection, the presence of tachyzoites at multiple foci (red arrows) were seen in untreated control group (Figure 2Av). However, young tissue cyst was found in liver sections of bLf and AEC-CCo-CP-bLf NC group with sign of inflammation (Figure 2Avi and Avii) but no sign of parasite multiplication tachyzoites or tissue cyst was detected in sulfadiazine group (Figure 2Aviii).
Spleen
Spleen histopathological analysis showed different results from liver section studies. Untreated control group has shown multiple sites of parasite multiplication (red arrows) along with inflammation at days 10 and 15 postinfection (Figure 2Bi and Bv). Whereas no histopathological abnormalities were observed in different treatment groups at days 10 and 15 postinfection (Figure 2Bii–Biv, Bvi, and Bvii) except sulfadiazine group which showed inflammatory signs in spleen sections at day 15 postinfection (Figure 2Bviii). These results indicate the anti-inflammatory property of bLf protein, which does not cause any tissue damage and help inhibiting the growth of parasite.
Quantitative assessment of parasite load in liver and spleen
Quantitative assessment of parasite load was performed by using RT-PCR through quantitative analysis, which was used to calculate the mean copy number of parasite from liver and spleen tissue lysates. The distribution of T. gondii copy number in different groups was represented by graph plots (Figure 3). There was significant difference found between copy number of treated groups and untreated group (P<0.05). Highest copy number of 2,833 was found in untreated group, followed by 2,199 in bLf, 2,083 in NCs group and least copy number of 1,846 found in sulphadiazine group. These results are in concordance with histopathological findings, which showed more number of parasites in untreated group. Sulfadiazine-treated mice showed least number of copy number followed by NCs group and bLf treatment group (Figure 3).
ROS and NO production in liver and spleen of mice
ROS and NO production was used as a marker to observe the intracellular killing of parasites, observed at days 10 and 15 postinfection. Intracellular ROS production levels were found to be highly elevated in liver and spleen cells of all treatment groups of mice. There was a significant increase in mean fluorescence intensity (MFI) two- to fourfold change between different groups (Figure 4A and B). Highest shift in MFI from untreated group was found in sulphadiazine group followed by bLf and NCs in liver. Whereas, highest shift in MFI was found in NCs, sulphadiazine group in spleen cells. High levels of ROS production can be correlated with histopathological and quantitative parasite load detection, which has shown very less amount of parasitemia in treated group of mice. Similarly, substantial amount of NO was found elevated in treatment groups as compared with untreated group when observed at days 10 and 15 postinfection (Figure 4C). NO levels were found to be significantly high in NCs group (P>0.005) compared with bLf group, which showed the potential of NCs in clearing the parasitemia by producing intracellular free radicle production.
Cytokine response
Cytokine production was checked in mice by examining various innate and adaptive cytokine responses in bLf, NCs, and sulfadiazine group. Various innate immunity cytokines were found to be elevated in treatment groups of bLf and NCs such as IL-6, which is required for B-cell proliferation. Whereas lower levels of various cytokines were observed in sulfadiazine group (Table 2). An IL-12 elevated level signified the proliferation of T helper cells production and was found to be highly elevated in NCs and bLf treatment group. All other innate immune cytokines such as IL-18, IFN-1α, IFN-1β, and IL-17 showed increased levels in bLf and AEC-CCo-Cp-bLf groups compared with untreated control and sulfadiazine group. Various adaptive immunity cytokines were studied such as IL-2, IL-4, IL-5, IL-13, and IFN-γ, which has shown elevated levels in NCs treatment group compared with untreated group. However, only IL-10 which is a marker of Th2 response was found to be elevated in NCs group as well as in untreated group, the reason for this high level is unknown. Cytokines of hematopoiesis like IL-7, IL-11, IL-3, and TGF-β showed increased concentration up to threefold in NCs followed by bLf-treated mice compared with untreated group and sulfadiazine. All these cytokine markers are helpful in cell survival, differentiation, and proliferation as they are the stem-cell markers (Table 2) and therefore these cytokine results showed enhanced Th1 type of response which is highly expressed in case of NCs group compared with untreated group leading to survival of mice for a longer time.
Regulation of miRNA and genes involved in iron metabolism
Iron is an essential nutrient critical for many cellular functions including DNA synthesis, ATP generation, and cellular proliferation. So, it was essential to study the various markers involved in iron regulations after treatment of mice with mono form of bLf, AEC-CCo-CP-bLf NCs having one Fe molecule. Various markers such as miR-Let7d, miR-122, miR-196, miR-200b, miR-210, miR-214, miR-320, miR-485, and miR-584 were studied in different mice organs like intestine, spleen, liver, and brain at day 15 postinfection. Lower levels of miR-Let7d were found in all organs compared with untreated group. MiR122 levels that were highly expressed in spleen and brain compared with other organs. High expression levels of miR 200 were observed in intestine and brain of mice. MiR196 levels showed high expression in intestine and liver. Elevated levels of miR485 were found in all organs involved in all treated group compared with untreated group (Figure 5A). When Fe metabolism was studied in bLf-treated mice, negligible expression was observed and therefore these results were not included in the present study. These elevated levels of different iron regulators showed that even in its native form, bLf is capable of maintaining the appropriate iron metabolism and hence can inhibit the parasite and also maintain the tissue integrity.
Biodistribution of bLf in tissue lysates
Biodistribution of bLf in various tissue lysates obtained from blood, liver, spleen, kidney, lung, heart, brain, stomach, small intestine, and large intestine was studied and compared between mice fed with native bLf diet and NCs treatment mice. Results revealed high levels of bLf concentration expressed in ng/mL in NCs treatment group compared with native protein group. Levels of bLf were found to be approximately twofold high in case of NCs in all organs, which show the promising bioavailability of the protein after encapsulation and target delivery (Figure 5B).
Immunoreactivity
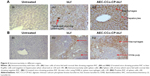
In order to assess the internalization of bLf and AEC-CP-bLf-NCs in the spleen and liver tissues, immunohistochemical observation for the bLf positivity was carried out. Interesting findings were observed when immunoreactivity was done in different organs. These observations imply the bioavailability of bLf at infected site.
Liver
Immunoreactivity was found to be positive only in macrophages of mouse liver cells (Kupffer cells) and negative in hepatocytes when observed from bLf- and NC-treated mice. Immunoreactivity was negative in hepatocytes and Kupffer cells of mice of untreated group as shown in Figure 6A. As histopathological findings have suggested that tissue cyst developed in Kupffer cells (macrophages) only, therefore, Lf must have accumulated at the site of parasite multiplication as shown in Figure 6Aii (positivity of tissue cyst with DAB) and hence showed its mechanism of action by converting actively multiplying tachyzoite form to dormant bradyzoite stage.
Spleen
Immunoreactivity of Lf was found to be positive in mouse spleen macrophages and negative in white pulp. Macrophage area showed positive brown color developed by Diaminobenzidine (DAB) chromogen, which was found to be negative in untreated group of mice (Figure 6B). This reactivity of Lf in spleen showed the accumulation and site of action of Lf inside the spleen macrophages, red pulp, which is the only site for multiplication of the parasite, T. gondii. These immunoreactivity results obtained from different organs have revealed the mode of mechanism of action of Lf protein against intracellular parasites. As Lf is found to be present in macrophages only, this proves that Lf has directly inhibited the parasite multiplication inside the macrophages and forced it to change to dormant tissue cyst stage.
Survival assay
To measure the survival tendency of mice treated with different formulation and untreated group, we kept the mice on normal diet as well as bLf, NC diet and sulfadiazine for 25–30 days (total period of experiment) and monitored them for remainder of their lifespan. As explained in Figure 7, mean and maximum life was extended by NC-treated mice till 25 days and was significantly different from untreated control group that survived till day 10 only. Survival among the NC and bLf groups was significantly high from untreated group who lived longer on their counterparts. Survival was found to be 100% in sulfadiazine group of mice. Two mice from bLf group and one from NCs group of mice died at day 20 postinfection (Figure 6). Whereas all mice died in untreated group by day 10 postinfection. Mortality of 6/6 was found in untreated group at day 10 postinfection. Whereas the mortality of 2/6 was observed at day 20 postinfection in bLf and 1/6 was observed in NCs treatment group (Figure 7).
Discussion
In order to be able to overcome the side effects of present-day’s toxic drugs, there is a need of natural potent molecule causing no side effects and help in developing immunity. To date, sulfadoxine/pyrimethamine has been tested successfully against toxoplasmosis. Although these drugs are effective, but cause many gastric problems. Efficacy of these drugs has been found to be low in subclinical infection. Suppression of tissue cyst with sulfadiazine in mouse has been found to be of little effect. Also these drugs are not able to cross the placenta efficiently for treatment of congenital toxoplasmosis. Due to over dose, side effects, and adverse reaction in immunocompromised host, these drugs need a replacement with natural therapy. Among the natural molecules used, Lf, a milk protein has been studied since so many decades for its antimicrobial properties. Recent study has shown the effect of this protein encapsulated in alginate chitosan NCs against cancer and osteoarthritis like diseases. These NCs have proven to be biologically safe and efficient to transport the drug across the mucosal, gastric barrier of human alimentary canal. These NCs are safe for oral route delivery and therefore, incorporated in animal diet. The alginate coating is not dissolved at lower pH in stomach but dissolve at higher pH (>6) in the intestine and due to mucoadhesive nature of the chitosan the remaining CCo-CP-bLf NCs stick to the intestine and pass through due to endocytosis and/or transcytosis.27 The efficacy of these NCs has already been studied against bacterial infection and cancer. Activity of the loaded Lf protein as well as efficacy has been shown previously by Kanwar et al and was found to be present in blood plasma at a concentration of 1,000 ng/mL after 8 hours postadministration and up to 100 ng/mL at 10 days postadministration, which has been found to be optimum to inhibit any tumor or microbial infection. The integrity of NCs in the blood has already been measured in our previous studies.13,14,33 In previous study by Gupta et al control release profile of bLf as well as NC group in different time intervals have been studied.33 In this study, administration of single dose of 100 mg/kg/body weight of NCs was given in mice and bLf controlled release has been studied in different time interval ranging from 0, 15, 30 minutes, 1, 3, 6, 8, 10, 12, 24, 48 hours to 5, 7, and 10 days. The maximum release has been observed at 8 hours postadministration (1,000 ng/mL) whereas 100 ng/mL has been observed at day 10 postinfection. Also the integrity of NCs in acidic and alkaline pH has been measured and NCs have been found to reach stomach and intestine and Lf was found to be intact in these NCs. Lf is an immunomodulatory protein, which has suppressed every tested disease so far. After taking into the mind so many advantages of Lf, we have targeted this protein against complex deadly coccidian protozoan parasite, T. gondii.
The present study has revealed the effect of various forms of bLf isolated from bovine colostrum and were checked using in vitro cell culture model for the anti-toxoplasma activity against T. gondii RH strain maintained in the J7741 macrophage cells. The optimum concentration that does not show any toxicity against macrophages was calculated by cytotoxicity experiments in our previous study.26 The concentration obtained in our previous study was found to be 20 μg/mL and when analyzed in present study this concentration was found to inhibit 50% of infected macrophages and therefore considered as IC50 dose. The anti-toxoplasmal activity of different forms of bLf at this dose was compared with standard drug used for treatment of toxoplasmosis ie, sulfadiazine. Effective anti-toxoplasmal activity was analyzed by counting the mean number of infected macrophages and mean number of tachyzoites present inside each macrophage and by determining the intracellular NO production that was measured from the culture supernatant after 24 hours posttreatment. In the present study, maximum inhibitory activity against Toxoplasma has been shown by mono form of bLf having less Fe content than holo form. The NO levels were also found to be higher by the same form of Lf. The amount of NO production inside the cell is directly proportional to the inhibition of intracellular tachyzoites.
The present results are in concordance with the previously published studies, which documented the role of iron as an essential micronutrient required for the growth of intracellular tachyzoites and its multiplications.28 The results of the present study also confirm the same findings as apo/mono form of Lf was found to be more potent compared with holo form against the parasite. Lf has been known to bind with two rhoptry proteins (ROP2 and ROP4) of T. gondii, which acts as a binding ligand for the parasite.29 Thus, binding of Lf with these rhoptry proteins may inhibit the adhesion and invasion of the parasite with the host cells.30,31 Similarly Eimeria stiedae when treated with Lf was found to be less invasive as compared with the untreated sporozoites.32 Previously, anti-toxoplasmal activity of human Lf has been documented and was found to inhibit the T. gondii tachyzoites at a concentration of 1 mg/mL. However, in this study no cytotoxicity was checked, whereas in our previous study we have analyzed the heavy dose cytotoxicity of Lf protein on macrophage cells and has shown degradation of macrophages at doses more than 50 μg/mL. The toxic effects of increased concentration of Lf have not been studied in the previously published studies. Thus, optimum concentration of Lf should be used if therapeutic effect of Lf has to be utilized against the T. gondii and prior host cells cytotoxicity must be studied, which we have studied previously.26
The mono form of bLf that was found to be effective in inducing NO in macrophages was further tested for in vivo activity against T. gondii using suitable BALB/c mouse model. To the best of our knowledge, the present study is the pioneer one to analyze the effect of bLf in mouse model infected with T. gondii targeting the tachyzoite form of parasite (acute infection). The inhibitory effect of NCs and bLf were demonstrated in the histopathological sections of spleen and the histopathological examination confirmed the finding that NCs and bLf are showing inhibitory effect to T. gondii. Due to the inhibitory effect of mono form of bLf, actively multiplying tachyzoites converted into bradyzoites which is a dormant stage of T. gondii. Moreover, there were no pathological abnormalities like inflammation or fibrosis present in and around liver and spleen among the NCs-treated group of mice this may be one of the evolving strategies of the parasite to defend itself from the inhibitory effect of bLf. Hisopathological findings were conciding with absolute quantification analysis in which least copy number of parasite was found in sulphadiazine group followed by NCs group and bLf group.
On the contrary sulfadiazine-treated group did not reveal tachyzoites or the bradyzoites formation. Thus, mechanism involved in inhibition by bLf is different from that of sulfadiazine. The results obtained from ROS production and serum levels of NO production have well confirmed the proposed mechanism of inhibition of intracellular tachyzoites. These findings were further confirmed by the use of nanoformulation of bLf in the mouse model, which showed more protection compared with native bLf. Due to the better bioavailability of nanoformulated Lf and elevated levels of NO and ROS production, it showed more protection/inhibition against parasite compared with native form of bLf.
High levels of innate cytokines such as IL-12, IL-18, IL-6, and Il-15 lead to B-cell proliferation, NK lymphocyte production enhanced antigen representation and ultimately leading to parasite clearance. The same results have been shown in our previous studies in which same type of innate cytokines was expressed up to four- to fivefolds as compared with untreated group. Adaptive cytokines such as IL-2 and 1L-17 were found to be highly expressed in NCs group followed by bLf group compared with untreated group. These cytokines are helping in maintain the balance between Th1and Th2 types of response as we have found high levels of IL-10 in untreated group. Hematopoietic stem cell markers such as IL-7 and TGF-β were found to be elevated and have shown importance in production of hematopoietic stem cells in bone marrow. These cytokines expression is in concordance with our previous studies, which have well documented against disease like colon cancer,13 arthritis,18 and bacterial infections.33 However in our previous study we did not find any significant difference between control group and NCs control group.14,33 Therefore, untreated group was kept as a control for comparison of cytokine response.
Biodistribution of bLf in various organs showed increased concentration of bLf in NCs group of mice compared with native protein mice. The concentration of bLf was found to be two- to threefold high in NCs group; therefore, NCs were found to be more effective in clearing the parasite. The biodistribution data can be correlated with our immunohistochemistry analysis, which has shown positivity in spleen and liver cells of mice of NCs group compared with native Lf-treated mice that showed lower levels of Lf content seen as light brown color developed compared with dark color developed in NCs group.
Fe is an essential micronutrient present in our body required for various cellular reactions. Maintenance and regulation of cellular iron metabolism is essential to keep the balance between iron deficiency and excess iron toxicity. Therefore, various iron regulators were studied after administration of NCs and were found to up regulated in different organs compared with untreated group. These levels of iron regulators have shown homeostasis maintained by different markers inside the mice treated with NCs and no sign of iron toxicity was found.
From the present study, we have hypothesized the mode of action of Lf on intracellular parasites in a complex way. After ingestion of Lf through oral route, it is digested in intestine and from there gets distributed to all the parts of the body. It accumulates in liver and spleen and distributes in blood as previous studies have shown its presence in blood after few hours of intake.34 As the parasite resides in reticuloendothelial organs of the body especially in macrophages there is possibility that Lf directly interacts with parasites present in macrophages, hence direct inhibition is resulting in less parasite count observed. This is for the first time that direct interactions of Lf with macrophages of visceral organs have been shown, which tells the story of its inhibitory action against intracellular parasite T. gondii.
Another possibility is that as Lf may also show interaction with nucleus of parasite so as it might show direct inhibition by binding with parasite DNA. Whereas the same scenario in a normal untreated Lf cell will not take place as there will not be any Lf accumulation and no inhibition of intracellular parasites will be observe and only rapid multiplication can be seen. So to summarize, present study has shown inhibition of Lf protein, which has been isolated from bovine colostrum on deadly parasite T. gondii. The nanoformulation was found to be better as compared with native protein in performing better inhibition and therefore should be recommending for drug therapy against T. gondii.
Conclusion
In the present study, we evaluated the inhibitory effect of natural milk protein Lf isolated from bovine colostrum against parasitic disease toxoplasmosis and compared its efficacy with standard drug sulfadiazine. In order to protect the Lf protein it was encapsulated inside alginate chitosan NCs. Efficacy of these NCs along with naïve protein was assessed by performing various in vitro and in vivo models. Various parameters like biodistribution, cytokine analysis, and Fe metabolism were studied to analyze the efficacy of different formulations. NCs were found to be accumulated inside various visceral organs maintained the Fe metabolism and have shown enhanced bioavailability of bLf compared with native bLf, which might have degraded though intestinal tract. Interesting finding of the study included the accumulation of Lf in macrophage cells of liver and spleen where T. gondii was found to be multiplying. So, we believed that from the enhanced bioavailability of NCs and accumulation of protein inside infected cell has diminished the parasite growth and cured the mice for a longer lifespan. Taken together, our results conclude that the NCs can be valuable tools in the development of potent drug molecule against parasitic diseases and other biomacromolecules including drugs and vaccines.
Acknowledgments
The authors would like to thank the Australia–India Strategic Research Fund (AISRF), Department of Biotechnology-India (BT/PR/12672/ICD/55/10/2009) and Dr Louis Balan for assisting in histopathological reporting. No writing assistance was utilized in the production of this manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
Gaddi PJ, Yap GS. Cytokine regulation of immunopathology in toxoplasmosis. Immunol Cell Biol. 2007;85(2):155–159. | ||
Alcantara CS, Yang C-H, Steiner TS, et al. Interleukin-8, tumor necrosis factor-α, and lactoferrin in immunocompetent hosts with experimental and Brazilian children with acquired cryptosporidiosis. Am J Trop Med Hygiene. 2003;68(3):325–358. | ||
Petersen E, Schmidt DR. Sulfadiazine and pyrimethamine in the postnatal treatment of congenital toxoplasmosis: what are the options? 2003; Expert Rev Anti Infect Ther. 2003;1(1):175–182. | ||
Baker EN, Baker HM. Molecular structure, binding properties and dynamics of lactoferrin. Cell Mol Life Sci. 2005;62(22):2531–2539. | ||
Kanwar JR, Kanwar RK, Sun X, et al. Molecular and biotechnological advances in milk proteins in relation to human health. Curr Protein Pept Sci. 2009;10(4):308–338. | ||
Kanyshkova TG, Buneva VN, Nevinsky GA. Lactoferrin and its biological functions. Biochemistry (Mosc). 2001;66(1):1–7. | ||
López-Soto F, León-Sicairos N, Nazmi K, Bolscher JG, de la Garza M. Microbicidal effect of the lactoferrin peptides lactoferricin 17–30, lactoferrampin 265–284, and lactoferrin chimera on the parasite Entamoeba histolytica. Biometals. 2010;23(3):563–568. | ||
Turchany JM, Aley SB, Gillin FD. Giardicidal activity of lactoferrin and N-terminal peptides. Infect Immun. 1995;63(11):4550–4552. | ||
Ochoa TJ, Cleary TG. Effect of lactoferrin on enteric pathogens. Biochimie. 2009;91(1):30–34. | ||
Tanaka T, Abe Y, Inoue N, et al. The detection of bovine lactoferrin binding protein on Trypanosoma brucei. J Vet Med Sci. 2004;66(6):619–625. | ||
Parveen S, Misra R, Sahoo SK. Nanoparticles: a boon to drug delivery, therapeutics, diagnostics and imaging. Nanomed Nanotechnol Biol Med. 2012;8(2):147–166. | ||
Huang R, Ke W, Han L, et al. Lactoferrin-modified nanoparticles could mediate efficient gene delivery to the brain in vivo. Brain Res Bull. 2010;81(6):600–604. | ||
Kanwar JR, Mahidhara G, Kanwar RK. Novel alginate-enclosed chitosan-calcium phosphate-loaded iron-saturated bovine lactoferrin nanocarriers for oral delivery in colon cancer therapy. Nanomedicine. 2012;7(10):1521–1550. | ||
Kanwar JR, Mahidhara G, Roy K, et al. Fe-bLf nanoformulation targets survivin to kill colon cancer stem cells and maintains absorption of iron, calcium and zinc. Nanomedicine. 2015;10(1):35–55. | ||
Kanwar JR, Palmano KP, Sun X, et al. ‘Iron-saturated’ lactoferrin is a potent natural adjuvant for augmenting cancer chemotherapy. Immunol Cell Biol. 2008;86(3):277–288. | ||
Das RK, Kasoju N, Bora U. Encapsulation of curcumin in alginate-chitosan-pluronic composite nanoparticles for delivery to cancer cells. Nanomed Nanotechnol Biol Med. 2010;6(1):153–160. | ||
Samarasinghe RM, Kanwar RK, Kanwar JR. The effect of oral administration of iron saturated-bovine lactoferrin encapsulated chitosan-nanocarriers on osteoarthritis. Biomaterials. 2014;35(26):7522–7534. | ||
Samarasinghe RM, Kanwar RK, Kumar K, Kanwar JR. Antiarthritic and chondroprotective activity of Lakshadi Guggul in novel alginate-enclosed chitosan calcium phosphate nanocarriers. Nanomedicine. 2014;9(6):819–837. | ||
Bates GW, Wernicke J. The kinetics and mechanism of iron (III) exchange between chelates and transferrin IV. The reaction of transferrin with iron (III) nitrilotriacetate. J Biol Chem. 1971;246(11):3679–3685. | ||
Feng M, van der Does L, Bantjes A. Preparation of apolactoferrin with a very low iron saturation. J Dairy Sci. 1995;78(11):2352–2537. | ||
Young D, Hicks JM. Method for the automatic determination of serum iron. J Clin Pathol. 1965;18(1):98–102. | ||
Amano F, Noda T. Improved detection of nitric oxide radical (NO) production in an activated macrophage culture with a radical scavenger, carboxy PTIO, and Griess reagent. FEBS Lett. 1995;368(3):425–458. | ||
Pascoe S, Gatehouse D. The use of a simple haematoxylin and eosin staining procedure to demonstrate micronuclei within rodent bone marrow. Mutat Res. 1986;164(4):237–243. | ||
Burg JL, Grover CM, Pouletty P, Boothroyd J. Direct and sensitive detection of a pathogenic protozoan, Toxoplasma gondii, by polymerase chain reaction. J Clin Microbiol. 1989;27(8):1787–1792. | ||
Singla B, Chakraborti A, Sharma BK, et al. Levels of hepatitis B virus replicative intermediate in serum samples of chronic hepatitis B patients. Mol Biol Rep. 2014:1–8. | ||
Anand N, Kanwar RK, Dubey ML, et al. Effect of Lactoferrin protein on RBCs and Macrophages: mechanism of parasite host interaction. Drug Design Devel Ther. 2015;9:3821–3835. | ||
Kanwar JR, Roy K, Patel Y, et al. Multifunctional iron bound lactoferrin and nanomedicinal approaches to enhance its bioactive functions. Molecules. 2015;20(6):9703–9731. | ||
Ortíz-Estrada G, Luna-Castro S, Piña-Vázquez C, et al. Iron-saturated lactoferrin and pathogenic protozoa: could this protein be an iron source for their parasitic style of life? Future Microbiol. 2012;7(1):149–164. | ||
Dziadek B, Brzostek A. Recombinant ROP2, ROP4, GRA4 and SAG1 antigen-cocktails as possible tools for immunoprophylaxis of toxoplasmosis: what’s next? Bioengineered. 2012;3(6):358–364. | ||
Dziadek B, Dziadek J, Dlugonska H. Identification of Toxoplasma gondii proteins binding human lactoferrin: a new aspect of rhoptry proteins function. Exp Parasitol. 2007;115(3):277–282. | ||
Dziadek B, Gatkowska J, Brzostek A, Dziadek J, Dzitko K, Dlugonska H. Toxoplasma gondii: The immunogenic and protective efficacy of recombinant ROP2 and ROP4 rhoptry proteins in murine experimental toxoplasmosis. Exp Parasitol. 2009;123(1):81–89. | ||
Omata Y, Satake M, Maeda R, et al. Reduction of the infectivity of Toxoplasma gondii and Eimeria stiedai sporozoites by treatment with bovine lactoferricin. J Vet Med Sci. 2001;63(2):187–190. | ||
Gupta I, Sehgal R, Kanwar RK, Punj V, Kanwar JR. Nanocapsules loaded with iron-saturated bovine lactoferrin have antimicrobial therapeutic potential and maintain calcium, zinc and iron metabolism. Nanomedicine. 2014(0):1–26. | ||
Baratchi S, Kanwar RK, Kanwar JR. Novel survivin mutant protects differentiated SK-N-SH human neuroblastoma cells from activated T-cell neurotoxicity. J Neuroimmunol. 2011;233(1):18–28. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.