Back to Journals » Clinical Ophthalmology » Volume 16
Optimization of Phacoemulsification Tip Gauge on the Oertli CataRhex3 in an in vitro Setting
Authors Peterson JS, Christensen MR , Cooper C, Mamalis C, Bernhisel AA , Zaugg B , Barlow WR , Ungricht E, Olson RJ, Pettey JH
Received 21 December 2021
Accepted for publication 17 March 2022
Published 8 April 2022 Volume 2022:16 Pages 1091—1097
DOI https://doi.org/10.2147/OPTH.S355094
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
John S Peterson,1,2 Michael R Christensen,1,3 Caitlynn Cooper,1,2 Christina Mamalis,1 Ashlie A Bernhisel,1 Brian Zaugg,1 William R Barlow,1 Emilie Ungricht,1,2 Randall J Olson,1 Jeff H Pettey1
1Department of Ophthalmology and Visual Sciences, John A. Moran Eye Center, University of Utah, Salt Lake City, UT, 84132, USA; 2University of Utah School of Medicine, Salt Lake City, UT, 84132, USA; 3Medical College of Virginia/Virginia Commonwealth University School of Medicine, Richmond, VA, 23298, USA
Correspondence: Jeff H Pettey, Department of Ophthalmology and Visual Sciences, John A. Moran Eye Center, University of Utah, 65 Mario Capecchi Drive, Salt Lake City, UT, 84132, USA, Tel +1 801 581 2352, Fax +1 801 581 3357, Email [email protected]
Purpose: To evaluate the effect of each of the tip sizes available for the Oertli CataRhex3® phacoemulsification machine on efficiency.
Methods: Porcine lenses were fixed in formalin for 2 hours, then cut into 3.0 mm cubes. We studied three Oertli tips, all of which had a 30-degree bevel: easyTip 2.2 mm (20G), easyTip 2.8 mm (19G), and CO-MICS (21G). For the 19G and 20G tips, vacuum was set at 600 mmHg, irrigation rate at 50 mL/min, continuous power 70%, and bottle height 85 cm. For the 21G tip, vacuum was set at 450 mmHg; irrigation and power settings were identical to those used for the easyTip tips. We measured time to removal and chatter events to determine efficiency.
Results: Results from 20 trials for each tip showed that the larger the gauge size, the more quickly lens fragments were removed. Chatter events demonstrated an increasing trend with smaller tip gauge. The 19G tip used an average time to fragment removal of 2.8 seconds; the 20G, 3.2 seconds; and the 21G, 4.6 seconds. Increasing tip diameter from 21G to 20G decreased emulsification time by 33% (P = 0.02). Increasing the diameter from 21G to 19G further decreased time to emulsification by 42% (P = 0.003). The 21G tip had a mean 1.4 events/cube; 20G, 0.35 events; and 19G, 0.1 events. Differences in mean chatter events for each tip were each statistically significant.
Conclusion: These data suggest that when evaluated by chatter events and emulsification time, the 2.8 mm (19G) easyTip proves to have greatest efficiency.
Keywords: phacoemulsification cataract, needle, tip size, efficiency, Oertli CataRhex3, longitudinal torsional power
Plain Language Summary
Cataracts are one of the main causes of low vision and blindness throughout the world. This is especially true in low- and middle-income countries where world access to health services is relatively limited. The Oertli CataRhex3 is a lightweight phacoemulsification platform making it ideal for use in settings where portability and reliability are prioritized. Until now, little research into how to most safely and efficiently perform surgery with this machine has been done. In this study, we looked at each of the surgical tip sizes—19G, 20G, and 21G—used by the CataRhex3 and tested to see which could remove the lens most efficiently. We found that the widest diameter tip (19G) was significantly more efficient than the others. This will help surgeons using this machine improve care for patients, particularly in low-resources settings.
Introduction
Unoperated cataracts account for visual impairment in 65.2 million of the estimated 2.2 billion people with moderate to severe vision impairment worldwide.1–4 Although manual small incision cataract surgery remains an important surgical approach in areas with high cataract burden, phacoemulsification (Phaco) utilization continues to increase throughout the world.5–7
Portable and reliable equipment remains one of the obstacles to phacoemulsification adoption as an approach in low resource settings. The Oertli CataRhex 3® (Oertli) phaco platform has a small profile, weighing only 5 kg, with dimensions of 22 × 24 x 18.3 cm and voltage capabilities between 100 and 240.8,9
Optimal phaco settings have been evaluated on many other commonly used machines.10–36 Due to the unique size and portability of the CataRhex platform, evaluating surgical settings on this system is an important area of research. Minimizing the power and duration of phaco engaged intraoperatively improves outcomes and minimizes complications. This paper is a key evaluation in our series to systematically evaluate the optimum settings and provide guidance for surgeons.9,37 It builds on previous studies that establish differences between efficiency and chatter using horizontal and longitudinal ultrasound with different gauged tips, efficiency being the shortest time possible to groove the lens.36 This study evaluates the efficiency and chatter events between the three tip diameters available on Oertli platform.
Materials and Methods
This study did not involve human subjects or laboratory animals, so Institutional Review Board/Institutional Animal Care and Use Committee approval did not apply.
We performed our experiments using the following Oertli tip diameters, all of which have a 30-degree bevel: easyTip 2.2 mm (20G), easyTip 2.8 mm (19G), and CO-MICS (21G). Our experimental design for the tips were: 600 mmHg vacuum, 50 mL/min irrigation rate, and 70% continuous power. However, at 600 mmHg vacuum pressure, the 21G tip impaled through the lens cubes, halting emulsification and aspiration of the piece. To allow the 21G tip to be included in the evaluations, we decreased the vacuum to 450 mmHg and proceeded with the study as outlined. Bottle height was maintained at 85 cm, measured from ¾ from the bottom of the drop chamber to the experimentation site. Aspiration and vacuum were set at relatively aggressive levels given the 3–4+ density of the pieces.
Lens Preparation
Intact porcine lenses purchased from a supplier (Visiontech, Inc., Sunnyvale, TX, USA) were dissected no longer than 48 hours after they were received. After hardening these lenses in formalin for 2 hours, they were washed 3 times and soaked in balanced salt solution (BSS) (Alcon Sterile Irrigating Solution 500 ml 0060795-50) for 24 hours, as previously described.38
A device which allows cutting of uniform lens cubes was used to produce 3.0 mm cubes. For this evaluation we chose the 3.0 mm cube in order to completely occlude all three tip gauges. Of note, the 3.0 mm cube also more closely replicates larger lens fragments more commonly seen in quadrant removal steps of cataract surgery.
Experimental Procedure
Twenty lens cubes were emulsified with each tip during the experiment, for a total of 60 experimental runs. Each run began with a lens cube that was selected at random and placed in the silicone testing chamber. Once the cube was in contact with the tip, phaco was turned to full power and the efficiency, defined as the time necessary for complete emulsification, was recorded. Throughout the test, the bevel was positioned upward so as to ensure that the irrigation flowed out laterally and was parallel to the base of the testing chamber. This technique replicated the clinical setting and allowed for consistency between each cube. Chatter events, defined as the number of incidents in which the cube was lost from the tip despite adequate aspiration, were recorded and time was stopped until the cube could be repositioned onto the tip. The surgeon used the same pedal maneuver and specified settings among all tips tested.
Statistical Analysis
After averaging efficiency times, means were calculated. Efficiency times for each tip gauge were compared using one-way ANOVA. When a significant P-value was found, we used Student’s t-tests in order to discern relationships among variables. Significance was set at a P-value of ≤0.05.
In our experimental design, it is possible for cubes to bounce from the tip for several seconds, accounting for the two data points that were more than two standard deviations from the mean. As in our previous investigations, these data were considered outliers and excluded from analyses, which were conducted using Microsoft Excel (Microsoft Corporation, Redmond, Washington, USA).13,14,17,26,28,33–35
Results
Our final analysis showed a significant difference in time required to emulsify the cubes, as well as a significant chatter events difference (Table 1).
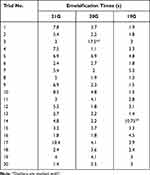 |
Table 1 Time to Complete Emulsification of 3 mm Lens Cube with Each Oertli Phaco Tip Size |
Our results showed an inverse relationship between gauge size and time to emulsification. The average cube emulsification time for the 19G tip was 2.8 seconds (s), 3.2 s for the 20G tip, and 4.6 s for the 21G tip (Figure 1). Increasing the gauge from 21G to 20G decreased time to emulsification by 33% (P=0.02). Increasing the gauge from 21G to 19G further decreased time to emulsification by 42% (P=0.003). Although the emulsification time decreased when increasing the tip gauge from 20G to 19G (13%), this was not found to be statistically significant (P=0.3).
 |
Figure 1 Average time to complete emulsification of 3 mm lens cube for each Oertli phaco tip size. |
There was a similar increasing trend in chatter events with decreasing tip gauge (Table 2). There was an average of 1.4 chatter events per cube when using the 21G tip, 0.35 when using the 20G tip, and 0.1 when using the 19G tip (Figure 2). The differences in mean chatter events seen between each tip gauge were statistically significant (21G to 20G: P=0.003, 21G to 19G: P=0.00008, 20G to 19G: P=0.04).
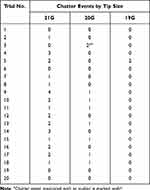 |
Table 2 Total Number of Chatter Events per 3 mm Lens Cube During Emulsification with Each Oertli Phaco Tip Size |
 |
Figure 2 Average number of chatter events per 3 mm lens cube during emulsification for each Oertli phaco tip size. |
Discussion
In this efficiency analysis of all available phaco tips in the Oertli system, emulsification time decreased as tip diameter increased; the 19G tip emulsified lens cubes significantly faster, on average, than the 20G and 21G tips. Likewise, there were, on average, significantly fewer chatter events per lens cube when using the 19G tip than when using the 21G tip. In our series of studies, machine settings that decreased chatter events consistently correlate with a decrease in the time required to remove lens fragments.
Our findings support the conclusion that optimal efficiency is achieved with the 19G gauge tip and is congruent with the mechanics of the CataRhex3.8,9 The Oertli system utilizes longitudinal ultrasound (US) to emulsify the lens, so as the gauge of the tip increases, the surface area making contact with the lens nucleus increases. This allows for greater transfer of energy to the lens. Additionally, the central channel through which lens material is aspirated increases proportionally with the tip gauge; thus, a larger-gauged tip is capable of aspirating higher volumes than tips with a smaller diameter gauge. Given the low incidence of chatter events per cube with the 19G tip, it seems reasonable to conclude that the 19G tip is the optimal one to decrease operative time and minimize phaco energy exposure while preserving surrounding ocular structures and tissues.39
In our previous study on a phacoemulsification platform, we used a similar experimental design to evaluate the effects of tip diameter on efficiency and chatter using torsional, transversal, and micropulsed US modalities. In the earlier study, we found that the differences in efficiency and chatter for the three tip sizes were much smaller than those that we observed in this study, and that the 0.9 mm tip (middle-size tested) was the most efficient.16 Our hypothesis for this discrepancy is that the relatively larger (3.0 mm) lens cubes used in this study allowed for a more clinically similar environment and likely contributed to the stepwise efficiency findings.
Decreasing the amount of time phaco is engaged during a surgery has been linked to decreased post-operative inflammation.40 This is of particular importance to cataract surgeons working in resource-constrained settings where dense opaque cataracts are more frequently encountered. Although very dense and brunescent cataracts may be better served with MSICS surgery, these data suggest that in cases of at least moderate nuclear sclerosis, emulsification time is likely to be halved by using the 20G tip as opposed to the narrower 21G, and with significantly less chatter.
Although the in vitro nature of this study constitutes a limitation, it would be exceedingly difficult to perform the number of surgeries or control for all the variables needed to determine the optimum settings for efficiency and chatter. The second limitation lies in our ability to adequately simulate uniform dense nuclear lenticular tissue. Our lens cube apparatus allowed us to make every effort to produce uniform 3.0 mm cubes. Admittedly, some variation still could exist among the cubes, but this variance was controlled by randomizing selection of cubes. The porcine model that we used simulated lens density with an equivalent hardness of 3 to 4 nuclear sclerosis,38 making it a suitable model for the types of cataracts a surgeon could encounter in medically underserved areas. Notwithstanding, we acknowledge that our method of fixing lenses in formalin was an imperfect replication of the extreme opacity and hardness of advanced cataracts. Nonetheless, we expect these trends in efficiency to hold in vivo.
Additionally, at high vacuum pressures (600 mmHg), the 21G CO_MICS tip punctured through the middle of the 3 mm cubes, which prohibited the runs from being included in the study. This is not entirely surprising given the combination of high vacuum through a small diameter tip on a large piece leads to such a “lollypop” phenomenon in clinical settings. Additional runs at a vacuum setting of 450 mmHg for the 21 G tip were included for evaluation. Further studies on the fluidics of the CO_MICS tips are warranted, though outside the scope of this current analysis.
With this information, surgeons working with this system can improve outcomes for patients, even for those with advanced nuclear cataracts. In addition, further research with various cube sizes could be performed, with the goal of further understanding the relationship of cube size, gauge size, time to removal, and chatter events. Furthermore, our optimized settings from our experiment may not be the ideal for a surgeon’s unique clinical scenario, and we recommend adjusting settings as they deem necessary.
Conclusion
There are three tip sizes compatible with the Oertli CataRhex3: 21G, 20G, and 19G. Using the average time required for complete emulsification of a 3 mm lens cube and the average number of chatter events per cube as surrogates for efficiency, each of these tips was compared head-to-head in a controlled setting. There was no significant difference between the average time required for a 19G tip or a 20G tip to emulsify a lens cube, but both tips averaged faster emulsification times than the 21G tip. The 19G tip had fewer chatter events per cube on average than the 20G and 21G tips. Given these findings, the utilization of the 19G tip is recommended when using the Oertli CataRhex3.
Abbreviations
phaco, phacoemulsification; Oertli, Oertli CataRhex3®; BSS, balanced salt solution; SD, standard deviation; s, seconds; US, ultrasound.
Ethics Approval and Informed Consent
Since no human subjects were involved, approval from the University of Utah Institutional Review Board was not obtained.
Acknowledgments
Susan Schulman assisted with manuscript editing and preparation.
Funding
This study was supported in part by an unrestricted grant from Research to Prevent Blindness, Inc., New York, New York, USA, to the Department of Ophthalmology and Visual Sciences, University of Utah, Salt Lake City, Utah, USA. The sponsor had no involvement in any of the stages from study design to submission of the manuscript for publication.
Disclosure
Dr. Olson is on the Board of Directors of Perceive Bio and the Scientific Advisory Board of Perfect Lens. Dr. Jeff Pettey reports a Consulting agreement for Lensar, outside the submitted work. The other authors report no conflicts of interest in this work.
References
1. Bourne RRA, Flaxman SR, Braithwaite T, et al. Magnitude, temporal trends, and projections of the global prevalence of blindness and distance and near vision impairment: a systematic review and meta-analysis. Lancet Glob Health. 2017;5(9):e888–e897. doi:10.1016/S2214-109X(17)30293-0
2. World Health Organization. World report on vision. licence: CC BY-NC-SA 3.0 IGO. Geneva, Switzerland; 2019. Available from: www.who.int/publications-detail/world-report-on-vision.
3. Flaxman SR, Bourne RRA, Resnikoff S, et al. Global causes of blindness and distance vision impairment 1990–2020: a systematic review and meta-analysis. Lancet Glob Health. 2017;5(12):e1221–e1234. doi:10.1016/S2214-109X(17)30393-5
4. Global Burden of Disease 2019 Blindness and Vision Impairment Collaborators; Vision Loss Expert Group of the Global Burden of Disease Study. Trends in prevalence of blindness and distance and near vision impairment over 30 years: an analysis for the Global Burden of Disease Study. Lancet Glob Health. 2021;9(2):e130–e143. doi:10.1016/S2214-109X(20)30425-3
5. Tabin G, Chen M, Espandar L. Cataract surgery for the developing world. Curr Opin Ophthalmol. 2008;19(1):55–59. doi:10.1097/ICU.0b013e3282f154bd
6. Menapace R. [Developments in modern cataract surgery - a critical overview]. Ther Umsch. 2016;73(2):53–59. German. doi:10.1024/0040-5930/a000756
7. Bernhisel A, Pettey J. Manual small incision cataract surgery. Curr Opin Ophthalmol. 2020;31(1):74–79. doi:10.1097/ICU.0000000000000624
8. Oertli. Making the difference in eye surgery. Berneck, Switzerland: Oertli Instrumente; 2021. Available from: https://www.oertli-instruments.com/en-us.
9. Stutz LA, Heczko JB, Bird BA, et al. Optimization of the Oertli CataRhex 3 phacoemulsification machine. Clin Ophthalmol. 2019;13:633–639. doi:10.2147/OPTH.S196281
10. Bird BA, Thomson RS, Stutz LA, et al. Effect of increasing flow when grooving during phacoemulsification. J Cataract Refract Surg. 2018;44(5):623–626. doi:10.1016/j.jcrs.2018.01.034
11. Bohner AD, Wright AJ, Ta BT, et al. Optimum on-time duty cycle for a transversal ultrasound machine. J Cataract Refract Surg. 2018;44(9):1140–1143. doi:10.1016/j.jcrs.2018.03.039
12. Boulter T, Jensen JD, Christensen MD, et al. Comparison of a torsional and a standard tip with a monitored forced infusion phacoemulsification system. J Cataract Refract Surg. 2016;42(4):613–617. doi:10.1016/j.jcrs.2015.11.048
13. Boulter T, Christensen MD, Jensen JD, et al. Optimization and comparison of a 0.7 mm tip with the 0.9 mm tip on an active-fluidics phacoemulsification platform. J Cataract Refract Surg. 2017;43(12):1591–1595. doi:10.1016/j.jcrs.2017.08.022
14. Cahoon JM, Gupta I, Gardiner G, et al. Comparison of venturi and peristaltic vacuum in phacoemulsification. J Cataract Refract Surg. 2015;41(2):428–432. doi:10.1016/j.jcrs.2014.12.043
15. DeMill DL, Zaugg BE, Pettey JH, et al. Objective comparison of 4 nonlongitudinal ultrasound modalities regarding efficiency and chatter. J Cataract Refract Surg. 2012;38(6):1065–1071. doi:10.1016/j.jcrs.2011.12.040
16. Farukhi AM, Stagg BC, Ronquillo C
17. Gardiner GL, Garff K, Gupta I, et al. Effect of pulsing ultrasound on phacoemulsification efficiency. J Cataract Refract Surg. 2015;41(11):2560–2564. doi:10.1016/j.jcrs.2015.06.030
18. Garff K, Jensen JD, Cahoon J, et al. Impact of micropulsed ultrasound power settings on the efficiency and chatter associated with lens-fragment removal. J Cataract Refract Surg. 2015;41(6):1264–1267. doi:10.1016/j.jcrs.2014.09.049
19. Gupta I, Cahoon JM, Gardiner G, et al. Effect of increased vacuum and aspiration rates on phacoemulsification efficiency. J Cataract Refract Surg. 2015;41(4):836–841. doi:10.1016/j.jcrs.2014.09.035
20. Gupta I, Zaugg B, Stagg BC, et al. Phacoemulsification efficiency with a radiused phaco tip. J Cataract Refract Surg. 2014;40(5):818–821. doi:10.1016/j.jcrs.2013.11.029
21. Jensen JD, Kirk KR, Gupta I, et al. Determining optimal ultrasound off time with micropulse longitudinal phacoemulsification. J Cataract Refract Surg. 2015;41(2):433–436. doi:10.1016/j.jcrs.2014.12.033
22. Jensen JD, Shi DS, Robinson MS, et al. Torsional power study using CENTURION phacoemulsification technology. Clin Exp Ophthalmol. 2016;44(8):710–713. doi:10.1111/ceo.12748
23. Ha L, Wright A, Wright DD, et al. High vacuum and aspiration on phacoemulsification efficiency and chatter for Centurion. Can J Ophthalmol. 2019;54(1):136–138. doi:10.1016/j.jcjo.2018.03.009
24. Kabbara SW, Heczko J, Ta B, et al. Determining optimal ultrasound percent on time with long-pulse torsional phacoemulsification. Can J Ophthalmol. 2019;54(3):395–398. doi:10.1016/j.jcjo.2018.07.005
25. Kabbara S, Heczko JB, Bernhisel AA, et al. Effect of high vacuum and aspiration on phacoemulsification efficiency and chatter using a transversal ultrasound machine. J Cataract Refract Surg. 2018;44(11):1378–1383. doi:10.1016/j.jcrs.2018.05.031
26. Kabbara SW, Heczko J, Ta B, et al. Impact of torsional micropulse on phacoemulsification efficiency and chatter. Can J Ophthalmol. 2019;54(5):560–564. doi:10.1016/j.jcjo.2019.02.016
27. Kirk KR, Ronquillo C
28. Ronquillo CC
29. Shi DS, Jensen JD, Kramer GD, et al. Comparison of vacuum and aspiration on phacoemulsification efficiency and chatter using a monitored forced infusion system. Am J Ophthalmol. 2016;169:162–167. doi:10.1016/j.ajo.2016.06.030
30. Stagg BC, Gupta I, Cahoon J, et al. Bent versus straight tips in micropulsed longitudinal phacoemulsification. Can J Ophthalmol. 2015;50(5):354–359. doi:10.1016/j.jcjo.2015.06.003
31. Wright AJ, Bohner AD, Bernhisel AA, et al. The effect of pulsing on transverse ultrasound efficiency and chatter. Am J Ophthalmol. 2017;183:107–110. doi:10.1016/j.ajo.2017.08.012
32. Wright AJ, Thomson RS, Bernhisel AA, et al. Effect of chamber stabilization software on efficiency and chatter in a porcine lens model. J Cataract Refract Surg. 2017;43(11):1464–1467. doi:10.1016/j.jcrs.2017.08.018
33. Wright DD, Wright AJ, Boulter TD, et al. Optimization of transversal phacoemulsification settings in peristaltic mode using a new transversal ultrasound machine. J Cataract Refract Surg. 2017;43(9):1202–1206. doi:10.1016/j.jcrs.2017.06.043
34. Bernhisel AA, Cahoon JM, Sella R, et al. Optimum on-time and off-time combinations for micropulse phacoemulsification in venturi vacuum mode. J Cataract Refract Surg. 2019;45(12):1797–1800. doi:10.1016/j.jcrs.2019.07.001
35. Bohner A, Peterson JS, Wright AJ, et al. Effects on phacoemulsification efficiency and chatter at variable longitudinal ultrasound settings when combined with constant torsional energy. J Cataract Refract Surg. 2020;46(5):774–777. doi:10.1097/j.jcrs.0000000000000150
36. Ramshekar A, Heczko J, Bernhisel A, et al. Optimizing tip diameter in phacoemulsification of varying lens sizes: an in vitro study. Clin Ophthalmol. 2021;15:4475–4484. doi:10.2147/OPTH.S333903
37. Thomson RS, Bird BA, Stutz LA, et al. The effect of increasing power when grooving using phacoemulsification. Clin Ophthalmol. 2019;13:611–615. doi:10.2147/OPTH.S194731
38. Oakey ZB, Jensen JD, Zaugg BE, Radmall BR, Pettey JH, Olson RJ. Porcine lens nuclei as a model for comparison of 3 ultrasound modalities regarding efficiency and chatter. J Cataract Refract Surg. 2013;39(8):1248–1253. doi:10.1016/j.jcrs.2013.01.049
39. Menapace R. Mini- and micro-incision cataract surgery: a critical review of current technologies. Eur Ophth Rev. 2009;3(2):52–57. doi:10.17925/EOR.2009.03.02.52
40. Kruger AJ, Schauersberger J, Abela-Formanek C, et al. [Effect of duration of phacoemulsification on postoperative inflammation–a retrospective study]. Klin Monbl Augenheilkd. 2001;218(4):204–208. German. doi:10.1055/s-2001-14914
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
