Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
Oil-in-water biocompatible microemulsion as a carrier for the antitumor drug compound methyl dihydrojasmonate
Authors Bevilacqua Rolfsen Ferreira da Silva G, Scarpa MV, Carlos IZ, Bassi Quilles M, Comeli Lia RC, Tabosa do Egito ES , Gomes de Oliveira A
Received 7 July 2014
Accepted for publication 27 September 2014
Published 12 January 2015 Volume 2015:10(1) Pages 585—594
DOI https://doi.org/10.2147/IJN.S67652
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Thomas Webster
Gisela Bevilacqua Rolfsen Ferreira da Silva,1 Maria Virginia Scarpa,1 Iracilda Zepone Carlos,2 Marcela Bassi Quilles,2 Raphael Carlos Comeli Lia,3 Eryvaldo Socrates Tabosa do Egito,4 Anselmo Gomes de Oliveira1
1Departamento de Fármacos e Medicamentos, 2Departamento de Análises Clínicas, UNESP–Universidade Estadual Paulista, Faculdade de Ciências Farmacêuticas, PPG em Nanotecnologia Farmacêutica, Rodovia Araraquara-Jaú Km 01, Araraquara, SP, Brazil; 3Instituto de Patologia Cirúrgica e Citopatologia (IPC), Araraquara, SP, Brazil; 4UFRN–Universidade Federal do Rio Grande do Norte, Programa de Pós-graduação em Ciências da Saúde, Natal, RN, Brazil
Abstract: Methyl dihydrojasmonate (MJ) has been studied because of its application as an antitumor drug compound. However, as MJ is a poorly water-soluble compound, a suitable oil-in-water microemulsion (ME) has been studied in order to provide its solubilization in an aqueous media and to allow its administration by the parenteral route. The ME used in this work was characterized on the pseudo-ternary phase diagram by dynamic light scattering and rheological measurements. Regardless of the drug presence, the droplet size was directly dependent on the oil/surfactant (O/S) ratio. Furthermore, the drug incorporation into the ME significantly increased the ME diameter, mainly at low O/S ratios. The rheological evaluation of the systems showed that in the absence of drug a Newtonian behavior was observed. On the other hand, in the presence of MJ the ME systems revealed pseudoplastic behavior, independently of the O/S ratio. The in vivo studies demonstrated that not only was the effect on the tumor inhibition inversely dependent on the MJ-loaded ME administered dose, but also it was slightly higher than the doxorubicin alone, which was used as the positive control. Additionally, a small antiangiogenic effect for MJ-loaded ME was found at doses in which it possesses antitumor activity. MJ revealed to be nontoxic at doses higher than 350 mg/kg, which was higher than the dose that provides tumor-inhibition effect in this study. Because the MJ-loaded ME was shown to have anticancer activity comparable to doxorubicin, the ME described here may be considered a suitable vehicle for parenteral administration of MJ.
Keywords: antitumor drug, nanocarrier, angiogenesis inhibition, antitumor activity, Ehrlich ascitic tumor
Introduction
Cancer is an increasingly diagnosed disease and is currently a serious public health problem worldwide. The main problem of most antineoplastic therapeutic compounds is their severe side effects.1–3 Drug delivery systems, which have the ability to compartmentalize drugs in nanostructures, are one of the technological strategies that can enable parenteral administration and reduce potential toxic effects. Doxorubicin (DOX), a widely used antitumor compound, generates heart and kidney toxicity. In addition to myelosuppression,1 cisplatin, another important antineoplastic agent, may cause nephrotoxicity.2 On the other hand, the monoclonal antibody bevacizumab may induce gastrointestinal hemorrhage and perforation.3
Jasmonate derivatives such as methyl dihydrojasmonate (MJ) have been widely studied as anticancer drugs, mainly against gastric,4 liver,5 cervical,6,7 multiple myeloma,8 breast,9 melanoma,10 and other types of cancers.11,12 The mechanism involved in the antitumor effect of MJ includes the bioenergetics in which the drug induces a severe depletion of adenosine tri-phosphate (ATP) in cancer cells via the mitochondrial perturbation. The reactive oxygen species from MJ induces apoptosis in cancer cells through the generation of hydrogen peroxide and pro-apoptotic proteins of the Bcl-2 family.13,14
The jasmonate derivatives have the ability to re-differentiate human myeloid leukemia cells through the activity of mitogen-activated protein kinase and cytokine isopentenyladenine.10 The inhibition of angiogenesis was also described to contribute in cancer therapy.15,16 Recently, it was described that sub-cytotoxic MJ attenuates the matrix metalloproteinases-14 expression via decreasing the Sp1 expression and binding on matrix metalloproteinases-14 promoter, thus inhibiting the migration, invasion, and angiogenesis of gastric cancer cells.5,17
However, MJ, a water-insoluble compound, requires a suitable lipid nanocarrier system such as microemulsions (MEs) and nanoemulsions for administering the drug by intravenous route. MEs have been proven a suitable vehicle for this purpose.18–20 In previous work, our research team studied anionic isotropic and anisotropic oil-in-water colloidal systems stabilized with a mixture of phosphatidylcholine (PC)/glycerol (GL)/sodium oleate as surfactant and cholesterol as oil phase. This colloidal dispersion system was able to incorporate MJ and to maintain its physical stability characteristics, suitable for intravenous administration.21
In this work, an efficient, safe, thermodynamic stable, and biocompatible oil-in-water ME system was studied to provide the solubilization of MJ in aqueous media intended for parenteral administration of this antitumor drug. These systems were characterized through pseudo-ternary phase diagrams, light scattering, and rheological analysis. The in vivo antineoplastic effect, angiogenesis and acute toxicity tests were also performed.
Materials and methods
Materials
Soya PC, conditional access system (CAS) number 97281-47-5 (Phospholipon 90G), was purchased from Lipoid GmbH (Ludwigshafen, Germany). MJ, CAS number 24851-98-7, GL, CAS number 56-81-5, Tween 20 (TW), CAS number 9005-64-5 (Polyoxyethylene-20-sorbitan monolaurate) and sodium hydroxide, CAS number 1310-73-2, were purchased from Sigma Chemical Co (St Louis, MO, USA). DOX hydrochloride was procured from Eurofarma (Brazil). Isotonic saline was prepared with 0.9% sodium chloride in water for injection and sterilized by filtration through sterile 0.22 μm PVDF Millipore Durapore® membrane. Pure soybean oil (O), CAS number 8001-22-7, came from Cargill Agrícola S/A (Brazil), and its main fatty acids percentage composition were: palmitic (16:0) 13.48%, stearic (18:0) 4.58%, oleic (18:1) 19.51%, linoleic (18:2) 51.00%, and linolenic (18:3) 9.56%, which represent a total of 98.13%. Coconut oil, CAS number 8001-31-8, was purchased from Audaz (São Paulo, Brazil), and its main fatty acids percentage composition were: caprylic (8:0) 6, capric (10:0) 5.54%, lauric (12:0) 48.79%, myristic (14:0) 19.90%, palmitic (16:0) 9.33%, stearic (18:0) 2.46%, oleic (18:1) 6.42%, and Linoleic (18:2) 1.44%, which represent a total of 99.88% and the weighted molecular weight of 210.83 g/mol. Coconut salt fatty acids (CSF) was obtained by saponification of coconut oil. Purified water (W) was obtained in a Milli-Q Plus system (Millipore) and its resistivity was 18.2 MΩ cm. All other solvents and chemicals were of analytical grade or better.
Methods
Phase diagram
The surfactant mixture (S) containing proportions of the surfactants CSF:GL:PC was experimentally established as 8:4:1 (w/w/w), in which PC and CSF are the primary surfactants and GL is a co-surfactant of the system. The O was added over the semisolid mixture of S at the ratios from 1:9 to 9:1 (w/w) to obtain the pseudo-ternary phase diagram, which was generated by carefully dripping W using a precision burette attached to an iron support, onto the mixture of S and O under constant gentle magnetic stirring. The regions of transition from semisolid mixture to clear systems and from clear to translucent colloidal systems were adequately reproducible within 0.05 mL of W. All determinations were performed at 25°C±0.1°C. For systems loaded with 20% (w/w) of MJ, the drug was added to the oil/surfactant (O/S) mixture and the procedure was followed as described for the MJ-unloaded system.
ME preparation
The systems were prepared as previously described.22 Briefly, CSF, GL, and PC were weighted and mixed to obtain S. To the mixture of S, an appropriate amount of O was added. To the mixture of S and O, the W was added. This sequence provides a complete homogenization of the components. Then, the system was kept under ultrasonic stirring (QSonica Sonicators with a standard probe, Model Q700) in a discontinuous mode for 20 minutes (1 minute of sonication and 30 seconds interval) in an ice bath. For the ME systems loaded with MJ, the drug was dissolved in the ME systems already prepared. The composition of the formulations studied in this work is described in Table 1.
Dynamic light scattering
The droplet size analysis was performed with a dynamic light scattering (DLS), Zetasizer Nano system ZS (Malvern Instruments, Worcestershire, UK), with He–Ne 4 mW laser source at 633 nm using a record angle of 173. The analysis was carried out at 25°C±0.2°C. The samples were diluted 10× with Milli-Q purified water. The results were obtained from ten determinations.
Rheological measurements
The analyses were performed using a Carrimed CSL 100 rheometer (TA Instruments, New Castle, DE, USA) equipped with a cone–plate geometry and a temperature controller. The cone–plate geometry was a 2° stainless steel cone with a 4 cm diameter and a gap of 105 μm between the cone and the plate. The results were obtained with a shear rate (γ) in the range of 0–300 s-1. The rheological analyses were performed for both up and up–down curves. The data of the shear cycle were fitted to the power-law model using the Rheology Solutions Software (Version Data V1.1.7, TA Instruments). All determinations were realized in triplicate at 25°C±0.2°C.
Antitumor activity
The Ehrlich tumor cells were maintained in vivo by weekly subcultures in mice by intraperitoneal inoculation of 0.5–1 mL of ascites. The mice were inoculated subcutaneously with 0.1 mL of tumor cell suspension adjusted to the concentration of 1×107 tumor cells/mL in RPMI 1640 for solid tumor induction in female Balb/c (18–25 g) mice. Treatments were performed with three doses (one dose every 7 days) by intraperitoneal injection. Each group consisted of five animals: Group I (Control) received 0.9% saline, 10 mL/kg; Group II (Positive control) received DOX, 5 mg/kg; Group III (Negative control) received MJ-unloaded ME, 10 mL/kg; Group IV (Test drug 1) received MJ-loaded ME, 2.5 mg/kg; Group V (Test drug 2) received MJ-loaded ME, 5 mg/kg; and Group VI (Test drug 3) received MJ-loaded ME, 10 mg/kg. For the in vivo experiments the formulation was selected from the range of formulations shown in Table 1 and represented in Figure 1. The stock MJ-unloaded ME formulation was composed of surfactant mixture PC (1.55%)/GL (6.15%)/CFS (12.3%), O (18%), and W (62%). For the MJ-Loaded ME, MJ was added to the previously prepared ME in the proportion of 20% (w/w), which was diluted with saline to the required doses. The tumors of the mice were removed after 20 days and then, the largest and smallest diameters were determined using a digital caliper, Mitutoyo Digimatic Caliper.
The tumor was also weighted and the volume was calculated by the Equation 1:23
TVo = 0.5 × larger diameter × (smaller diameter)2 | (1) |
The larger and the smaller diameters were taken in mm.
The tumor inhibition rate was calculated from the Equation 2:24
|
|
where, TIR is the percent of tumor inhibition rate, TVoT is the tumor volume of the Treated Group, and TVoC is the tumor volume of the Control Group.
Microscopic angiogenesis grading system
From the tumors removed from the animals, 5 μm sections of breast tumor tissue were fixed in formaldehyde, dehydrated with ethanol and xylene, embedded in paraffin, and subsequently stained with hematoxylin and eosin. The slides were then photographed and analyzed histopathologically by using a light microscope with image capture (Nikon Eclipse E200). The microscopic angiogenesis grading system (MAGS) score was determined following the previously established procedure.25,26 The vessel proliferation (KnN), hyperplasia of endothelial cells (KeE), and endothelial cytology (KxX) were analyzed by optical microscopy using a magnification of 400×. The score was calculated as:
MAGS = KnN + KeE + KxX | (3) |
where the constants are: Kn =1, Ke =3 and Kx =6.
The KnN has a maximum value of 40 and N is the number of capillaries viewed under magnification of 400-fold in each field. The KeE has a maximum value of 30, and E is the number of cells lining the capillary cross section with a maximum value of 10. KxX has a maximum value of 30, and X has values from 0 to 5. The X value indicates the histological appearance of endothelial cells at the zone of hyperplasia. X =0 implies a normal result; X =1 implies cells increasing, but with a normal and clear nucleus; X =2 implies cells increasing, but with a clear and prominent nucleus; X =3 implies cells with a large and hyperchromatic nucleus; X =4 implies bizarre endothelial cells; and X =5 implies the cells showing a mitotic shape. The MAGS gradation was calculated by this formula ranging between 0 and 100, on an arbitrary scale. The results were expressed in arbitrary values obtained from ten random fields in each of the blades out of five animals in each studied group.
Acute toxicity
Fifty-five male Swiss mice, weighing about 30 g, were divided into eleven groups of five animals: four experimental groups for MJ-loaded ME; four experimental groups for MJ dispersed in 3% TW solution; three control groups (one for saline, one for 3% TW solution, and another for empty ME). The experimental groups received, by single intraperitoneal injection, doses of 70 mg/kg, 140 mg/kg, 280 mg/kg, and 350 mg/kg of MJ. The control groups received the empty ME or saline solution. The size of needles used for intraperitoneal injections was 0.45×13 mm. The animals were observed for 14 days after administrations for any obvious symptoms and mortality. The results were considered positive if death occurred in 50% of test animals with one of the administered doses.
Statistical analysis
The results were presented as mean + standard deviation (SD) of n independent experiments. Statistical significance was assessed using analysis of variance (one-factor ANOVA) and the pairwise comparisons of specific means were performed using the Tukey test. P<0.05 was considered statistically significant.
Ethics statement
All procedures were carried out in accordance with the guidelines for the care and use of laboratory animals of the National Council for the Control of Animal Experimentation Control (CONCEA), Brazil. All the experimental protocols were previously approved by the Ethics Research Committee of the UNESP–School of Pharmaceutical, protocol number 041/2012.
Results and discussion
The pseudo-ternary phase diagrams can describe the proportions of the components of the formulations which give rise to different systems from the mixture thereof.27 The interest region in the pseudo-ternary phase diagram, which includes the biocompatible liquid ME (LME) domain, is depicted in Figure 1. These systems are characterized by high W proportions, around 80%, and low proportions of O and S, which render them suitable for intravenous administration.
In the absence of the drug, it was possible to verify a liquid ME region built to the limits over 60% of the aqueous phase, up to 37% of the oil phase, and up to 60% of the surfactant. This LME region is surrounded by regions of conventional liquid emulsions and gel phases. However, in the presence of the drug, the domain region of LME was not only reduced, but also related to the surfactant proportion and enlarged regarding the oil phase proportion. Thus, the domain of the LME region was obtained over 33% of aqueous phase, up to 50% of oil phase, and up to 37% of surfactant. Similarly, this region of the LME was also surrounded by regions of conventional liquid emulsions. However, a region of gel emulsion was also defined within the limits of up to 60% of aqueous phase, between 5% and 50% of oil phase, and from 5% to 17% of surfactant.
It is important to note that high proportions of oil phase and surfactant significantly increase the viscosity of the system and, thus, the emulsions and the MEs became predominant as a gel phase system. This phenomenon may be related to the hydrophobicity of PC that can form the gel phase when it absorbs a small proportion of the aqueous phase. Furthermore, it is also widely known that increasing the oil phase proportion in liquid–liquid dispersed systems also leads to increased viscosity of the system.21
Suitable conditions for thermodynamic stability of the MEs include the combination of lipophilic surfactants such as phosphatidylcholine (HLB [hydrophilic-lipophilic balance] =4.5), hydrophilic surfactants such as sodium salt of fatty acids (HLB =20.4), and co-surfactants with a short chain structure such as GL (HLB =11.2).27
The pseudo-ternary phase diagrams in the absence and in the presence of MJ allowed assessment of the effect of the drug in the domain area of MEs, which also represented an important source of help for the selection of the composition of the MEs used for further studies. The specific compositions of MEs are detailed in Table 1.
The data in Figure 2 show that in the absence of the drug the O/S ratio appears to be negligible on the droplet size diameters. In fact, the size of the droplet diameters decreases from about 100 nm at very low O/S ratio to 80 nm at high O/S values. This phenomenon can be understood because even at O/S ratios the proportion of surfactant in the mixture was high enough to cover the entire interfacial area created by the formation of the dispersed droplets into the ME system. In fact, such conditions allow stabilizing the diameter values. In addition, further experiments replacing the soybean oil as the oil phase of the ME by the oily MJ drug revealed diameters of a magnitude similar to those presented by the incorporation of the drug into the soybean oil droplets at low O/S ratio.
Actually, 20% (w/w) of MJ incorporated into ME led to a dramatic increase in the diameter of droplets, especially at low O/S ratios. The droplet diameters increased from about 80 nm to 580 nm at very low O/S ratios such as 0.125. This effect was similar when MJ was directly used as the oil phase of the ME demonstrating that the local volume of the oil phase of the ME was not able to completely dissolve all the added MJ. Therefore, a substantial fraction of the drug stayed in the aqueous phase as dispersed droplets forming a mixed emulsion with soybean oil droplets incorporated of MJ that present a larger size than the empty ME. This feature also shows that the surfactant mixture, even at high proportions, has no ability to allow the expansion of the interfacial area from the oily drug to form nanometric sized droplets, which demonstrates the important role of the soybean oil in the ME composition.
On the other hand, the diameter of the droplets decreased with increasing the O/S ratio, converging to similar values of those of the empty ME at high O/S ratios in which the oil fraction predominates in the oil–surfactant mixture (Figure 2). Since the oil phase droplets of the ME can be considered as a separated phase from the aqueous phase, the incorporation of the drug into the droplets should be driven the hydrophobic effect in which the lipophilic drug, which also presents low polarity, interacts with the oily droplets. The literature shows that regardless of the electric charge of the carrier, for neutral lipophilic drugs such as MJ, the hydrophobic effect is the parameter that controls its process of incorporation.27–29
In fact, our experimental results clearly showed that the drug fraction, which formed dispersed droplets of greater size (about 580 nm), was able to incorporate into the dispersed soybean oil nanodroplets when the O/S ratio increased, leading to a gradual decrease in the droplets diameter with the drug incorporation.
The results of rheological analyses are shown in Figure 3.
As show in Figure 3, there is a strong dependence of the rheological profiles on the O/S ratios as well as the presence of the drug. In the absence of the drug, a Newtonian behavior was observed regardless of the O/S ratios (Figure 3A). A small area of antithixotropy with time dependent reconstruction was found at high O/S ratios in which the oil phase proportion predominates in the mixture. However, at smaller O/S values in which the O/S proportions are balanced or the surfactant fraction is predominant, the thixotropy disappears and only the feature of Newtonian flow is shown overlapping the upward and downward curves in the rheograms (Figure 3A).
The incorporation of the drug into the ME system changed its Newtonian profile compared to the empty ME. In this case, the ME revealed a pseudoplastic behavior for O/S ratios >0.125. There is a clear trend towards an increase in the shear-thinning property of the ME by increasing the O/S ratio. A comparison of the results of Figures 3A and B, shows that while in the absence of drug the pseudoplastic profile appeared only when the oil phase predominates in the O/S ratio, in the presence of drug the effect already exist from small proportions of the oil phase. This may be due to the presence of an additional fraction of lipophilic drug, which is dispersed as droplets and then, also plays a role in the oil phase, at a low proportion to the soybean oil. On the other hand, it is important to highlight the greater intensity in shear stress when the drug was incorporated into ME (Figure 3A and B), indicating a trend towards increase in the viscosity of the system when the drug is present.
Furthermore, the antithixotropic property was presented for all O/S ratios with a clear trend towards an increase in the hysteresis loop with increasing the oil phase. Therefore, the ME system containing 18% (w/w) of oil phase, 20% (w/w) of surfactant mixture, 62% (w/w) of aqueous phase (Table 1 and Figure 1) and a O/S ratio of 0.9 was chosen for further studies. It is important to note that this ME possesses hydrophilic characteristic, with a clear aspect and low viscosity, and it has a droplet diameter size of around 220 nm. Additionally, this stock formulation may be safely diluted with aqueous phase maintaining its feature of LME, as shown in the phase diagram of Figure 1. Although conceptually droplets with these dimensions do not properly fit in the general definition of ME, the system described here has optical transparency and is formed spontaneously by gentle stirring. Therefore, it has thermodynamic stability and, in the pharmaceutical field, can be safely used by any route of administration including intraperitoneal and intravenous ones.
Regardless of the carrier in which MJ is administered, the literature shows that for most of the in vitro experiments the biological activity was evaluated from a range of concentration between some micrometers (μM) to a few millimeters (mM).4,5,14,31 Usually to solubilize MJ at such concentrations for in vivo antitumor activity evaluation, parenteral nutrition emulsions,12 vegetable oil,8 and ethanol17 have been used. In this work, we have determined the antitumor activity of MJ at concentrations similar to the other antitumor substances, such as DOX.
The in vivo antitumor activity of the MJ-loaded ME and the positive and negative controls was evaluated using Balb/c mice. The results of the tumor growth inhibition are shown in Figure 4. The tumor tissues were carefully removed from mice after complete treatment and timing of expected tumor growth. After removal, the tumor tissues were weighed and measured to calculate tumor volume to obtain the tumor growth inhibition.
The volume of the tumor mass can provide adequate information about the tendency of treatment related to tumor progression. Thus, the volume was used as reference to calculate the extent of TIR (see Equation 2).
The Control Group that received the saline solution showed the highest tumor mass of 7.0+3.9 g and tumor volume of 6.6 mm3, which were used as reference values for the absence of tumor inhibition growth and used for normalization of the results of Figure 4.
On the other hand, an inhibition of 62.1% in the tumor volume was verified for the positive control, DOX. It should be emphasized that it was not possible to test the MJ alone due to its negligible solubility in aqueous medium. However, the treatment groups of MJ-loaded ME containing 2.5 mg/kg, 5 mg/kg, and 10 mg/kg of MJ revealed an inhibition of tumor mass of approximately 43.9%, 68.2%, and 66.7%, respectively. These results show that a dose-dependent effect from 2.5 mg/kg to 5 mg/kg remained constant up to the test limit dose of 10 mg/kg.
These results were of utmost importance because the values were comparable to those obtained for the positive control, DOX, and higher than those obtained for the negative control of saline solution, as well as the MJ-unloaded ME. Statistical difference was observed only between the groups treated with MJ-unloaded ME and MJ-loaded ME with doses from 5 mg/kg (P<0.05). The differences in the effect obtained between the DOX and MJ-loaded ME was statistically insignificant for all administered doses of MJ. The results in Figure 4 also show a small tumor growth inhibition for empty ME when compared to the negative saline control, but it was not statistically significant for P<0.05. However, such negligible inhibition can be understood as the surface-active stabilizer mixture of ME when in the bloodstream of animals can increase the membrane permeability, impairing the growth of tumor cells.
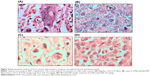
After tumor growth with subsequent treatment, the animals of each group were sacrificed and the tumors were removed for analysis. Histological analysis by optical microscopy to evaluate the presence of typical and atypical mitoses and determination of the inhibition of tumor angiogenesis were also performed.
It is well known that cell division is a phenomenon associated with cell proliferation, and then the presence of mitoses indicates that the tumor cells are in constant division and are consequently increasing the tumor mass. The analysis of tumor tissues revealed a high frequency of mitosis (circled area), mainly in tumor sections of the negative controls of saline and MJ-unloaded ME (Figure 5A and B). However, similar histological pattern also occurred, with less intensity, with the positive control, DOX, and those treated with MJ-loaded ME (Figure 5C and D). Figure 5C shows the presence of blood vessels around the tumor cells that are characteristic of angiogenesis. Angiogenesis is a common feature in cancer tissues because the tumor cells need to be fed to keep growing. In addition, angiogenesis may cause tumor cells to spread out to other sites in the body, by the same route through which the nutrients arrived, resulting in metastasis.16
The degree of angiogenesis was determined from the histological analysis of tumor tissue sections. The results of the MAGS score are shown in Figure 6 and are expressed as the mean ± SD in arbitrary values obtained from ten random field determinations in each blade of five animals.
  | Figure 6 Determination of the angiogenesis using the MAGS score. |
The Control Group of saline solution and MJ-unloaded ME showed a similar degree of angiogenesis of 37.1 and 40.8, respectively. The positive control of DOX showed a degree of angiogenesis of approximately 34.9, which was a value slightly lower than the negative control, indicating a weak effect on the angiogenesis.
The groups treated with MJ-loaded ME had degrees of angiogenesis of 39.1 (2.5 mg/kg), 37.1 (5 mg/kg), and 33.9 (10 mg/kg). The difference between the effects on the angiogenesis observed at the lower and higher dose intervals of MJ-loaded ME was dose dependent, with the values decreasing with increasing the administered dose in animals. However, this result was not statistically significant when compared to DOX, saline solution, or MJ-unloaded ME (Figure 6). Although angiogenesis inhibition is not the main mechanism of action of both DOX and MJ, the results of this study show that both compounds inhibited the growth of new blood vessels in tumor tissues.
When administered at the highest dose (10 mg/kg), the MJ-loaded ME was more effective than DOX in inhibiting angiogenesis. Indeed, the antimetastatic and antiangiogenic effects of MJ were previously described in the literature.17,33 It is well known that the antitumor effect of MJ involves various mechanisms. However, the inhibition of angiogenesis was described as decisively contributing to cancer therapy.15,16 In addition, recent published studies have described that the MJ abolishes the migration, invasion, and angiogenesis in gastric cancer cells.4 Therefore, jasmonate derivatives are effective therapeutic agents against cancer.
The evaluation of acute toxicity is an important step to evaluate the toxicological profile of a substance. It also provides information about the risk to human health resulting from the administration of the drug. The 50% lethal dose (LD50) may be defined as a single dose causing death in 50% of the tested animals and is expressed as mg/kg of the test drug. In this study we have tested doses within the minimum limits of 70 mg/kg and up to 350 mg/kg. The experimental animal groups that received MJ-loaded ME, MJ-unloaded ME, and MJ dispersed in TW solution did not show any signs of toxicity and no death was observed during the 14 days after administration. These data demonstrate that for both the vehicle and the MJ-loaded ME, and MJ-unloaded ME and empty ME vehicle, the systems had a nontoxic behavior in the conditions of this study. Besides, due to the overall survival of the animals in all treatments, it was impossible to calculate the LD50 value. However, we can suggest that for both MJ-loaded ME and MJ-unloaded ME (dispersed in TW surfactant solution) the LD50 was greater than 350 mg/kg. These results are in agreement with the literature, which shows that jasmonate derivatives are nontoxic at doses higher than the therapeutic ones of other antitumor compounds. Moreover, doses from nanomoles (nM) to a few micromoles (mM) can be used.14 The administration by intravenous route of doses of 75 mg/kg in mouse and 236 mg/kg in adult mice proved to be nontoxic.12,14,33 Presumably, the drug dose administration by intraperitoneal route should have higher values than that administrated by intravenous route.
Furthermore, it is important to note that after treatment, most animals showed transient drowsiness with full recovery hours later. This symptom may be related to the deregulation of the cellular bioenergetics with ATP depletion involved in the mechanism of action of jasmonate derivatives.10
Another important point to be considered is related to the negligible solubility of MJ in aqueous medium. Data from the literature have reported the use of inappropriate procedures for determination of the in vivo effect of jasmonate derivatives, such as adding the drug into commercial emulsions,12,33 the use of a solution in vegetable oil for intraperitoneal administration,34 or the use of ethanol solutions of jasmonates.17,33 All these techniques for mixing a drug with an established formulation can lead to formulation instability and are completely unsuitable for in vivo studies. However, it seems evident that the negligible water solubility of MJ is one of the limiting factors for its application as a therapeutic agent and for in vitro tests. To circumvent this problem, emulsions designed for use in parenteral nutrition and vegetal oil has been used as a vehicle for administration of jasmonate derivatives.17,33 However, this is not a recommended procedure because the lipid characteristic of these compounds can destabilize the emulsions, leading to formation of aggregates, increasing the droplet size, or even inducing phase separation in a short time, which is unsuitable for parenteral administration. In this work, a safe vehicle, thermodynamically stable, represented by a biocompatible O/W ME was described. Thus, a significant contribution of this study was to provide a biocompatible nanostructured carrier with ability to solubilize MJ proportions for obtaining the in vivo antitumor activity.
Conclusion
The negligible water solubility of MJ is one of the limiting factors for its administration as a therapeutic agent by parenteral route. This paper presented a biocompatible ME system, which is clear and stable either in the absence or in the presence of MJ. Due to its characteristic of aqueous nanodroplets dispersion, this system can provide conditions for the parenteral delivery of the drug. In fact, the O/W ME system has the ability to dissolve about 20% (w/w) of MJ, which is much higher than the dose required to achieve the MJ in vivo antitumor activity. The in vivo antitumor activity revealed that the effect of the MJ-loaded ME was comparable to DOX effect by inhibiting tumor growth and killing tumor cells. A remarkable absence of toxicity was achieved for MJ-unloaded ME, MJ-loaded ME, and the empty vehicle. Moreover, its stable aqueous dispersion characteristic, its high intensity ability of tumor-inhibiting effect and its lack of toxicity makes the ME system here presented a promising carrier for MJ administration by parenteral route.
Acknowledgments
The authors gratefully acknowledge the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-CAPES, the Conselho Nacional de Desenvolvimento Científico and Tecnologico-CNPq for the financial support, and Glenn Hawes, M Ed English, from the University of Georgia, for editing this manuscript.
Disclosure
The authors report no conflicts of interest.
References
Chen X, Ding G, Gao Q, et al. A human anti-c-Met Fab fragment conjugated with doxorubicin as targeted chemotherapy for hepatocellular carcinoma. PLoS ONE. 2013;8(5):e63093. | ||
Máthé C, Szénási G, Sebestény A, et al. Protective effect of CV247 against cisplatin nephrotoxicity in rats. Hum Exp Toxicol. 2013;33(8): 789–799. | ||
Islam R, Chyou P, Burmester JK. Modeling efficacy of bevacizumab treatment for metastatic colon cancer. J Cancer. 2013;4:330–335. | ||
Zheng L, Li D, Xiang X, et al. Methyl jasmonate abolishes the migration, invasion and angiogenesis of gastric cancer cells through down-regulation of matrix metalloproteinase 14. BMC Cancer. 2013;13:74. | ||
Park C, Jin CY, Hwang HJ, et al. J7, a methyl jasmonate derivative, enhances TRAIL-mediated apoptosis through up-regulation of reactive oxygen species generation in human hepatoma HepG2 cells. Toxicol In Vitro. 2012;26:86–93. | ||
Milrot E, Jackman A, Kniazhanski T, Gonen P, Flescher E, Sherman L. Methyl jasmonate reduces the survival of cervical cancer cells and downregulates HPV E6 and E7, and surviving. Cancer Lett. 2012;319: 31–38. | ||
Kniazhanski T, Jackman A, Heyfets A, Gonen P, Flescher E, Sherman L. Methyl jasmonate induces cell death with mixed characteristics of apoptosis and necrosis in cervical cancer cells. Cancer Lett. 2008;271:34–46. | ||
Klippel S, Jakubikova J, Delmore J, et al. Methyljasmonate displays in vitro and in vivo activity against multiple myeloma cells. Br J Haematol. 2012;159:340–351. | ||
Yeruva L, Elegbedea JA, Carper SW. Methyl jasmonate decreases membrane fluidity and induces apoptosis via tumor necrosis factor receptor 1 in breast cancer cells. Anticancer Drugs. 2008;19:766–776. | ||
Cohen S, Flescher E. Methyl jasmonate: a plant stress hormone as an anti-cancer drug. Phytochemistry. 2009;70:1600–1609. | ||
Flescher E. Jasmonates in cancer therapy. Cancer Lett. 2007;245: 1–10. | ||
Fingrut O, Flescher E. Plant stress hormones suppress the proliferation and induce apoptosis in human cancer cells. Leukemia. 2002;16: 608–616. | ||
Rotem R, Heyfets A, Fingrut O, Blickstein D, Shaklai M, Flescher E. Jasmonates: novel anticancer agents acting directly and selectively on human cancer cell mitochondria. Cancer Res. 2005;65:1984–1993. | ||
Cesari MC, Carvalho E, Rodrigues MF, Mendonça BS, Amôedo ND, Rumjanek FD. Mehtyl Jamonate: putative mechanisms of action on cancer cells cycle, metabolism, and apoptosis. Int J Cell Biol. 2014;2014:25. [Article ID 572097]. | ||
Takara K, Hatakeyama H, Kibria G, Ohga N, Hida K, Harashima H. Size-controlled, dual-ligand modified liposomes that target the tumor vasculature. J Control Release. 2012;162:225–232. | ||
George DJ, Moore C. Angiogenesis inhibitors in clinical oncology. Update Cancer Ther. 2006;1:429–434. | ||
Pereira Lopes J, Barbosa M, Stella C, et al. In vivo anti-angiogenic effects further support the promise of the antineoplasic activity of methyl jasmonate. Braz J Biol. 2010;70:443–449. | ||
Formariz TP, Chiavacci LA, Sarmento VHS, Franzini CM, Silva-Jr AA, Oliveira AG. Structural changes of biocompatible neutral microemulsions stabilized by mixed surfactant containing soya phosphatidylcholine and its relationship with doxorubicin release. Colloids Surf B. 2008;63:287–295. | ||
Franzini MC, Pestana KC, Molina EF, Scarpa MV, Egito EST, Oliveira AG. Structural properties induced by the composition of biocompatible phospholipid-based microemulsion and amphotericin b association. J Biomed Nanotech. 2012;8:350–359. | ||
Bottos KM, Oliveira AG, Bersanetti PA, et al. Corneal absorption of a new riboflavin-nanostructured system for transepithelial collagen cross-linking. PLoS One. 2013;8(6):e66408. | ||
Silva GBRF, Scarpa MV, Rossanezi G, Egito EST, Oliveira AG. Development and characterization of biocompatible isotropic and anisotropic oil-in-water colloidal dispersions as a new delivery system for methyl dihydrojasmonate antitumor drug. Int J Nanomedicine. 2014;9: 867–876. | ||
Oliveira AG, Rossanezzi G, Formariz TP, Egito EST, Scarpa MV. Effect of cationic micelles on the decomposition of alpha-aminophenyl cephalosporins. Colloids Surf B. 2010;79:372–376. | ||
Zhou J, Mukherjee P, Gugger E, Tanaka T, Blackburn GL, Clinton SK. Inhibition of murino bladder tumorigenesis by soy isoflavones via alterations in cell cycle, apoptosis, and angiogenesis. Cancer Res. 1998;58: 5231–5238. | ||
Sorensen AG, Patel S, Harmath C, et al. Comparison of diameter and perimeter methods for tumor volume calculation. J Clinical Oncol. 2001;19: 551–557. | ||
Brem S, Cotran R, Folkman J. Tumor angiogenesis: a quantitative method for histological grading. J Nat Cancer Inst. 1972;48:347–356. | ||
Shankar R, Tiwary SK, Khanna R, Kumar M, Khanna AK. Tumor Angiogenesis: determined By VEGF Expression, MAGS Scoring, Doppler Study, As Prognostic Indicator In Carcinoma Breast. Internet J Surg. 2006;8(1):5. | ||
Oliveira AG, Chiavachi LA, Scarpa MV, Egito EST. Microemulsions: physico-chemical approaches on the systems for pharmaceutical applications. In: Songjun L, editor. Current Focus on Colloids and Surfaces. Vol. 1. Kerala: Transword Research Network; 2009:57–84. | ||
Oliveira AG, Nothenberg MS, Cuccovia IM, Chaimovich H. Micellar catalysis of the intramolecular aminolysis of the β-lactam antibiotic cephaclor. J Phys Org Chem. 1991;4:19–24. | ||
Correa MA, Scarpa MV, Franzini MC, Oliveira AG. On the incorporation of the non-steroidal anti-inflammatory naproxen into cationic O/W microemulsions. Colloids Surf B Biointerfaces. 2005;43:108–114. | ||
Park C, Jin C-Y, Kim G-Y. A methyl jasmonate derivative, J-7, induces apoptosis in human hepatocarcinoma Hep3B cells in vitro. Toxicol In Vitro. 2010;24(7):1920–1926. | ||
Reischer D, Heyfets A, Shimony S, Nordenberg J, Kashman Y, Flescher E. Effects of natural and novel synthetic jasmonates in experimental metastatic melanoma. Br J Pharmacol. 2007;150:739–749. | ||
Klipel S, Jakubikova J, Delmore J. Methyl jasmonates displays in vitro and in vivo activity against multiple myeloma cells. Br J Haemathol. 2012; 159:340–351. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.